Key Points
-
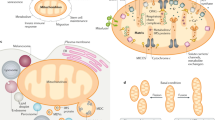
Mitochondria are organelles with key roles in cellular metabolism. They have unique cellular dynamics to ensure their proper distribution to dividing cells and high fidelity of inheritance of their genome in a maternal mode of transmission.
-
In mammals, mitochondrial segregation during cell division seems to be primarily a passive process. Mitochondrial fusion, fission, transport, contacts with the endoplasmic reticulum and mitophagy all play a part in maintaining a homogeneous population that is spatially well distributed in the cell soma and that can thus be partitioned equally to daughter cells.
-
Mitochondrial DNA (mtDNA) inheritance from one generation to another is strongly influenced by mtDNA bottlenecks and genetic selection that occur during oogenesis and early embryonic development. Quality-control mechanisms are probably present to minimize the accumulation of pathogenic mutations, which lead to a class of diseases termed mitochondrial encephalomyopathies.
-
The depletion of paternal mitochondria during fertilization is nearly universal in metozoans, although its timing and mechanisms vary substantially between species. Proteasome-dependent degradation, mtDNA degradation and mitophagy have been implicated in this process in different organisms.
Abstract
During cell division, it is critical to properly partition functional sets of organelles to each daughter cell. The partitioning of mitochondria shares some common features with that of other organelles, particularly in the use of interactions with cytoskeletal elements to facilitate delivery to the daughter cells. However, mitochondria have unique features — including their own genome and a maternal mode of germline transmission — that place additional demands on this process. Consequently, mechanisms have evolved to regulate mitochondrial segregation during cell division, oogenesis, fertilization and tissue development, as well as to ensure the integrity of these organelles and their DNA, including fusion–fission dynamics, organelle transport, mitophagy and genetic selection of functional genomes. Defects in these processes can lead to cell and tissue pathologies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Scheffler, I. E. Mitochondria (John Wiley & Sons, 2009).
Stehling, O., Wilbrecht, C. & Lill, R. Mitochondrial iron-sulfur protein biogenesis and human disease. Biochimie 100, 61–77 (2014).
Tait, S. W. & Green, D. R. Mitochondria and cell death: outer membrane permeabilization and beyond. Nature Rev. Mol. Cell Biol. 11, 621–632 (2010).
Cloonan, S. M. & Choi, A. M. Mitochondria: sensors and mediators of innate immune receptor signaling. Curr. Opin. Microbiol. 16, 327–338 (2013).
Gray, M. W., Burger, G. & Lang, B. F. Mitochondrial evolution. Science 283, 1476–1481 (1999).
Chan, D. C. Fusion and fission: interlinked processes critical for mitochondrial health. Annu. Rev. Genet. 46, 265–287 (2012).
Ball, E. H. & Singer, S. J. Mitochondria are associated with microtubules and not with intermediate filaments in cultured fibroblasts. Proc. Natl Acad. Sci. USA 79, 123–126 (1982).
Heggeness, M. H., Simon, M. & Singer, S. J. Association of mitochondria with microtubules in cultured cells. Proc. Natl Acad. Sci. USA 75, 3863–3866 (1978).
Sheng, Z. H. & Cai, Q. Mitochondrial transport in neurons: impact on synaptic homeostasis and neurodegeneration. Nature Rev. Neurosci. 13, 77–93 (2012).
Tanaka, Y. et al. Targeted disruption of mouse conventional kinesin heavy chain, kif5B, results in abnormal perinuclear clustering of mitochondria. Cell 93, 1147–1158 (1998).
Tanaka, K., Sugiura, Y., Ichishita, R., Mihara, K. & Oka, T. KLP6: a newly identified kinesin that regulates the morphology and transport of mitochondria in neuronal cells. J. Cell Sci. 124, 2457–2465 (2011).
Hirokawa, N. & Noda, Y. Intracellular transport and kinesin superfamily proteins, KIFs: structure, function, and dynamics. Physiol. Rev. 88, 1089–1118 (2008).
Nangaku, M. et al. KIF1B, a novel microtubule plus end-directed monomeric motor protein for transport of mitochondria. Cell 79, 1209–1220 (1994). Shows that the kinesin family member KIF1B mediates the transport of mitochondria in the cytoplasm in vitro and in vivo.
