Key Points
-
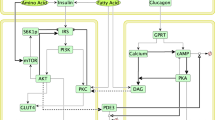
Cell-signalling networks are complex systems that allow for the specific biological response to a defined stimulus. We argue that it is possible to qualitatively identify the important mediators of any ligand–receptor system through a method that conceptualizes the most important genes as critical nodes.
-
A 'critical node' is defined as a group of related proteins (for example, gene isoforms) that are essential for the receptor-mediated signal, and in which two or more of these related proteins might have unique biological roles within a signalling network and therefore serve as a source of divergence within the signalling system. The node is highly regulated, both positively and negatively, and is a junction for potential crosstalk with other signalling systems.
-
Here, we use the insulin-signalling pathway as a model system. Three groups of genes are used as examples of 'critical nodes': the insulin receptor and its substrates (IR/IRS), phosphatidylinositol 3-kinase (PI3K) and AKT/protein kinase B (PKB). Although many proteins could be considered to be significant players in insulin signalling, only these three have attained sufficient in vivo and in vitro evidence to be considered 'critical nodes'.
-
The IRSs are a critical node because six isoforms exist, with most of insulin's signal being mediated by IRS1 and IRS2. These IRS proteins, and others, have been shown, through knockout and RNAi studies, to have different biological functions in vivo. These proteins are regulated, both positively by the insulin receptor, and negatively through serine phosphorylation and downregulation of protein levels. The IRS proteins also mediate crosstalk with other signalling systems, particularly with the inflammation pathways.
-
PI3K is also a critical node because both of its subunits, p85 and p110, have many different isoforms, many of which have unique biological functions. The p85α isoform is of particular interest because it negatively regulates insulin action independently of its function in the PI3K holoenzyme. This p85α subunit might also mediate crosstalk with the inflammatory pathways through the activation of c-Jun N-terminal kinase (JNK).
-
AKT/PKB is a critical node because it has been shown to be crucial for glucose homeostasis. AKT comes in three isoforms, and is subject to considerable positive and negative regulation, both by other kinases and by proteins that bind to and inhibit AKT. Many of insulin's downstream functions are mediated by AKT, including the activation of glycogen synthesis, the suppression of gluconeogenesis and the regulation of cell growth and cell size.
Abstract
Physiologically important cell-signalling networks are complex, and contain several points of regulation, signal divergence and crosstalk with other signalling cascades. Here, we use the concept of 'critical nodes' to define the important junctions in these pathways and illustrate their unique role using insulin signalling as a model system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Avruch, J. Insulin signal transduction through protein kinase cascades. Mol. Cell Biochem. 182, 31–48 (1998).
Ullrich, A. & Schlessinger, J. Signal transduction by receptors with tyrosine kinase activity. Cell 61, 203–212 (1990).
Elchebly, M. et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 283, 1544–1548 (1999).
Ueki, K., Kondo, T. & Kahn, C. R. Suppressor of cytokine signaling 1 (SOCS-1) and SOCS-3 cause insulin resistance through inhibition of tyrosine phosphorylation of insulin receptor substrate proteins by discrete mechanisms. Mol. Cell Biol. 24, 5434–5446 (2004).
Emanuelli, B. et al. SOCS-3 inhibits insulin signaling and is up-regulated in response to tumor necrosis factor-α in the adipose tissue of obese mice. J. Biol. Chem. 276, 47944–47949 (2001).
Friedman, J. E. et al. Reduced insulin receptor signaling in the obese spontaneously hypertensive Koletsky rat. Am. J. Physiol. 273, E1014–E1023 (1997).
Sun, X. J. et al. Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein. Nature 352, 73–77 (1991).
Sun, X. J. et al. Role of IRS-2 in insulin and cytokine signalling. Nature 377, 173–177 (1995).
Lavan, B. E., Lane, W. S. & Lienhard, G. E. The 60-kDa phosphotyrosine protein in insulin-treated adipocytes is a new member of the insulin receptor substrate family. J. Biol. Chem. 272, 11439–11443 (1997).
Fantin, V. R. et al. Characterization of insulin receptor substrate 4 in human embryonic kidney 293 cells. J. Biol. Chem. 273, 10726–10732 (1998).
