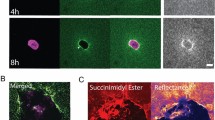
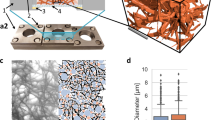
Key Points
-
Different tissues have unique and specialized extracellular matrix (ECM) components and organization, which enables each ECM to carry out tissue-specific roles, including structural support, the transmission of forces and macromolecular filtration. The architecture of the ECM is highly organized, which partly arises from the innate properties of its constituent molecules and their interactions and partly from the activities of the resident cells.
-
The fibrous (collagens and elastin) and glycoprotein (fibronectin, proteoglycans and laminins) macromolecules that constitute the ECM have evolved structures and chemical properties that are particularly suited to their specific biological functions in their respective tissues. Each class of ECM molecule is designed to interact with another class to produce unique physical and signalling properties that support tissue structure, growth and function. Small, modular subunits form homopolymers and heteropolymers that become supramolecular assemblies with highly specialized organization.
-
Collagens are the major proteins of the ECM. The structural hallmark of all collagens is the triple helix, which is a right-handed helix of three polypeptide α-chains (homotrimers and heterotrimers), each of which contains one or more regions that are characterized by the repeating amino acid motif Gly-X-Y, where X and Y can be any amino acid.
-
The assembly of fibrillar collagen involves multiple complex intracellular and extracellular post-translational steps from the translational product to a fibrillar structure that is capable of withstanding tensile forces. The unique mechanical properties of fibrillar collagen are mainly controlled by the collagen structure, which shows the importance of the relationship between three-dimensional protein structure and the resulting ECM function.
-
The primary biological function of proteoglycans derives from the biochemical and hydrodynamic characteristics of the glycosaminoglycan (GAG) components of the molecules, which are long, negatively charged, linear chains of disaccharide repeats that bind water to provide hydration and compressive resistance. Heparan sulphate proteoglycans (HSPGs) are a major part of the basement membrane and chondroitin sulphate proteoglycans (CSPGs) can be found in cartilage and in neural ECMs.
-
The laminin family of large, mosaic glycoproteins are primarily located in basal lamina and some mesenchymal compartments, and they mediate interactions between cells via cell surface receptors (such as integrins and dystroglycan) and other components of the ECM through the modular domains within the laminin molecule. Similarly, many ECM proteins interact with cells through crucial connections with the multidomain protein fibronectin, which is secreted as a large glycoprotein that assembles via cell-mediated processes into fibrillar structures around cells.
-
The production and assembly of the ECM follow different temporal and spatial patterns in various tissues, with load-bearing tissues such as tendons showing highly ordered, fibrillar structures and the continually evolving brain showing a less organized, GAG-rich ECM. Therefore, disruption of the relative abundance of ECM proteins or their interactions with one another has important consequences for the behaviour and the fate of cells within that tissue.
Abstract
The biochemical and biophysical properties of the extracellular matrix (ECM) dictate tissue-specific cell behaviour. The molecules that are associated with the ECM of each tissue, including collagens, proteoglycans, laminins and fibronectin, and the manner in which they are assembled determine the structure and the organization of the resultant ECM. The product is a specific ECM signature that is comprised of unique compositional and topographical features that both reflect and facilitate the functional requirements of the tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mecham, R. P. Overview of extracellular matrix. Curr. Protoc. Cell Biol. 10, Unit 10.1 (2012).
Lu, P., Takai, K., Weaver, V. M. & Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 3, a005058 (2011).
Rozario, T. & DeSimone, D. W. The extracellular matrix in development and morphogenesis: a dynamic view. Dev. Biol. 341, 126–140 (2010).
Naba, A. et al. The matrisome: in silico definition and in vivo characterization by proteomics of normal and tumor extracellular matrices. Mol. Cell. Proteomics 11, M111.014647 (2012).
Jakeman, L. B., Williams, K. E. & Brautigam, B. In the presence of danger: the extracellular matrix defensive response to central nervous system injury. Neural Regen. Res. 9, 377–384 (2014).
Fratzl, P. et al. Fibrillar structure and mechanical properties of collagen. J. Struct. Biol. 122, 119–122 (1998).
Bonnans, C., Chou, J. & Werb, Z. Remodeling the extracellular matrix in development and diseases. Nature Rev. Cell Mol. Biol. http://dx.doi.org/10.1038/nrm3904 (2014).
Humphrey, J. D., Dufresne, E. R. & Schwartz, A. M. Mechanotransduction and extracellular matrix homeostasis. Nature Rev. Cell Mol. Biol. http://dx.doi.org/10.1038/nrm3896 (2014).
Rowe, R. G. & Weiss, S. J. Breaching the basement membrane: who, when and how? Trends Cell Biol. 18, 560–574 (2008).
Schaefer, L. & Schaefer, R. M. Proteoglycans: from structural compounds to signaling molecules. Cell Tissue Res. 339, 237–246 (2010). This paper shows the biological structure and functions of the primarily extracellular SLRPs and highlights SLRP-associated genetic diseases and signalling events.