Saxton, W. M. & Hollenbeck, P. J. The axonal transport of mitochondria. J. Cell Sci. 125, 2095–2104 (2012).
Stowers, R. S., Megeath, L. J., Gorska-Andrzejak, J., Meinertzhagen, I. A. & Schwarz, T. L. Axonal transport of mitochondria to synapses depends on milton, a novel Drosophila protein. Neuron 36, 1063–1077 (2002).
Guo, X. et al. The GTPase dMiro is required for axonal transport of mitochondria to Drosophila synapses. Neuron 47, 379–393 (2005).
Wang, X. & Schwarz, T. L. The mechanism of Ca2+-dependent regulation of kinesin-mediated mitochondrial motility. Cell 136, 163–174 (2009). Shows that the Milton–kinesin complex mediates calcium-dependent motility of mitochondria in neurons.
Kang, J. S. et al. Docking of axonal mitochondria by syntaphilin controls their mobility and affects short-term facilitation. Cell 132, 137–148 (2008).
Chen, Y. & Sheng, Z. H. Kinesin-1–syntaphilin coupling mediates activity-dependent regulation of axonal mitochondrial transport. J. Cell Biol. 202, 351–364 (2013).
Glater, E. E., Megeath, L. J., Stowers, R. S. & Schwarz, T. L. Axonal transport of mitochondria requires milton to recruit kinesin heavy chain and is light chain independent. J. Cell Biol. 173, 545–557 (2006).
van Spronsen, M. et al. TRAK/Milton motor-adaptor proteins steer mitochondrial trafficking to axons and dendrites. Neuron 77, 485–502 (2013).
Youle, R. J. & van der Bliek, A. M. Mitochondrial fission, fusion, and stress. Science 337, 1062–1065 (2012).
Chen, H. et al. Mitochondrial fusion is required for mtDNA stability in skeletal muscle and tolerance of mtDNA mutations. Cell 141, 280–289 (2010). Shows that mitochondrial fusion is important for the tolerance of mtDNA mutations.
Chen, H. et al. Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J. Cell Biol. 160, 189–200 (2003).
Ishihara, N. et al. Mitochondrial fission factor Drp1 is essential for embryonic development and synapse formation in mice. Nature Cell Biol. 11, 958–966 (2009).
Wakabayashi, J. et al. The dynamin-related GTPase Drp1 is required for embryonic and brain development in mice. J. Cell Biol. 186, 805–816 (2009).
Chen, H., McCaffery, J. M. & Chan, D. C. Mitochondrial fusion protects against neurodegeneration in the cerebellum. Cell 130, 548–562 (2007).
Chen, H., Chomyn, A. & Chan, D. C. Disruption of fusion results in mitochondrial heterogeneity and dysfunction. J. Biol. Chem. 280, 26185–26192 (2005). Shows that the deletion of the MFN1 and MFN2 genes leads to heterogeneity in mitochondrial membrane potential and defects in oxidative phosphorylation.
Weaver, D. et al. Distribution and apoptotic function of outer membrane proteins depend on mitochondrial fusion. Mol. Cell 54, 870–878 (2014).
Twig, G. et al. Fission and selective fusion govern mitochondrial segregation and elimination by autophagy. EMBO J. 27, 433–446 (2008).
Ishihara, N., Fujita, Y., Oka, T. & Mihara, K. Regulation of mitochondrial morphology through proteolytic cleavage of OPA1. EMBO J. 25, 2966–2977 (2006).
Frank, S. et al. The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Dev. Cell 1, 515–525 (2001).
Gomes, L. C., Di Benedetto, G. & Scorrano, L. During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nature Cell Biol. 13, 589–598 (2011).
Rambold, A. S., Kostelecky, B., Elia, N. & Lippincott-Schwartz, J. Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc. Natl Acad. Sci. USA 108, 10190–10195 (2011).
Mitra, K., Wunder, C., Roysam, B., Lin, G. & Lippincott-Schwartz, J. A hyperfused mitochondrial state achieved at G1–S regulates cyclin E buildup and entry into S phase. Proc. Natl Acad. Sci. USA 106, 11960–11965 (2009).
