Key Points
-
The reversible attachment of ubiquitin chains regulates the activity, localization and/or stability of a myriad of cellular proteins.
-
The formation of ubiquitin chains requires the sequential action of three types of enzymes: ubiquitin-activating enzymes (E1s), ubiquitin-conjugating enzymes (E2s) and ubiquitin ligases (E3s).
-
Humans express at least 38 E2 genes. E2s contain a highly conserved catalytic ubiquitin-conjugating (UBC) domain that interacts with E1s and E3s. Several E2s possess amino- or carboxy-terminal appendices that modulate their interaction with E3s or enable their association with E2 cofactors.
-
E2s are key regulators of ubiquitin chain assembly, and E2s with specific roles in ubiquitin chain initiation or elongation have been described. Some E2s catalyse both initiation and elongation with high specificity and efficiency.
-
E2s can determine the processivity of ubiquitin chain formation. They have evolved distinct strategies to increase the processivity of chain formation, including the recognition of substrate motifs or the preassembly of ubiquitin chains on their active sites.
-
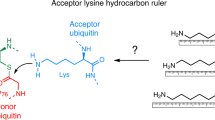
E2s are crucial regulators of ubiquitin chain topology. Most linkage-specific E2s bind to the acceptor ubiquitin in a non-covalent manner to orient a particular Lys residue relative to the E2 active site (charged with the donor ubiquitin).
Abstract
The modification of proteins with ubiquitin chains can change their localization, activity and/or stability. Although ubiquitylation requires the concerted action of ubiquitin-activating enzymes (E1s), ubiquitin-conjugating enzymes (E2s) and ubiquitin ligases (E3s), it is the E2s that have recently emerged as key mediators of chain assembly. These enzymes are able to govern the switch from ubiquitin chain initiation to elongation, regulate the processivity of chain formation and establish the topology of assembled chains, thereby determining the consequences of ubiquitylation for the modified proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kerscher, O., Felberbaum, R. & Hochstrasser, M. Modification of proteins by ubiquitin and ubiquitin-like proteins. Annu. Rev. Cell Dev. Biol. 22, 159–180 (2006).
Mukhopadhyay, D. & Riezman, H. Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science 315, 201–205 (2007).
Pickart, C. M. & Fushman, D. Polyubiquitin chains: polymeric protein signals. Curr. Opin. Chem. Biol. 8, 610–616 (2004).
Li, W. & Ye, Y. Polyubiquitin chains: functions, structures, and mechanisms. Cell. Mol. Life Sci. 65, 2397–2406 (2008).
Tokunaga, F. et al. Involvement of linear polyubiquitylation of NEMO in NF-κB activation. Nature Cell Biol. 11, 123–132 (2009).
Xu, P. et al. Quantitative proteomics reveals the function of unconventional ubiquitin chains in proteasomal degradation. Cell 137, 133–145 (2009). Demonstrates the existence and importance of non-canonical ubiquitin chains in vivo .
Kim, H. T. et al. Certain pairs of ubiquitin-conjugating enzymes (E2s) and ubiquitin-protein ligases (E3s) synthesize nondegradable forked ubiquitin chains containing all possible isopeptide linkages. J. Biol. Chem. 282, 17375–17386 (2007).
Kirkpatrick, D. S. et al. Quantitative analysis of in vitro ubiquitinated cyclin B1 reveals complex chain topology. Nature Cell Biol. 8, 700–710 (2006).
Ben-Saadon, R., Zaaroor, D., Ziv, T. & Ciechanover, A. The polycomb protein Ring1B generates self atypical mixed ubiquitin chains required for its in vitro histone H2A ligase activity. Mol. Cell 24, 701–711 (2006).
Dikic, I., Wakatsuki, S. & Walters, K. J. Ubiquitin binding domains — from structures to functions. Nature Rev. Mol. Cell Biol. 10, 659–671 (2009).
Hurley, J. H., Lee, S. & Prag, G. Ubiquitin-binding domains. Biochem. J. 399, 361–372 (2006).
Verma, R., Oania, R., Graumann, J. & Deshaies, R. J. Multiubiquitin chain receptors define a layer of substrate selectivity in the ubiquitin-proteasome system. Cell 118, 99–110 (2004).
Kim, I., Mi, K. & Rao, H. Multiple interactions of rad23 suggest a mechanism for ubiquitylated substrate delivery important in proteolysis. Mol. Biol. Cell 15, 3357–3365 (2004).
