Key Points
-
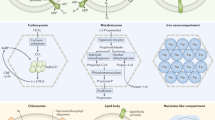
The complexity of bacterial cells is discussed, together with an explanation of the advantages that can be provided by an organelle that sequesters related enzymes in a pathway. The carboxysome is introduced as a bacterial microcompartment that functions to enhance CO2 fixation.
-
The discovery and early characterization of the carboxysome is discussed. Evidence for the function of the carboxysome is reviewed, together with the first isolation and purification of carboxysomes and identification of the proteins that constitute the shell.
-
The two types of carboxysome (α and β) are introduced. The important role of carbonic anhydrase is discussed, together with experiments that identify which genes encode carbonic anhydrase in the two types of carboxysome.
-
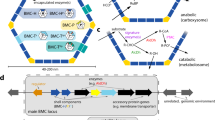
The main shell proteins and their genes are introduced for the two types of carboxysome. Other proteins that are associated with the shell are also introduced, most of which are not yet fully understood.
-
A discussion of the main shell protein from the carboxysome, which belongs to a family of proteins (the bacterial microcompartment domain family) that is widespread among bacteria is provided. Diverse microcompartments exist, with presumably similar shells, but distinct metabolic enzymes are encapsulated inside particular microcompartments. The list of organisms with microcompartments includes Escherichia coli and serovars of Salmonella enterica.
-
Recent structural studies on the carboxysome are highlighted. These include moderate-resolution reconstructions on the basis of cryo-electron tomography and high-resolution crystal structures of several shell proteins. The emerging principles of assembly and transport across the shell are presented.
-
Outstanding questions and future studies are indicated, the role of structure-guided mutagenesis is noted and the long-term goal of numerically modelling the behaviour of microcompartments is suggested, together with the long-term prospect for protein-engineering studies.
Abstract
Many bacteria contain intracellular microcompartments with outer shells that are composed of thousands of protein subunits and interiors that are filled with functionally related enzymes. These microcompartments serve as organelles by sequestering specific metabolic pathways in bacterial cells. The carboxysome, a prototypical bacterial microcompartment that is found in cyanobacteria and some chemoautotrophs, encapsulates ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO) and carbonic anhydrase, and thereby enhances carbon fixation by elevating the levels of CO2 in the vicinity of RuBisCO. Evolutionarily related, but functionally distinct, microcompartments are present in diverse bacteria. Although bacterial microcompartments were first observed more than 40 years ago, a detailed understanding of how they function is only now beginning to emerge.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gitai, Z. The new bacterial cell biology: moving parts and subcellular architecture. Cell 120, 577–586 (2005).
Shively, J. M. (ed.) Microbiology Monographs: Complex Intracellular Structures in Prokaryotes (Springer, Berlin/Heidelberg, 2006).
Lowe, J. & Amos, L. A. Crystal structure of the bacterial cell-division protein FtsZ. Nature 391, 203–206 (1998).
van den Ent, F., Amos, L. & Lowe, J. Bacterial ancestry of actin and tubulin. Curr. Opin. Microbiol. 4, 634–638 (2001).
van den Ent, F., Amos, L. A. & Lowe, J. Prokaryotic origin of the actin cytoskeleton. Nature 413, 39–44 (2001).
Cannon, G. C. et al. Microcompartments in prokaryotes: carboxysomes and related polyhedra. Appl. Environ. Microbiol. 67, 5351–5361 (2001).
Price, G. D., Sültemeyer, D., Klughammer, B., Ludwig, M. & Badger, M. R. The functioning of the CO2 concentrating mechanism in several cyanobacterial strains: a review of general physiological characteristics, genes, proteins, and recent advances. Can. J. Bot. 76, 973–1002 (1998).
Bobik, T. A. Polyhedral organelles compartmenting bacterial metabolic processes. Appl. Microbiol. Biotechnol. 70, 517–525 (2006).
Huang, X., Holden, H. M. & Raushel, F. M. Channeling of substrates and intermediates in enzyme-catalyzed reactions. Annu. Rev. Biochem. 70, 149–180 (2001).
Drews, G. & Niklowitz, W. Beiträge zur Cytologie der Blaualgen. II. Zentroplasma und granulare Einschlüsse von Phormidium uncinatum. Arch. Mikrobiol. 24, 147–162 (1956).
Shively, J. M. Inclusion bodies of prokaryotes. Annu. Rev. Microbiol. 28, 167–187 (1974).
