Key Points
-
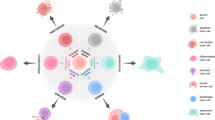
Fusion of one or more cells can occur during development as well as in mature individuals. Fusion of two or more cells of the same type yields a syncytium. Syncytia are commonly found in placenta, bone, cartilage, muscle and multi-nucleated giant cells.
-
Fusion of two or more cells of different types can yield multi-nucleated cells called heterokaryons. Heterokaryons have been observed after cells of bone marrow origin fused with hepatocytes, cardiomyocytes and Purkinje neurons to form multi-nucleated cells.
-
Fusion of two or more cells of the same or different type and fusion of nuclei can yield synkaryons. Synkaryons have been found among hepatocytes, kidney tubules and B cells.
-
Cell fusion can cause a change in the phenotype and/or the function of cells. Cell fusion could therefore explain transdifferentiation of committed somatic cells.
-
Cell fusion can reverse or repair injury to tissues. In this context, cell fusion facilitates the regeneration of tissues.
-
Cell fusion can promote the transmission of viruses. Fusion of cells of individuals of disparate species might explain the transmission of viruses between species and the genesis of new pathogens.
-
Cell fusion and nuclear fusion that occur in the formation of synkaryons can allow the re-sorting and recombination of chromosomal DNA. Fusion of nuclei of an immature cell, such as a stem cell, and a mature cell might generate proliferation and malignancy.
Abstract
Until recently, cells were thought to be integral and discrete components of tissues, and their state was determined by cell differentiation. However, under some conditions, stem cells or their progeny can fuse with cells of other types, mixing cytoplasmic and even genetic material of different (heterotypic) origins. The fusion of heterotypic cells could be of central importance for development, repair of tissues and the pathogenesis of disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hooke, R. Micrographia: or some Physiological Descriptions of Minute Bodies made by Magnifying Glasses with Observations and Inquiries thereupon. (J. Martyn and J. Allestry, London, 1665).
van Leeuwenhoek, A. The Collected Letters of Antoni van Leeuwenhoek. (Swetz & Zeitlinger, Amsterdam, 1939–1967).
Baker, J. R. The cell theory: a restatement, history and critique. Part IV. The multiplication of cells. Q. J. Microsc. Sci. 94, 407–440 (1953).
Schleiden, M. J. Beitrage zur Phytogenesis. Arch. Anat. Physiol. 137–176 (1838).
Schwann, T. Mikroskopische Untersuchungen uber die Ubereinstimmung in der Struktur und dem Wachsten der Thiere und Pflanzen. (Saunderschen Buchhandlung, Berlin, 1839).
Wolpert, L. The evolution of 'the cell theory'. Curr. Biol. 6, 225–228 (1996).
Huppertz, B., Frank, H. G., Kingdom, J. C., Reister, F. & Kaufmann, P. Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochem. Cell Biol. 110, 495–508 (1998).
Vignery, A. Osteoclasts and giant cells: macrophage–macrophage fusion mechanism. Int. J. Exp. Pathol. 81, 291–304 (2000).
Kahn, A. J., Teitelbaum, S. L., Malone, J. D. & Krukowski, M. The relationship of monocytic cells to the differentiation and resorption of bone. Prog. Clin. Biol. Res. B. 110, 239–248 (1982).
Loutit, J. F. & Nisbet, N. W. The origin of osteoclasts. Immunobiology 161, 193–203 (1982).
Huppertz, B., Tews, D. S. & Kaufmann, P. Apoptosis and syncytial fusion in human placental trophoblast and skeletal muscle. Int. Rev. Cytol. 205, 215–253 (2001).
Taylor, M. V. Muscle development: molecules of myoblast fusion. Curr. Biol. 10, R646–R648 (2000).