Myllyharju, J. & Kivirikko, K. I. Collagens, modifying enzymes and their mutations in humans, flies and worms. Trends Genet. 20, 33–43 (2004).
Ricard-Blum, S. & Ruggiero, F. The collagen superfamily: from the extracellular matrix to the cell membrane. Pathol. Biol. (Paris). 53, 430–442 (2005).
Brodsky, B. & Persikov, A. V. Molecular structure of the collagen triple helix. Adv. Protein Chem. 70, 301–339 (2005).
Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 3, a004978 (2011).
Hulmes, D. J. S. in Collagen Struct. Mech. (ed. Fratzl, P.) 15–47 (Springer, 2008). This is a comprehensive review of intracellular and extracellular collagen biosynthesis and collagen fibril assembly.
Hulmes, D. J. S. Building collagen molecules, fibrils, and suprafibrillar structures. J. Struct. Biol. 137, 2–10 (2002).
Myllyharju, J. Intracellular post-translational modifications of collagens. Top Curr. Chem. 247, 115–147 (2005).
Bella, J., Eaton, M., Brodsky, B. & Berman, H. M. Crystal and molecular structure of a collagen-like peptide at 1.9 A resolution. Science 266, 75–81 (1994).
Persikov, A. V., Ramshaw, J. A. M., Kirkpatrick, A. & Brodsky, B. Electrostatic interactions involving lysine make major contributions to collagen triple-helix stability. Biochemistry 44, 1414–1422 (2005).
Bourhis, J.-M. et al. Structural basis of fibrillar collagen trimerization and related genetic disorders. Nature Struct. Mol. Biol. 19, 1031–1036 (2012). This paper elucidates the crystal structure of the procollagen C propeptide to reveal an exquisite structural mechanism of α-chain selectivity during intracellular trimerization.
McLaughlin, S. H. & Bulleid, N. J. Molecular recognition in procollagen chain assembly. Matrix Biol. 16, 369–377 (1998).
Lees, J. F., Tasab, M. & Bulleid, N. J. Identification of the molecular recognition sequence which determines the type-specific assembly of procollagen. EMBO J. 16, 908–916 (1997).
Boudko, S. P., Engel, J. & Bächinger, H. P. The crucial role of trimerization domains in collagen folding. Int. J. Biochem. Cell Biol. 44, 21–32 (2012).
Ellgaard, L. & Ruddock, L. W. The human protein disulphide isomerase family: substrate interactions and functional properties. EMBO Rep. 6, 28–32 (2005).
Wess, T. J. Collagen fibril form and function. Adv. Protein Chem. 70, 341–374 (2005).
Shoulders, M. D. & Raines, R. T. Collagen structure and stability. Annu. Rev. Biochem. 78, 929–958 (2009).
Trelstad, R. L. & Hayashi, K. Tendon collagen fibrillogenesis: intracellular subassemblies and cell surface changes associated with fibril growth. Dev. Biol. 71, 228–242 (1979).
Birk, D. E. & Trelstad, R. L. Extracellular compartments in tendon morphogenesis: collagen fibril, bundle, and macroaggregate formation. J. Cell Biol. 103, 231–240 (1986).
Birk, D. E., Zycband, E. I., Winkelmann, D. A. & Trelstad, R. L. Collagen fibrillogenesis in situ: fibril segments are intermediates in matrix assembly. Proc. Natl Acad. Sci. USA 86, 4549–4553 (1989).
Canty, E. G. et al. Coalignment of plasma membrane channels and protrusions (fibripositors) specifies the parallelism of tendon. J. Cell Biol. 165, 553–563 (2004). This paper shows how embryonic fibroblasts produce an ECM that is rich in elongated, parallel, collagen fibrils, through the initiation of collagen fibrillogenesis by targeting collagen fibril-containing Golgi-to-plasma membrane carriers (GPCs) to plasma membrane protrusions.
Starborg, T. et al. Using transmission electron microscopy and 3View to determine collagen fibril size and three-dimensional organization. Nature Protoc. 8, 1433–1448 (2013).
Kalson, N. S. et al. Nonmuscle myosin II powered transport of newly formed collagen fibrils at the plasma membrane. Proc. Natl Acad. Sci. USA 110, E4743–E4752 (2013).
Porter, S., Clark, I. M., Kevorkian, L. & Edwards, D. R. The ADAMTS metalloproteinases. Biochem. J. 386, 15–27 (2005).
Hopkins, D. R., Keles, S. & Greenspan, D. S. The bone morphogenetic protein 1/tolloid-like metalloproteinases. Matrix Biol. 26, 508–523 (2007).
Seidah, N. G. & Prat, A. Precursor convertases in the secretory pathway, cytosol and extracellular milieu. Essays Biochem. 38, 79–94 (2002).
Pappano, W. N., Steiglitz, B. M., Scott, I. C., Keene, D. R. & Greenspan, D. S. Use of BMP1/TLL1 doubly homozygous null mice and proteomics to identify and validate in vivo substrates of bone morphogenetic protein 1/tolloid-like metalloproteinases. Mol. Cell. Biol. 23, 4428–4438 (2003).