Richly, H. et al. A series of ubiquitin binding factors connects CDC48/p97 to substrate multiubiquitylation and proteasomal targeting. Cell 120, 73–84 (2005).
Chen, Z. J. Ubiquitin signalling in the NF-κB pathway. Nature Cell Biol. 7, 758–765 (2005).
Rahighi, S. et al. Specific recognition of linear ubiquitin chains by NEMO is important for NF-κB activation. Cell 136, 1098–1109 (2009).
Hoege, C., Pfander, B., Moldovan, G. L., Pyrowolakis, G. & Jentsch, S. RAD6-dependent DNA repair is linked to modification of PCNA by ubiquitin and SUMO. Nature 419, 135–141 (2002).
Sims, J. J. & Cohen, R. E. Linkage-specific avidity defines the lysine 63-linked polyubiquitin-binding preference of rap80. Mol. Cell 33, 775–783 (2009).
Schulman, B. A. & Harper, J. W. Ubiquitin-like protein activation by E1 enzymes: the apex for downstream signalling pathways. Nature Rev. Mol. Cell Biol. 10, 319–331 (2009).
Deshaies, R. J. & Joazeiro, C. A. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 78, 399–434 (2009).
Scheffner, M., Nuber, U. & Huibregtse, J. M. Protein ubiquitination involving an E1–E2–E3 enzyme ubiquitin thioester cascade. Nature 373, 81–83 (1995).
Michelle, C., Voure'h, P., Mignon, L. & Andres, C. R. What was the set of ubiquitin and ubiquitin-like conjugating enzymes in the eukaryote common ancestor? J. Mol. Evol. 68, 616–628 (2009).
Lin, Y., Hwang, W. C. & Basavappa, R. Structural and functional analysis of the human mitotic-specific ubiquitin-conjugating enzyme, UbcH10. J. Biol. Chem. 277, 21913–21921 (2002).
Ozkan, E., Yu, H. & Deisenhofer, J. Mechanistic insight into the allosteric activation of a ubiquitin-conjugating enzyme by RING-type ubiquitin ligases. Proc. Natl Acad. Sci. USA 102, 18890–18895 (2005). Suggests that RING E3s mediate the allosteric activation of E2s.
Eddins, M. J., Carlile, C. M., Gomez, K. M., Pickart, C. M. & Wolberger, C. Mms2–Ubc13 covalently bound to ubiquitin reveals the structural basis of linkage-specific polyubiquitin chain formation. Nature Struct. Mol. Biol. 13, 915–920 (2006). Together with reference 58, reveals the mechanism of linkage-specific ubiquitin chain formation, in this case for Lys63-linked chains.
Haas, A. L., Bright, P. M. & Jackson, V. E. Functional diversity among putative E2 isozymes in the mechanism of ubiquitin-histone ligation. J. Biol. Chem. 263, 13268–13275 (1988).
Lois, L. M. & Lima, C. D. Structures of the SUMO E1 provide mechanistic insights into SUMO activation and E2 recruitment to E1. EMBO J. 24, 439–451 (2005).
Huang, D. T. et al. Basis for a ubiquitin-like protein thioester switch toggling E1–E2 affinity. Nature 445, 394–398 (2007). An elegant structural study that defines the molecular basis underlying the communication between E1s and E2s.
Lee, I. & Schindelin, H. Structural insights into E1-catalyzed ubiquitin activation and transfer to conjugating enzymes. Cell 134, 268–278 (2008).
Jin, J., Li, X., Gygi, S. P. & Harper, J. W. Dual E1 activation systems for ubiquitin differentially regulate E2 enzyme charging. Nature 447, 1135–1138 (2007).
Huang, D. T. et al. A unique E1–E2 interaction required for optimal conjugation of the ubiquitin-like protein NEDD8. Nature Struct. Mol. Biol. 11, 927–935 (2004).
Huang, D. T., Zhuang, M., Ayrault, O. & Schulman, B. A. Identification of conjugation specificity determinants unmasks vestigial preference for ubiquitin within the NEDD8 E2. Nature Struct. Mol. Biol. 15, 280–287 (2008). Provides striking insight into the role of N- or C-terminal appendices of E2s in determining E2 specificity.
Rape, M. & Kirschner, M. W. Autonomous regulation of the anaphase-promoting complex couples mitosis to S-phase entry. Nature 432, 588–595 (2004).
Summers, M. K., Pan, B., Mukhyala, K. & Jackson, P. K. The unique N terminus of the UbcH10 E2 enzyme controls the threshold for APC activation and enhances checkpoint regulation of the APC. Mol. Cell 31, 544–556 (2008).