Taguchi, N., Ishihara, N., Jofuku, A., Oka, T. & Mihara, K. Mitotic phosphorylation of dynamin-related GTPase Drp1 participates in mitochondrial fission. J. Biol. Chem. 282, 11521–11529 (2007).
Kim, I., Rodriguez-Enriquez, S. & Lemasters, J. J. Selective degradation of mitochondria by mitophagy. Arch. Biochem. Biophys. 462, 245–253 (2007).
Rodriguez-Enriquez, S., He, L. & Lemasters, J. J. Role of mitochondrial permeability transition pores in mitochondrial autophagy. Int. J. Biochem. Cell Biol. 36, 2463–2472 (2004).
Kanki, T. & Klionsky, D. J. Mitophagy in yeast occurs through a selective mechanism. J. Biol. Chem. 283, 32386–32393 (2008).
Schon, E. A., DiMauro, S. & Hirano, M. Human mitochondrial DNA: roles of inherited and somatic mutations. Nature Rev. Genet. 13, 878–890 (2012).
Houlden, H. & Singleton, A. B. The genetics and neuropathology of Parkinson's disease. Acta Neuropathol. 124, 325–338 (2012).
Clark, I. E. et al. Drosophila Pink1 is required for mitochondrial function and interacts genetically with parkin. Nature 441, 1162–1166 (2006).
Park, J. et al. Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by Parkin. Nature 441, 1157–1161 (2006).
Yang, Y. et al. Mitochondrial pathology and muscle and dopaminergic neuron degeneration caused by inactivation of Drosophila Pink1 is rescued by Parkin. Proc. Natl Acad. Sci. USA 103, 10793–10798 (2006).
Narendra, D., Tanaka, A., Suen, D. F. & Youle, R. J. Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J. Cell Biol. 183, 795–803 (2008). Shows that parkin translocates to mitochondria with depolarized membranes and promotes their removal by mitophagy.
Narendra, D. P. et al. PINK1 is selectively stabilized on impaired mitochondria to activate parkin. PLoS Biol. 8, e1000298 (2010).
Chan, N. C. et al. Broad activation of the ubiquitin–proteasome system by parkin is critical for mitophagy. Hum. Mol. Genet. 20, 1726–1737 (2011).
Sarraf, S. A. et al. Landscape of the PARKIN-dependent ubiquitylome in response to mitochondrial depolarization. Nature 496, 372–376 (2013).
Yoshii, S. R., Kishi, C., Ishihara, N. & Mizushima, N. Parkin mediates proteasome-dependent protein degradation and rupture of the outer mitochondrial membrane. J. Biol. Chem. 286, 19630–19640 (2011).
Tanaka, A. et al. Proteasome and p97 mediate mitophagy and degradation of mitofusins induced by parkin. J. Cell Biol. 191, 1367–1380 (2010).
Xu, S., Peng, G., Wang, Y., Fang, S. & Karbowski, M. The AAA-ATPase p97 is essential for outer mitochondrial membrane protein turnover. Mol. Biol. Cell 22, 291–300 (2011).
Suen, D. F., Narendra, D. P., Tanaka, A., Manfredi, G. & Youle, R. J. Parkin overexpression selects against a deleterious mtDNA mutation in heteroplasmic cybrid cells. Proc. Natl Acad. Sci. USA 107, 11835–11840 (2010).
Vincow, E. S. et al. The PINK1–parkin pathway promotes both mitophagy and selective respiratory chain turnover in vivo. Proc. Natl Acad. Sci. USA 110, 6400–6405 (2013).
Mortensen, M. et al. Loss of autophagy in erythroid cells leads to defective removal of mitochondria and severe anemia in vivo. Proc. Natl Acad. Sci. USA 107, 832–837 (2010).
Matsui, M., Yamamoto, A., Kuma, A., Ohsumi, Y. & Mizushima, N. Organelle degradation during the lens and erythroid differentiation is independent of autophagy. Biochem. Biophys. Res. Commun. 339, 485–489 (2006).
Zhang, J. et al. Mitochondrial clearance is regulated by Atg7-dependent and -independent mechanisms during reticulocyte maturation. Blood 114, 157–164 (2009).
