Key Points
-
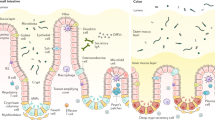
Restitution is the initial repair process of mucosa whereby epithelial cells spread and migrate across the basement membrane to re-establish surface-cell continuity. Trefoil proteins (TFFs) are essential for restitution to occur.
-
The TFFs are 7–12-kDa protease-resistant proteins that are secreted by mucus-secreting cells of the stomach (TFF1 and TFF2) and intestine (TFF3). All three TFFs are encoded together as a contig on human chromosome 21.
-
The trefoil domain is a motif of 38 or 39 amino acids containing six cysteine residues forming three disulphide-linked loops. In vivo, TFFs form two trefoil domains through interchain disulphide bonds (TFF1 and TFF3) or genomic duplication (TFF2). Dimer formation seems to be essential for some TFF functions but not for cell migration.
-
Tff1-null mice show gastric hyperproliferation and adenoma formation. Tff2-null mice seem normal, but show increased acid secretion and are susceptible to injury by non-steroidal anti-inflammatory drugs. Tff3-null mice also seem normal but are fatally susceptible to colonic damage.
-
Ulcer-associated mucosal epithelia show a substantial and coordinated increase in expression of the TFF family with the spread of expression into adjacent, non-metaplastic glands. New enhancer elements within TFF1 and TFF2 promoters have been reported, including elements that confer co-ordinated expression. TFF3 goblet-cell-specific transcription is conferred through a goblet-cell response element (GCRE) and an upstream silencer inhibitor. The GCRE is also found in the promoter of the mucin MUC2, which is consistent with the co-regulation of mucins and TFFs.
-
TFFs cause mucosal healing through unique mechanisms. Unlike other motogens, TFFs are not mitogenic and no receptor has yet been demonstrated. However, TFF2 and TFF3 activate signal-transduction events, including the induction of epidermal growth factor (EGF) receptor and β-catenin phosphorylation, activation of extracellular signal-regulated kinase (ERK)/mitogen-activated protein kinase (MAPK) and prevention of apoptotic signalling.
-
TFF1 mRNA and protein expression is frequently reduced in gastric cancer, often associated with somatic mutations in TFF1. Present data do not indicate that TFF2 or TFF3 have tumour-suppressor roles.
-
TFFs show many properties that are necessary for therapeutic efficacy. TFFs are compact, stable molecules and exert a protective effect when given orally. Possible therapeutic targets include prevention of the mucositis that follows radiotherapy and chemotherapy, which results in substantial morbidity in cancer patients.
Abstract
Maintaining the integrity of the gastrointestinal tract, despite the continual presence of microbial flora and injurious agents, is essential. Epithelial continuity depends on a family of small, yet abundant, secreted proteins — the trefoil factors (TFFs). TFFs protect mucous epithelia from a range of insults and contribute to mucosal repair, although the signalling events that mediate these responses are only partially understood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Notes
‡ Please note that this entry has been corrected and differs from the print version, in which HER2 has been omitted.
References
Podolsky, D. K. Mucosal immunity and inflammation. V. Innate mechanisms of mucosal defense and repair: the best offense is a good defense. Am. J. Physiol. 277, G495–G499 (1999).
Yamaguchi, Y. & Yoshikawa, K. Cutaneous wound healing: an update. J. Dermatol. 28, 521–523 (2001).
Nusrat, A. et al. Hepatocyte growth factor/scatter factor effects on epithelia. Regulation of intercellular junctions in transformed and nontransformed cell lines, basolateral polarisation of c-met receptor in transformed and natural intestinal epithelia, and induction of rapid wound repair in a transformed model epithelium. J. Clin. Invest. 93, 2056–2065 (1994).
Santos, M. F. et al. Rho proteins play a critical role in cell migration during the early phase of mucosal restitution. J. Clin. Invest. 100, 216–225 (1997).
Thim, L. A new family of growth-factor-like peptides. 'Trefoil' disulphide loop structures as a common feature in breast cancer associated peptide (pS2), pancreatic spasmolytic polypeptide (PSP), and frog skin peptides (spasmolysins). FEBS Lett. 250, 85–90 (1989). In this paper, Thim first identified the structural homologies between TFF1 and TFF2 (and the Xenopus spasmolysins), and put forward the hypothesis (since largely proven to be correct) that the trefoil domain was essential for resistance to the gut milieu and for function.