Skowyra, D., Craig, K. L., Tyers, M., Elledge, S. J. & Harper, J. W. F-box proteins are receptors that recruit phosphorylated substrates to the SCF ubiquitin-ligase complex. Cell 91, 209–219 (1997).
Williamson, A. et al. Identification of a physiological E2 module for the human anaphase promoting complex. Proc. Natl Acad. Sci. USA 12 Oct 2009 (doi: 10.1073/pnas.0907887106). Defines the molecular mechanism underlying the formation of Lys11-linked ubiquitin chains by APC/C.
Deng, L. et al. Activation of the IκB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 103, 351–361 (2000).
Fang, S. et al. The tumor autocrine motility factor receptor, gp78, is a ubiquitin protein ligase implicated in degradation from the endoplasmic reticulum. Proc. Natl Acad. Sci. USA 98, 14422–14427 (2001).
Chen, B. et al. The activity of a human endoplasmic reticulum-associated degradation E3, gp78, requires its Cue domain, RING finger, and an E2-binding site. Proc. Natl Acad. Sci. USA 103, 341–346 (2006).
Bailly, V., Lamb, J., Sung, P., Prakash, S. & Prakash, L. Specific complex formation between yeast RAD6 and RAD18 proteins: a potential mechanism for targeting RAD6 ubiquitin-conjugating activity to DNA damage sites. Genes Dev. 8, 811–820 (1994).
Xin, H. et al. The human RAD18 gene product interacts with HHR6A and HHR6B. Nucleic Acids Res. 28, 2847–2854 (2000).
Machida, Y. J. et al. UBE2T is the E2 in the Fanconi anemia pathway and undergoes negative autoregulation. Mol. Cell 23, 589–96 (2006).
Alpi, A. F., Pace, P. E., Babu, M. M. & Patel, K. J. Mechanistic insight into site-restricted monoubiquitination of FANCD2 by Ube2t, FANCL, and FANCI. Mol. Cell 32, 767–777 (2008).
Zheng, N., Wang, P., Jeffrey, P. D. & Pavletich, N. P. Structure of a c-Cbl-UbcH7 complex: RING domain function in ubiquitin-protein ligases. Cell 102, 533–539 (2000). First structural study to illustrate the interaction of an E2 with a RING E3.
Dominguez, C. et al. Structural model of the UbcH5B/CNOT4 complex revealed by combining NMR, mutagenesis, and docking approaches. Structure 12, 633–644 (2004).
Zhang, M. et al. Chaperoned ubiquitylation — crystal structures of the CHIP U box E3 ubiquitin ligase and a CHIP-Ubc13-Uev1a complex. Mol. Cell 20, 525–538 (2005).
Yin, Q. et al. E2 interaction and dimerization in the crystal structure of TRAF6. Nature Struct. Mol. Biol. 16, 658–666 (2009).
Bailly, V., Prakash, S. & Prakash, L. Domains required for dimerization of yeast Rad6 ubiquitin-conjugating enzyme and Rad18 DNA binding protein. Mol. Cell Biol. 17, 4536–4543 (1997).
Kolman, C. J., Toth, J. & Gonda, D. K. Identification of a portable determinant of cell cycle function within the carboxyl-terminal domain of the yeast CDC34 (UBC3) ubiquitin conjugating (E2) enzyme. EMBO J. 11, 3081–3090 (1992).
Silver, E. T., Gwozd, T. J., Ptak, C., Goebl, M. & Ellison, M. J. A chimeric ubiquitin conjugating enzyme that combines the cell cycle properties of CDC34 (UBC3) and the DNA repair properties of RAD6 (UBC2): implications for the structure, function and evolution of the E2s. EMBO J. 11, 3091–3098 (1992).
Li, W. et al. Mechanistic insights into active site-associated polyubiquitination by the ubiquitin-conjugating enzyme Ube2g2. Proc. Natl Acad. Sci. USA 106, 3722–3727 (2009).
Das, R. et al. Allosteric activation of E2-RING finger-mediated ubiquitylation by a structurally defined specific E2-binding region of gp78. Mol. Cell 34, 674–685 (2009).
Petroski, M. D. & Deshaies, R. J. Mechanism of lysine 48-linked ubiquitin-chain synthesis by the cullin-RING ubiquitin-ligase complex SCF-Cdc34. Cell 123, 1107–1120 (2005). A thorough dissection of ubiquitin chain formation by Cdc34 that led to the concept that chain initiation and elongation are carried out by distinct mechanisms.