Schweers, R. L. et al. NIX is required for programmed mitochondrial clearance during reticulocyte maturation. Proc. Natl Acad. Sci. USA 104, 19500–19505 (2007).
Sandoval, H. et al. Essential role for Nix in autophagic maturation of erythroid cells. Nature 454, 232–235 (2008).
Novak, I. et al. Nix is a selective autophagy receptor for mitochondrial clearance. EMBO Rep. 11, 45–51 (2010).
Liu, L. et al. Mitochondrial outer-membrane protein FUNDC1 mediates hypoxia-induced mitophagy in mammalian cells. Nature Cell Biol. 14, 177–185 (2012).
Chen, G. et al. A regulatory signaling loop comprising the PGAM5 phosphatase and CK2 controls receptor-mediated mitophagy. Mol. Cell 54, 362–377 (2014).
Okamoto, K., Kondo-Okamoto, N. & Ohsumi, Y. Mitochondria-anchored receptor Atg32 mediates degradation of mitochondria via selective autophagy. Dev. Cell 17, 87–97 (2009).
Kanki, T., Wang, K., Cao, Y., Baba, M. & Klionsky, D. J. Atg32 is a mitochondrial protein that confers selectivity during mitophagy. Dev. Cell 17, 98–109 (2009).
Mao, K., Wang, K., Liu, X. & Klionsky, D. J. The scaffold protein Atg11 recruits fission machinery to drive selective mitochondria degradation by autophagy. Dev. Cell 26, 9–18 (2013).
Mendl, N. et al. Mitophagy in yeast is independent of mitochondrial fission and requires the stress response gene WHI2. J. Cell Sci. 124, 1339–1350 (2011).
He, Y. et al. Heteroplasmic mitochondrial DNA mutations in normal and tumour cells. Nature 464, 610–614 (2010).
Payne, B. A. et al. Universal heteroplasmy of human mitochondrial DNA. Hum. Mol. Genet. 22, 384–390 (2013).
Yu-Wai-Man, P., Griffiths, P. G. & Chinnery, P. F. Mitochondrial optic neuropathies – disease mechanisms and therapeutic strategies. Prog. Retin Eye Res. 30, 81–114 (2011).
Carelli, V. et al. Retinal ganglion cell neurodegeneration in mitochondrial inherited disorders. Biochim. Biophys. Acta 1787, 518–528 (2009).
Chomyn, A. et al. MELAS mutation in mtDNA binding site for transcription termination factor causes defects in protein synthesis and in respiration but no change in levels of upstream and downstream mature transcripts. Proc. Natl Acad. Sci. USA 89, 4221–4225 (1992).
Hayashi, J. et al. Introduction of disease-related mitochondrial DNA deletions into HeLa cells lacking mitochondrial DNA results in mitochondrial dysfunction. Proc. Natl Acad. Sci. USA 88, 10614–10618 (1991).
Rossignol, R. et al. Mitochondrial threshold effects. Biochem. J. 370, 751–762 (2003).
Nakada, K. et al. Inter-mitochondrial complementation: mitochondria-specific system preventing mice from expression of disease phenotypes by mutant mtDNA. Nature Med. 7, 934–940 (2001).
Ono, T., Isobe, K., Nakada, K. & Hayashi, J. I. Human cells are protected from mitochondrial dysfunction by complementation of DNA products in fused mitochondria. Nature Genet. 28, 272–275 (2001).
Koehler, C. M. et al. Replacement of bovine mitochondrial DNA by a sequence variant within one generation. Genetics 129, 247–255 (1991).
Jenuth, J. P., Peterson, A. C., Fu, K. & Shoubridge, E. A. Random genetic drift in the female germline explains the rapid segregation of mammalian mitochondrial DNA. Nature Genet. 14, 146–151 (1996).
Ghosh, S. S., Fahy, E., Bodis-Wollner, I., Sherman, J. & Howell, N. Longitudinal study of a heteroplasmic 3460 Leber hereditary optic neuropathy family by multiplexed primer-extension analysis and nucleotide sequencing. Am. J. Hum. Genet. 58, 325–334 (1996).
