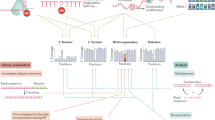
Key Points
-
Two recent independent studies demonstrated that N6-methyladenosine (m6A) is a widespread base modification in the mammalian transcriptome, which exhibits a unique enrichment near the stop codon and in the untranslated regions (UTRs) of mRNAs.
-
Recent studies have identified methyltransferase-like 3 (METTL3), METTL14 and Wilms tumour 1-associated protein (WTAP) as components of an m6A methyltransferase complex. Further characterization of this complex will be needed to understand the dynamics and specificity of adenosine methylation in various classes of cellular RNA.
-
FTO (fat mass and obesity-associated protein) and ALKBH5 (α-ketoglutarate-dependent dioxygenase alkB homologue 5) are the two m6A demethylating enzymes identified to date. Based on studies of the mRNA targeting specificity and tissue-specific expression patterns of these enzymes, however, it is likely that additional m6A demethylases exist.
-
m6A probably functions by recruiting m6A binding proteins, which influence RNA processing and regulation. Although a small number of m6A-binding proteins have been identified, much work remains to understand the full repertoire of m6A-binding proteins and how they contribute to mRNA regulation.
-
Although m6A probably has many functions, studies so far suggest that it has a role in splicing regulation and mRNA stability.
Abstract
N6-methyladenosine (m6A) is a modified base that has long been known to be present in non-coding RNAs, ribosomal RNA, polyadenylated RNA and at least one mammalian mRNA. However, our understanding of the prevalence of this modification has been fundamentally redefined by transcriptome-wide m6A mapping studies, which have shown that m6A is present in a large subset of the transcriptome in specific regions of mRNA. This suggests that mRNA may undergo post-transcriptional methylation to regulate its fate and function, which is analogous to methyl modifications in DNA. Thus, the pattern of methylation constitutes an mRNA 'epitranscriptome'. The identification of adenosine methyltransferases ('writers'), m6A demethylating enzymes ('erasers') and m6A-binding proteins ('readers') is helping to define cellular pathways for the post-transcriptional regulation of mRNAs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lewis, J. D. et al. Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69, 905–914 (1992).
Pawson, T. & Scott, J. D. Protein phosphorylation in signaling — 50 years and counting. Trends Biochem. Sci. 30, 286–290 (2005).
Meyer, K. D. et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell 149, 1635–1646 (2012). Provides the first demonstration that m6A is a widespread modification in mammalian mRNAs and reveals that m6A is highly enriched in sites surrounding stop codons and in UTRs. Also identifies many methylated ncRNAs, which were not previously known to contain m6A.
Dominissini, D. et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A–seq. Nature 485, 201–206 (2012). Demonstrates, together with reference 3, that m6A is a pervasive feature of the transcriptome, which exhibits a unique distribution within mRNAs. Identifies YTHDF2, YTFDF3 and HuR as potential m6A binding proteins.
Jia, G. et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nature Chem. Biol. 7, 885–887 (2011). Reveals that the obesity-associated protein FTO is capable of demethylating m6A residues in mRNA and points to the reversibility of this modification.
Zheng, G. et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol. Cell 49, 18–29 (2013).
Perry, R. P. & Kelley, D. E. Existence of methylated messenger RNA in mouse L cells. Cell 1, 37–42 (1974).
Desrosiers, R., Friderici, K. & Rottman, F. Identification of methylated nucleosides in messenger RNA from Novikoff hepatoma cells. Proc. Natl Acad. Sci. USA 71, 3971–3975 (1974).
Lavi, S. & Shatkin, A. J. Methylated simian virus 40-specific RNA from nuclei and cytoplasm of infected BSC-1 cells. Proc. Natl Acad. Sci. USA 72, 2012–2016 (1975).
Wei, C. M. & Moss, B. Methylated nucleotides block 5′-terminus of vaccinia virus messenger RNA. Proc. Natl Acad. Sci. USA 72, 318–322 (1975).
Furuichi, Y. et al. Methylated, blocked 5 termini in HeLa cell mRNA. Proc. Natl Acad. Sci. USA 72, 1904–1908 (1975).