Cai, D., Dhe-Paganon, S., Melendez, P. A., Lee, J. & Shoelson, S. E. Two new substrates in insulin signaling, IRS5/DOK4 and IRS6/DOK5. J. Biol. Chem. 278, 25323–25330 (2003).
Lehr, S. et al. Identification of major tyrosine phosphorylation sites in the human insulin receptor substrate Gab-1 by insulin receptor kinase in vitro. Biochemistry 39, 10898–10907 (2000).
Baumann, C. A. et al. CAP defines a second signalling pathway required for insulin-stimulated glucose transport. Nature 407, 202–207 (2000).
Gustafson, T. A., He, W., Craparo, A., Schaub, C. D. & O'Neill, T. J. Phosphotyrosine-dependent interaction of SHC and insulin receptor substrate 1 with the NPEY motif of the insulin receptor via a novel non-SH2 domain. Mol. Cell Biol. 15, 2500–2508 (1995).
Virkamaki, A., Ueki, K. & Kahn, C. R. Protein–protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. J. Clin. Invest. 103, 931–943 (1999).
Myers, M. G. Jr et al. The COOH-terminal tyrosine phosphorylation sites on IRS-1 bind SHP-2 and negatively regulate insulin signaling. J. Biol. Chem. 273, 26908–26914 (1998).
Algenstaedt, P., Antonetti, D. A., Yaffe, M. B. & Kahn, C. R. Insulin receptor substrate proteins create a link between the tyrosine phosphorylation cascade and the Ca2+-ATPases in muscle and heart. J. Biol. Chem. 272, 23696–23702 (1997).
Fei, Z. L., D'Ambrosio, C., Li, S., Surmacz, E. & Baserga, R. Association of insulin receptor substrate 1 with simian virus 40 large T antigen. Mol. Cell Biol. 15, 4232–4239 (1995).
Zick, Y. Ser/Thr phosphorylation of IRS proteins: a molecular basis for insulin resistance. Sci. STKE 2005, PE4 (2005).
Bouzakri, K. et al. Reduced activation of phosphatidylinositol-3 kinase and increased serine 636 phosphorylation of insulin receptor substrate-1 in primary culture of skeletal muscle cells from patients with type 2 diabetes. Diabetes 52, 1319–1325 (2003).
Harrington, L. S. et al. The TSC1–2 tumor suppressor controls insulin-PI3K signaling via regulation of IRS proteins. J. Cell Biol. 166, 213–223 (2004).
Miller, B. S. et al. Activation of cJun NH2-terminal kinase/stress-activated protein kinase by insulin. Biochemistry 35, 8769–8775 (1996).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKK-β and NF-κB. Nature Med. 11, 183–190 (2005).
Hirosumi, J. et al. A central role for JNK in obesity and insulin resistance. Nature 420, 333–336 (2002).
Aguirre, V., Uchida, T., Yenush, L., Davis, R. & White, M. F. The c-Jun NH(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of Ser(307). J. Biol. Chem. 275, 9047–9054 (2000). An important paper that established the connection between Ser307 phosphorylation, JNK and insulin resistance.
Craparo, A., Freund, R. & Gustafson, T. A. 14-3-3 (ε) interacts with the insulin-like growth factor I receptor and insulin receptor substrate I in a phosphoserine-dependent manner. J. Biol. Chem. 272, 11663–11669 (1997).
Bard-Chapeau, E. A. et al. Deletion of Gab1 in the liver leads to enhanced glucose tolerance and improved hepatic insulin action. Nature Med. 11, 567–571 (2005).
Hirashima, Y. et al. Insulin down-regulates insulin receptor substrate-2 expression through the phosphatidylinositol 3-kinase/Akt pathway. J. Endocrinol. 179, 253–266 (2003).
Rui, L., Yuan, M., Frantz, D., Shoelson, S. & White, M. F. SOCS-1 and SOCS-3 block insulin signaling by ubiquitin-mediated degradation of IRS1 and IRS2. J. Biol. Chem. 277, 42394–42398 (2002).