Harris, H. & Watkins, J. F. Hybrid cells derived from mouse and man: artificial heterokaryons of mammalian cells from different species. Nature 205, 640–646 (1965). Sendai virus was used to fuse murine Ehrlich ascites cells and human HeLa cells in vitro . The cells formed by fusion were heterokaryons that remained stable over time and exhibited functions and a phenotype that was distinct from either fusion partner. This paper was the first to report the distinct properties of heterokaryons.
Harris, H., Sidebottom, E., Grace, D. M. & Bramwell, M. E. The expression of genetic information: a study with hybrid animal cells. J. Cell Sci. 4, 499–525 (1969).
Ringertz, N. R., Carlsson, S. A., Ege, T. & Bolund, L. Detection of human and chick nuclear antigens in nuclei of chick erythrocytes during reactivation in heterokaryons with HeLa cells. Proc. Natl Acad. Sci. USA 68, 3228–3232 (1971).
Carlsson, S. A., Luger, O., Ringertz, N. R. & Savage, R. E. Phenotypic expression in chick erythrocyte x rat myoblast hybrids and in chick myoblast x rat myoblast hybrids. Exp. Cell Res. 84, 47–55 (1974).
Lawrence, J. B. & Coleman, J. R. Extinction of muscle-specific properties in somatic cell heterokaryons. Dev. Biol. 101, 463–476 (1984).
Wright, W. E. & Aronoff, J. The suppression of myogenic functions in heterokaryons formed by fusing chick myocytes to diploid rat fibroblasts. Cell Differ. 12, 299–306 (1983).
Konieczny, S. F., Lawrence, J. B. & Coleman, J. R. Analysis of muscle protein expression in polyethylene glycol-induced chicken: rat myoblast heterokaryons. J. Cell Biol. 97, 1348–1355 (1983).
Ringertz, N. R. & Savage, R. E. Cell Hybrids (Academic Press, New York, 1976).
Blau, H. M. & Blakely, B. T. Plasticity of cell fate: insights from heterokaryons. Sem.Cell Dev. Biol. 10, 267–272 (1999).
Terada, N. et al. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 416, 542–545 (2002).
Ying, Q. L., Nichols, J., Evans, E. P. & Smith, A. G. Changing potency by spontaneous fusion. Nature 416, 545–548 (2002). Neuronal stem cells were fused with embryonic stem cells and the fused cells injected into murine blastocysts. The fused cells acquired tissue-specific properties and differentiation potential in the hosts. These authors suggest that cell fusion could account for transdifferentiation.
Gibson, A. J. et al. Dermal fibroblasts convert to a myogenic lineage in mdx mouse muscle. J. Cell Sci. 108, 207–214 (1995). The authors transplanted dermal fibroblasts into mice with defective myotubes and found that the fibroblasts fused with myotubes. This study was the first to show that fusion of cells can contribute to the regeneration of tissues.
Gussoni, E. et al. Long-term persistence of donor nuclei in a Duchenne muscular dystrophy patient receiving bone marrow transplantation. J. Clin. Invest. 110, 807–814 (2002). Describes a human patient with Duchenne muscular dystrophy who received a bone marrow transplant. The bone marrow cells fused with skeletal muscle cells to yield heterokaryons. This paper is the first report of heterokaryon formation in a human subject.
Wang, X. et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 422, 897–900 (2003). Bone-marrow-derived cells were shown to fuse with hepatocytes to repair liver damage. This paper shows that cell fusion can contribute to the regeneration of a parenchymal organ and that fusion can eventuate reduction division.
Vassilopoulos, G., Wang, P. R. & Russell, D. W. Transplanted bone marrow regenerates liver by cell fusion. Nature 422, 901–904 (2003).
Weimann, J. M., Johansson, C. B., Trejo, A. & Blau, H. M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nature Cell Biol. 5, 959–966 (2003).
Alvarez-Dolado, M. et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425, 968–973 (2003). Bone-marrow-derived cells were shown to fuse with hepatocytes, Purkinje neurons and cardiac muscle cells to yield heterokaryons in mice. The studies were done in healthy mice that had not suffered tissue injury, which raises the possibility that cell fusion can contribute to the development and maintenance of certain tissues.