Larsson, N. G. et al. Segregation and manifestations of the mtDNA tRNA(Lys) A-->G(8344) mutation of myoclonus epilepsy and ragged-red fibers (MERRF) syndrome. Am. J. Hum. Genet. 51, 1201–1212 (1992).
Cree, L. M. et al. A reduction of mitochondrial DNA molecules during embryogenesis explains the rapid segregation of genotypes. Nature Genet. 40, 249–254 (2008).
Cao, L. et al. New evidence confirms that the mitochondrial bottleneck is generated without reduction of mitochondrial DNA content in early primordial germ cells of mice. PLoS Genet. 5, e1000756 (2009).
Wai, T., Teoli, D. & Shoubridge, E. A. The mitochondrial DNA genetic bottleneck results from replication of a subpopulation of genomes. Nature Genet. 40, 1484–1488 (2008).
Bogenhagen, D. & Clayton, D. A. Mouse L cell mitochondrial DNA molecules are selected randomly for replication throughout the cell cycle. Cell 11, 719–727 (1977).
Sato, A. et al. Deletion-mutant mtDNA increases in somatic tissues but decreases in female germ cells with age. Genetics 177, 2031–2037 (2007).
Fan, W. et al. A mouse model of mitochondrial disease reveals germline selection against severe mtDNA mutations. Science 319, 958–962 (2008).
Stewart, J. B. et al. Strong purifying selection in transmission of mammalian mitochondrial DNA. PLoS Biol. 6, e10 (2008). References 84 and 85 provide evidence for the existence of a mechanism of purifying selection for mtDNA mutations in the mouse female germ line.
Stewart, J. B., Freyer, C., Elson, J. L. & Larsson, N. G. Purifying selection of mtDNA and its implications for understanding evolution and mitochondrial disease. Nature Rev. Genet. 9, 657–662 (2008).
Chomyn, A. & Attardi, G. mtDNA mutations in aging and apoptosis. Biochem. Biophys. Res. Commun. 304, 519–529 (2003).
Piko, L. & Taylor, K. D. Amounts of mitochondrial DNA and abundance of some mitochondrial gene transcripts in early mouse embryos. Dev. Biol. 123, 364–374 (1987).
Lee, H. S. et al. Rapid mitochondrial DNA segregation in primate preimplantation embryos precedes somatic and germline bottleneck. Cell Rep. 1, 506–515 (2012).
Jenuth, J. P., Peterson, A. C. & Shoubridge, E. A. Tissue-specific selection for different mtDNA genotypes in heteroplasmic mice. Nature Genet. 16, 93–95 (1997).
Battersby, B. J. & Shoubridge, E. A. Selection of a mtDNA sequence variant in hepatocytes of heteroplasmic mice is not due to differences in respiratory chain function or efficiency of replication. Hum. Mol. Genet. 10, 2469–2479 (2001).
Sharpley, M. S. et al. Heteroplasmy of mouse mtDNA is genetically unstable and results in altered behavior and cognition. Cell 151, 333–343 (2012).
Boynton, J. E., Harris, E. H., Burkhart, B. D., Lamerson, P. M. & Gillham, N. W. Transmission of mitochondrial and chloroplast genomes in crosses of Chlamydomonas. Proc. Natl Acad. Sci. USA 84, 2391–2395 (1987).
Schwartz, M. & Vissing, J. Paternal inheritance of mitochondrial DNA. N. Engl. J. Med. 347, 576–580 (2002).
DeLuca, S. Z. & O'Farrell, P. H. Barriers to male transmission of mitochondrial DNA in sperm development. Dev. Cell 22, 660–668 (2012).
Hecht, N. B., Liem, H., Kleene, K. C., Distel, R. J. & Ho, S. M. Maternal inheritance of the mouse mitochondrial genome is not mediated by a loss or gross alteration of the paternal mitochondrial DNA or by methylation of the oocyte mitochondrial DNA. Dev. Biol. 102, 452–461 (1984).
Sutovsky, P. et al. Ubiquitin tag for sperm mitochondria. Nature 402, 371–372 (1999).
Al Rawi, S. et al. Postfertilization autophagy of sperm organelles prevents paternal mitochondrial DNA transmission. Science 334, 1144–1147 (2011).