Shimomura, I. et al. Decreased IRS-2 and increased SREBP-1c lead to mixed insulin resistance and sensitivity in livers of lipodystrophic and ob/ob mice. Mol. Cell 6, 77–86 (2000).
Sesti, G. et al. Defects of the insulin receptor substrate (IRS) system in human metabolic disorders. FASEB J. 15, 2099–2111 (2001).
Araki, E. et al. Alternative pathway of insulin signaling in targeted disruption of the IRS-1 gene. Nature 372, 186–190 (1994).
Kubota, N. et al. Insulin receptor substrate 2 plays a crucial role in β cells and the hypothalamus. J. Clin. Invest. 114, 917–927 (2004).
Withers, D. J. et al. Disruption of IRS-2 causes type 2 diabetes in mice. Nature 391, 900–904 (1998).
Tseng, Y. H. et al. Prediction of preadipocyte differentiation by gene expression reveals role of insulin receptor substrates and necdin. Nature Cell Biol. 7, 601–611 (2005).
Miki, H. et al. Essential role of insulin receptor substrate 1 (IRS-1) and IRS-2 in adipocyte differentiation. Mol. Cell Biol. 21, 2521–2532 (2001).
Huang, C., Thirone, A. C., Huang, X. & Klip, A. Differential contribution of insulin receptor substrates 1 versus 2 to insulin signaling and glucose uptake in l6 myotubes. J. Biol. Chem. 280, 19426–19435 (2005).
Taniguchi, C. M., Ueki, K. & Kahn, R. Complementary roles of IRS-1 and IRS-2 in the hepatic regulation of metabolism. J. Clin. Invest. 115, 718–727 (2005).
Sun, X. J. et al. The IRS-2 gene on murine chromosome 8 encodes a unique signaling adapter for insulin and cytokine action. Mol. Endocrinol. 11, 251–262 (1997).
Miura, A. et al. Insulin substrates 1 and 2 are corequired for activation of atypical protein kinase C and Cbl-dependent phosphatidylinositol 3-kinase during insulin action in immortalized brown adipocytes. Biochemistry 43, 15503–15509 (2004).
Tsuruzoe, K., Emkey, R., Kriauciunas, K. M., Ueki, K. & Kahn, C. R. Insulin receptor substrate 3 (IRS-3) and IRS-4 impair IRS-1- and IRS-2-mediated signaling. Mol. Cell Biol. 21, 26–38 (2001).
Inoue, G., Cheatham, B., Emkey, R. & Kahn, C. R. Dynamics of insulin signaling in 3T3-L1 adipocytes. Differential compartmentalization and trafficking of insulin receptor substrate (IRS)-1 and IRS-2. J. Biol. Chem. 273, 11548–11555 (1998).
Ogihara, T. et al. Insulin receptor substrate (IRS)-2 is dephosphorylated more rapidly than IRS-1 via its association with phosphatidylinositol 3-kinase in skeletal muscle cells. J. Biol. Chem. 272, 12868–12873 (1997).
Sawka-Verhelle, D., Tartare-Deckert, S., White, M. F. & Van Obberghen, E. Insulin receptor substrate-2 binds to the insulin receptor through its phosphotyrosine-binding domain and through a newly identified domain comprising amino acids 591–786. J. Biol. Chem. 271, 5980–5983 (1996).
Myers, M. G. Jr et al. IRS-1 activates phosphatidylinositol 3′-kinase by associating with src homology 2 domains of p85. Proc. Natl Acad. Sci. USA 89, 10350–10354 (1992).
Cheatham, B. et al. Phosphatidylinositol 3-kinase activation is required for insulin stimulation of pp70 S6 kinase, DNA synthesis, and glucose transporter translocation. Mol. Cell Biol. 14, 4902–4911 (1994).
Alessi, D. R. et al. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Bα. Curr. Biol. 7, 261–269 (1997). Describes the discovery of PDK1.
Le Good, J. A. et al. Protein kinase C isotypes controlled by phosphoinositide 3-kinase through the protein kinase PDK1. Science 281, 2042–2045 (1998).