Pomerantz, J. & Blau, H. M. Nuclear reprogramming: a key to stem cell function in regenerative medicine. Nature Cell Biol. 6, 810–816 (2004).
Tsai, R. Y., Kittappa, R. & McKay, R. D. Plasticity, niches, and the use of stem cells. Dev. Cell 2, 707–712 (2002).
Alison, M. R. et al. Recipes for adult stem cell plasticity: fusion cuisine or readymade? J. Clin. Pathol. 57, 113–120 (2004).
Ogle, B. M. et al. Spontaneous fusion of cells between species yields transdifferentiation and retroviral in vivo. FASEB J. 18, 548–550 (2004). Human bone marrow cells were engrafted into fetal pigs that after birth contained synkaryons with both porcine and human DNA. The synkaryons had porcine endogenous retrovirus that could be transmitted to human cells. This study shows how cell fusion can transfer viruses from one species to another.
Barski, G., Sorieul, S. & Cornefert, F. Production dans des cultures in vitro de deux souches cellulaires en association de cellules de caratere hybride. C. R. Hebd. Seances Acad. des Sci. 251, 1825–1827 (1960). When cells of two different tumour lines are mixed in culture, approximately 30% of one cell type fuse with the other to yield a cell with a single hybrid nucleus. This study was the first to report the fusion of cells and the fusion of nuclei in vitro.
Kohler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Hernandez, L. D., Hoffman, L. R., Wolfsberg, T. G. & White, J. M. Virus–cell and cell–cell fusion. Annu. Rev. Cell Dev. Biol. 12, 627–661 (1996).
Kozlov, M. M. & Markin, V. S. Possible mechanism of membrane fusion. Biofizika 28, 242–247 (1983) (in Russian).
Leikina, E. & Chernomordik, L. V. Reversible merger of membranes at the early stage of influenza hemagglutinin-mediated fusion. Mol. Biol. Cell 11, 2359–2371 (2000).
Yang, L. & Huang, H. W. Observation of a membrane fusion intermediate structure. Science 297, 1877–1879 (2002).
Chernomordik, L., Kozlov, M. M. & Zimmerberg, J. Lipids in biological membrane fusion. J. Membr. Biol. 146, 1–14 (1995).
Chernomordik, L. V. & Kozlov, M. M. Protein–lipid interplay in fusion and fission of biological membranes. Annu. Rev. Biochem. 72, 175–207 (2003).
Muller, M., Katsov, K. & Schick, M. A new mechanism of model membrane fusion determined from Monte Carlo simulation. Biophys. J. 85, 1611–1623 (2003).
Katsov, K., Muller, M. & Schick, M. Field theoretic study of bilayer membrane fusion. I. Hemifusion mechanism. Biophys. J. 87, 3277–3290 (2004).
Shemer, G. & Podbilewicz, B. The story of cell fusion: big lessons from little worms. BioEssays 25, 672–682 (2003). This paper provides an authoritative review of the mechanics of cell fusion.
Blond, J. L. et al. An envelope glycoprotein of the human endogenous retrovirus HERV-W is expressed in the human placenta and fuses cells expressing the type D mammalian retrovirus receptor. J. Virol. 74, 3321–3329 (2000).
Mi, S. et al. Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature 403, 785–789 (2000).
Inoue, N., Ikawa, M., Isotani, A. & Okabe, M. The immunoglobulin superfamily protein Izumo is required for sperm to fuse with eggs. Nature 434, 234–238 (2005).
Gilpin, B. J. et al. A novel, secreted form of human ADAM12 (meltrin-α) provokes myogenesis in vivo. J. Biol. Chem. 273, 157–166 (1998).
Galliano, M. F. et al. Binding of ADAM12, a marker of skeletal muscle regeneration, to the muscle-specific actin-binding protein, α-actinin-2, is required for myoblast fusion. J. Biol. Chem. 275, 13933–13939 (2000).