Sato, M. & Sato, K. Degradation of paternal mitochondria by fertilization-triggered autophagy in C. elegans embryos. Science 334, 1141–1144 (2011). References 98 and 99 demonstrate a role for autophagy in the removal of paternal mitochondria in the early nematode embryo.
Zhou, Q., Li, H. & Xue, D. Elimination of paternal mitochondria through the lysosomal degradation pathway in C. elegans. Cell Res. 21, 1662–1669 (2011).
Luo, S. M. et al. Unique insights into maternal mitochondrial inheritance in mice. Proc. Natl Acad. Sci. USA 110, 13038–13043 (2013).
Meeusen, S. et al. Mitochondrial inner-membrane fusion and crista maintenance requires the dynamin-related GTPase Mgm1. Cell 127, 383–395 (2006).
Song, Z., Ghochani, M., McCaffery, J. M., Frey, T. G. & Chan, D. C. Mitofusins and OPA1 mediate sequential steps in mitochondrial membrane fusion. Mol. Biol. Cell 20, 3525–3532 (2009).
Loson, O. C., Song, Z., Chen, H. & Chan, D. C. Fis1, Mff, MiD49, and MiD51 mediate Drp1 recruitment in mitochondrial fission. Mol. Biol. Cell 24, 659–667 (2013).
Otera, H. et al. Mff is an essential factor for mitochondrial recruitment of Drp1 during mitochondrial fission in mammalian cells. J. Cell Biol. 191, 1141–1158 (2010).
Rafelski, S. M. et al. Mitochondrial network size scaling in budding yeast. Science 338, 822–824 (2012).
McConnell, S. J., Stewart, L. C., Talin, A. & Yaffe, M. P. Temperature-sensitive yeast mutants defective in mitochondrial inheritance. J. Cell Biol. 111, 967–976 (1990).
Evangelista, M., Pruyne, D., Amberg, D. C., Boone, C. & Bretscher, A. Formins direct Arp2/3-independent actin filament assembly to polarize cell growth in yeast. Nature Cell Biol. 4, 260–269 (2002).
Fagarasanu, A., Mast, F. D., Knoblach, B. & Rachubinski, R. A. Molecular mechanisms of organelle inheritance: lessons from peroxisomes in yeast. Nature Rev. Mol. Cell Biol. 11, 644–654 (2010).
Lazzarino, D. A., Boldogh, I., Smith, M. G., Rosand, J. & Pon, L. A. Yeast mitochondria contain ATP-sensitive, reversible actin-binding activity. Mol. Biol. Cell 5, 807–818 (1994).
Drubin, D. G., Jones, H. D. & Wertman, K. F. Actin structure and function: roles in mitochondrial organization and morphogenesis in budding yeast and identification of the phalloidin-binding site. Mol. Biol. Cell 4, 1277–1294 (1993).
Boldogh, I. R. et al. Arp2/3 complex and actin dynamics are required for actin-based mitochondrial motility in yeast. Proc. Natl Acad. Sci. USA 98, 3162–3167 (2001).
Boldogh, I. R., Ramcharan, S. L., Yang, H. C. & Pon, L. A. A type V myosin (Myo2p) and a Rab-like G-protein (Ypt11p) are required for retention of newly inherited mitochondria in yeast cells during cell division. Mol. Biol. Cell 15, 3994–4002 (2004).
Lambrechts, A., Gevaert, K., Cossart, P., Vandekerckhove, J. & Van Troys, M. Listeria comet tails: the actin-based motility machinery at work. Trends Cell Biol. 18, 220–227 (2008).
Altmann, K., Frank, M., Neumann, D., Jakobs, S. & Westermann, B. The class V myosin motor protein, Myo2, plays a major role in mitochondrial motility in Saccharomyces cerevisiae. J. Cell Biol. 181, 119–130 (2008). Identifies a myosin family member, myosin 2, as a mitochondrial motor protein.
Chernyakov, I., Santiago-Tirado, F. & Bretscher, A. Active segregation of yeast mitochondria by Myo2 is essential and mediated by Mmr1 and Ypt11. Curr. Biol. 23, 1818–1824 (2013).
Fortsch, J., Hummel, E., Krist, M. & Westermann, B. The myosin-related motor protein Myo2 is an essential mediator of bud-directed mitochondrial movement in yeast. J. Cell Biol. 194, 473–488 (2011).