Kadler, K. E., Holmes, D. F., Trotter, J. A. & Chapman, J. A. Collagen fibril formation. Biochem. J. 316, 1–11 (1996).
Wenstrup, R. J. et al. Type V collagen controls the initiation of collagen fibril assembly. J. Biol. Chem. 279, 53331–53337 (2004).
Chapman, J. A. The staining pattern of collagen fibrils. I. An analysis of electron micrographs. Connect. Tissue Res. 2, 137–150 (1974).
Olsen, B. R. Electron microscope studies on collagen. I. Native collagen fibrils. Z. Zellforsch. Mikrosk. Anat. 59, 184–198 (1963).
Petruska, J. A. & Hodge, A. J. A subunit model for the tropocollagen macromolecule. Proc. Natl Acad. Sci. USA 51, 871–876 (1964).
Hulmes, D. J. & Miller, A. Quasi-hexagonal molecular packing in collagen fibrils. Nature 282, 878–880 (1979).
Hulmes, D. J., Wess, T. J., Prockop, D. J. & Fratzl, P. Radial packing, order, and disorder in collagen fibrils. Biophys. J. 68, 1661–1670 (1995).
Orgel, J. P. R. O., Irving, T. C., Miller, A. & Wess, T. J. Microfibrillar structure of type I collagen in situ. Proc. Natl Acad. Sci. USA 103, 9001–9005 (2006).
Schmitt, F. O., Hall, C. E. & Jakus, M. A. Electron microscope investigations of the structure of collagen. J. Cell. Comp. Physiol. 20, 11–33 (1942).
Bruckner, P. Suprastructures of extracellular matrices: paradigms of functions controlled by aggregates rather than molecules. Cell Tissue Res. 339, 7–18 (2010).
Ameye, L. & Young, M. F. Mice deficient in small leucine-rich proteoglycans: novel in vivo models for osteoporosis, osteoarthritis, Ehlers–Danlos syndrome, muscular dystrophy, and corneal diseases. Glycobiology 12, 107R–116R (2002).
Kalamajski, S. & Oldberg, A. The role of small leucine-rich proteoglycans in collagen fibrillogenesis. Matrix Biol. 29, 248–253 (2010).
Molnar, J. et al. Structural and functional diversity of lysyl oxidase and the LOX-like proteins. Biochim. Biophys. Acta 1647, 220–224 (2003).
Eyre, D. R., Weis, M. A. & Wu, J.-J. Advances in collagen cross-link analysis. Methods 45, 65–74 (2008).
Eyre, D. R. & Glimcher, M. J. Collagen cross-linking. Isolation of cross-linked peptides from collagen of chicken bone. Biochem. J. 135, 393–403 (1973).
Lucero, H. A. & Kagan, H. M. Lysyl oxidase: an oxidative enzyme and effector of cell function. Cell. Mol. Life Sci. 63, 2304–2316 (2006).
Rucker, R. B. & Murray, J. Cross-linking amino acids in collagen and elastin. Am. J. Clin. Nutr. 31, 1221–1236 (1978).
Orgel, J. P., Wess, T. J. & Miller, A. The in situ conformation and axial location of the intermolecular cross-linked non-helical telopeptides of type I collagen. Structure 8, 137–142 (2000).
O'Leary, L. E. R., Fallas, J. A., Bakota, E. L., Kang, M. K. & Hartgerink, J. D. Multi-hierarchical self-assembly of a collagen mimetic peptide from triple helix to nanofibre and hydrogel. Nature Chem. 3, 821–828 (2011).
Bandtlow, C. E. & Zimmermann, D. R. Proteoglycans in the developing brain: new conceptual insights for old proteins. Physiol. Rev. 80, 1267–1290 (2000).
Knudson, C. B. & Knudson, W. Cartilage proteoglycans. Semin. Cell Dev. Biol. 12, 69–78 (2001).
Cui, H., Freeman, C., Jacobson, G. A. & Small, D. H. Proteoglycans in the central nervous system: role in development, neural repair, and Alzheimer's disease. IUBMB Life 65, 108–120 (2013).
Schwartz, N. B. & Domowicz, M. Proteoglycans in brain development. Glycoconj. J. 21, 329–341 (2004).
Zhang, L. (ed.) Glycosaminoglycans in Development, Health and Disease (Academic, 2010).
Deepa, S. S. et al. Composition of perineuronal net extracellular matrix in rat brain: a different disaccharide composition for the net-associated proteoglycans. J. Biol. Chem. 281, 17789–17800 (2006).
Simon Davis, D. A. & Parish, C. R. Heparan sulfate: a ubiquitous glycosaminoglycan with multiple roles in immunity. Front. Immunol. 4, 470 (2013).
Bishop, J. R., Schuksz, M. & Esko, J. D. Heparan sulphate proteoglycans fine-tune mammalian physiology. Nature 446, 1030–1037 (2007).
