Key Points
-
The conserved Polycomb group (PcG) and Trithorax group (TrxG) proteins are transcriptional regulators, with several hundred target genes. Disruption of PcG or TrxG protein function can cause developmental defects and cancer.
-
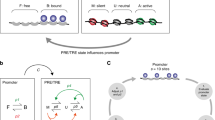
The PcG and TrxG proteins are recruited to cis-regulatory elements called Polycomb/Trithorax response elements (PREs). The PcG and TrxG proteins act antagonistically through PREs to maintain silent and active gene expression states, respectively. These states are initiated by transcription factors that act at enhancers and are maintained by PcG and TrxG proteins at the PRE.
-
Epigenetic memory is defined as the maintenance of gene expression states through cell generations in the absence of the initiating signals. PREs were first discovered in flies, in which they can mediate epigenetic memory of both silent and active states of their associated genes. Epigenetic memory has not yet been tested for mammalian PREs.
-
Silent and activated states are each ensured by several self-reinforcing molecular mechanisms that act on chromatin and are mediated by the PcG and TrxG proteins. These include covalent modifications of chromatin, structural changes and modulation of RNA polymerase II activity. Mechanisms of silencing and activation antagonize each other.
-
During the cell cycle, replication and mitosis are the stages at which epigenetic memory is most likely to be erased. Each stage presents a different molecular challenge to memory.
-
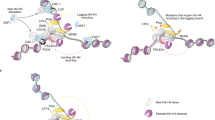
During replication, newly assembled chromatin is acetylated and may favour an active PRE but challenge a silent one. Several PcG and TrxG proteins remain bound to replicating chromatin and may transmit memory to daughter strands.
-
During mitosis, many proteins dissociate from chromatin and transcription shuts down. This may favour a silent PRE but challenge an active one. Several PcG and TrxG proteins remain bound to mitotic chromatin and may 'bookmark' silent and active states.
-
Many PcG and TrxG target genes are dynamically regulated during development and do not display long-term epigenetic memory. For these targets, the PcG and TrxG proteins may reinforce or amplify transcriptional levels over shorter timescales. This may add robustness to gene regulation.
Abstract
In any biological system with memory, the state of the system depends on its history. Epigenetic memory maintains gene expression states through cell generations without a change in DNA sequence and in the absence of initiating signals. It is immensely powerful in biological systems — it adds long-term stability to gene expression states and increases the robustness of gene regulatory networks. The Polycomb group (PcG) and Trithorax group (TrxG) proteins can confer long-term, mitotically heritable memory by sustaining silent and active gene expression states, respectively. Several recent studies have advanced our understanding of the molecular mechanisms of this epigenetic memory during DNA replication and mitosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ringrose, L. & Paro, R. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu. Rev. Genet. 38, 413–443 (2004).
Ringrose, L. Polycomb comes of age: genome-wide profiling of target sites. Curr. Opin. Cell Biol. 19, 290–297 (2007).
Schuettengruber, B., Chourrout, D., Vervoort, M., Leblanc, B. & Cavalli, G. Genome regulation by Polycomb and Trithorax proteins. Cell 128, 735–745 (2007).
Margueron, R. & Reinberg, D. The Polycomb complex PRC2 and its mark in life. Nature 469, 343–349 (2011).
Schumacher, A. & Magnuson, T. Murine Polycomb- and Trithorax-group genes regulate homeotic pathways and beyond. Trends Genet. 13, 167–170 (1997).
Kennison, J. A. Introduction to Trx-G and Pc-G genes. Methods Enzymol. 377, 61–70 (2004).
Christophersen, N. S. & Helin, K. Epigenetic control of embryonic stem cell fate. J. Exp. Med. 207, 2287–2295 (2010).
Dillon, N. Factor mediated gene priming in pluripotent stem cells sets the stage for lineage specification. Bioessays 34, 194–204 (2012).
Maeda, R. K. & Karch, F. The ABC of the BX-C: the bithorax complex explained. Development 133, 1413–1422 (2006).
Soshnikova, N. & Duboule, D. Epigenetic regulation of vertebrate Hox genes: a dynamic equilibrium. Epigenetics 4, 537–540 (2009).
Mendenhall, E. M. & Bernstein, B. E. Chromatin state maps: new technologies, new insights. Curr. Opin. Genet. Dev. 18, 109–115 (2008).
Simon, J., Chiang, A., Bender, W., Shimell, M. J. & O'Connor, M. Elements of the Drosophila bithorax complex that mediate repression by Polycomb group products. Dev. Biol. 158, 131–144 (1993). First study to show that the PRE from one regulatory domain of the BX-C maintains the pattern set by an enhancer from a different regulatory domain, which demonstrates that the PRE is a memory element and not a position-specific enhancer.
Chan, C. S., Rastelli, L. & Pirrotta, V. A. Polycomb response element in the Ubx gene that determines an epigenetically inherited state of repression. EMBO J. 13, 2553–2564 (1994). First report to show that a PRE is required for the transfer of epigenetic memory of transcriptional patterns that are set during embryogenesis in larval tissues, thereby demonstrating long-term epigenetic memory.
Chiang, A., O'Connor, M. B., Paro, R., Simon, J. & Bender, W. Discrete Polycomb-binding sites in each parasegmental domain of the bithorax complex. Development 121, 1681–1689 (1995).
Poux, S., Kostic, C. & Pirrotta, V. Hunchback-independent silencing of late Ubx enhancers by a Polycomb group response element. EMBO J. 15, 4713–4722 (1996).
Hagstrom, K., Muller, M. & Schedl, P. A Polycomb and GAGA dependent silencer adjoins the Fab-7 boundary in the Drosophila bithorax complex. Genetics 146, 1365–1380 (1997).
Busturia, A., Wightman, C. D. & Sakonju, S. A silencer is required for maintenance of transcriptional repression throughout Drosophila development. Development 124, 4343–4350 (1997). Shows, for the first time, that the PRE is required for maintenance of repression, using FLP-mediated excision of a PRE, thus excluding models in which memory is maintained by heterochromatin-like structures that act independently of the DNA sequence. Also carries out a PRE 'swap' experiment.