Makino, S. et al. Cardiomyocytes can be generated from marrow stromal cells in vitro. J. Clin. Invest. 103, 697–705 (1999).
Cho, C., Turner, L., Primakoff, P. & Myles, D. G. Genomic organization of the mouse fertilin β gene that encodes an ADAM family protein active in sperm–egg fusion. Dev. Genet. 20, 320–328 (1997).
Evans, J. P., Schultz, R. M. & Kopf, G. S. Roles of the disintegrin domains of mouse fertilins α and β in fertilization. Biol. Reprod. 59, 145–152 (1998).
Primakoff, P. & Myles, D. G. The ADAM gene family: surface proteins with adhesion and protease activity. Trends Genet. 16, 83–87 (2000).
Blumenthal, R., Clague, M. J., Durell, S. R. & Epand, R. M. Membrane fusion. Chem. Rev. 103, 53–69 (2003).
Nishikawa, S. et al. Nep98p is a component of the yeast spindle pole body and essential for nuclear division and fusion. J. Biol. Chem. 278, 9938–9943 (2003).
Bucci, M. & Wente, S. R. In vivo dynamics of nuclear pore complexes in yeast. J. Cell Biol. 136, 1185–1199 (1997).
Rose, M. D. Nuclear fusion in the yeast Saccharomyces cerevisiae. Annu. Rev. Cell Dev. Biol. 12, 663–695 (1996).
Chen, Y., Zhang, L., Zhou, Y., Geng, Y. & Chen, Z. Inducing somatic meiosis-like reduction at high frequency by caffeine in root-tip cells of Vicia faba. Mutat. Res. 452, 67–72 (2000).
Brinkley, B. R. Managing the centrosome numbers game: from chaos to stability in cancer cell division. Trends Cell Biol. 11, 18–21 (2001).
Mohler, W. A. et al. The type I membrane protein EFF-1 is essential for developmental cell fusion. Dev. Cell 2, 355–362 (2002).
Bernirschke, K. & Kaufmann, P. Pathology of the Human Placenta (Springer, New York, 2000).
Hoshina, M., Boothby, M. & Boime, I. Cytological localization of chorionic gonadotropin α and placental lactogen mRNAs during development of the human placenta. J. Cell Biol. 93, 190–198 (1982).
Hoshina, M., Boime, I. & Mochizuki, M. Cytological localization of hPL, hCG, and mRNA in chorionic tissue using in situ hybridization. Nippon Sanka Fujinka Gakkai Zasshi 36, 397–404 (1984).
Johansen, M., Redman, C. W., Wilkins, T. & Sargent, I. L. Trophoblast deportation in human pregnancy — its relevance for pre-eclampsia. Placenta 20, 531–539 (1999).
Redman, C. W. & Sargent, I. L. Placental debris, oxidative stress and pre-eclampsia. Placenta 21, 597–602 (2000).
Blau, H. M., Chiu, C. P. & Webster, C. Cytoplasmic activation of human nuclear genes in stable heterocaryons. Cell 32, 1171–1180 (1983).
Blau, H. M. Stem-cell fusion: a twist of fate. Nature 419, 437 (2002).
Weimann, J. M., Charlton, C. A., Brazelton, T. R., Hackman, R. C. & Blau, H. M. Contribution of transplanted bone marrow cells to Purkinje neurons in human adult brains. Proc. Natl Acad. Sci. USA 100, 2088–2093 (2003).
van Bekkum, D. W. Phylogenetic aspects of tissue regeneration: role of stem cells. A concise overview. Blood Cells Mol. Dis. 32, 11–16 (2004).
Relvas, J. B., Aldridge, H., Wells, K. E., Wells, D. J. & Watt, D. J. Exogenous genes are expressed in mdx muscle fibres following implantation of primary mouse skin cells. Basic App. Myol. 7, 211–219 (1997).
Ferrari, G. et al. Muscle regeneration by bone marrow-derived myogenic progenitors. Science 279, 1528–1530 (1998).