Esko, J. D., Kimata, K. & Lindahl, U. in Essentials of Glycobiology. 2nd edn Ch. 16 (Cold Spring Harbor, 2009).
Wade, A., McKinney, A. & Phillips, J. J. Matrix regulators in neural stem cell functions. Biochim. Biophys. Acta http://dx.doi.org/10.1016/j.bbagen.2014.01.017 (2014).
Johnson, K. G. et al. Axonal heparan sulfate proteoglycans regulate the distribution and efficiency of the repellent slit during midline axon guidance. Curr. Biol. 14, 499–504 (2004).
Häcker, U., Nybakken, K. & Perrimon, N. Heparan sulphate proteoglycans: the sweet side of development. Nature Rev. Mol. Cell. Biol. 6, 530–541 (2005).
Maeda, N., Ishii, M., Nishimura, K. & Kamimura, K. Functions of chondroitin sulfate and heparan sulfate in the developing brain. Neurochem. Res. 36, 1228–1240 (2011).
Yamaguchi, Y. Lecticans: organizers of the brain extracellular matrix. Cell. Mol. Life Sci. 57, 276–289 (2000).
Lundell, A. et al. Structural basis for interactions between tenascins and lectican C-type lectin domains: evidence for a crosslinking role for tenascins. Structure 12, 1495–1506 (2004).
Dellett, M., Hu, W., Papadaki, V. & Ohnuma, S. Small leucine rich proteoglycan family regulates multiple signalling pathways in neural development and maintenance. Dev. Growth Differ. 54, 327–340 (2012).
McEwan, P. A., Scott, P. G., Bishop, P. N. & Bella, J. Structural correlations in the family of small leucine-rich repeat proteins and proteoglycans. J. Struct. Biol. 155, 294–305 (2006).
Schaefer, L. & Iozzo, R. V. Biological functions of the small leucine-rich proteoglycans: from genetics to signal transduction. J. Biol. Chem. 283, 21305–21309 (2008).
Chen, S. & Birk, D. E. The regulatory roles of small leucine-rich proteoglycans in extracellular matrix assembly. FEBS J. 280, 2120–2137 (2013).
Oohira, A., Matsui, F., Tokita, Y., Yamauchi, S. & Aono, S. Molecular interactions of neural chondroitin sulfate proteoglycans in the brain development. Arch. Biochem. Biophys. 374, 24–34 (2000).
Timpl, R. & Brown, J. C. The laminins. Matrix Biol. 14, 275–281 (1994).
Aumailley, M. et al. A simplified laminin nomenclature. Matrix Biol. 24, 326–332 (2005).
Miner, J. H. & Yurchenco, P. D. Laminin functions in tissue morphogenesis. Annu. Rev. Cell Dev. Biol. 20, 255–284 (2004). This paper discusses how different laminin proteins affect invertebrate and vertebrate tissue morphogenesis through the induction and the maintenance of cell polarity, the establishment of barriers between tissue compartments and the protection of adherent cells from anoikis.
Mercurio, A. M. & Shaw, L. M. Laminin binding proteins. Bioessays 13, 469–473 (1991).
Yurchenco, P. D., Smirnov, S. & Mathus, T. Analysis of basement membrane self-assembly and cellular interactions with native and recombinant glycoproteins. Methods Cell Biol. 69, 111–144 (2002).
Schwarzbauer, J. E. & DeSimone, D. W. Fibronectins, their fibrillogenesis, and in vivo functions. Cold Spring Harb. Perspect. Biol. 3, a005041 (2011).
Singh, P., Carraher, C. & Schwarzbauer, J. E. Assembly of fibronectin extracellular matrix. Annu. Rev. Cell Dev. Biol. 26, 397–419 (2010). This is an excellent review highlighting the major steps, molecular interactions and cellular mechanisms involved in assembling fibronectin into a fibrillar matrix.
Hynes, R. O. in Guidebook to the Extracellular Matrix, Anchor and Adhesion Proteins. 2nd edn (eds Kreis, T. & Vale, R.) 422–425 (Oxford University Press, 1999).
García, A. J., Schwarzbauer, J. E. & Boettiger, D. Distinct activation states of α5β1 integrin show differential binding to RGD and synergy domains of fibronectin. Biochemistry 41, 9063–9069 (2002).
Ilic´, D. et al. FAK promotes organization of fibronectin matrix and fibrillar adhesions. J. Cell Sci. 117, 177–187 (2004).
Hay, E. D. Extracellular matrix alters epithelial differentiation. Curr. Opin. Cell Biol. 5, 1029–1035 (1993).
Birk, D. E., Zycband, E. I., Woodruff, S., Winkelmann, D. A. & Trelstad, R. L. Collagen fibrillogenesis in situ: fibril segments become long fibrils as the developing tendon matures. Dev. Dyn. 208, 291–298 (1997).
Zhang, G. et al. Development of tendon structure and function: regulation of collagen fibrillogenesis. J. Musculoskelet. Neuronal Interact. 5, 5–21 (2005).
Wang, J. H.-C. Mechanobiology of tendon. J. Biomech. 39, 1563–1582 (2006).