Cavalli, G. & Paro, R. The Drosophila Fab-7 chromosomal element conveys epigenetic inheritance during mitosis and meiosis. Cell 93, 505–518 (1998). First study to show that a PRE can maintain induced active transcriptional states (previous work had focused on the maintenance of silencing). Reports that active states are maintained across generations in a proportion of flies.
Cavalli, G. & Paro, R. Epigenetic inheritance of active chromatin after removal of the main transactivator. Science 286, 955–958 (1999).
Rank, G., Prestel, M. & Paro, R. Transcription through intergenic chromosomal memory elements of the Drosophila bithorax complex correlates with an epigenetic switch. Mol. Cell. Biol. 22, 8026–8034 (2002).
Americo, J. et al. A complex array of DNA-binding proteins required for pairing-sensitive silencing by a Polycomb group response element from the Drosophila engrailed gene. Genetics 160, 1561–1571 (2002).
Maurange, C. & Paro, R. A cellular memory module conveys epigenetic inheritance of hedgehog expression during Drosophila wing imaginal disc development. Genes Dev. 16, 2672–2683 (2002).
Perez, L. et al. Enhancer-PRE communication contributes to the expansion of gene expression domains in proliferating primordia. Development 138, 3125–3134 (2011). Describes, together with reference 18, how reporter genes that are linked to PREs are activated. In reference 18, activation of the reporter is artificially induced at high levels by heat shock. In reference 23, activation is achieved using developmental enhancers, which reveals a function for PRE-mediated memory of active states during tissue growth.
Muller, J. & Bienz, M. Long range repression conferring boundaries of Ultrabithorax expression in the Drosophila embryo. EMBO J. 10, 3147–3155 (1991).
Busturia, A. & Bienz, M. Silencers in abdominal-B, a homeotic Drosophila gene. EMBO J. 12, 1415–1425 (1993).
Mihaly, J., Hogga, I., Gausz, J., Gyurkovics, H. & Karch, F. In situ dissection of the Fab-7 region of the bithorax complex into a chromatin domain boundary and a Polycomb-response element. Development 124, 1809–1820 (1997).
Shimell, M. J., Peterson, A. J., Burr, J., Simon, J. A. & O'Connor, M. B. Functional analysis of repressor binding sites in the iab-2 regulatory region of the abdominal-A homeotic gene. Dev. Biol. 218, 38–52 (2000).
Barges, S. et al. The Fab-8 boundary defines the distal limit of the bithorax complex iab-7 domain and insulates iab-7 from initiation elements and a PRE in the adjacent iab-8 domain. Development 127, 779–790 (2000).
Perez-Lluch, S., Cuartero, S., Azorin, F. & Espinas, M. L. Characterization of new regulatory elements within the Drosophila bithorax complex. Nucleic Acids Res. 36, 6926–6933 (2008).
Zink, B., Engstrom, Y., Gehring, W. J. & Paro, R. Direct interaction of the Polycomb protein with Antennapedia regulatory sequences in polytene chromosomes of Drosophila melanogaster. EMBO J. 10, 153–162 (1991).
Gindhart, J. G. Jr & Kaufman, T. C. Identification of Polycomb and trithorax group responsive elements in the regulatory region of the Drosophila homeotic gene Sex combs reduced. Genetics 139, 797–814 (1995).
Ringrose, L. & Paro, R. Polycomb/Trithorax response elements and epigenetic memory of cell identity. Development 134, 223–232 (2007).
Kassis, J. A. & Brown, J. L. Polycomb group response elements in Drosophila and vertebrates. Adv. Genet. 81, 83–118 (2013).
DeVido, S. K., Kwon, D., Brown, J. L. & Kassis, J. A. The role of Polycomb-group response elements in regulation of engrailed transcription in Drosophila. Development 135, 669–676 (2008).
Okulski, H., Druck, B., Bhalerao, S. & Ringrose, L. Quantitative analysis of Polycomb response elements (PREs) at identical genomic locations distinguishes contributions of PRE sequence and genomic environment. Epigenet. Chromatin 4, 4 (2011).
Beuchle, D., Struhl, G. & Muller, J. Polycomb group proteins and heritable silencing of Drosophila Hox genes. Development 128, 993–1004 (2001).
Ringrose, L. & Paro, R. Remembering silence. Bioessays 23, 566–570 (2001).
Sipos, L., Kozma, G., Molnar, E. & Bender, W. In situ dissection of a Polycomb response element in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 104, 12416–12421 (2007).
Kozma, G., Bender, W. & Sipos, L. Replacement of a Drosophila Polycomb response element core, and in situ analysis of its DNA motifs. Mol. Genet. Genom. 279, 595–603 (2008).
Sing, A. et al. A vertebrate Polycomb response element governs segmentation of the posterior hindbrain. Cell 138, 885–897 (2009).
Mendenhall, E. M. et al. GC-rich sequence elements recruit PRC2 in mammalian ES cells. PLoS Genet. 6, e1001244 (2010).
Woo, C. J., Kharchenko, P. V., Daheron, L., Park, P. J. & Kingston, R. E. A region of the human HOXD cluster that confers Polycomb-group responsiveness. Cell 140, 99–110 (2010).
Meng, S. et al. Identification and characterization of Bmi-1-responding element within the human p16 promoter. J. Biol. Chem. 285, 33219–33229 (2010).
Lynch, M. D. et al. An interspecies analysis reveals a key role for unmethylated CpG dinucleotides in vertebrate Polycomb complex recruitment. EMBO J. 31, 317–329 (2011).
Cabianca, D. S. et al. A long ncRNA links copy number variation to a Polycomb/Trithorax epigenetic switch in FSHD muscular dystrophy. Cell 149, 819–831 (2012).
Arnold, P. et al. Modeling of epigenome dynamics identifies transcription factors that mediate Polycomb targeting. Genome Res. 23, 60–73 (2012).
Woo, C. J., Kharchenko, P. V., Daheron, L., Park, P. J. & Kingston, R. E. Variable requirements for DNA-binding proteins at Polycomb-dependent repressive regions in human HOX clusters. Mol. Cell. Biol. 33, 3274–3285 (2013).
Lanzuolo, C. & Orlando, V. Memories from the Polycomb group proteins. Annu. Rev. Genet. 46, 561–589 (2012).