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Maehama, T. & Dixon, J. E. PTEN: a tumour suppressor that functions as a phospholipid phosphatase. Trends Cell Biol. 9, 125–128 (1999).
Wijesekara, N. et al. Muscle-specific Pten deletion protects against insulin resistance and diabetes. Mol. Cell Biol. 25, 1135–1145 (2005).
Tang, X., Powelka, A. M., Soriano, N. A., Czech, M. P. & Guilherme, A. PTEN, but not SHIP2, suppresses insulin signaling through the phosphatidylinositol 3-kinase/Akt pathway in 3T3-L1 adipocytes. J. Biol. Chem. 280, 22523–22529 (2005).
Sleeman, M. W. et al. Absence of the lipid phosphatase SHIP2 confers resistance to dietary obesity. Nature Med. 11, 199–205 (2005).
Shepherd, P. R., Withers, D. J. & Siddle, K. Phosphoinositide 3-kinase: the key switch mechanism in insulin signalling. Biochem. J. 333, 471–490 (1998).
Yu, J. et al. Regulation of the p85/p110 phosphatidylinositol 3′-kinase: stabilization and inhibition of the p110α catalytic subunit by the p85 regulatory subunit. Mol. Cell Biol. 18, 1379–1387 (1998). Helped to define the molecular and functional relationships between the regulatory and catalytic subunits of PI3K.
Asano, T. et al. p110β is up-regulated during differentiation of 3T3-L1 cells and contributes to the highly insulin-responsive glucose transport activity. J. Biol. Chem. 275, 17671–17676 (2000).
Tanti, J. F., Gremeaux, T., Van Obberghen, E. & Le Marchand-Brustel, Y. Insulin receptor substrate 1 is phosphorylated by the serine kinase activity of phosphatidylinositol 3-kinase. Biochem. J. 304, 17–21 (1994).
Tanti, J. F., Gremeaux, T., van Obberghen, E. & Le Marchand-Brustel, Y. Serine/threonine phosphorylation of insulin receptor substrate 1 modulates insulin receptor signaling. J. Biol. Chem. 269, 6051–6057 (1994).
Antonetti, D. A., Algenstaedt, P. & Kahn, C. R. Insulin receptor substrate 1 binds two novel splice variants of the regulatory subunit of phosphatidylinositol 3-kinase in muscle and brain. Mol. Cell Biol. 16, 2195–2203 (1996).
Ueki, K. et al. Increased insulin sensitivity in mice lacking p85β subunit of phosphoinositide 3-kinase. Proc. Natl Acad. Sci. USA 99, 419–424 (2002).
Terauchi, Y. et al. Increased insulin sensitivity and hypoglycaemia in mice lacking the p85 α subunit of phosphoinositide 3-kinase. Nature Genet. 21, 230–235 (1999). The first description of the paradoxical negative regulation of insulin signalling by the regulatory subunit p85α.
Chen, D. et al. p50α/p55α phosphoinositide 3-kinase knockout mice exhibit enhanced insulin sensitivity. Mol. Cell Biol. 24, 320–329 (2004).
Fruman, D. A. et al. Hypoglycaemia, liver necrosis and perinatal death in mice lacking all isoforms of phosphoinositide 3-kinase p85 α. Nature Genet. 26, 379–382 (2000).
Mauvais-Jarvis, F. et al. Reduced expression of the murine p85α subunit of phosphoinositide 3-kinase improves insulin signaling and ameliorates diabetes. J. Clin. Invest. 109, 141–149 (2002).
Barbour, L. A. et al. Human placental growth hormone increases expression of the p85 regulatory unit of phosphatidylinositol 3-kinase and triggers severe insulin resistance in skeletal muscle. Endocrinology 145, 1144–1150 (2004). Shows that the physiologic upregulation of p85α in vivo induces insulin resistance.
Bandyopadhyay, G. K., Yu, J. G., Ofrecio, J. & Olefsky, J. M. Increased p85/55/50 expression and decreased phosphatidylinositol 3-kinase activity in insulin-resistant human skeletal muscle. Diabetes 54, 2351–2359 (2005).