Adams, J. M. & Cory, S. Modified nucleosides and bizarre 5′-termini in mouse myeloma mRNA. Nature 255, 28–33 (1975).
Dubin, D. T. & Taylor, R. H. The methylation state of poly A-containing messenger RNA from cultured hamster cells. Nucleic Acids Res. 2, 1653–1668 (1975).
Wei, C., Gershowitz, A. & Moss, B. N6, O2'-dimethyladenosine a novel methylated ribonucleoside next to the 5′ terminal of animal cell and virus mRNAs. Nature 257, 251–253 (1975).
Schmidt, W., Arnold, H. H. & Kersten, H. Biosynthetic pathway of ribothymidine in B. subtilis and M. lysodeikticus involving different coenzymes for transfer RNA and ribosomal RNA. Nucleic Acids Res. 2, 1043–1051 (1975).
Tanaka, T. & Weisblum, B. Systematic difference in the methylation of ribosomal ribonucleic acid from gram-positive and gram-negative bacteria. J. Bacteriol. 123, 771–774 (1975).
Munns, T. W., Sims, H. F. & Liszewski, M. K. Immunospecific retention of oligonucleotides possessing N6-methyladenosine and 7-methylguanosine. J. Biol. Chem. 252, 3102–3104 (1977).
Epstein, P., Reddy, R., Henning, D. & Busch, H. The nucleotide sequence of nuclear U6 (4.7 S) RNA. J. Biol. Chem. 255, 8901–8906 (1980).
Harada, F., Kato, N. & Nishimura, S. The nucleotide sequence of nuclear 4.8S RNA of mouse cells. Biochem. Biophys. Res. Commun. 95, 1332–1340 (1980).
Dimock, K. & Stoltzfus, C. M. Sequence specificity of internal methylation in B77 avian sarcoma virus RNA subunits. Biochemistry 16, 471–478 (1977).
Beemon, K. & Keith, J. Localization of N6-methyladenosine in the Rous sarcoma virus genome. J. Mol. Biol. 113, 165–179 (1977).
Furuichi, Y., Shatkin, A. J., Stavnezer, E. & Bishop, J. M. Blocked, methylated 5′-terminal sequence in avian sarcoma virus RNA. Nature 257, 618–620 (1975).
Sommer, S. et al. The methylation of adenovirus-specific nuclear and cytoplasmic RNA. Nucleic Acids Res. 3, 749–765 (1976).
Canaani, D., Kahana, C., Lavi, S. & Groner, Y. Identification and mapping of N6-methyladenosine containing sequences in simian virus 40 RNA. Nucleic Acids Res. 6, 2879–2899 (1979).
Perry, R. P., Kelley, D. E., Friderici, K. & Rottman, F. The methylated constituents of L cell messenger RNA: evidence for an unusual cluster at the 5′ terminus. Cell 4, 387–394 (1975).
Choi, Y. C. & Busch, H. Modified nucleotides in T1 RNase oligonucleotides of 18S ribosomal RNA of the Novikoff hepatoma. Biochemistry 17, 2551–2560 (1978).
Bringmann, P. & Luhrmann, R. Antibodies specific for N6-methyladenosine react with intact snRNPs U2 and U4/U6. FEBS Lett. 213, 309–315 (1987).
Shimba, S., Bokar, J. A., Rottman, F. & Reddy, R. Accurate and efficient N-6-adenosine methylation in spliceosomal U6 small nuclear RNA by HeLa cell extract in vitro. Nucleic Acids Res. 23, 2421–2426 (1995).
Perlman, S., Abelson, H. T. & Penman, S. Mitochondrial protein synthesis: RNA with the properties of eukaryotic messenger RNA. Proc. Natl Acad. Sci. USA 70, 350–353 (1973).
Nagaike, T., Suzuki, T. & Ueda, T. Polyadenylation in mammalian mitochondria: insights from recent studies. Biochim. Biophys. Acta 1779, 266–269 (2008).
Slomovic, S., Laufer, D., Geiger, D. & Schuster, G. Polyadenylation of ribosomal RNA in human cells. Nucleic Acids Res. 34, 2966–2975 (2006).