Wu, P. Y. et al. A conserved catalytic residue in the ubiquitin-conjugating enzyme family. EMBO J. 22, 5241–5250 (2003). A biochemical study providing crucial insight into the mechanism of E2-dependent ubiquitin transfer.
Huang, D. T. et al. Structural basis for recruitment of Ubc12 by an E2 binding domain in NEDD8's E1. Mol. Cell 17, 341–350 (2005).
Eletr, Z. M., Huang, D. T., Duda, D. M., Schulman, B. A. & Kuhlman, B. E2 conjugating enzymes must disengage from their E1 enzymes before E3-dependent ubiquitin and ubiquitin-like transfer. Nature Struct. Mol. Biol. 12, 933–934 (2005).
Soucy, T. A. et al. An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature 458, 732–736 (2009).
VanDemark, A. P., Hofmann, R. M., Tsui, C., Pickart, C. M. & Wolberger, C. Molecular insights into polyubiquitin chain assembly: crystal structure of the Mms2/Ubc13 heterodimer. Cell 105, 711–720 (2001).
Biederer, T., Volkwein, C. & Sommer, T. Role of Cue1p in ubiquitination and degradation at the ER surface. Science 278, 1806–1809 (1997).
Kostova, Z., Mariano, J., Scholz, S., Koenig, C. & Weissman, A. M. A Ubc7p-binding domain in Cue1p activates ER-associated protein degradation. J. Cell Sci. 122, 1374–1381 (2009).
Bazirgan, O. A. & Hampton, R. Y. Cue1p is an activator of Ubc7p E2 activity in vitro and in vivo. J. Biol. Chem. 283, 12797–12810 (2008).
Ravid, T. & Hochstrasser, M. Autoregulation of an E2 enzyme by ubiquitin-chain assembly on its catalytic residue. Nature Cell Biol. 9, 422–427 (2007).
Polo, S. et al. A single motif responsible for ubiquitin recognition and monoubiquitination in endocytic proteins. Nature 416, 451–455 (2002).
Hoeller, D. et al. E3-independent monoubiquitination of ubiquitin-binding proteins. Mol. Cell 26, 891–898 (2007).
Plafker, S. M., Plafker, K. S., Weissman, A. M. & Macara, I. G. Ubiquitin charging of human class III ubiquitin-conjugating enzymes triggers their nuclear import. J. Cell Biol. 167, 649–659 (2004).
Rodrigo-Brenni, M. C. & Morgan, D. O. Sequential E2s drive polyubiquitin chain assembly on APC targets. Cell 130, 127–139 (2007). An elegant study demonstrating the role of distinct ubiquitin chain-initiating E2s and ubiquitin chain-elongating E2s for the yeast APC/C.
Jin, L., Williamson, A., Banerjee, S., Philipp, I. & Rape, M. Mechanism of ubiquitin-chain formation by the human anaphase-promoting complex. Cell 133, 653–665 (2008). Identifies the first role for Lys11-linked ubiquitin chains in controlling cell cycle progression and provides a mechanism by which an E2 and an E3 can cooperate in efficient ubiquitin chain initiation.
Christensen, D. E., Brzovic, P. S. & Klevit, R. E. E2-BRCA1 RING interactions dictate synthesis of mono- or specific polyubiquitin chain linkages. Nature Struct. Mol. Biol. 14, 941–948 (2007).
Petroski, M. D. et al. Substrate modification with lysine 63-linked ubiquitin chains through the UBC13-UEV1A ubiquitin-conjugating enzyme. J. Biol. Chem. 282, 29936–29945 (2007).
Windheim, M., Peggie, M. & Cohen, P. Two different classes of E2 ubiquitin-conjugating enzymes are required for the mono-ubiquitination of proteins and elongation by polyubiquitin chains with a specific topology. Biochem. J. 409, 723–729 (2008).
Hofmann, R. M. & Pickart, C. M. Noncanonical MMS2-encoded ubiquitin-conjugating enzyme functions in assembly of novel polyubiquitin chains for DNA repair. Cell 96, 645–653 (1999).
Haldeman, M. T., Xia, G., Kasperek, E. M. & Pickart, C. M. Structure and function of ubiquitin conjugating enzyme E2–25K: the tail is a core-dependent activity element. Biochemistry 36, 10526–10537 (1997).