Shively, J. M., Ball, F., Brown, D. H. & Saunders, R. E. Functional organelles in prokaryotes: polyhedral inclusions (carboxysomes) of Thiobacillus neapolitanus. Science 182, 584–586 (1973). Reported the first isolation and description of polyhedral bodies and proposed the name carboxysome.
Codd, G. A. Carboxysomes and ribulose bisphosphate carboxylase/oxygenase. Adv. Microbial. Physiol. 29, 115–164 (1988).
Codd, G. A. & Marsden, W. J. N. The carboxysomes (polyhedral bodies) of autotrophic prokaryotes. Biol. Rev. 59, 389–422 (1984).
Shively, J. M. et al. Functional inclusions in prokaryotic cells. Int. Rev. Cytol. 113, 35–100 (1988).
Heinhorst, S., Cannon, G. C. & Shively, J. M. in Complex Intracellular Structures in Prokaryotes (ed. Shively, J. M.) 141–165 (Springer-Verlag, Berlin, 2006).
Shively, J. M., Ball, F. L. & Kline, B. W. Electron microscopy of the carboxysomes (polyhedral bodies) of Thiobacillus neapolitanus. J. Bacteriol. 116, 1405–1411 (1973).
Shively, J. M. & English, R. S. The carboxysome, a prokaryotic organelle: a mini-review. Can. J. Bot. 69, 957–962 (1991).
Cannon, G. C., English, R. S. & Shively, J. M. In situ assay of ribulose-1,5-bisphosphate carboxylase/oxygenase in Thiobacillus neapolitanus. J. Bacteriol. 173, 1565–1568 (1991). The first direct evidence that carboxysomal RuBisCO exists in a fully activated form in vitro , which demonstrated that carboxysomes are metabolically active and are not just storage bodies for the enzyme.
Cannon, G. C. & Shively, J. M. Characterization of a homogeneous preparation of carboxysomes from Thiobacillus neapolitanus. Arch. Microbiol. 134, 52–59 (1983). Reported the detailed biochemical characterization of carboxysomes that were purified almost to homogeneity.
Buedeker, R. F., Cannon, G. C., Kuenen, J. G. & Shively, J. M. Relations between D-ribulose-1,5 bisphosphate carboxylase, carboxysomes, and CO2 fixing capacity in the obligate chemolithotroph Thiobacillus neapolitanus grown under different limitations in the chemostat. Arch. Microbiol. 124, 185–189 (1980).
Turpin, D. H., Miller, A. G. & Canvin, D. T. Carboxysome content of Synechococcus leopoliensis (cyanophyta) in response to inorganic carbon. J. Phycol. 20, 249–253 (1984).
Price, G. D. & Badger, M. R. Isolation and characterization of high CO2-requiring-mutants of the cyanobacterium Synechococcus PCC7942: two phenotypes that accumulate inorganic carbon but are apparently unable to generate CO2 within the carboxysome. Plant Physiol. 91, 514–525 (1989).
Reinhold, L., Zviman, M. & A, Kaplan, A. quantitative model for inorganic carbon fluxes and photosynthesis in cyanobacteria. Plant Physiol. Biochem. 27, 945–954 (1989).
Price, G. D. & Badger, M. R. Expression of human carbonic anhydrase in the cyanobacterium Synechococcus PCC7942 creates a high CO2-requiring phenotype: evidence for a central role for carboxysomes in the CO2 concentrating mechanism. Plant Physiol. 91, 505–513 (1989). Experimentally established that carbonic anhydrase activity must be localized to the carboxysome rather than to the cytosol for CO 2 fixation.
Price, G. D., Howitt, S. M., Harrison, K. & Badger, M. R. Analysis of a genomic DNA region from the cyanobacterium Synechococcus sp. strain PCC7942 involved in carboxysome assembly and function. J. Bacteriol. 175, 2871–2879 (1993). Identified several genes that are required for carboxysome formation in cyanobacteria.
Suzuki, E., Fukuzawa, H. & Miyachi, S. Identification of a genomic region that complements a temperature-sensitive, high CO2-requiring mutant of the cyanobacterium, Synechococcus sp. PCC7942. Mol. Gen. Genet. 226, 401–408 (1991).
English, R. S., Jin, S. & Shively, J. M. Use of electroporation to generate a Thiobacillus neapolitanus carboxysome mutant. Appl. Environ. Microbiol. 61, 3256–3260 (1995).
Holthuizen, Y. A., Van Breemen, J. F. L., Kuenen, J. G. & Konigs, W. N. Protein composition of the carboxysomes of Thiobacillus neapolitanus. Arch. Microbiol. 144, 398–404 (1986).