Polshakov, V. I. et al. High-resolution solution structure of human pNR-2/pS2: a single trefoil motif protein. J. Mol. Biol. 267, 418–432 (1997).
Kinoshita, K., Taupin, D. R., Itoh, H. & Podolsky, D. K. Distinct pathways of cell migration and antiapoptotic response to epithelial injury: structure-function analysis of human intestinal trefoil factor. Mol. Cell. Biol. 20, 4680–4690 (2000).
Chadwick, M. P., Westley, B. R. & May, F. E. Homodimerization and hetero-oligomerization of the single-domain trefoil protein pNR-2/pS2 through cysteine 58. Biochem. J. 327, 117–123 (1997).
Prest, S. J., May, F. E. & Westley, B. R. The estrogen-regulated protein, TFF1, stimulates migration of human breast cancer cells. FASEB J. 16, 592–594 (2002).
Beck, S., Schmitt, H., Shizuya, H., Blin, N. & Gott, P. Cloning of contiguous genomic fragments from human chromosome 21 harbouring three trefoil peptide genes. Hum. Genet. 98, 233–235 (1996). Although these authors had previously mapped the TFFs to 21q22.3, this report showed that the three TFFs were contiguously encoded.
Sands, B. E. & Podolsky, D. K. The trefoil peptide family. Annu. Rev. Physiol. 58, 253–273 (1996).
Mashimo, H., Wu, D. -C., Podolsky, D. K. & Fishman, M. C. Impaired defense of intestinal mucosa in mice lacking intestinal trefoil factor. Science 274, 262–265 (1996).
Lefebvre, O. et al. Gastric mucosa abnormalities and tumorigenesis in mice lacking the pS2 trefoil protein. Science 274, 259–262 (1996). References 12 and 13 first showed crucial roles for the TFFs in vivo . Tff3 was shown to be essential for restitution in the mouse (and restitution was also proven to be essential to the health of the animal). Tff1 -null mice ( pS2 -null) displayed gastric adenomas, a surprising finding supported by reports indicating a tumour–suppressor role in humans (see also references 14 and 62).
Wang, T. C. & Goldenring, J. R. Inflammation intersection: gp130 balances gut irritation and stomach cancer. Nature Med. 8, 1080–1082 (2002).
Masiakowski, P. et al. Cloning of cDNA sequences of hormone-regulated genes from the MCF-7 human breast cancer cell line. Nucl. Acids Res. 10, 7895–7903 (1982).
Brown, A. M. C., Jeltsch, J. M., Roberts, M. & Chambon, P. Activation of pS2 gene transcription is a primary response to estrogen in the human breast cancer cell line MCF-7. Proc. Natl Acad. Sci. USA 81, 6344–6348 (1984).
Jakowlew, S. B., Breathnach, R., Jeltsch, J. -M., Masiakowski, P. & Chambon, P. Sequence of the pS2 mRNA induced by estrogen in the human breast cancer cell line MCF-7. Nucl. Acids Res. 12, 2861–2878 (1984).
Rio, M. -C. et al. Breast cancer-associated pS2 protein: synthesis and secretion by normal stomach mucosa. Science 241, 705–708 (1988).
Nunez, A. M., Berry, M., Imler, J. L. & Chambon, P. The 5′ flanking region of the pS2 gene contains a complex enhancer region responsive to oestrogens, epidermal growth factor, a tumour promoter (TPA), the c-Ha-ras oncoprotein and the c-jun protein. EMBO J. 8, 823–829 (1989).
Luqmani, Y. et al. Expression of the pS2 gene in normal, benign and neoplastic human stomach. Int. J. Cancer 15, 806–812 (1989).
Taupin, D. et al. Augmented intestinal trefoil factor (TFF3) and loss of pS2 (TFF1) expression precedes metaplastic differentiation of gastric epithelium. Lab. Invest. 81, 397–408 (2001).
Park, W. S. et al. Somatic mutations of the trefoil factor family 1 gene in gastric cancer. Gastroenterology 119, 691–698 (2000). The first report indicating a potential tumour-suppressor role for TFF1 in gastric cancer.
Jørgensen, K. D., Diamant, B., Jørgensen, K. H. & Thim, L. Pancreatic spasmolytic polypeptide (PSP): III. Pharmacology of a new porcine pancreatic polypeptide with spasmolytic and gastric acid secretion inhibitory effects. Regulatory Peptides 3, 231–243 (1982).