Ueki, K., Algenstaedt, P., Mauvais-Jarvis, F. & Kahn, C. R. Positive and negative regulation of phosphoinositide 3-kinase-dependent signaling pathways by three different gene products of the p85α regulatory subunit. Mol. Cell Biol. 20, 8035–8046 (2000).
Luo, J., Field, S. J., Lee, J. Y., Engelman, J. A. & Cantley, L. C. The p85 regulatory subunit of phosphoinositide 3-kinase down-regulates IRS-1 signaling via the formation of a sequestration complex. J. Cell Biol. 170, 455–464 (2005).
Ueki, K. et al. Positive and negative roles of p85 α and p85 β regulatory subunits of phosphoinositide 3-kinase in insulin signaling. J. Biol. Chem. 278, 48453–48466 (2003). The first published connection between the p85α regulatory subunit and JNK activation.
Carpenter, C. L. & Cantley, L. C. Phosphoinositide kinases. Curr. Opin. Cell Biol. 8, 153–158 (1996).
Fang, D. & Liu, Y. C. Proteolysis-independent regulation of PI3K by Cbl-b-mediated ubiquitination in T cells. Nature Immunol. 2, 870–875 (2001).
Zheng, Y., Bagrodia, S. & Cerione, R. A. Activation of phosphoinositide 3-kinase activity by Cdc42Hs binding to p85. J. Biol. Chem. 269, 18727–18730 (1994).
Cross, D. A., Alessi, D. R., Cohen, P., Andjelkovich, M. & Hemmings, B. A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378, 785–789 (1995).
Frame, S. & Cohen, P. GSK3 takes centre stage more than 20 years after its discovery. Biochem. J. 359, 1–16 (2001).
Sano, H. et al. Insulin-stimulated phosphorylation of a Rab GTPase-activating protein regulates GLUT4 translocation. J. Biol. Chem. 278, 14599–14602 (2003).
Harris, T. E. & Lawrence, J. C. Jr. TOR signaling. Sci. STKE 2003, RE15 (2003).
Tran, H., Brunet, A., Griffith, E. C. & Greenberg, M. E. The many forks in FOXO's road. Sci. STKE 2003, RE5 (2003).
Puigserver, P. et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1α interaction. Nature 423, 550–555 (2003).
Nakae, J. et al. The forkhead transcription factor Foxo1 regulates adipocyte differentiation. Dev. Cell 4, 119–129 (2003).
Wolfrum, C., Asilmaz, E., Luca, E., Friedman, J. M. & Stoffel, M. Foxa2 regulates lipid metabolism and ketogenesis in the liver during fasting and in diabetes. Nature 432, 1027–1032 (2004).
Brazil, D. P., Yang, Z. Z. & Hemmings, B. A. Advances in protein kinase B signalling: AKTion on multiple fronts. Trends Biochem. Sci. 29, 233–242 (2004).
Gao, T., Furnari, F. & Newton, A. C. PHLPP: a phosphatase that directly dephosphorylates Akt, promotes apoptosis, and suppresses tumor growth. Mol. Cell 18, 13–24 (2005).
Du, K., Herzig, S., Kulkarni, R. N. & Montminy, M. TRB3: a tribbles homolog that inhibits Akt/PKB activation by insulin in liver. Science 300, 1574–1577 (2003).
Koo, S. H. et al. PGC-1 promotes insulin resistance in liver through PPAR-α-dependent induction of TRB-3. Nature Med. 10, 530–534 (2004).
Chen, W. S. et al. Growth retardation and increased apoptosis in mice with homozygous disruption of the Akt1 gene. Genes Dev. 15, 2203–2208 (2001).
Cho, H., Thorvaldsen, J. L., Chu, Q., Feng, F. & Birnbaum, M. J. Akt1/PKBα is required for normal growth but dispensable for maintenance of glucose homeostasis in mice. J. Biol. Chem. 276, 38349–38352 (2001).
Cho, H. et al. Insulin resistance and a diabetes mellitus-like syndrome in mice lacking the protein kinase Akt2 (PKBβ). Science 292, 1728–1731 (2001).
George, S. et al. A family with severe insulin resistance and diabetes due to a mutation in AKT2. Science 304, 1325–1328 (2004).