Rape, M., Reddy, S. K. & Kirschner, M. W. The processivity of multiubiquitination by the APC determines the order of substrate degradation. Cell 124, 89–103 (2006).
Verma, R., Feldman, R. M. & Deshaies, R. J. SIC1 is ubiquitinated in vitro by a pathway that requires CDC4, CDC34, and cyclin/CDK activities. Mol. Biol. Cell 8, 1427–1437 (1997).
Saha, A. & Deshaies, R. J. Multimodal activation of the ubiquitin ligase SCF by Nedd8 conjugation. Mol. Cell 32, 21–31 (2008).
Duda, D. M. et al. Structural insights into NEDD8 activation of cullin-RING ligases: conformational control of conjugation. Cell 134, 995–1006 (2008).
Gazdoiu, S., Yamoah, K., Wu, K. & Pan, Z. Q. Human Cdc34 employs distinct sites to coordinate attachment of ubiquitin to a substrate and assembly of polyubiquitin chains. Mol. Cell Biol. 27, 7041–7052 (2007).
Thrower, J. S., Hoffman, L., Rechsteiner, M. & Pickart, C. M. Recognition of the polyubiquitin proteolytic signal. EMBO J. 19, 94–102 (2000).
Komander, D., Clague, M. J. & Urbe, S. Breaking the chains: structure and function of the deubiquitinases. Nature Rev. Mol. Cell Biol. 10, 550–563 (2009).
Hochstrasser, M. Lingering mysteries of ubiquitin-chain assembly. Cell 124, 27–34 (2006).
Baer, R. & Ludwig, T. The BRCA1/BARD1 heterodimer, a tumor suppressor complex with ubiquitin E3 ligase activity. Curr. Opin. Genet. Dev. 12, 86–91 (2002).
Joukov, V. et al. The BRCA1/BARD1 heterodimer modulates ran-dependent mitotic spindle assembly. Cell 127, 539–552 (2006).
Brzovic, P. S., Lissounov, A., Christensen, D. E., Hoyt, D. W. & Klevit, R. E. A UbcH5/ubiquitin noncovalent complex is required for processive BRCA1-directed ubiquitination. Mol. Cell 21, 873–880 (2006).
Li, W., Tu, D., Brunger, A. T. & Ye, Y. A ubiquitin ligase transfers preformed polyubiquitin chains from a conjugating enzyme to a substrate. Nature 446, 333–337 (2007). Together with references 51 and 62, shows that ubiquitin chains can be preassembled on a catalytic Cys of E2 and subsequently be transferred en bloc to a substrate protein.
Cao, J. et al. Ufd1 is a cofactor of gp78 and plays a key role in cholesterol metabolism by regulating the stability of HMG-CoA reductase. Cell Metab. 6, 115–128 (2007).
Chen, Z. & Pickart, C. M. A 25-kilodalton ubiquitin carrier protein (E2) catalyzes multi-ubiquitin chain synthesis via lysine 48 of ubiquitin. J. Biol. Chem. 265, 21835–21842 (1990).
Van Nocker, S. & Vierstra, R. D. Cloning and characterization of a 20-kDa ubiquitin carrier protein from wheat that catalyzes multiubiquitin chain formation in vitro. Proc. Natl Acad. Sci. USA 88, 10297–10301 (1991).
Haas, A. L., Reback, P. B. & Chau, V. Ubiquitin conjugation by the yeast RAD6 and CDC34 gene products. Comparison to their putative rabbit homologs, E220K and E232K. J. Biol. Chem. 266, 5104–5112 (1991).
Merkley, N. & Shaw, G. S. Solution structure of the flexible class II ubiquitin-conjugating enzyme Ubc1 provides insights for polyubiquitin chain assembly. J. Biol. Chem. 279, 47139–47147 (2004).
Wang, M., Cheng, D., Peng, J. & Pickart, C. M. Molecular determinants of polyubiquitin linkage selection by an HECT ubiquitin ligase. EMBO J. 25, 1710–1719 (2006).
Martinez-Noel, G., Muller, U. & Harbers, K. Identification of molecular determinants required for interaction of ubiquitin-conjugating enzymes and RING finger proteins. Eur. J. Biochem. 268, 5912–5919 (2001).
Markson, G. et al. Analysis of the human E2 ubiquitin conjugating enzyme protein interaction network. Genome Res. Jun 23 2009 (doi: 10.1101/gr.093963.109).
Gazdoiu, S. et al. Proximity-induced activation of human Cdc34 through heterologous dimerization. Proc. Natl Acad. Sci. USA 102, 15053–15058 (2005).