Simon, J. A. & Kingston, R. E. Occupying chromatin: Polycomb mechanisms for getting to genomic targets, stopping transcriptional traffic, and staying put. Mol. Cell 49, 808–824 (2013).
Simon, J. A. & Kingston, R. E. Mechanisms of Polycomb gene silencing: knowns and unknowns. Nature Rev. Mol. Cell Biol. 10, 697–708 (2009).
Schuettengruber, B., Martinez, A. M., Iovino, N. & Cavalli, G. Trithorax group proteins: switching genes on and keeping them active. Nature Rev. Mol. Cell Biol. 12, 799–814 (2011).
O'Meara, M. M. & Simon, J. A. Inner workings and regulatory inputs that control Polycomb repressive complex 2. Chromosoma 121, 221–234 (2012).
Poux, S., Horard, B., Sigrist, C. J. & Pirrotta, V. The Drosophila trithorax protein is a coactivator required to prevent re-establishment of Polycomb silencing. Development 129, 2483–2493 (2002).
Klymenko, T. & Muller, J. The histone methyltransferases Trithorax and Ash1 prevent transcriptional silencing by Polycomb group proteins. EMBO Rep. 5, 373–377 (2004). References 53 and 54 demonstrate antagonism between PcG and TrxG proteins for target gene regulation in vivo.
Yuan, W. et al. H3K36 methylation antagonizes PRC2-mediated H3K27 methylation. J. Biol. Chem. 286, 7983–7989 (2011).
Schmitges, F. W. et al. Histone methylation by PRC2 is inhibited by active chromatin marks. Mol. Cell 42, 330–341 (2011).
Tie, F. et al. CBP-mediated acetylation of histone H3 lysine 27 antagonizes Drosophila Polycomb silencing. Development 136, 3131–3141 (2009).
Tie, F. et al. Trithorax monomethylates histone H3K4 and interacts directly with CBP to promote H3K27 acetylation and antagonize Polycomb silencing. Development 141, 1129–1139 (2014).
Pasini, D. et al. Characterization of an antagonistic switch between histone H3 lysine 27 methylation and acetylation in the transcriptional regulation of Polycomb group target genes. Nucleic Acids Res. 38, 4958–4969 (2010).
Jung, H. R., Pasini, D., Helin, K. & Jensen, O. N. Quantitative mass spectrometry of histones H3.2 and H3.3 in Suz12-deficient mouse embryonic stem cells reveals distinct, dynamic post-translational modifications at Lys-27 and Lys-36. Mol. Cell Proteom. 9, 838–850 (2010).
Stock, J. K. et al. Ring1-mediated ubiquitination of H2A restrains poised RNA polymerase II at bivalent genes in mouse ES cells. Nature Cell Biol. 9, 1428–1435 (2007).
Brookes, E. et al. Polycomb associates genome-wide with a specific RNA polymerase II variant, and regulates metabolic genes in ESCs. Cell Stem Cell 10, 157–170 (2012).
Devaiah, B. N. et al. BRD4 is an atypical kinase that phosphorylates serine2 of the RNA polymerase II carboxy-terminal domain. Proc. Natl Acad. Sci. USA 109, 6927–6932 (2012).
Francis, N. J., Kingston, R. E. & Woodcock, C. L. Chromatin compaction by a Polycomb group protein complex. Science 306, 1574–1577 (2004).
Eskeland, R. et al. Ring1B compacts chromatin structure and represses gene expression independent of histone ubiquitination. Mol. Cell 38, 452–464 (2010).
Endoh, M. et al. Histone H2A mono-ubiquitination is a crucial step to mediate PRC1-dependent repression of developmental genes to maintain ES cell identity. PLoS Genet. 8, e1002774 (2012).
Zentner, G. E. & Henikoff, S. Regulation of nucleosome dynamics by histone modifications. Nature Struct. Mol. Biol. 20, 259–266 (2013).
Sengupta, A. K., Kuhrs, A. & Muller, J. General transcriptional silencing by a Polycomb response element in Drosophila. Development 131, 1959–1965 (2004).
Papp, B. & Muller, J. Histone trimethylation and the maintenance of transcriptional ON and OFF states by trxG and PcG proteins. Genes Dev. 20, 2041–2054 (2006).
Beisel, C. et al. Comparing active and repressed expression states of genes controlled by the Polycomb/Trithorax group proteins. Proc. Natl Acad. Sci. USA 104, 16615–16620 (2007).
Schwartz, Y. B. et al. Alternative epigenetic chromatin states of Polycomb target genes. PLoS Genet. 6, e1000805 (2010).
Dejardin, J. & Cavalli, G. Chromatin inheritance upon Zeste-mediated Brahma recruitment at a minimal cellular memory module. EMBO J. 23, 857–868 (2004).
Langlais, K. K., Brown, J. L. & Kassis, J. A. Polycomb group proteins bind an engrailed PRE in both the “ON” and “OFF” transcriptional states of engrailed. PLoS ONE 7, e48765 (2012).
Simon, J., Chiang, A. & Bender, W. Ten different Polycomb group genes are required for spatial control of the abdA and AbdB homeotic products. Development 114, 493–505 (1992).
Birve, A. et al. Su(z)12, a novel Drosophila Polycomb group gene that is conserved in vertebrates and plants. Development 128, 3371–3379 (2001).
Bloyer, S., Cavalli, G., Brock, H. W. & Dura, J. M. Identification and characterization of polyhomeotic PREs and TREs. Dev. Biol. 261, 426–442 (2003).
Breen, T. R., Chinwalla, V. & Harte, P. J. Trithorax is required to maintain engrailed expression in a subset of engrailed-expressing cells. Mech. Dev. 52, 89–98 (1995).
Petruk, S. et al. Trithorax and dCBP acting in a complex to maintain expression of a homeotic gene. Science 294, 1331–1334 (2001).
Tanaka, Y. et al. Dual function of histone H3 lysine 36 methyltransferase ASH1 in regulation of Hox gene expression. PLoS ONE 6, e28171 (2011).
Cao, R., Tsukada, Y. & Zhang, Y. Role of Bmi-1 and Ring1A in H2A ubiquitylation and Hox gene silencing. Mol. Cell 20, 845–854 (2005).