Tschopp, O. et al. Essential role of protein kinase B γ (PKB γ/Akt3) in postnatal brain development but not in glucose homeostasis. Development 132, 2943–2954 (2005).
Chan, T. O., Rittenhouse, S. E. & Tsichlis, P. N. AKT/PKB and other D3 phosphoinositide-regulated kinases: kinase activation by phosphoinositide-dependent phosphorylation. Annu. Rev. Biochem. 68, 965–1014 (1999).
Bae, S. S., Cho, H., Mu, J. & Birnbaum, M. J. Isoform-specific regulation of insulin-dependent glucose uptake by Akt/protein kinase B. J. Biol. Chem. 278, 49530–49536 (2003).
Jiang, Z. Y. et al. Insulin signaling through Akt/protein kinase B analyzed by small interfering RNA-mediated gene silencing. Proc. Natl Acad. Sci. USA 100, 7569–7574 (2003).
Calera, M. R. et al. Insulin increases the association of Akt-2 with Glut4-containing vesicles. J. Biol. Chem. 273, 7201–7204 (1998).
Yamada, E. et al. Akt2 phosphorylates Synip to regulate docking and fusion of GLUT4-containing vesicles. J. Cell Biol. 168, 921–928 (2005).
Masure, S. et al. Molecular cloning, expression and characterization of the human serine/threonine kinase Akt-3. Eur. J. Biochem. 265, 353–360 (1999).
Standaert, M. L., Bandyopadhyay, G., Kanoh, Y., Sajan, M. P. & Farese, R. V. Insulin and PIP3 activate PKC-ζ by mechanisms that are both dependent and independent of phosphorylation of activation loop (T410) and autophosphorylation (T560) sites. Biochemistry 40, 249–255 (2001).
Farese, R. V. Function and dysfunction of aPKC isoforms for glucose transport in insulin-sensitive and insulin-resistant states. Am. J. Physiol. Endocrinol. Metab. 283, E1–E11 (2002).
Farese, R. V., Sajan, M. P. & Standaert, M. L. Atypical protein kinase C in insulin action and insulin resistance. Biochem. Soc. Trans. 33, 350–353 (2005).
Pouyssegur, J., Volmat, V. & Lenormand, P. Fidelity and spatio-temporal control in MAP kinase (ERKs) signalling. Biochem. Pharmacol. 64, 755–763 (2002).
Pages, G. et al. Defective thymocyte maturation in p44 MAP kinase (Erk 1) knockout mice. Science 286, 1374–1377 (1999).
Bost, F. et al. The extracellular signal-regulated kinase isoform ERK1 is specifically required for in vitro and in vivo adipogenesis. Diabetes 54, 402–411 (2005).
Hancock, J. F. & Parton, R. G. Ras plasma membrane signalling platforms. Biochem. J. 389, 1–11 (2005).
Rodriguez-Viciana, P. et al. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370, 527–532 (1994).
Wellbrock, C., Karasarides, M. & Marais, R. The RAF proteins take centre stage. Nature Rev. Mol. Cell Biol. 5, 875–885 (2004).
Matos, P. et al. Small GTPase Rac1: structure, localization, and expression of the human gene. Biochem. Biophys. Res. Commun. 277, 741–751 (2000).
Marks, P. W. & Kwiatkowski, D. J. Genomic organization and chromosomal location of murine Cdc42. Genomics 38, 13–18 (1996).
Marcusohn, J., Isakoff, S. J., Rose, E., Symons, M. & Skolnik, E. Y. The GTP-binding protein Rac does not couple PI 3-kinase to insulin-stimulated glucose transport in adipocytes. Curr. Biol. 5, 1296–1302 (1995).
Usui, I., Imamura, T., Huang, J., Satoh, H. & Olefsky, J. M. Cdc42 is a Rho GTPase family member that can mediate insulin signaling to glucose transport in 3T3-L1 adipocytes. J. Biol. Chem. 278, 13765–13774 (2003).
Ip, Y. T. & Davis, R. J. Signal transduction by the c-Jun N-terminal kinase (JNK) — from inflammation to development. Curr. Opin. Cell Biol. 10, 205–219 (1998).