Salditt-Georgieff, M. et al. Methyl labeling of HeLa cell hnRNA: a comparison with mRNA. Cell 7, 227–237 (1976).
Okazaki, Y. et al. Analysis of the mouse transcriptome based on functional annotation of 60,770 full-length cDNAs. Nature 420, 563–573 (2002).
Numata, K. et al. Identification of putative noncoding RNAs among the RIKEN mouse full-length cDNA collection. Genome Res. 13, 1301–1306 (2003).
Ravasi, T. et al. Experimental validation of the regulated expression of large numbers of non-coding RNAs from the mouse genome. Genome Res. 16, 11–19 (2006).
Jacquier, A. The complex eukaryotic transcriptome: unexpected pervasive transcription and novel small RNAs. Nature Rev. Genet. 10, 833–844 (2009).
Csepany, T., Lin, A., Baldick, C. J. Jr & Beemon, K. Sequence specificity of mRNA N6-adenosine methyltransferase. J. Biol. Chem. 265, 20117–20122 (1990).
Kane, S. E. & Beemon, K. Inhibition of methylation at two internal N6-methyladenosine sites caused by GAC to GAU mutations. J. Biol. Chem. 262, 3422–3427 (1987).
Kane, S. E. & Beemon, K. Precise localization of m6A in Rous sarcoma virus RNA reveals clustering of methylation sites: implications for RNA processing. Mol. Cell. Biol. 5, 2298–2306 (1985).
Carroll, S. M., Narayan, P. & Rottman, F. M. N6-methyladenosine residues in an intron-specific region of prolactin pre-mRNA. Mol. Cell. Biol. 10, 4456–4465 (1990).
Horowitz, S., Horowitz, A., Nilsen, T. W., Munns, T. W. & Rottman, F. M. Mapping of N6-methyladenosine residues in bovine prolactin mRNA. Proc. Natl Acad. Sci. USA 81, 5667–5671 (1984).
Hess, M. E. et al. The fat mass and obesity associated gene (FTO) regulates activity of the dopaminergic midbrain circuitry. Nature Neurosci. 16, 1042–1048 (2013).
Saletore, Y. et al. The birth of the epitranscriptome: deciphering the function of RNA modifications. Genome Biol. 13, 175 (2012).
Grimson, A. et al. MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol. Cell 27, 91–105 (2007).
Baltz, A. G. et al. The mRNA-bound proteome and its global occupancy profile on protein-coding transcripts. Mol. Cell 46, 674–690 (2012).
Li, F. et al. Global analysis of RNA secondary structure in two metazoans. Cell Rep. 1, 69–82 (2012).
Elemento, O., Slonim, N. & Tavazoie, S. A universal framework for regulatory element discovery across all genomes and data types. Mol. Cell 28, 337–350 (2007).
Wei, C. M., Gershowitz, A. & Moss, B. 5′-terminal and internal methylated nucleotide sequences in HeLa cell mRNA. Biochemistry 15, 397–401 (1976).
Wei, C. M. & Moss, B. Nucleotide sequences at the N6-methyladenosine sites of HeLa cell messenger ribonucleic acid. Biochemistry 16, 1672–1676 (1977).
Schibler, U., Kelley, D. E. & Perry, R. P. Comparison of methylated sequences in messenger RNA and heterogeneous nuclear RNA from mouse L cells. J. Mol. Biol. 115, 695–714 (1977).
Harper, J. E., Miceli, S. M., Roberts, R. J. & Manley, J. L. Sequence specificity of the human mRNA N6-adenosine methylase in vitro. Nucleic Acids Res. 18, 5735–5741 (1990).
Bokar, J. A., Rath-Shambaugh, M. E., Ludwiczak, R., Narayan, P. & Rottman, F. Characterization and partial purification of mRNA N6-adenosine methyltransferase from HeLa cell nuclei. Internal mRNA methylation requires a multisubunit complex. J. Biol. Chem. 269, 17697–17704 (1994).