Pengelly, A. R., Copur, O., Jackle, H., Herzig, A. & Muller, J. A histone mutant reproduces the phenotype caused by loss of histone-modifying factor Polycomb. Science 339, 698–699 (2013). Uses genetic techniques in D. melanogaster to create flies that carry clones of cells in which all H3 proteins are mutated at Lys27. These cells show upregulation of PcG targets, which demonstrates that H3K27 is essential for PcG-mediated silencing.
Muller, J. Transcriptional silencing by the Polycomb protein in Drosophila embryos. EMBO J. 14, 1209–1220 (1995).
Roseman, R. R. et al. Long-range repression by multiple polycomb group (PcG) proteins targeted by fusion to a defined DNA-binding domain in Drosophila. Genetics 158, 291–307 (2001).
Poux, S., McCabe, D. & Pirrotta, V. Recruitment of components of Polycomb group chromatin complexes in Drosophila. Development 128, 75–85 (2001).
Bunker, C. A. & Kingston, R. E. Transcriptional repression by Drosophila and mammalian Polycomb group proteins in transfected mammalian cells. Mol. Cell. Biol. 14, 1721–1732 (1994).
Mohd-Sarip, A., Venturini, F., Chalkley, G. E. & Verrijzer, C. P. Pleiohomeotic can link Polycomb to DNA and mediate transcriptional repression. Mol. Cell. Biol. 22, 7473–7483 (2002).
Hansen, K. H. et al. A model for transmission of the H3K27me3 epigenetic mark. Nature Cell Biol. 10, 1291–1300 (2008). Demonstrates PRC2 binding to trimethylated H3K27, which the authors propose propagates this mark after replication. Shows that tethering the PRC2 component EED or EZH2 to a reporter gene is sufficient to recruit other PRC2 components and to silence the reporter in HEK293 cells.
van der Vlag, J. & Otte, A. P. Transcriptional repression mediated by the human Polycomb-group protein EED involves histone deacetylation. Nature Genet. 23, 474–478 (1999).
Hsieh, J. J., Ernst, P., Erdjument-Bromage, H., Tempst, P. & Korsmeyer, S. J. Proteolytic cleavage of MLL generates a complex of N- and C-terminal fragments that confers protein stability and subnuclear localization. Mol. Cell. Biol. 23, 186–194 (2003).
Takeda, S. et al. Proteolysis of MLL family proteins is essential for taspase1-orchestrated cell cycle progression. Genes Dev. 20, 2397–2409 (2006).
Capotosti, F., Hsieh, J. J. & Herr, W. Species selectivity of mixed-lineage leukemia/Trithorax and HCF proteolytic maturation pathways. Mol. Cell. Biol. 27, 7063–7072 (2007).
Probst, A. V., Dunleavy, E. & Almouzni, G. Epigenetic inheritance during the cell cycle. Nature Rev. Mol. Cell Biol. 10, 192–206 (2009).
Alabert, C. & Groth, A. Chromatin replication and epigenome maintenance. Nature Rev. Mol. Cell Biol. 13, 153–167 (2012).
Mikkelsen, T. S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Enderle, D. et al. Polycomb preferentially targets stalled promoters of coding and noncoding transcripts. Genome Res. 21, 216–226 (2011).
Adelman, K. & Lis, J. T. Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans. Nature Rev. Genet. 13, 720–731 (2012).
Lanzuolo, C., Lo Sardo, F., Diamantini, A. & Orlando, V. PcG complexes set the stage for epigenetic inheritance of gene silencing in early S phase before replication. PLoS Genet. 7, e1002370 (2011).
Lo, S. M. et al. A bridging model for persistence of a Polycomb group protein complex through DNA replication in vitro. Mol. Cell 46, 784–796 (2012). In vitro experiments demonstrate dynamic contacts between D. melanogaster PRC1 (through Psc) and replicating chromatin, which provides a mechanism for continued attachment of PRC1, despite passage of the replication machinery.
Scharf, A. N., Barth, T. K. & Imhof, A. Establishment of histone modifications after chromatin assembly. Nucleic Acids Res. 37, 5032–5040 (2009).
Xu, M., Wang, W., Chen, S. & Zhu, B. A model for mitotic inheritance of histone lysine methylation. EMBO Rep. 13, 60–67 (2011). References 99 and 100 use quantitative mass spectrometry to examine the kinetics of histone methylation mark restoration after replication in HeLa cells, which shows that these marks are not fully restored until after mitosis.
Petruk, S. et al. TrxG and PcG proteins but not methylated histones remain associated with DNA through replication. Cell 150, 922–933 (2012). In vivo experiments using D. melanogaster embryos show that PcG and TrxG proteins are associated within 200 bp of the replication fork. PcG and TrxG proteins associate with newly replicated chromatin ahead of the modifications that they catalyse.
Radman-Livaja, M. et al. Patterns and mechanisms of ancestral histone protein inheritance in budding yeast. PLoS Biol. 9, e1001075 (2011). Uses epitope tagging to monitor parental and new H3 across the yeast genome over six generations. Ancestral H3 is preferentially retained at the 5′ ends of most genes. Mathematical modelling shows that the data are best explained by a model in which most maternal histones are reincorporated within 400 bp of their pre-replication locus during replication.
Leung, K. H., El Hassan, M. A. & Bremner, R. A rapid and efficient method to purify proteins at replication forks under native conditions. Biotechniques 55, 204–206 (2013). A simplified and more sensitive adaptation of the isolation of protein on nascent DNA (iPOND) technique. Detects the PRC2 components suppressor of zeste 12 homologue (SUZ12), EZH2, Rb-associated protein 46 (RbAp46; also known as RBBP7) and RbAp48 (also known as RBBP4) at replication forks in mammalian cells, which are not detected with classic iPOND.
Lo Sardo, F. et al. PcG-mediated higher-order chromatin structures modulate replication programs at the Drosophila BX-C. PLoS Genet. 9, e1003283 (2013).
Krajewski, W. A., Nakamura, T., Mazo, A. & Canaani, E. A motif within SET-domain proteins binds single-stranded nucleic acids and transcribed and supercoiled DNAs and can interfere with assembly of nucleosomes. Mol. Cell. Biol. 25, 1891–1899 (2005).