Ishikawa, K. et al. Identification of an organelle-specific myosin V receptor. J. Cell Biol. 160, 887–897 (2003).
Tang, F. et al. Regulated degradation of a class V myosin receptor directs movement of the yeast vacuole. Nature 422, 87–92 (2003).
Govindan, B., Bowser, R. & Novick, P. The role of Myo2, a yeast class V myosin, in vesicular transport. J. Cell Biol. 128, 1055–1068 (1995).
Pruyne, D. W., Schott, D. H. & Bretscher, A. Tropomyosin-containing actin cables direct the Myo2p-dependent polarized delivery of secretory vesicles in budding yeast. J. Cell Biol. 143, 1931–1945 (1998).
Rossanese, O. W. et al. A role for actin, Cdc1p, and Myo2p in the inheritance of late Golgi elements in Saccharomyces cerevisiae. J. Cell Biol. 153, 47–62 (2001).
Hoepfner, D., van den Berg, M., Philippsen, P., Tabak, H. F. & Hettema, E. H. A role for Vps1p, actin, and the Myo2p motor in peroxisome abundance and inheritance in Saccharomyces cerevisiae. J. Cell Biol. 155, 979–990 (2001).
Itoh, T., Toh, E. A. & Matsui, Y. Mmr1p is a mitochondrial factor for Myo2p-dependent inheritance of mitochondria in the budding yeast. EMBO J. 23, 2520–2530 (2004).
Itoh, T., Watabe, A., Toh, E. A. & Matsui, Y. Complex formation with Ypt11p, a rab-type small GTPase, is essential to facilitate the function of Myo2p, a class V myosin, in mitochondrial distribution in Saccharomyces cerevisiae. Mol. Cell. Biol. 22, 7744–7757 (2002).
Kelleher, J. F. et al. Myosin VI is required for asymmetric segregation of cellular components during C. elegans spermatogenesis. Curr. Biol. 10, 1489–1496 (2000).
Klecker, T., Scholz, D., Fortsch, J. & Westermann, B. The yeast cell cortical protein Num1 integrates mitochondrial dynamics into cellular architecture. J. Cell Sci. 126, 2924–2930 (2013).
Lackner, L. L., Ping, H., Graef, M., Murley, A. & Nunnari, J. Endoplasmic reticulum-associated mitochondria-cortex tether functions in the distribution and inheritance of mitochondria. Proc. Natl Acad. Sci. USA 110, E458–E467 (2013).
de Brito, O. M. & Scorrano, L. Mitofusin 2 tethers endoplasmic reticulum to mitochondria. Nature 456, 605–610 (2008).
Rizzuto, R., Brini, M., Murgia, M. & Pozzan, T. Microdomains with high Ca2+ close to IP3-sensitive channels that are sensed by neighboring mitochondria. Science 262, 744–747 (1993).
Achleitner, G. et al. Association between the endoplasmic reticulum and mitochondria of yeast facilitates interorganelle transport of phospholipids through membrane contact. Eur. J. Biochem. 264, 545–553 (1999).
Rusinol, A. E., Cui, Z., Chen, M. H. & Vance, J. E. A unique mitochondria-associated membrane fraction from rat liver has a high capacity for lipid synthesis and contains pre-Golgi secretory proteins including nascent lipoproteins. J. Biol. Chem. 269, 27494–27502 (1994).
Friedman, J. R. et al. ER tubules mark sites of mitochondrial division. Science 334, 358–362 (2011). Shows that ER tubules are involved in the constriction of mitochondria at fission sites.
Korobova, F., Ramabhadran, V. & Higgs, H. N. An actin-dependent step in mitochondrial fission mediated by the ER-associated formin INF2. Science 339, 464–467 (2013).
Kornmann, B. et al. An ER–mitochondria tethering complex revealed by a synthetic biology screen. Science 325, 477–481 (2009). Reports the use of a genetic synthetic biology screen to identify components of the ERMES (ER–mitochondria encounter structure) complex in S. cerevisiae.