English, R. S., Lorbach, S. C., Qin, X. & Shively, J. M. Isolation and characterization of a carboxysome shell gene from Thiobacillus neapolitanus. Mol. Microbiol. 12, 647–654 (1994). The first study to correlate a shell protein with its corresponding gene.
Cannon, G. C., Heinhorst, S., Bradburne, C. E. & Shively, J. M. Carboxysome genomics: a status report. Funct. Plant. Biol. 29, 175–182 (2002). Pointed out that Prochlorococcus marine cyanobacteria contain carboxysome operons that are related to those of chemolithotrophic bacteria.
Shively, J. M., Lorbach, S. C., Jin, S. & Baker, S. H. in Microbial Growth on C1 Compounds (eds Lidstrom, M. & Tabita, F. R.) 56–63 (Kluwer, Dordrecht, 1996).
Baker, S. H. et al. The correlation of the gene csoS2 of the carboxysome operon with two polypeptides of the carboxysome in Thiobacillus neapolitanus. Arch. Microbiol. 172, 233–239 (1999).
Baker, S. H., Williams, D. S., Aldrich, H. C., Gambrell, A. C. & Shively, J. M. Identification and localization of the carboxysome peptide Csos3 and its corresponding gene in Thiobacillus neapolitanus. Arch. Microbiol. 173, 278–283 (2000).
Tanaka, S. et al. Atomic-level models of the bacterial carboxysome shell. Science 319, 1083–1086 (2008). Showed that certain previously uncharacterized carboxysome proteins assemble as pentamers, postulated that these proteins form the vertices of the carboxysome shell and provided the first atomic level models for the assembled shell.
Field, C. B., Behrenfeld, M. J., Randerson, J. T. & Falkowski, P. Primary production of the biosphere: integrating terrestrial and oceanic components. Science 281, 237–240 (1998).
Badger, M. R., Hanson, D. T. & Price, G. D. Evolution and diversity of CO2 concentrating mechanisms in cyanobacteria. Funct. Plant Biol. 29, 407–416 (2002).
Badger, M. R. & Price, G. D. CO2 concentrating mechanisms in cyanobacteria: molecular components, their diversity and evolution. J. Exp. Bot. 54, 609–622 (2003).
So, A. K. et al. A novel evolutionary lineage of carbonic anhydrase (ɛ class) is a component of the carboxysome shell. J. Bacteriol. 186, 623–630 (2004). The first study to show the presence of carbonic anhydrase in carboxysomes as a protein that is tightly associated with, or is part of, the carboxysome shell. Owing to sequence divergence, CsoS3 could not be shown to be homologous to known families of carbonic anhydrases.
Sawaya, M. R. et al. The structure of β-carbonic anhydrase from the carboxysomal shell reveals a distinct subclass with one active site for the price of two. J. Biol. Chem. 281, 7546–7555 (2006). The structure reported in this paper revealed that the α-carboxysome carbonic anhydrase (CscoS3 or CsoSCA) is a member of the β-family of carbonic anhydrases, but with the loss of an active site and addition of a domain of unknown function.
Price, G. D., Coleman, J. R. & Badger, M. R. Association of carbonic anhydrase activity with carboxysomes isolated from the cyanobacterium Synechococcus PCC7942. Plant Physiol. 100, 784–793 (1992).
Cot, S. S., So, A. K. & Espie, G. S. A multiprotein bicarbonate dehydration complex essential to carboxysome function in cyanobacteria. J. Bacteriol. 190, 936–945 (2008). Used various approaches to identify several protein–protein interactions in the β-carboxysome, identified CcmM as a key organizing component and argued that the CcaA carbonic anhydrase is a component of the Synechocystis sp. PCC 6803 carboxysome.
Long, B. M., Badger, M. R., Whitney, S. M. & Price, G. D. Analysis of carboxysomes from Synechococcus PCC7942 reveals multiple Rubisco complexes with carboxysomal proteins CcmM and CcaA. J. Biol. Chem. 282, 29323–29335 (2007). Identified protein–protein complexes between proteins from the β-carboxysome that involve RuBisCO, CcaA and CcmM.
Reinhold, L., Kosloff, R. & Kaplan, A. A model for inorganic carbon fluxes and photosynthesis in cyanobacterial carboxysomes. Can. J. Bot. 69, 984–988 (1991).