Kannus, P. Structure of the tendon connective tissue. Scand. J. Med. Sci. Sports 10, 312–320 (2000).
Tresoldi, I. et al. Tendon's ultrastructure. Muscles. Ligaments Tendons J. 3, 2–6 (2013).
Mienaltowski, M. J. & Birk, D. E. Structure, physiology, and biochemistry of collagens. Adv. Exp. Med. Biol. 802, 5–29 (2014).
Craig, A. S., Birtles, M. J., Conway, J. F. & Parry, D. A. An estimate of the mean length of collagen fibrils in rat tail-tendon as a function of age. Connect. Tissue Res. 19, 51–62 (1989).
Tozer, S. & Duprez, D. Tendon and ligament: development, repair and disease. Birth Defects Res. C. Embryo Today 75, 226–236 (2005).
Richardson, S. H. et al. Tendon development requires regulation of cell condensation and cell shape via cadherin-11-mediated cell-cell junctions. Mol. Cell. Biol. 27, 6218–6228 (2007). This paper describes how the precise regulation of cell shape via cell–cell junctions and the coaxial alignment of plasma membrane channels in longitudinally stacked cells drives tendon formation in the developing limb.
Sasaki, N. & Odajima, S. Elongation mechanism of collagen fibrils and force-strain relations of tendon at each level of structural hierarchy. J. Biomech. 29, 1131–1136 (1996).
Graham, H. K., Holmes, D. F., Watson, R. B. & Kadler, K. E. Identification of collagen fibril fusion during vertebrate tendon morphogenesis. The process relies on unipolar fibrils and is regulated by collagen-proteoglycan interaction. J. Mol. Biol. 295, 891–902 (2000).
Paulsson, M. Basement membrane proteins: structure, assembly, and cellular interactions. Crit. Rev. Biochem. Mol. Biol. 27, 93–127 (1992).
Martin Rupert, G. R. T. Laminin and other basement membrane components. Annu. Rev. Cell Biol. 3, 57–85 (1987).
Kalluri, R. Basement membranes: structure, assembly and role in tumour angiogenesis. Nature Rev. Cancer 3, 422–433 (2003). This is an excellent review highlighting the constituents, structure and assembly of the basement membrane, with emphasis on angiogenesis and vascular basement membranes.
Yurchenco, P. D. Basement membranes: cell scaffoldings and signaling platforms. Cold Spring Harb. Perspect. Biol. 3, a004911 (2011).
Yurchenco, P. D. & Schittny, J. C. Molecular architecture of basement membranes. FASEB J. 4, 1577–1590 (1990).
Sanes, J. R. The basement membrane/basal lamina of skeletal muscle. J. Biol. Chem. 278, 12601–12604 (2003).
Yurchenco, P. D. & Patton, B. L. Developmental and pathogenic mechanisms of basement membrane assembly. Curr. Pharm. Des. 15, 1277–1294 (2009).
Takagi, J., Yang, Y., Liu, J.-H., Wang, J.-H. & Springer, T. A. Complex between nidogen and laminin fragments reveals a paradigmatic β-propeller interface. Nature 424, 969–974 (2003).
Pöschl, E. et al. Collagen IV is essential for basement membrane stability but dispensable for initiation of its assembly during early development. Development 131, 1619–1628 (2004).
Iozzo, R. V., Zoeller, J. J. & Nyström, A. Basement membrane proteoglycans: modulators Par Excellence of cancer growth and angiogenesis. Mol. Cells 27, 503–513 (2009).
Costello, I., Biondi, C. A., Taylor, J. M., Bikoff, E. K. & Robertson, E. J. Smad4-dependent pathways control basement membrane deposition and endodermal cell migration at early stages of mouse development. BMC Dev. Biol. 9, 54 (2009).
Wiradjaja, F., DiTommaso, T. & Smyth, I. Basement membranes in development and disease. Birth Defects Res. C. Embryo Today 90, 8–31 (2010).
Paralkar, V. M., Vukicevic, S. & Reddi, A. H. Transforming growth factor-β type 1 binds to collagen IV of basement membrane matrix: Implications for development. Dev. Biol. 143, 303–308 (1991).
Streuli, C. H. Control of mammary epithelial differentiation: basement membrane induces tissue-specific gene expression in the absence of cell–cell interaction and morphological polarity. J. Cell Biol. 115, 1383–1395 (1991).
Camelo, A., Dunmore, R., Sleeman, M. A. & Clarke, D. L. The epithelium in idiopathic pulmonary fibrosis: breaking the barrier. Front. Pharmacol. 4, 173 (2014).
Torricelli, A. A. M., Singh, V., Santhiago, M. R. & Wilson, S. E. The corneal epithelial basement membrane: structure, function, and disease. Invest. Ophthalmol. Vis. Sci. 54, 6390–6400 (2013).
Brownell, A. G. & Slavkin, H. C. Role of basal lamina in tissue interactions. Ren. Physiol. 3, 193–204 (1980).