Bittner, R. E. et al. Recruitment of bone-marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anat. Embryol. (Berl.) 199, 391–396 (1999).
Gussoni, E. et al. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature 401, 390–394 (1999).
Nygren, J. M. et al. Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nature Med. 10, 494–501 (2004).
Balsam, L. B. et al. Hematopoietic stem cells adopt mature hematopoietic fates in ischemic myocardium. Nature 428, 668–673 (2004).
Murry, C. E. et al. Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature 428, 664–668 (2004).
Deb, A. et al. Bone marrow-derived cardiomyocytes are present in adult human heart: a study of gender-mismatched bone marrow transplantation patients. Circulation 107, 1247–1249 (2003).
Quaini, F. et al. Chimerism of the transplanted heart. N. Engl. J. Med. 346, 5–15 (2002).
Poulsom, R., Alison, M. R., Forbes, S. J. & Wright, N. A. Adult stem cell plasticity. J. Pathol. 197, 441–456 (2002).
Ianus, A., Holz, G. G., Theise, N. D. & Hussain, M. A. In vivo derivation of glucose-competent pancreatic endocrine cells from bone marrow without evidence of cell fusion. J. Clin. Invest. 111, 843–850 (2003).
Camargo, F. D., Green, R., Capetenaki, Y., Jackson, K. A. & Goodell, M. A. Single hematopoietic stem cells generate skeletal muscle through myeloid intermediates. Nature Med. 9, 1520–1527 (2003).
Camargo, F. D., Finegold, M. & Goodell, M. A. Hematopoietic myelomonocytic cells are the major source of hepatocyte fusion partners. J. Clin. Invest. 113, 1266–1270 (2004).
Willenbring, H. et al. Myelomonocytic cells are sufficient for therapeutic cell fusion in liver. Nature Med. 10, 744–748 (2004).
Chapman, L. E. et al. Xenotransplantation and xenogeneic infections. N. Eng. J. Med. 333, 1498–1501 (1995).
Patience, C., Takeuchi, Y. & Weiss, R. A. Infection of human cells by an endogenous retrovirus of pigs. Nature Med. 3, 282–286 (1997).
Paradis, K. et al. Search for cross-species transmission of porcine endogenous retrovirus in patients treated with living pig tissue. Science 285, 1236–1241 (1999).
Urnovitz, H. B. & Murphy, W. H. Human endogenous retroviruses: nature, occurrence, and clinical implications in human disease. Clin. Microbiol. Rev. 9, 72–99 (1996).
Goldenberg, D. M., Pavia, R. A. & Tsao, M. C. In vivo hybridisation of human tumour and normal hamster cells. Nature 250, 649–651 (1974).
Condamine, H., Custer, R. P. & Mintz, B. Pure-strain and genetically mosaic liver tumors histochemically identified with the-glucuronidase marker in allophenic mice. Proc. Natl Acad. Sci. USA 68, 2032–2036 (1971).
Houghton, J. et al. Gastric cancer originating from bone marrow-derived cells. Science 306, 1568–1571 (2004). Reports that transplanted bone marrow cells can promote the formation of Helicobacter felis -induced gastric epithelial cancers in mice. A possible interpretation is that the bone marrow cells fused with gastric epithelium to yield synkaryons with genomic instability and that the synkaryons became tumorigenic.
Acknowledgements
Work in the authors' laboratory is supported by grants from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Glossary
- DIFFERENTIATION
-
The acquisition of 'mature' cellular characteristics, which can be connected with the acquisition of the cell's mature function. Differentiation can be terminal, which means that the cell adopts a different set of characteristics and will not proliferate.
- HETEROKARYON
-
A cell formed by the fusion of two or more cells of different types and having two or more distinct nuclei.
- SYNKARYON
-
A cell formed by the fusion of two or more cells and having one common nucleus, formed by nuclear fusion.