Heinhorst, S. et al. Characterization of the carboxysomal carbonic anhydrase CsoSCA from Halothiobacillus neapolitanus. J. Bacteriol. 188, 8087–8094 (2006). The first study to provide biochemical evidence that the carboxysome shell acts as a barrier for CO 2.
Dou, Z. et al. CO2 fixation kinetics of Halothiobacillus neapolitanus mutant carboxysomes lacking carbonic anhydrase suggest the shell acts as a diffusional barrier for CO2 . J. Biol. Chem. 283, 10377–10384 (2008).
Cai, F., Heinhorst, S., Shively, J. M. & Cannon, G. C. Transcript analysis of the Halothiobacillus neapolitanus cso operon. Arch. Microbiol. 189, 141–150 (2008).
Chen, P., Andersson, D. I. & Roth, J. R. The control region of the pdu/cob regulon in Salmonella typhimurium. J. Bacteriol. 176, 5474–5482 (1994). First report of the presence and putative function of a bacterial microcompartment shell protein gene in a heterotrophic organism.
Havemann, G. D., Sampson, E. M. & Bobik, T. A. PduA is a shell protein of polyhedral organelles involved in coenzyme B12-dependent degradation of 1,2-propanediol in Salmonella enterica serovar Typhimurium LT2. J. Bacteriol. 184, 1253–1261 (2002). Showed that bacterial microcompartments were formed by S. typhimurium in the presence of 1,2-propanediol, which confirmed that bacterial microcompartments are involved in metabolic processes other than CO 2 fixation. PduA was also localized to the shell.
Penrod, J. T. & Roth, J. R. Conserving a volatile metabolite: a role for carboxysome-like organelles in Salmonella enterica. J. Bacteriol. 188, 2865–2874 (2006).
Stojiljkovic, I., Baumler, A. J. & Heffron, F. Ethanolamine utilization in Salmonella typhimurium: nucleotide sequence, protein expression, and mutational analysis of the cchA cchB eutE eutJ eutG eutH gene cluster. J. Bacteriol. 177, 1357–1366 (1995).
Brinsmade, S. R., Paldon, T. & Escalante-Semerena, J. C. Minimal functions and physiological conditions required for growth of Salmonella enterica on ethanolamine in the absence of the metabolosome. J. Bacteriol. 187, 8039–8046 (2005).
Havemann, G. D. & Bobik, T. A. Protein content of polyhedral organelles involved in coenzyme B12-dependent degradation of 1,2-propanediol in Salmonella enterica serovar Typhimurium LT2. J. Bacteriol. 185, 5086–5095 (2003). Purified and characterized the Pdu microcompartment.
Bateman, A. et al. The Pfam protein families database. Nucleic Acids Res. 32, D138–D141 (2004).
Wackett, L. P., Frias, J. A., Seffernick, J. L., Sukovich, D. J. & Cameron, S. M. Genomic and biochemical studies demonstrating the absence of an alkane-producing phenotype in Vibrio furnissii M1. Appl. Environ. Microbiol. 73, 7192–7198 (2007).
Schmid, M. F. et al. Structure of Halothiobacillus neapolitanus carboxysomes by cryo-electron tomography. J. Mol. Biol. 364, 526–535 (2006). Provided the first three-dimensional reconstructions of an α-carboxysome by cryo-electron tomography.
Ebert, A. Ribulose-1,5-bisphosphate carboxylase in Nitrobacter. Thesis, Univ. Hamburg (1982).
Shively, J. M., Bock, E., Westphal, K. & Cannon, G. C. Icosahedral inclusions (carboxysomes) of Nitrobacter agilis. J. Bacteriol. 132, 673–675 (1977).
Iancu, C. V. et al. The structure of isolated Synechococcus strain WH8102 carboxysomes as revealed by electron cryotomography. J. Mol. Biol. 372, 764–773 (2007). Provided three-dimensional reconstructions of α-carboxysomes by cryo-electron tomography, together with models for the packing of RuBisCO enzymes inside the carboxysome.
Kerfeld, C. A. et al. Protein structures forming the shell of primitive bacterial organelles. Science 309, 936–938 (2005). Provided the first atomic resolution crystal structures of the proteins that constitute the bulk of the carboxysome shell, which revealed hexameric shell protein assemblies that were arranged in tightly packed two-dimensional layers, with small pores that were suggestive of molecular transport.
Tsai, Y. et al. Structural analysis of CsoS1A and the protein shell of the Halothiobacillus neapolitanus carboxysome. PLoS Biol. 5, 1345–1354 (2007). Provided the first crystal structure of a shell protein from an α-carboxysome, together with a comparison of the structure and packing of shell proteins from the previously elucidated β-carboxysome.