Martin, C. & Zhang, Y. The diverse functions of histone lysine methylation. Nature Rev. Mol. Cell Biol. 6, 838–849 (2005).
Bokar, J. A., Shambaugh, M. E., Polayes, D., Matera, A. G. & Rottman, F. M. Purification and cDNA cloning of the AdoMet-binding subunit of the human mRNA (N6-adenosine)-methyltransferase. RNA 3, 1233–1247 (1997). Identifies METTL3 as a key component of the m6A methyltransferase complex.
Rottman, F. M., Bokar, J. A., Narayan, P., Shambaugh, M. E. & Ludwiczak, R. N6-adenosine methylation in mRNA: substrate specificity and enzyme complexity. Biochimie 76, 1109–1114 (1994).
Narayan, P., Ludwiczak, R. L., Goodwin, E. C. & Rottman, F. M. Context effects on N6-adenosine methylation sites in prolactin mRNA. Nucleic Acids Res. 22, 419–426 (1994).
Zhong, S. et al. MTA is an Arabidopsis messenger RNA adenosine methylase and interacts with a homolog of a sex-specific splicing factor. Plant Cell 20, 1278–1288 (2008).
Clancy, M. J., Shambaugh, M. E., Timpte, C. S. & Bokar, J. A. Induction of sporulation in Saccharomyces cerevisiae leads to the formation of N6-methyladenosine in mRNA: a potential mechanism for the activity of the IME4 gene. Nucleic Acids Res. 30, 4509–4518 (2002).
Hongay, C. F. & Orr-Weaver, T. L. Drosophila inducer of meiosis 4 (IME4) is required for Notch signaling during oogenesis. Proc. Natl Acad. Sci. USA 108, 14855–14860 (2011).
Havugimana, P. C. et al. A census of human soluble protein complexes. Cell 150, 1068–1081 (2012).
Ping, X. L. et al. Mammalian WTAP is a regulatory subunit of the RNA N6-methyladenosine methyltransferase. Cell Res. 24, 177–189 (2014).
Liu, J. et al. A METTL3–METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nature Chem. Biol. 10, 93–95 (2014). Identifies, together with reference 61, WTAP as an additional component of the mammalian m6A mRNA methyltransferase complex.
Wang, Y. et al. N6-methyladenosine modification destabilizes developmental regulators in embryonic stem cells. Nature Cell Biol. 16, 191–198 (2014). Identifies, together with references 61 and 62, METTL14 as an adenosine methyltransferase and a component of the m6A mRNA methyltransferase complex. Also shows that m6A is required for stem cell differentiation in mouse embryonic stem cells.
Agarwala, S. D., Blitzblau, H. G., Hochwagen, A. & Fink, G. R. RNA methylation by the MIS complex regulates a cell fate decision in yeast. PLoS Genet. 8, e1002732 (2012).
Horiuchi, K. et al. Identification of Wilms' tumor 1-associating protein complex and its role in alternative splicing and the cell cycle. J. Biol. Chem. 288, 33292–33302 (2013).
Narayan, P. & Rottman, F. M. An in vitro system for accurate methylation of internal adenosine residues in messenger RNA. Science 242, 1159–1162 (1988).
Uhlen, M. et al. Towards a knowledge-based Human Protein Atlas. Nature Biotech. 28, 1248–1250 (2010).
Leach, R. A. & Tuck, M. T. Methionine depletion induces transcription of the mRNA (N6-adenosine)methyltransferase. Int. J. Biochem. Cell Biol. 33, 1116–1128 (2001).
Leach, R. A. & Tuck, M. T. Expression of the mRNA (N6-adenosine)-methyltransferase S-adenosyl-l-methionine binding subunit mRNA in cultured cells. Int. J. Biochem. Cell Biol. 33, 984–999 (2001).
Petry, S., Weixlbaumer, A. & Ramakrishnan, V. The termination of translation. Curr. Opin. Struct. Biol. 18, 70–77 (2008).
Czaplinski, K. et al. The surveillance complex interacts with the translation release factors to enhance termination and degrade aberrant mRNAs. Genes Dev. 12, 1665–1677 (1998).