Zarubin, T. & Han, J. Activation and signaling of the p38 MAP kinase pathway. Cell Res. 15, 11–18 (2005).
Furtado, L. M., Somwar, R., Sweeney, G., Niu, W. & Klip, A. Activation of the glucose transporter GLUT4 by insulin. Biochem. Cell Biol. 80, 569–578 (2002).
Carlson, C. J., Koterski, S., Sciotti, R. J., Poccard, G. B. & Rondinone, C. M. Enhanced basal activation of mitogen-activated protein kinases in adipocytes from type 2 diabetes: potential role of p38 in the downregulation of GLUT4 expression. Diabetes 52, 634–641 (2003).
Chiang, S. H. et al. TCGAP, a multidomain Rho GTPase-activating protein involved in insulin-stimulated glucose transport. EMBO J. 22, 2679–2691 (2003).
Thien, C. B. & Langdon, W. Y. Cbl: many adaptations to regulate protein tyrosine kinases. Nature Rev. Mol. Cell Biol. 2, 294–307 (2001).
Molero, J. C. et al. c-Cbl-deficient mice have reduced adiposity, higher energy expenditure, and improved peripheral insulin action. J. Clin. Invest. 114, 1326–1333 (2004).
Zhou, Q. L. et al. Analysis of insulin signalling by RNAi-based gene silencing. Biochem. Soc. Trans. 32, 817–821 (2004).
Stegmeier, F., Hu, G., Rickles, R. J., Hannon, G. J. & Elledge, S. J. A lentiviral microRNA-based system for single-copy polymerase II-regulated RNA interference in mammalian cells. Proc. Natl Acad. Sci. USA 102, 13212–13217 (2005).
Shinagawa, T. & Ishii, S. Generation of Ski-knockdown mice by expressing a long double-strand RNA from an RNA polymerase II promoter. Genes Dev. 17, 1340–1345 (2003).
Ding, S. et al. Efficient transposition of the piggyBac (PB) transposon in mammalian cells and mice. Cell 122, 473–483 (2005).
Becker, A. B. & Roth, R. A. Insulin receptor structure and function in normal and pathological conditions. Annu. Rev. Med. 41, 99–115 (1990).
Goren, H. J., White, M. F. & Kahn, C. R. Separate domains of the insulin receptor contain sites of autophosphorylation and tyrosine kinase activity. Biochemistry 26, 2374–2382 (1987).
Sesti, G. et al. Tissue-specific expression of two alternatively spliced isoforms of the human insulin receptor protein. Acta Diabetol. 31, 59–65 (1994).
Yamaguchi, Y. et al. Functional properties of two naturally occurring isoforms of the human insulin receptor in Chinese hamster ovary cells. Endocrinology 129, 2058–2066 (1991).
Leibiger, B. et al. Selective insulin signaling through A and B insulin receptors regulates transcription of insulin and glucokinase genes in pancreatic β cells. Mol. Cell 7, 559–570 (2001).
Bhalla, U. S. & Iyengar, R. Emergent properties of networks of biological signaling pathways. Science 283, 381–387 (1999).
Acknowledgements
This work was supported by National Institutes of Health grants, and a Joslin Diabetes and Endocrinology Research Center grant (to C.R.K.). C.M.T. acknowledges support from the American Diabetes Association Medical Scholars Award and the Medical Scientist Training Program (Harvard Medical School). B.E. was supported by a National Institutes of Health grant. We apologize to the authors whose work could not be cited in this review due to space limitations, but appreciate all of the many contributions to the large body of literature we have tried to summarize here.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Critical node
-
A point in a signalling network that is essential for the biological function of a ligand–receptor interaction, but also allows divergence of the signal to facilitate crosstalk between systems and/or to fine-tune the response to stimuli.
- Insulin receptor substrate protein
-
(IRS protein). A large protein scaffold that serves as a docking platform for other signalling proteins that contain Src-homology-2 domains. The IRS proteins are required for a complete insulin signal.