Lau, L. W., Cua, R., Keough, M. B., Haylock-Jacobs, S. & Yong, V. W. Pathophysiology of the brain extracellular matrix: a new target for remyelination. Nature Rev. Neurosci. 14, 722–729 (2013). This paper shows the organization of the brain ECM, emphasizing the pathophysiological roles of CSPGs after neural injury.
Plantman, S. Proregenerative properties of ECM molecules. Biomed. Res. Int. 2013, 981695 (2013).
Ruoslahti, E. Brain extracellular matrix. Glycobiology 6, 489–492 (1996).
Barros, C. S., Franco, S. J. & Müller, U. Extracellular matrix: functions in the nervous system. Cold Spring Harb. Perspect. Biol. 3, a005108 (2011).
Chiquet-Ehrismann, R. & Tucker, R. P. Tenascins and the importance of adhesion modulation. Cold Spring Harb. Perspect. Biol. 3, a004960 (2011).
Dityatev, A. Remodeling of extracellular matrix and epileptogenesis. Epilepsia 51 (Suppl. 3), 61–65 (2010).
Lajtha, A. & Banik, N. Handbook of Neurochemistry and Molecular Neurobiology: Neural Protein Metabolism and Function. Vol. 3 (Springer, 2007).
Howell, M. D. & Gottschall, P. E. Lectican proteoglycans, their cleaving metalloproteinases, and plasticity in the central nervous system extracellular microenvironment. Neuroscience 217, 6–18 (2012).
Carulli, D., Rhodes, K. E. & Fawcett, J. W. Upregulation of aggrecan, link protein 1, and hyaluronan synthases during formation of perineuronal nets in the rat cerebellum. J. Comp. Neurol. 501, 83–94 (2007).
Frischknecht, R. & Gundelfinger, E. D. The brain's extracellular matrix and its role in synaptic plasticity. Adv. Exp. Med. Biol. 970, 153–171 (2012).
Kwok, J. C. F., Dick, G., Wang, D. & Fawcett, J. W. Extracellular matrix and perineuronal nets in CNS repair. Dev. Neurobiol. 71, 1073–1089 (2011).
Wang, D. & Fawcett, J. The perineuronal net and the control of CNS plasticity. Cell Tissue Res. 349, 147–160 (2012).
Romberg, C. et al. Depletion of perineuronal nets enhances recognition memory and long-term depression in the perirhinal cortex. J. Neurosci. 33, 7057–7065 (2013).
Grimpe, B. & Silver, J. The extracellular matrix in axon regeneration. Prog. Brain Res. 137, 333–349 (2002).
Fitch, M. T. & Silver, J. Glial cell extracellular matrix: boundaries for axon growth in development and regeneration. Cell Tissue Res. 290, 379–384 (1997).
Ishii, M. & Maeda, N. Oversulfated chondroitin sulfate plays critical roles in the neuronal migration in the cerebral cortex. J. Biol. Chem. 283, 32610–32620 (2008).
Sharma, K., Selzer, M. E. & Li, S. Scar-mediated inhibition and CSPG receptors in the CNS. Exp. Neurol. 237, 370–378 (2012).
Bartus, K., James, N. D., Bosch, K. D. & Bradbury, E. J. Chondroitin sulphate proteoglycans: key modulators of spinal cord and brain plasticity. Exp. Neurol. 235, 5–17 (2012).
Li, A.-H., Liu, P. P., Villarreal, F. J. & Garcia, R. A. Dynamic changes in myocardial matrix and relevance to disease: translational perspectives. Circ. Res. 114, 916–927 (2014).
Page-McCaw, A., Ewald, A. J. & Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nature Rev. Mol. Cell Biol. 8, 221–233 (2007).
Fan, D., Takawale, A., Lee, J. & Kassiri, Z. Cardiac fibroblasts, fibrosis and extracellular matrix remodeling in heart disease. Fibrogen. Tissue Repair 5, 15 (2012).
Nickla, H., Klug, W. S. & Cummings, M. R. Concepts of Genetics. (Prentice Hall, 1997).
Jäälinoja, J., Ylöstalo, J., Beckett, W., Hulmes, D. J. S. & Ala-Kokko, L. Trimerization of collagen IX α-chains does not require the presence of the COL1 and NC1 domains. Biochem. J. 409, 545–554 (2008).
Liu, Y., Ramanath, H. S. & Wang, D.-A. Tendon tissue engineering using scaffold enhancing strategies. Trends Biotechnol. 26, 201–209 (2008).
Edwards, I. J. Proteoglycans in prostate cancer. Nature Rev. Urol. 9, 196–206 (2012).
Rudd, T. R. et al. A key biological signalling system promises new routes to intervention in medically important processes. Diamond Science [online], (2014).
Marinkovich, M. P. Tumour microenvironment: laminin 332 in squamous-cell carcinoma. Nature Rev. Cancer 7, 370–380 (2007).
Bezakova, G. & Ruegg, M. A. New insights into the roles of agrin. Nature Rev. Mol. Cell. Biol. 4, 295–308 (2003).