Adams, J. The development of proteasome inhibitors as anticancer drugs. Cancer Cell 5, 417–421 (2004).
Yang, Y. et al. Inhibitors of ubiquitin-activating enzyme (E1), a new class of potential cancer therapeutics. Cancer Res. 67, 9472–9481 (2007).
Acknowledgements
We thank the members of our laboratories for many stimulating discussions. We are grateful to J. Schaletzky for discussions and critically reading the manuscript and C. Wolberger (Johns Hopkins University, Maryland, USA) for providing the coordinates of the Mms2-bound ubiquitin. The work in our laboratories is funded by a National Institutes of Health Director's New Innovator Award (M.R.), RO1 5R01GM083064-02 (M.R.), a March of Dimes grant (M.R.), and the intramural research programme of the National Institute of Diabetes and Digestive and Kidney Diseases, NIH (Y.Y.). M.R. is a Pew Scholar.
Author information
Authors and Affiliations
Supplementary information
41580_2009_BFnrm2780_MOESM1_ESM.pdf
Supplementary information S1 (table) | A comprehensive list of the known human E2 enzymes and their key features. (PDF 246 kb)
Related links
Glossary
- Ubiquitin-interacting motif
-
A small motif that mediates the interaction of a protein with the hydrophobic patch of ubiquitin around Ile44.
- Ubiquitin-associated (UBA) domain
-
A protein domain that forms a three-helix bundle and interacts with hydrophobic regions of ubiquitin.
- 26S proteasome
-
A multisubunit protease that degrades proteins with attached ubiquitin chains. It contains a barrel-like 20S proteolytic core particle that houses the active sites and a 19S regulatory particle that governs substrate recognition and entry into the 20S core particle.
- HECT domain
-
A domain of ∼40 kDa (350 amino acids) that is found at the C terminus of HECT E3s. It contains a catalytic Cys residue that accepts ubiquitin from an E2 to form a ubiquitin thioester intermediate before transferring the ubiquitin to substrates.
- RING domain
-
A domain that is present in most E3s and is defined by the consensus sequence CX2CX(9–39)CX(1–3)HX(2–3)C/HX2CX(4–48)CX2C (where X means any amino acid). It coordinates two structural zinc cations.
- Ubiquitin-conjugating (UBC) domain
-
A conserved core domain of ∼150 residues that is found in all E2s, including those for UBLs. It contains the catalytic Cys residue of E2s.
- 310-helix
-
A type of secondary protein structure in which the amino acids are in a right-handed helical arrangement. The hydrogen bonds are formed between the NH group of an amino acid and the CO group of the amino acid three residues earlier (as opposed to four residues earlier in an α-helix).
- Ubiquitin fold domain
-
A domain found in E1s that mediates binding to an E2 and forms a similar structure to ubiquitin.
- TEK box
-
A Lys-rich region in APC/C substrates downstream of initial APC/C recognition sites (such as the D box or KEN box), which promotes initiation of ubiquitin chain formation.
- D box
-
The amino acid sequence RXXL(X)nN (where X means any amino acid), which mediates binding of an APC/C substrate to the co-activators Cdc20 and Cdh1 and potentially also to subunits of the core APC/C.
- KEN box
-
The amino acid sequence KEN(X)nP (where X means any amino acid), which mediates binding of APC/C substrates to the co-activator Cdh1.
Rights and permissions
About this article
Cite this article
Ye, Y., Rape, M. Building ubiquitin chains: E2 enzymes at work. Nat Rev Mol Cell Biol 10, 755–764 (2009). https://doi.org/10.1038/nrm2780
Issue Date:
DOI: https://doi.org/10.1038/nrm2780
This article is cited by
-
Structure-guided engineering enables E3 ligase-free and versatile protein ubiquitination via UBE2E1
Nature Communications (2024)
-
UBE2J1 is the E2 ubiquitin-conjugating enzyme regulating androgen receptor degradation and antiandrogen resistance
Oncogene (2024)
-
Ubiquitination Process Mediates Prostate Cancer Development and Metastasis through Multiple Mechanisms
Cell Biochemistry and Biophysics (2024)
-
Exploiting E3 ubiquitin ligases to reeducate the tumor microenvironment for cancer therapy
Experimental Hematology & Oncology (2023)
-
Identification of transcriptome characteristics of granulosa cells and the possible role of UBE2C in the pathogenesis of premature ovarian insufficiency
Journal of Ovarian Research (2023)