Francis, N. J., Follmer, N. E., Simon, M. D., Aghia, G. & Butler, J. D. Polycomb proteins remain bound to chromatin and DNA during DNA replication in vitro. Cell 137, 110–122 (2009).
Fischle, W. et al. Molecular basis for the discrimination of repressive methyl-lysine marks in histone H3 by Polycomb and HP1 chromodomains. Genes Dev. 17, 1870–1881 (2003).
Min, J., Zhang, Y. & Xu, R. M. Structural basis for specific binding of Polycomb chromodomain to histone H3 methylated at Lys 27. Genes Dev. 17, 1823–1828 (2003).
Margueron, R. et al. Role of the Polycomb protein EED in the propagation of repressive histone marks. Nature 461, 762–767 (2009). Demonstrates the activation of EZH2 histone methyltransferase activity by PRC2 binding to trimethylated H3K27, which the authors propose propagates this mark after replication. Shows that the PRC2 subunit EED is responsible for binding of PRC2 to trimethylated H3K27 and provides a co-crystal structure of the C-terminal domain of EED that contains WD40 repeats with a trimethylated H3K27 peptide. Mutations in the gene encoding the fly homologue of EED (Esc) that disrupt trimethylated H3K27 binding also disrupt development.
Yuan, W. et al. Dense chromatin activates Polycomb repressive complex 2 to regulate H3 lysine 27 methylation. Science 337, 971–975 (2012).
Pena-Diaz, J. et al. Transcription profiling during the cell cycle shows that a subset of Polycomb-targeted genes is upregulated during DNA replication. Nucleic Acids Res. 41, 2846–2856 (2013).
Lanzuolo, C., Lo Sardo, F. & Orlando, V. Concerted epigenetic signatures inheritance at PcG targets through replication. Cell Cycle 11, 1296–1300 (2012).
Bermejo, R., Lai, M. S. & Foiani, M. Preventing replication stress to maintain genome stability: resolving conflicts between replication and transcription. Mol. Cell 45, 710–718 (2012).
Gottesfeld, J. M. & Forbes, D. J. Mitotic repression of the transcriptional machinery. Trends Biochem. Sci. 22, 197–202 (1997).
Kruhlak, M. J. et al. Regulation of global acetylation in mitosis through loss of histone acetyltransferases and deacetylases from chromatin. J. Biol. Chem. 276, 38307–38319 (2001).
Woodcock, C. L. & Ghosh, R. P. Chromatin higher-order structure and dynamics. Cold Spring Harb. Perspect. Biol. 2, a000596 (2010).
Phillips, R., Kondev, J. & Theriot, J. in Physical Biology of the Cell 1st edn 292–304 (Garland Science, 2009).
Chen, D. et al. Condensed mitotic chromatin is accessible to transcription factors and chromatin structural proteins. J. Cell Biol. 168, 41–54 (2005).
Ohta, S. et al. The protein composition of mitotic chromosomes determined using multiclassifier combinatorial proteomics. Cell 142, 810–821 (2010).
Martínez-Balbás, M. A., Dey, A., Rabindran, S. K., Ozato, K. & Wu, C. Displacement of sequence-specific transcription factors from mitotic chromatin. Cell 83, 29–38 (1995).
Kadauke, S. & Blobel, G. A. Mitotic bookmarking by transcription factors. Epigenet. Chromatin 6, 6 (2013).
Spencer, C. A., Kruhlak, M. J., Jenkins, H. L., Sun, X. & Bazett-Jones, D. P. Mitotic transcription repression in vivo in the absence of nucleosomal chromatin condensation. J. Cell Biol. 150, 13–26 (2000).
Niessen, H. E., Demmers, J. A. & Voncken, J. W. Talking to chromatin: post-translational modulation of Polycomb group function. Epigenet. Chromatin 2, 10 (2009).
Chen, S. et al. Cyclin-dependent kinases regulate epigenetic gene silencing through phosphorylation of EZH2. Nature Cell Biol. 12, 1108–1114 (2010).
Kaneko, S. et al. Phosphorylation of the PRC2 component Ezh2 is cell cycle-regulated and up-regulates its binding to ncRNA. Genes Dev. 24, 2615–2620 (2010).
Buchenau, P., Hodgson, J., Strutt, H. & Arndt-Jovin, D. J. The distribution of Polycomb-group proteins during cell division and development in Drosophila embryos: impact on models for silencing. J. Cell Biol. 141, 469–481 (1998).
Schwendemann, A. & Lehmann, M. Pipsqueak and GAGA factor act in concert as partners at homeotic and many other loci. Proc. Natl Acad. Sci. USA 99, 12883–12888 (2002).
Aoto, T., Saitoh, N., Sakamoto, Y., Watanabe, S. & Nakao, M. Polycomb group protein-associated chromatin is reproduced in post-mitotic G1 phase and is required for S phase progression. J. Biol. Chem. 283, 18905–18915 (2008).
Fanti, L. et al. The Trithorax group and Pc group proteins are differentially involved in heterochromatin formation in Drosophila. Chromosoma 117, 25–39 (2008).
Vincenz, C. & Kerppola, T. K. Different Polycomb group CBX family proteins associate with distinct regions of chromatin using nonhomologous protein sequences. Proc. Natl Acad. Sci. USA 105, 16572–16577 (2008).
Blobel, G. A. et al. A reconfigured pattern of MLL occupancy within mitotic chromatin promotes rapid transcriptional reactivation following mitotic exit. Mol. Cell 36, 970–983 (2009). Genome-wide location of the Trx homologue MLL on mitotic chromatin shows that it preferentially occupies genes that are highly transcribed during interphase, and that MLL is required for rapid transcription reactivation of these genes after mitotic exit.
Dey, A., Nishiyama, A., Karpova, T., McNally, J. & Ozato, K. Brd4 marks select genes on mitotic chromatin and directs postmitotic transcription. Mol. Biol. Cell 20, 4899–4909 (2009). Shows that the mammalian TrxG protein BRD4 preferentially binds to genes that are expressed at the M–G1 transition. By contrast, BRD4 does not bind to genes that are expressed at later phases of the cell cycle. Brd4 -knockdown causes impaired post-mitotic reactivation of these genes.