Dahlberg, J. E. & Lund, E. Does protein synthesis occur in the nucleus? Curr. Opin. Cell Biol. 16, 335–338 (2004).
Bujnicki, J. M., Feder, M., Radlinska, M. & Blumenthal, R. M. Structure prediction and phylogenetic analysis of a functionally diverse family of proteins homologous to the MT-A70 subunit of the human mRNA: m6A methyltransferase. J. Mol. Evol. 55, 431–444 (2002).
Tuck, M. T. Partial purification of a 6-methyladenine mRNA methyltransferase which modifies internal adenine residues. Biochem. J. 288, 233–240 (1992).
Schwartz, S. et al. High-resolution mapping reveals a conserved, widespread, dynamic mRNA methylation program in yeast meiosis. Cell 155, 1409–1421 (2013).
Sanchez-Pulido, L. & Andrade-Navarro, M. A. The FTO (fat mass and obesity associated) gene codes for a novel member of the non-heme dioxygenase superfamily. BMC Biochem. 8, 23 (2007).
Gerken, T. et al. The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase. Science 318, 1469–1472 (2007).
Falnes, P. O., Johansen, R. F. & Seeberg, E. AlkB-mediated oxidative demethylation reverses DNA damage in Escherichia coli. Nature 419, 178–182 (2002).
Trewick, S. C., Henshaw, T. F., Hausinger, R. P., Lindahl, T. & Sedgwick, B. Oxidative demethylation by Escherichia coli AlkB directly reverts DNA base damage. Nature 419, 174–178 (2002).
Jia, G. et al. Oxidative demethylation of 3-methylthymine and 3-methyluracil in single-stranded DNA and RNA by mouse and human FTO. FEBS Lett. 582, 3313–3319 (2008).
Maden, B. E., Corbett, M. E., Heeney, P. A., Pugh, K. & Ajuh, P. M. Classical and novel approaches to the detection and localization of the numerous modified nucleotides in eukaryotic ribosomal RNA. Biochimie 77, 22–29 (1995).
Cheung, M. K., Gulati, P., O'Rahilly, S. & Yeo, G. S. FTO expression is regulated by availability of essential amino acids. Int. J. Obes. 37, 744–747 (2012).
Vujovic, P. et al. Fasting induced cytoplasmic Fto expression in some neurons of rat hypothalamus. PLoS ONE 8, e63694 (2013).
Ito, S. et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333, 1300–1303 (2011).
Chen, B. et al. Development of cell-active N6-methyladenosine RNA demethylase FTO inhibitor. J. Am. Chem. Soc. 134, 17963–17971 (2012).
Wang, X. et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature 505, 117–120 (2014). Shows that the YTHDF2 m6A-binding protein can destabilize target mRNAs by recruiting them to cellular mRNA decay sites.
Zhang, Z. et al. The YTH domain is a novel RNA binding domain. J. Biol. Chem. 285, 14701–14710 (2010).
Fu, Y. et al. FTO-mediated formation of N6-hydroxymethyladenosine and N6-formyladenosine in mammalian RNA. Nature Commun. 4, 1798 (2013).
Yang, X., Li, H., Huang, Y. & Liu, S. The dataset for protein-RNA binding affinity. Protein Sci. 22, 1808–1811 (2013).
Zhang, C. & Darnell, R. B. Mapping in vivo protein-RNA interactions at single-nucleotide resolution from HITS–CLIP data. Nature Biotech. 29, 607–614 (2011).
Konig, J. et al. iCLIP reveals the function of hnRNP particles in splicing at individual nucleotide resolution. Nature Struct. Mol. Biol. 17, 909–915 (2010).
Sugimoto, Y. et al. Analysis of CLIP and iCLIP methods for nucleotide-resolution studies of protein-RNA interactions. Genome Biol. 13, R67 (2012).
Botuyan, M. V. et al. Structural basis for the methylation state-specific recognition of histone H4-K20 by 53BP1 and Crb2 in DNA repair. Cell 127, 1361–1373 (2006).
Li, H. et al. Structural basis for lower lysine methylation state-specific readout by MBT repeats of L3MBTL1 and an engineered PHD finger. Mol. Cell 28, 677–691 (2007).