- HYBRID CELL
-
A cell that arises from the fusion of two different cells.
- STALK
-
A structure that is formed when a fusion protein from one cell interacts with a receptor on a neighbouring cell, and the attractive force exceeds the repulsive force caused by hydration.
- PORE
-
A structure that is formed on completion of the fusion of cell membranes; the 'stalk' elongates to form a fusion pore.
- FUSOGEN
-
A protein or molecule that directly mediates the merging of cell membranes (for example, polyethylene glycol and calcium).
- OSTEOCLAST
-
A bone-tissue-resorbing cell that is formed by the fusion of mononuclear cell precursors to yield multinucleated cells. Osteoclasts contribute to bone development and homeostasis, including the formation of canals and cavities.
- ADAM FAMILY
-
A group of transmembrane proteins that contains a disintegrin and metalloprotease domain. Proteins of the ADAM family can promote cell adhesion and/or catalyse proteolysis and might promote or enable cells to fuse.
- PLOIDY
-
The number of single sets of chromosomes in a cell.
- TROPHOBLAST
-
A cell from the outermost layer of cells of the blastocyst, constituting the interface between the foetus and the mother. Cells of the trophoblast (cytotrophoblast) ultimately fuse to form the syncytiotrophoblast of the placenta.
- SYNCYTIUM
-
A cell containing two or more nuclei, usually having a large, continuous mass of cytoplasm. A syncytium is generally formed by fusion of two or more cells of the same type.
- MATERNAL-FETAL BARRIER
-
The placenta allows the exchange of oxygen and nutrients but restricts the interchange of cells between the foetus and the mother's uterus.
- TRANSDIFFERENTIATION
-
A change from one differentiated state of a cell to another. Transdifferentiation is thought to be rare and possibly limited to stem cells.
- FISH
-
Fluorescence in situ hybridization. A technique in which one or more genes are located in chromosomes by hybridization of fluorescently labelled complementary DNA. FISH can be used to determine the number of copies of a specific sequence of DNA in a cell.
- HAEMATOPOIETIC
-
Related to the blood or blood cells. Haematopoietic stem cells give rise to cells of the various lineages of the blood and have been found to fuse with cells from non-haematopoietic tissues.
- PARENCHYMAL CELL
-
A cell that contributes to the essential structure and/or function of an organ, in contrast to stroma or blood vessels.
- MYOTUBE
-
A developing fibre of skeletal muscle with a tubular appearance.
- HEPATOCYTE
-
A parenchymal cell of the liver. Hepatocytes are the only cells in mature humans that can undergo mitosis without cytokinesis to form multinucleated cell masses.
- MYELOMONOCYTIC CELL
-
An immature white blood cell of the granulocyte–monocyte–histiocyte lineage. These cells typically have cytoplasmic granules, a large horse-shoe-shaped nucleus and cell markers characteristic of immature myeloid cells. The term can refer to leukaemia cells exhibiting both myeloid and monocytic properties.
- PORCINE ENDOGENOUS RETROVIRUS
-
A retrovirus in germline DNA that is dormant in normal pigs and exists as DNA copies in every cell.
- ALLOPHENIC MICE
-
Tetraparental mice formed by the aggregation of blastocyst cells or cells from early embryos of two genetically pure lines of mice. The cell aggregates are implanted in the uterus of a foster mother.
Rights and permissions
About this article
Cite this article
Ogle, B., Cascalho, M. & Platt, J. Biological implications of cell fusion. Nat Rev Mol Cell Biol 6, 567–575 (2005). https://doi.org/10.1038/nrm1678
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm1678
This article is cited by
-
Primary aldosteronism: molecular medicine meets public health
Nature Reviews Nephrology (2023)
-
Asymmetric chromatin retention and nuclear envelopes separate chromosomes in fused cells in vivo
Communications Biology (2022)
-
Deletion of ER-retention motif on SARS-CoV-2 spike protein reduces cell hybrid during cell–cell fusion
Cell & Bioscience (2021)