Zhao, R., Nakamura, T., Fu, Y., Lazar, Z. & Spector, D. L. Gene bookmarking accelerates the kinetics of post-mitotic transcriptional re-activation. Nature Cell Biol. 13, 1295–1304 (2011). Defines the real time kinetics of BRD4 binding to chromatin during mitosis using live imaging of a tagged locus. BRD4 is required to accelerate post-mitotic reactivation of the locus.
Fonseca, J. P. et al. In vivo Polycomb kinetics and mitotic chromatin binding distinguish stem cells from differentiated cells. Genes Dev. 26, 857–871 (2012). Defines mitotic and interphase chromatin binding parameters for the D. melanogaster Pc and Ph proteins, and reveals cell lineage-specific kinetics, using FRAP in combination with the quantification of endogenous protein molecule numbers in living animals.
Follmer, N. E., Wani, A. H. & Francis, N. J. A Polycomb group protein is retained at specific sites on chromatin in mitosis. PLoS Genet. 8, e1003135 (2013). First study to carry out ChIP analysis on a PcG protein in mitotic chromatin. Mitotic cells are isolated by elutriation, thus avoiding chemical treatments. Genome-wide analysis reveals retention of D. melanogaster Psc at a subset of binding sites from interphase to mitosis. Proposes that PcG proteins are stored at these sites during mitosis for rapid reoccupancy of targets during interphase.
Steffen, P. A. et al. Quantitative in vivo analysis of chromatin binding of Polycomb and Trithorax group proteins reveals retention of ASH1 on mitotic chromatin. Nucleic Acids Res. 41, 5235–5250 (2013). Defines mitotic and interphase chromatin binding parameters for several D. melanogaster PcG and TrxG proteins. Shows that the TrxG protein Ash1 binds robustly to mitotic chromatin in living animals using FRAP and fluorescence correlation spectroscopy (FCS) in combination with quantification of endogenous protein molecule numbers.
Laue, K. et al. The multidomain protein Brpf1 binds histones and is required for Hox gene expression and segmental identity. Development 135, 1935–1946 (2008).
Wang, F. & Higgins, J. M. G. Histone modifications and mitosis: countermarks, landmarks, and bookmarks. Trends Cell Biol. 23, 175–184 (2012).
Sawicka, A. & Seiser, C. Histone H3 phosphorylation — a versatile chromatin modification for different occasions. Biochimie 94, 2193–2201 (2012).
Varier, R. A. et al. A phospho/methyl switch at histone H3 regulates TFIID association with mitotic chromosomes. EMBO J. 29, 3967–3978 (2010).
Hirota, T., Lipp, J. J., Toh, B. H. & Peters, J. M. Histone H3 serine 10 phosphorylation by Aurora B causes HP1 dissociation from heterochromatin. Nature 438, 1176–1180 (2005).
Fischle, W. et al. Regulation of HP1–chromatin binding by histone H3 methylation and phosphorylation. Nature 438, 1116–1122 (2005).
Gehani, S. S. et al. Polycomb group protein displacement and gene activation through MSK-dependent H3K27me3S28 phosphorylation. Mol. Cell 39, 886–900 (2010).
Ng, R. K. & Gurdon, J. B. Epigenetic memory of active gene transcription is inherited through somatic cell nuclear transfer. Proc. Natl Acad. Sci. USA 102, 1957–1962 (2005).
Ng, R. K. & Gurdon, J. B. Epigenetic memory of an active gene state depends on histone H3.3 incorporation into chromatin in the absence of transcription. Nature Cell Biol. 10, 102–109 (2008).
Muramoto, T., Muller, I., Thomas, G., Melvin, A. & Chubb, J. R. Methylation of H3K4 is required for inheritance of active transcriptional states. Curr. Biol. 20, 397–406 (2010). This reference, together with references 143 and 144, uses histone point mutants to demonstrate a requirement for H3K4 (reference 144 and 146) or H3.3K4 (reference 143) in the epigenetic memory of active states.
Hodl, M. & Basler, K. Transcription in the absence of histone H3.2 and H3K4 methylation. Curr. Biol. 22, 2253–2257 (2012). Uses genetic techniques in D. melanogaster to create flies that carry clones of cells in which H3 is mutated at Lys4. These cells show little impairment of transcriptional upregulation after signalling, which demonstrates that H3K4 is not essential for transcriptional activation.
Hathaway, N. A. et al. Dynamics and memory of heterochromatin in living cells. Cell 149, 1447–1460 (2012). Uses chemical tethering to demonstrate that ectopic induction of H3K9 methylation is sufficient to establish a silenced domain and that the domain of trimethylated H3K9 is maintained over several cell divisions after withdrawal of the initiating signal.
Lempradl, A. & Ringrose, L. How does noncoding transcription regulate Hox genes? Bioessays 30, 110–121 (2008).
Hekimoglu, B. & Ringrose, L. Non-coding RNAs in Polycomb/Trithorax regulation. RNA Biol. 6, 129–137 (2009).
Rinn, J. L. & Chang, H. Y. Genome regulation by long noncoding RNAs. Annu. Rev. Biochem. 81, 145–166 (2012).
Michelotti, E. F., Sanford, S. & Levens, D. Marking of active genes on mitotic chromosomes. Nature 388, 895–899 (1997). Demonstrates that DNA conformation may contribute to epigenetic memory. Uses potassium permanganate sensitivity mapping to show that DNA at transcription start sites of genes, that are scheduled for reactivation, is conformationally distorted during mitosis. These conformations may be favourable to transcription after mitosis.
Juan, G., Pan, W. & Darzynkiewicz, Z. DNA segments sensitive to single-strand-specific nucleases are present in chromatin of mitotic cells. Exp. Cell Res. 227, 197–202 (1996).
John, S. & Workman, J. L. Bookmarking genes for activation in condensed mitotic chromosomes. BioEssays 20, 275–279 (1998).
Ringrose, L., Rehmsmeier, M., Dura, J. & Paro, R. Genome-wide prediction of Polycomb/Trithorax response elements in Drosophila melanogaster. Dev. Cell 5, 759–771 (2003).
Light, W. H. et al. A conserved role for human Nup98 in altering chromatin structure and promoting epigenetic transcriptional memory. PLoS Biol. 11, e1001524 (2013).