Meier, T. & Wallace, B. G. Formation of the neuromuscular junction: molecules and mechanisms. Bioessays 20, 819–829 (1998).
McMahan, U. J. et al. Agrin isoforms and their role in synaptogenesis. Curr. Opin. Cell Biol. 4, 869–874 (1992).
Fisher, L. W., Torchia, D. A., Fohr, B., Young, M. F. & Fedarko, N. S. Flexible structures of SIBLING proteins, bone sialoprotein, and osteopontin. Biochem. Biophys. Res. Commun. 280, 460–465 (2001).
Bellahcène, A., Castronovo, V., Ogbureke, K. U. E., Fisher, L. W. & Fedarko, N. S. Small integrin-binding ligand N-linked glycoproteins (SIBLINGs): multifunctional proteins in cancer. Nature Rev. Cancer 8, 212–226 (2008).
Ogata, Y. Bone sialoprotein and its transcriptional regulatory mechanism. J. Periodontal Res. 43, 127–135 (2008).
Deák, F., Wagener, R., Kiss, I. & Paulsson, M. The matrilins: a novel family of oligomeric extracellular matrix proteins. Matrix Biol. 18, 55–64 (1999).
Klatt, A. R., Becker, A.-K. A., Neacsu, C. D., Paulsson, M. & Wagener, R. The matrilins: modulators of extracellular matrix assembly. Int. J. Biochem. Cell Biol. 43, 320–330 (2011).
Wagener, R. et al. The matrilins — adaptor proteins in the extracellular matrix. FEBS Lett. 579, 3323–3329 (2005).
Chen, Q., Johnson, D. M., Haudenschild, D. R. & Goetinck, P. F. Cartilage matrix protein: expression patterns in chicken, mouse, and human. Ann. NY Acad. Sci. 785, 238–240 (1996).
Mithieux, S. M. & Weiss, A. S. Elastin. Adv. Protein Chem. 70, 437–461 (2005).
Halper, J. & Kjaer, M. Basic components of connective tissues and extracellular matrix: elastin, fibrillin, fibulins, fibrinogen, fibronectin, laminin, tenascins and thrombospondins. Adv. Exp. Med. Biol. 802, 31–47 (2014).
Pepe, A., Bochicchio, B. & Tamburro, A. M. Supramolecular organization of elastin and elastin-related nanostructured biopolymers. Nanomed. 2, 203–218 (2007).
Wagenseil, J. E. & Mecham, R. P. New insights into elastic fiber assembly. Birth Defects Res. C. Embryo Today 81, 229–240 (2007).
Weisel, J. W. Fibrinogen and fibrin. Adv. Protein Chem. 70, 247–299 (2005).
Mosesson, M. W., Siebenlist, K. R. & Meh, D. A. The structure and biological features of fibrinogen and fibrin. Ann. NY Acad. Sci. 936, 11–30 (2001).
Ramirez, F., Sakai, L. Y., Dietz, H. C. & Rifkin, D. B. Fibrillin microfibrils: multipurpose extracellular networks in organismal physiology. Physiol. Genom. 19, 151–154 (2004).
Kielty, C. M. et al. Fibrillin: from microfibril assembly to biomechanical function. Phil. Trans. R. Soc. 357, 207–217 (2002).
Timpl, R., Sasaki, T., Kostka, G. & Chu, M.-L. Fibulins: a versatile family of extracellular matrix proteins. Nature Rev. Mol. Cell. Biol. 4, 479–489 (2003).
De Vega, S., Iwamoto, T. & Yamada, Y. Fibulins: multiple roles in matrix structures and tissue functions. Cell. Mol. Life Sci. 66, 1890–1902 (2009).
Cirulli, V. & Yebra, M. Netrins: beyond the brain. Nature Rev. Mol. Cell Biol. 8, 296–306 (2007).
Lai Wing Sun, K., Correia, J. P. & Kennedy, T. E. Netrins: versatile extracellular cues with diverse functions. Development 138, 2153–2169 (2011).
Bradshaw, A. D. Diverse biological functions of the SPARC family of proteins. Int. J. Biochem. Cell Biol. 44, 480–488 (2012).
Brekken, R. A. & Sage, E. H. SPARC, a matricellular protein: at the crossroads of cell-matrix communication. Matrix Biol. 19, 816–827 (2001).
Giachelli, C. M. & Steitz, S. Osteopontin: a versatile regulator of inflammation and biomineralization. Matrix Biol. 19, 615–622 (2000).
Hunter, G. K. Role of osteopontin in modulation of hydroxyapatite formation. Calcif. Tissue Int. 93, 348–354 (2013).
Fatemi, S. H. Reelin glycoprotein: structure, biology and roles in health and disease. Mol. Psychiatry 10, 251–257 (2005).
Magdaleno, S. M. & Curran, T. Brain development: integrins and the Reelin pathway. Curr. Biol. 11, R1032–R1035 (2001).
Vincent, T. L., Woolfson, D. N. & Adams, J. C. Prediction and analysis of higher-order coiled-coils: insights from proteins of the extracellular matrix, tenascins and thrombospondins. Int. J. Biochem. Cell Biol. 45, 2392–2401 (2013).