Kierzek, E. & Kierzek, R. The thermodynamic stability of RNA duplexes and hairpins containing N6-alkyladenosines and 2-methylthio-N6-alkyladenosines. Nucleic Acids Res. 31, 4472–4480 (2003).
Hibio, N., Hino, K., Shimizu, E., Nagata, Y. & Ui-Tei, K. Stability of miRNA 5′terminal and seed regions is correlated with experimentally observed miRNA-mediated silencing efficacy. Sci. Rep. 2, 996 (2012).
Brown, J. A., Valenstein, M. L., Yario, T. A., Tycowski, K. T. & Steitz, J. A. Formation of triple-helical structures by the 3′-end sequences of MALAT1 and MENβ noncoding RNAs. Proc. Natl Acad. Sci. USA 109, 19202–19207 (2012).
Heilman, K. L., Leach, R. A. & Tuck, M. T. Internal 6-methyladenine residues increase the in vitro translation efficiency of dihydrofolate reductase messenger RNA. Int. J. Biochem. Cell Biol. 28, 823–829 (1996).
Kariko, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Lewis, B. P., Burge, C. B. & Bartel, D. P. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120, 15–20 (2005).
Yeo, G. S. FTO and obesity: a problem for a billion people. J. Neuroendocrinol. 24, 393–394 (2012).
Garcia-Closas, M. et al. Genome-wide association studies identify four ER negative-specific breast cancer risk loci. Nature Genet. 45, 392–398 (2013).
Tuck, M. T., James, C. B., Kelder, B. & Kopchick, J. J. Elevation of internal 6-methyladenine mRNA methyltransferase activity after cellular transformation. Cancer Lett. 103, 107–113 (1996).
Bodi, Z., Button, J. D., Grierson, D. & Fray, R. G. Yeast targets for mRNA methylation. Nucleic Acids Res. 38, 5327–5335 (2010).
Loos, R. J. & Bouchard, C. FTO: the first gene contributing to common forms of human obesity. Obes Rev. 9, 246–250 (2008).
Fischer, J. et al. Inactivation of the FTO gene protects from obesity. Nature 458, 894–898 (2009).
Church, C. et al. Overexpression of FTO leads to increased food intake and results in obesity. Nature Genet. 42, 1086–1092 (2010).
Iles, M. M. et al. A variant in FTO shows association with melanoma risk not due to BMI. Nature Genet. 45, 428–432, 432e1 (2013).
Fustin, J.-M. et al. RNA-methylation-dependent RNA processing controls the speed of the circadian clock. Cell 155, 793–806 (2013). Demonstrates that inhibition of adenosine methylation alters the circadian period of target mRNAs and disrupts mRNA processing.
Bodi, Z. et al. Adenosine methylation in Arabidopsis mRNA is associated with the 3′ end and reduced levels cause developmental defects. Front. Plant Sci. 3, 48 (2012).
Gu, M. & Lima, C. D. Processing the message: structural insights into capping and decapping mRNA. Curr. Opin. Struct. Biol. 15, 99–106 (2005).
Muthukrishnan, S., Both, G. W., Furuichi, Y. & Shatkin, A. J. 5′-Terminal 7-methylguanosine in eukaryotic mRNA is required for translation. Nature 255, 33–37 (1975).
Shatkin, A. J. et al. 5′-Terminal caps, cap-binding proteins and eukaryotic mRNA function. Biochem. Soc. Symp. 47, 129–143 (1982).
Schibler, U. & Perry, R. P. The 5′-termini of heterogeneous nuclear RNA: a comparison among molecules of different sizes and ages. Nucleic Acids Res. 4, 4133–4149 (1977).
Ishikawa, M., Murai, R., Hagiwara, H., Hoshino, T. & Suyama, K. Preparation of eukaryotic mRNA having differently methylated adenosine at the 5′-terminus and the effect of the methyl group in translation. Nucleic Acids Symp. Ser. 129–130 (2009).