- Receptor tyrosine kinase
-
(RTK). A cell-surface receptor with an intracellular tyrosine kinase domain. The ligand-mediated activation of an RTK results in the activation of the intracellular kinase domain.
- Insulin resistance
-
Insulin resistance is a condition in which normal concentrations of insulin produce a subnormal biological response. It is common in many physiological and pathological states, including obesity, type-2 diabetes, metabolic syndrome, polycystic ovarian disease, pregnancy and puberty.
- Pleckstrin-homology domain
-
(PH domain). A protein domain of ∼100 amino acids with homology to pleckstrin that mediates the binding to membrane phospholipids, such as PIP3.
- Phosphotyrosine-binding domain
-
(PTB domain). A domain that mediates the binding to specific phosphotyrosine residues. The affinity of a PTB domain for a phosphotyrosine depends on the residues that surround the phosphotyrosine. For instance, the PTB domain of the IRS proteins binds to an NPXpY motif.
- Src-homology-2 domain
-
(SH2 domain). A domain of ∼100 amino acids that binds to phosphotyrosine residues in proteins. Every protein's SH2 domain might have a slightly different phosphotyrosine-binding motif.
- Transphosphorylation
-
The transfer of a phosphate by a protein kinase to a residue within the same kinase molecule, or to a different kinase molecule of the same kind.
- Phosphotyrosine motif
-
A phosphorylated tyrosine residue that is flanked by a few different amino-acid residues. Different phosphotyrosine-binding proteins have varying affinities for different phosphotyrosine motifs, which explains, in part, the specificity of phosphotyrosine-binding proteins.
- AGC kinases
-
A large family of serine/threonine kinases that includes members as diverse as the cAMP-dependent protein kinases, and protein kinase C. The members of this AGC subgroup include AKT/PKB, serum/glucocorticoid kinases and the atypical PKCs.
- Src-homology-3 domain
-
(SH3 domain). A protein sequence of ∼50 amino acids that facilitates the binding to proline-rich regions of proteins. Binding of an SH3 domain to a proline-rich region can occur in an intramolecular or intermolecular fashion.
- Breakpoint-cluster region homology domain
-
A ∼150-residue region of p85a with homology to the breakpoint-cluster region gene and homology to Rho-GTPase activating proteins, which decrease the activity of small GTPases. Despite this homology, the breakpoint-cluster region homology domain has not been demonstrated to have GTPase activity.
- Diacylglycerol
-
(DAG). DAG is a lipid second messenger that is produced by the cleavage of PIP2 by phospholipase C. DAG is involved in the activation of conventional and novel protein kinase Cs (PKCs).
- GLUT4 translocation
-
Relocalization and fusion of an intracellular vesicle that contains the glucose transporter GLUT4 to the plasma membrane. It allows the uptake of extracellular glucose that is stimulated by insulin in muscle and adipose tissues.
Rights and permissions
About this article
Cite this article
Taniguchi, C., Emanuelli, B. & Kahn, C. Critical nodes in signalling pathways: insights into insulin action. Nat Rev Mol Cell Biol 7, 85–96 (2006). https://doi.org/10.1038/nrm1837
Issue Date:
DOI: https://doi.org/10.1038/nrm1837
This article is cited by
-
Integrated traditional Chinese and Western medicine in the prevention and treatment of non-alcoholic fatty liver disease: future directions and strategies
Chinese Medicine (2024)
-
Effects of swimming training in hot and cold temperatures combined with cinnamon supplementation on HbA1C levels, TBC1D1, and TBC1D4 in diabetic rats
Nutrition & Diabetes (2024)
-
Effects of Inorganic Arsenic on Type 2 Diabetes Mellitus In Vivo: the Roles and Mechanisms of miRNAs
Biological Trace Element Research (2024)
-
Ab initio modeling of human IRS1 protein to find novel target to dock with drug MH to mitigate T2DM diabetes by insulin signaling
3 Biotech (2024)
-
Prenylated flavonoid fractions from Glycyrrhiza glabra alleviate insulin resistance in HepG2 cells by regulating the ERK/IRS-1 and PI3K/Akt signaling pathways
Archives of Pharmacal Research (2024)