Laine, J. P., Singh, B. N., Krishnamurthy, S. & Hampsey, M. A physiological role for gene loops in yeast. Genes Dev. 23, 2604–2609 (2009).
Caravaca, J. M. et al. Bookmarking by specific and nonspecific binding of FoxA1 pioneer factor to mitotic chromosomes. Genes Dev. 27, 251–260 (2013). Shows that ∼15% of mammalian FOXA1 interphase target sites are bound in mitosis and that FOXA1 is required for rapid post-mitotic reactivation of these genes.
Kadauke, S. et al. Tissue-specific mitotic bookmarking by hematopoietic transcription factor GATA1. Cell 150, 725–737 (2012). Reveals that mitotically bound mammalian GATA1-target genes are preferentially and rapidly activated after mitosis. Mitosis-specific destruction of GATA1 by fused mitotic degron sequences delayed reactivation of genes that retain GATA1 during mitosis.
Yan, J. et al. Transcription factor binding in human cells occurs in dense clusters formed around cohesin anchor sites. Cell 154, 801–813 (2013). Defines a potential role for cohesin in marking sites of transcription factor binding for reassociation after replication and mitosis.
Strubbe, G. et al. Polycomb purification by in vivo biotinylation tagging reveals cohesin and Trithorax group proteins as interaction partners. Proc. Natl Acad. Sci. USA 108, 5572–5577 (2011).
Kellner, W. A. et al. Distinct isoforms of the Drosophila Brd4 homologue are present at enhancers, promoters and insulator sites. Nucleic Acids Res. 41, 9274–9283 (2013).
Schaaf, C. A. et al. Cohesin and Polycomb proteins functionally interact to control transcription at silenced and active genes. PLoS Genet. 9, e1003560 (2013).
Mishra, R. K. et al. The iab-7 Polycomb response element maps to a nucleosome-free region of chromatin and requires both GAGA and pleiohomeotic for silencing activity. Mol. Cell. Biol. 21, 1311–1318 (2001).
Mito, Y., Henikoff, J. G. & Henikoff, S. Histone replacement marks the boundaries of cis-regulatory domains. Science 315, 1408–1411 (2007).
Cao, R. et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298, 1039–1043 (2002).
Czermin, B. et al. Drosophila enhancer of Zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal Polycomb sites. Cell 111, 185–196 (2002).
Muller, J. et al. Histone methyltransferase activity of a Drosophila Polycomb group repressor complex. Cell 111, 197–208 (2002).
Kuzmichev, A., Nishioka, K., Erdjument-Bromage, H., Tempst, P. & Reinberg, D. Histone methyltransferase activity associated with a human multiprotein complex containing the Enhancer of Zeste protein. Genes Dev. 16, 2893–2905 (2002).
Scheuermann, J. C., Gutierrez, L. & Muller, J. Histone H2A monoubiquitination and Polycomb repression: the missing pieces of the puzzle. Fly 6, 162–168 (2012).
Tanaka, Y., Katagiri, Z., Kawahashi, K., Kioussis, D. & Kitajima, S. Trithorax-group protein ASH1 methylates histone H3 lysine 36. Gene 397, 161–168 (2007).
An, S., Yeo, K. J., Jeon, Y. H. & Song, J. J. Crystal structure of the human histone methyltransferase ASH1L catalytic domain and its implications for the regulatory mechanism. J. Biol. Chem. 286, 8369–8374 (2011).
Milne, T. A. et al. MLL targets SET domain methyltransferase activity to Hox gene promoters. Mol. Cell 10, 1107–1117 (2002).
Nakamura, T. et al. ALL-1 is a histone methyltransferase that assembles a supercomplex of proteins involved in transcriptional regulation. Mol. Cell 10, 1119–1128 (2002).
Smith, S. T. et al. Modulation of heat shock gene expression by the TAC1 chromatin-modifying complex. Nature Cell Biol. 6, 162–167 (2004).
Whitehouse, I. & Smith, D. J. Chromatin dynamics at the replication fork: there's more to life than histones. Curr. Opin. Genet. Dev. 23, 140–146 (2013).
Almouzni, G., Clark, D. J., Mechali, M. & Wolffe, A. P. Chromatin assembly on replicating DNA in vitro. Nucleic Acids Res. 18, 5767–5774 (1990).
Smith, S. & Stillman, B. Stepwise assembly of chromatin during DNA replication in vitro. EMBO J. 10, 971–980 (1991).
Steffen, P. A., Fonseca, J. P. & Ringrose, L. Epigenetics meets mathematics: Towards a quantitative understanding of chromatin biology. Bioessays 34, 901–913 (2012).
Acknowledgements
The authors apologize to colleagues whose publications were not cited owing to space limitations. They would like to thank M. Rehmsmeier and members of the Ringrose laboratory for discussions and feedback during the preparation of this manuscript. P.A.S. was funded by the EU FP6 Network of Excellence, 'The Epigenome'and the EU FP7 Network of Excellence 'Epigenesys'. The L.R. laboratory is supported by the Austrian Academy of Sciences. This article is dedicated to the memory of Audrey Ringrose (23.03.1931–01.02.2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
Molecular components of silent and active PREs and their potential for memory. (PDF 762 kb)
Glossary
- Epigenetic memory
-
The maintenance of active or silent gene expression states through cell generations, without a change in DNA sequence and in the absence of the initiating signals.
- Genome-wide profiling
-
Mapping of binding sites for proteins or sites of histone or DNA modification on a genome-wide scale. It uses tiling microarrays or high-throughput sequencing and is usually based on chromatin immunoprecipitation.
- Gal4–UAS system
-
(Gal4–upstream activating sequence system). A dual system for gene regulation, which is mostly used in flies. The UAS is placed upstream of the gene of interest and is only activated after expression of the yeast Gal4 protein, which is placed under the control of a promoter of choice.
- Bistable
-
A system that exhibits bistable behaviour has two extreme states that are stable, whereas intermediate states are unstable. Thus, the system can switch between these two stable states.