Murley, A. et al. ER-associated mitochondrial division links the distribution of mitochondria and mitochondrial DNA in yeast. Elife 2, e00422 (2013). Provides a model linking mitochondrial division with inheritance of mtDNA nucleoids in yeast.
Berger, K. H., Sogo, L. F. & Yaffe, M. P. Mdm12p, a component required for mitochondrial inheritance that is conserved between budding and fission yeast. J. Cell Biol. 136, 545–553 (1997).
Sogo, L. F. & Yaffe, M. P. Regulation of mitochondrial morphology and inheritance by Mdm10p, a protein of the mitochondrial outer membrane. J. Cell Biol. 126, 1361–1373 (1994).
Acknowledgements
P.M. is supported by a Baxter Postdoctoral Fellowship. Work in laboratory of D.C.C. is supported by the US National Institutes of Health grants GM062967 and GM110039, the Muscular Dystrophy Association and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Oxidative phosphorylation
-
(OXPHOS). A biochemical pathway within mitochondria that generates ATP through the oxidation of nutrients.
- Endosymbiotic theory
-
A theory postulating that genome-containing organelles, such as mitochondria and chloroplasts, are derived from bacteria that underwent endosymbiosis with ancestral eukaryotic cells.
- Genetic bottlenecks
-
Drastic reductions in the genetic diversity of a population.
- Motor proteins
-
Molecular motors that use ATP hydrolysis to power the movement of cargo along substrate surfaces.
- Kinesins
-
A family of ATP-dependent molecular motors that transport cargo along microtubule filaments, usually towards the plus end.
- Dyneins
-
A family of ATP-dependent molecular motors that transport cargo along microtubule filaments towards the minus end.
- Formin proteins
-
A group of proteins that regulate the actin cytoskeleton and cell signalling.
- Myosin
-
A family of ATP-dependent molecular motors that transport cargo along actin filaments.
- Dynamin-related GTPases
-
A family of large GTP-hydrolysing enzymes related to 'classical' dynamin, which use GTP hydrolysis to mechanically remodel membranes.
- mtDNA nucleoid
-
A punctate structure within mitochondria that contains one or more copies of the mitochondrial DNA (mtDNA) genome in complex with proteins.
- Macroautophagy
-
The most commonly studied autophagic process, whereby cellular components are incorporated into autophagosomes and ultimately degraded or recycled through the lysosome. Macroautophagy is commonly referred to as simply 'autophagy'.
- Autophagosomes
-
Double-membrane structures that engulf cellular components for delivery to lysosomes.
- Lysosomes
-
Organelles that degrade cellular components using acid hydrolases.
- Cytoplasmic hybrid (cybrid) cells
-
Hybrid cells created by the fusion of a cell with an enucleated second cell, thereby combining the nucleus and mitochondria from two distinct cells.
- Heteroplasmic
-
A state in which more than one haplotype of mitochondrial DNA exists in a cell or an organism.
- Homoplasmy
-
A state in which a single haplotype of mitochondrial DNA exists in a cell or an organism.
- Genetic drift
-
A change in the allele or haplotype frequencies within a population owing to stochastic forces.
- Purifying selection
-
A mode of natural selection in which detrimental genetic variants are selected against.
Rights and permissions
About this article
Cite this article
Mishra, P., Chan, D. Mitochondrial dynamics and inheritance during cell division, development and disease. Nat Rev Mol Cell Biol 15, 634–646 (2014). https://doi.org/10.1038/nrm3877
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3877
This article is cited by
-
Preneoplastic cells switch to Warburg metabolism from their inception exposing multiple vulnerabilities for targeted elimination
Oncogenesis (2024)
-
Structural insights into the specific recognition of mitochondrial ribosome-binding factor hsRBFA and 12 S rRNA by methyltransferase METTL15
Cell Discovery (2024)
-
Toxic interactions between dopamine, α-synuclein, monoamine oxidase, and genes in mitochondria of Parkinson’s disease
Journal of Neural Transmission (2024)
-
Mitophagy plays a “double-edged sword” role in the radiosensitivity of cancer cells
Journal of Cancer Research and Clinical Oncology (2024)
-
Mitochondria in Mesenchymal Stem Cells: Key to Fate Determination and Therapeutic Potential
Stem Cell Reviews and Reports (2024)