Johnson, J. E. & Speir, J. A. Quasi-equivalent viruses: a paradigm for protein assemblies. J. Mol. Biol. 269, 665–675 (1997).
Caspar, D. L. & Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 27, 1–24 (1962).
Eisenhut, M. et al. The plant-like C2 glycolate cycle and the bacterial-like glycerate pathway cooperate in phosphoglycolate metabolism in cyanobacteria. Plant Physiol. 142, 333–342 (2006).
Eisenhut, M. et al. Long-term response toward inorganic carbon limitation in wild type and glycolate turnover mutants of the cyanobacterium Synechocystis sp. strain PCC 6803. Plant Physiol. 144, 1946–1959 (2007).
Liu, Y. et al. PduL is an evolutionarily distinct phosphotransacylase involved in B12-dependent 1,2-propanediol degradation by Salmonella enterica serovar Typhimurium LT2. J. Bacteriol. 189, 1589–1596 (2007).
Kofoid, E., Rappleye, C., Stojiljkovic, I. & Roth, J. The 17-gene ethanolamine (eut) operon of Salmonella typhimurium encodes five homologues of carboxysome shell proteins. J. Bacteriol. 181, 5317–5329 (1999).
DeLano, W. L. The PyMOL User's Manual (DeLano Scientific, San Carlos, California, 2002).
So, A. K. & Espie, G. S. Cloning, characterization and expression of carbonic anhydrase from the cyanobacterium Synechocystis PCC6803. Plant Mol. Biol. 37, 202–215 (1998).
Acknowledgements
The authors thank S. Tanaka for figure preparation and T. Bobik for discussions. T.O.Y. and C.A.K. have been supported by grants from the BER programme of the Department of Energy Office of Science and the US Department of Agriculture, G.C.C. and S.H. have been supported by the National Science Foundation (grant numbers MCB-0444568 and DMR-0213883, repectively) and G.C.C. and S.H. have been supported by the T.W. Bennett Foundation at The University of Southern Mississippi.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Entrez Genome Project
Prochlorococcus marinus str. MIT 9313
Prochlorococcus marinus subsp. pastoris str. CCMP1986
Rhodopseudomonas palustris BisB18
Salmonella enterica serovar Typhimurium
Synechococcus elongatus PCC 7942
FURTHER INFORMATION
Glossary
- Bacterial microcompartment
-
A large, polyhedral, proteinaceous structure that functions as an organelle by encapsulating specific enzymes inside a protein shell that is reminiscent of a viral capsid.
- Carboxysome
-
A polyhedral bacterial microcompartment that enhances carbon fixation by encapsulating the ribulose-1,5-bisphosphate carboxylase/oxygenase and carbonic anhydrase enzymes.
- CO2 fixation
-
The process by which carbon in the biosphere is converted from an inorganic form (for example, CO2) into organic molecules.
- Ribulose-1,5-bisphosphate carboxylase/oxygenase
-
The enzyme that fixes carbon by combining CO2 with the five-carbon compound ribulose-1,5-bisphosphate to form two molecules of the three-carbon compound phosphoglycerate.
- Carbonic anhydrase
-
An enzyme that catalyses the interconversion of bicarbonate and CO2.
- Icosahedron
-
A regular geometric solid that has 20 triangular faces and 12 vertices; a large icosahedron can be constructed by assembling hexagons together on the triangular faces with pentagons at the 12 vertices.
Rights and permissions
About this article
Cite this article
Yeates, T., Kerfeld, C., Heinhorst, S. et al. Protein-based organelles in bacteria: carboxysomes and related microcompartments. Nat Rev Microbiol 6, 681–691 (2008). https://doi.org/10.1038/nrmicro1913
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro1913
This article is cited by
-
Tandem-biocatalysis reactors constructed by topological evolution of CaCO3 particles into hollow metal hydroxide spheres
Nature Communications (2023)
-
Towards engineering a hybrid carboxysome
Photosynthesis Research (2023)
-
Trade-offs and design principles in the spatial organization of catalytic particles
Nature Physics (2022)
-
Varying protein architectures in 3-dimensions for scaffolding and modulating properties of catalytic gold nanoparticles
Amino Acids (2022)
-
Role of carbon-dioxide sequestering bacteria for clean air environment and prospective production of biomaterials: a sustainable approach
Environmental Science and Pollution Research (2022)