- Polycomb repressive complex 1
-
(PRC1). In flies, PRC1 contains Polycomb (Pc), Posterior Sex comb (Psc), Polyhomeotic (Ph), Sex combs extra (Sce) and accessory proteins. In vertebrates, PRC1 contains chromobox homologue (CBX) proteins, B lymphoma Mo-MLV insertion region 1 homologue (BMI1), PH, RING finger protein 1A (RING1A) or RING1B, and accessory proteins. Vertebrate PRC1 proteins are highly diverse.
- Polycomb repressive complex 2
-
(PRC2). In flies, PRC2 contains enhancer of zeste (E(z)), Suppressor of zeste 12 (SU(z)12), Extra sex combs (Esc) and Nucleosome-remodelling factor 55 kDa subunit (Nurf55; also known as Caf1). In vertebrates, PRC2 contains enhancer of zeste homologue 2 (EZH2), suppressor of zeste homologue 12 (SUZ12), embryonic ectoderm development (EED), Rb-associated protein 46 (RbAp46; also known as RBBP7), RbAp48 (also known as RBBP4) and Jumonji/ARID domain-containing protein 2 (JARID2).
- Histone Lys methylation
-
Covalent modification of specific Lys residues of histone proteins by addition of up to three methyl groups.
- Histone Lys acetylation
-
Covalent addition of an acetyl group to Lys residues of histones. Many Lys residues can be acetylated and, unlike methylation, acetylation changes the charge of the Lys. By masking the positive charge of Lys, acetylation reduces electrostatic interactions between DNA and histone tails, thus increasing accessibility.
- Histone Lys ubiquitylation
-
Covalent addition of an ubiquitin molecule to Lys residues of histones. The Polycomb repressive complex 1(PRC1) monoubiquitylates histone H2A at Lys118 (Lys119 in vertebrates). This adds a large (76 amino acid) molecule to the DNA–histone interface.
- FLP recombinase
-
A site-specific recombinase that is used to engineer specific DNA rearrangements in living organisms.
- Chromatin immunoprecipitation
-
(ChIP). Method for detecting sites of chromatin binding. Cells or tissues are crosslinked to preserve chromatin–protein interactions, and the protein of interest is immunoprecipitated with an antibody. Quantification of the precipitated DNA by PCR or profiling methods indicates enrichments of the protein of interest.
- Chromatin remodelers
-
Enzymes or complexes that alter nucleosomal positions, composition, conformation or presence using ATP hydrolysis, which enables changes in the accessibility of DNA to other molecules.
- CREB-binding protein
-
(CBP). Transcriptional activator that couples chromatin remodelling to transcription factor recognition. CBP and its close relative p300 have histone acetyltransferase activity.
- SET domain
-
(Su(var)3–9, E(z), Trx domain). A conserved 130–140 amino acid domain, which was initially characterized in the Drosophila melanogaster proteins Suppressor of variegation 3–9 (Su(var)3–9), Enhancer of zeste (E(z)) and Trithorax (Trx). These and other SET domains have histone methyltransferase activity with different specificities and may also methylate non-histone proteins.
- Nucleosomes
-
The basic packaging units of DNA. These consist of two copies each of histones H2A, H2B, H3 and H4 that are wrapped by 147 bp of DNA, which passes 1.67 times around the histone octamer.
- Bivalent chromatin
-
Bivalent chromatin contains histone modifications that are catalysed by both activators (trimethylated Lys4 of histone 3 (H3K4)) and repressors (trimethylated H3K27). It is thought to be 'poised' to become stably activated or repressed after differentiation when one of the marks is removed.
- Elutriation
-
A process for separating particles on the basis of their size, shape and density. Cell cultures can be fractionated into cells that are in G1, S phase, and G2 or M phase, which have increasingly large size. Elutriation bypasses the drug treatments that are required for cell synchronization.
- Chromodomain
-
(Chromatin organization modifier domain). A conserved domain of ∼60 amino acids, which was originally identified in Drosophila melanogaster proteins that modify variegation. Different chromodomains bind to specifically modified histone tails and/or RNA.
- H3–H4 tetramers
-
H3 and H4 histones are assembled into new chromatin as an H3–H4 tetramer.
- H2A–H2B dimers
-
H2A and H2B histones are assembled into new chromatin as two H2A–H2B dimers per nucleosome.
- Cdc2–cyclin B kinase
-
Cdc2 is a cyclin-dependent kinase which, as a heterodimer with cyclin B, is responsible for the execution of the M phase of the cell cycle.
- FRAP
-
(Fluorescene recovery after photobleaching). A method for determining kinetic parameters of fluorescently tagged proteins in living cells. The speed with which fluorescent proteins move into a region in which fluorescence has been bleached provides information on binding and diffusion behaviour.
- Residence time
-
Average time that a given molecule spends in an interaction. For proteins, residence time can be extracted from fluorescence recovery after photobleaching (FRAP) experiments and is inversely proportional to the dissociation rate.
- Heterochromatin protein 1
-
(HP1). An essential component of heterochromatin in fission yeast (Swi6), mammals (HP1) and Drosophila melanogaster (HP1). It contains a chromodomain (chromatin organization modifier domain) that specifically binds to methylated Lys9 of histone H3.
- Reprogramming by nuclear transfer
-
Somatic cell nuclei are introduced into enucleated cells of a different identity. This can erase epigenetic memory and alter the identity of the donor nucleus.
- H3.3
-
A variant of histone H3, which is expressed throughout the cell cycle and is incorporated into chromatin independently of replication, for example at sites of active transcription. H3.3 and H3 differ by only four amino acids.
Rights and permissions
About this article
Cite this article
Steffen, P., Ringrose, L. What are memories made of? How Polycomb and Trithorax proteins mediate epigenetic memory. Nat Rev Mol Cell Biol 15, 340–356 (2014). https://doi.org/10.1038/nrm3789
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3789
This article is cited by
-
RNA polymerase II associates with active genes during DNA replication
Nature (2023)
-
Regulation, functions and transmission of bivalent chromatin during mammalian development
Nature Reviews Molecular Cell Biology (2023)
-
Polycomb Repressive Complexes and Their Roles in Plant Developmental Programs, Particularly Floral Transition
Journal of Plant Biology (2023)
-
Maternal SMCHD1 regulates Hox gene expression and patterning in the mouse embryo
Nature Communications (2022)
-
Genomic and functional conservation of lncRNAs: lessons from flies
Mammalian Genome (2022)