Marcotrigiano, J., Gingras, A. C., Sonenberg, N. & Burley, S. K. Cocrystal structure of the messenger RNA 5′ cap-binding protein (eIF4E) bound to 7-methyl-GDP. Cell 89, 951–961 (1997).
Kong, H. et al. Functional analysis of putative restriction-modification system genes in the Helicobacter pylori J99 genome. Nucleic Acids Res. 28, 3216–3223 (2000).
Dai, Q. et al. Identification of recognition residues for ligation-based detection and quantitation of pseudouridine and N6-methyladenosine. Nucleic Acids Res. 35, 6322–6329 (2007).
Harcourt, E. M., Ehrenschwender, T., Batista, P. J., Chang, H. Y. & Kool, E. T. Identification of a selective polymerase enables detection of N6-methyladenosine in RNA. J. Am. Chem. Soc. 135, 19079–19082 (2013).
Liu, N. et al. Probing N6-methyladenosine RNA modification status at single nucleotide resolution in mRNA and long noncoding RNA. RNA 19, 1848–1856 (2013).
Levene, M. J. et al. Zero-mode waveguides for single-molecule analysis at high concentrations. Science 299, 682–686 (2003).
Vilfan, I. D. et al. Analysis of RNA base modification and structural rearrangement by single-molecule real-time detection of reverse transcription. J. Nanobiotechnol. 11, 8 (2013).
Roberts, A., Trapnell, C., Donaghey, J., Rinn, J. L. & Pachter, L. Improving RNA–Seq expression estimates by correcting for fragment bias. Genome Biol. 12, R22 (2011).
Acknowledgements
The authors thank O. Elemento and members of the Jaffrey laboratory for helpful comments and suggestions. This work was supported by US National Institutes of Health (NIH) grant R01 DA037150 (S.R.J.) and the Revson Senior Fellowship in Biomedical Sciences to K.D.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
Internal mammalian mRNA base modifications (PDF 595 kb)
Glossary
- Non-coding RNAs
-
(ncRNAs). RNAs that are not translated into proteins. They include functional RNAs such as tRNAs, microRNAs and long ncRNAs.
- Ribosomal RNA
-
(rRNA). A highly abundant species of cellular RNA that functions in complex with ribosomes to mediate mRNA translation.
- Small nucleolar RNAs
-
(snoRNAs). This class of RNAs primarily functions to guide chemical modification of other RNAs, such as ribosomal RNAs, small nuclear RNAs and transfer RNAs.
- Zero-mode waveguides
-
Nanostructures with highly confined optical observation volumes.
- Nuclear speckles
-
Small, dynamic, subnuclear structures that are enriched in pre-mRNA splicing factors.
- Small nuclear RNAs
-
(snRNAs). RNAs that associate with specific proteins and that are frequently involved in pre-mRNA processing events such as splicing.
- Fe(II)- and 2-oxoglutarate dependent oxygenases
-
(Fe(II)/2-OG-dependent oxygenases). A family of proteins that catalyses cellular oxidative reactions, most notably hydroxylation. Several members of this family are involved in nucleic acid demethylation.
Rights and permissions
About this article
Cite this article
Meyer, K., Jaffrey, S. The dynamic epitranscriptome: N6-methyladenosine and gene expression control. Nat Rev Mol Cell Biol 15, 313–326 (2014). https://doi.org/10.1038/nrm3785
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3785
This article is cited by
-
Circular RNA-circPan3 attenuates cardiac hypertrophy via miR-320-3p/HSP20 axis
Cellular & Molecular Biology Letters (2024)
-
Sex differences in energy metabolism: natural selection, mechanisms and consequences
Nature Reviews Nephrology (2024)
-
The m6A demethylases FTO and ALKBH5 aggravate the malignant progression of nasopharyngeal carcinoma by coregulating ARHGAP35
Cell Death Discovery (2024)
-
Epitranscriptomic modifications in mesenchymal stem cell differentiation: advances, mechanistic insights, and beyond
Cell Death & Differentiation (2024)
-
METTL3-mediated m6A modification of lncRNA TSPAN12 promotes metastasis of hepatocellular carcinoma through SENP1-depentent deSUMOylation of EIF3I
Oncogene (2024)