Tucker, R. P. & Chiquet-Ehrismann, R. The regulation of tenascin expression by tissue microenvironments. Biochim. Biophys. Acta 1793, 888–892 (2009).
Leavesley, D. I. et al. Vitronectin — master controller or micromanager? IUBMB Life 65, 807–818 (2013).
Preissner, K. T. & Reuning, U. Vitronectin in vascular context: facets of a multitalented matricellular protein. Semin. Thromb. Hemost. 37, 408–424 (2011).
Acknowledgements
The authors apologize to all colleagues whose work cannot be cited owing to space limitations. This work was supported by DOD Breast Cancer Research Program (BCRP) grant W81XWH-07-1-0538 (to J.K.M.), US National Science Foundation (NSF) Graduate Research Fellowship (to G.O.), DOD BCRP grants W81XWH-05-1-0330 and W81XWH-13-1-0216 (to V.M.W.), US National Institutes of Health National Cancer Institute (NCI) grants R01 CA138818, U54 CA143836, R01 CA085492 and U01 ES019458 (to V.M.W.), and Susan G. Komen grant KG110560PP (to V.M.W.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Topographies
-
The three-dimensional qualities of surfaces or structures, including contours and relief. In the context of the ECM this includes features such as peaks and valleys, changes in roughness and geometric features.
- Morphogenesis
-
The process of cell movement during embryonic development that controls the size, the shape and the patterning of tissues and organs.
- Proteoglycans
-
(PGs). Glycoproteins that consist of a core protein to which one or more glycosaminoglycan chain is attached.
- Glycosaminoglycans
-
(GAGs). Long, linear, charged polysaccharides that comprise repeating pairs of sugars, of which one is an amino sugar.
- Fibrillar collagen
-
Polymerized, supramolecular collagen that has been organized into fibrils; collagen types I, II and III form fibrils.
- Tensile forces
-
The forces required to exert a certain amount of tension (which is a type of normal stress) on a one-dimensional object. In this context, a pulling force on a cable tending to cause extension of the cable.
- Procollagen
-
A trimeric collagen precursor molecule containing large amino- and carboxy-terminal propeptide domains.
- Lysyl oxidase
-
An enzyme that participates in the formation of collagen polymers by facilitating oxidative deamination of peptidyl lysine residues on collagen monomers.
- Pericellular space
-
The space surrounding a cell.
- Fibripositors
-
(Also known as fibropositors). Plasma membrane protrusions projecting from the cell surface, where procollagen can potentially be processed, assembled and organized by individual cells.
- Fibrillogenesis
-
The development or formation of fibrils, used to refer to collagen- or fibronectin-rich structures.
- Small leucine-rich repeat proteoglycans
-
(SLRPs). A family of proteoglycans that share a common leucine-rich-repeat motif in their conserved carboxy termini; they have been strongly implicated in modulating fibrillar collagen assembly.
- FACIT
-
(Fibril-associated collagens with interrupted triple helices). A type of collagen that does not form fibrils by itself but that is associated with the surface of fibrillar collagen.
- Basement membrane
-
A thin, complex extracellular matrix that separates endothelial and epithelial cells from their subjacent connective tissues. It is composed of various collagens, proteoglycans and adhesive glycoproteins.
- Syndecans
-
One of the two major families of heparin sulphate proteoglycans.
- Glypicans
-
One of the two major families of heparin sulphate proteoglycans.
- Epimerization
-
The formation of stereoisomers.
- Integrins
-
A large family of heterodimeric transmembrane proteins, which are present in the plasma membrane as heterodimers of α- and β-subunits and function as receptors for cell adhesion molecules.
- Nidogens
-
(Also known as entactins). Glycoproteins and key parts of the basement membrane.
- Fibrillar adhesions
-
Cell–matrix connections that are typified by their location near the centre of the cell and their higher length to width ratio compared with focal adhesions at the periphery of cells.
- Fascicles
-
Bundles of fibres.
- Reticular lamina
-
A collagen-rich layer that is often found below the basal lamina that connects the combined basement membrane to the connective tissue.
Rights and permissions
About this article
Cite this article
Mouw, J., Ou, G. & Weaver, V. Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol 15, 771–785 (2014). https://doi.org/10.1038/nrm3902
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3902
This article is cited by
-
Collagen in the central nervous system: contributions to neurodegeneration and promise as a therapeutic target
Molecular Neurodegeneration (2024)
-
Multiomics analysis of cultured mouse periodontal ligament cell-derived extracellular matrix
Scientific Reports (2024)
-
Therapeutic potential of melatonin in targeting molecular pathways of organ fibrosis
Pharmacological Reports (2024)
-
The value of a dual-energy CT Iodine map radiomics model for the prediction of collagen fiber content in the ccRCC tumor microenvironment
BMC Medical Imaging (2023)
-
Critical contributions of protein cargos to the functions of macrophage-derived extracellular vesicles
Journal of Nanobiotechnology (2023)