Baker, M. E. Oestrogen-induced pS2 protein is similar to pancreatic spasmolytic polypeptide and the kringle domain. Biochem. J. 253, 307–309 (1988).
Lefebvre, O. et al. The mouse one P-domain (pS2) and two P-domain (raSP) genes exhibit distinct patterns of expression. J. Cell. Biol. 122, 191–198 (1993).
Hanby, A. M. et al. The expression of the trefoil peptides pS2 and human spasmolytic polypeptide (hSP) in gastric metaplasia of the proximal duodenum: implications for the nature of 'gastric metaplasia'. J. Pathol. 169, 355–360 (1993).
Jeffrey, G. P., Oates, P. S., Wang, T. C., Babyatsky, M. W. & Brand, S. J. Spasmolytic polypeptide: a trefoil peptide secreted by rat gastric mucous cells. Gastroenterology 106, 336–345 (1994).
Tomasetto, C. et al. hSP, the domain-duplicated homolog of pS2 protein, is co-expressed with pS2 in stomach but not in breast carcinoma. EMBO J. 9, 407–414 (1990).
Farrell, J. J. et al. TFF2/SP-deficient mice show decreased gastric proliferation, increased acid secretion, and increased susceptibility to NSAID injury. J. Clin. Invest. 109, 193–204 (2002).
Suemori, S., Lynch-Devaney, K. & Podolsky, D. K. Identification and characterisation of rat intestinal trefoil factor: tissue and cell-specific member of the trefoil protein family. Proc. Natl Acad. Sci. USA 88, 11017–11021 (1991).
Podolsky, D. K. et al. Identification of human intestinal trefoil factor. Goblet cell-specific expression of a peptide targeted for apical secretion. J. Biol. Chem. 268, 6694–6702 (1993).
Schwarz, H., Jagla, W., Wiede, A. & Hoffmann, W. Ultrastructural co-localization of TFF3-peptide and oxytocin in the neural lobe of the porcine pituitary. Cell Tissue Res. 305, 411–416 (2001).
dos Santos Silva, E., Ulrich, M., Doring, G., Botzenhart, K. & Gott, P. Trefoil factor family domain peptides in the human respiratory tract. J. Pathol. 190, 133–142 (2000).
Lee, F. D. Pyloric metaplasia in the small intestine. Science 87, 267–279 (1964).
Taylor, P. R. et al. Gastric carcinogenesis in the rat induced by duodenogastric reflux without carcinogens: morphology, mucin histochemistry, polyamine metabolism, and labelling index. Gut 32, 1447–1454 (1991).
Wright, N. A. et al. Epidermal growth factor (EGF/URO) induces expression of regulatory peptides in damaged human gastrointestinal tissues. Science 162, 279–284 (1990).
Wright, N. A. et al. Trefoil peptide gene expression in gastrointestinal epithelial cells in inflammatory bowel disease. Scand. J. Gastroenterol. 193 (Suppl.), 76–82 (1992). This was the first of several clinical and in vivo reports to show rapid upregulation of TFF genes in injured mucosa of the gastrointestinal tract.
Alison, M. R. et al. Experimental ulceration leads to sequential expression of spasmolytic polypeptide, intestinal trefoil factor, epidermal growth factor and transforming growth factor α mRNAs in rat stomach. J. Pathol. 175, 405–414 (1995)
Emerson, B. M. Specificity of gene regulation. Cell 109, 267–270 (2002).
Itoh, H., Inoue, N. & Podolsky, D. K. Goblet-cell-specific transcription of mouse intestinal trefoil factor gene results from collaboration of complex series of positive and negative regulatory elements. Biochem. J. 341, 461–472 (1999).
Ogata, H., Inoue, N. & Podolsky, D. K. Identification of a goblet cell-specific enhancer element in the rat intestinal trefoil factor gene promoter bound by a goblet cell nuclear protein. J. Biol. Chem. 273, 3060–3067 (1998).
Iwakiri, D. & Podolsky D. K. A silencer inhibitor confers specific expression of intestinal trefoil factor in gobletlike cell lines. Am. J. Physiol. Gastrointest. Liver Physiol. 280, G1114–G1123 (2001).
Iwakiri, D. & Podolsky, D. K. Keratinocyte growth factor promotes goblet cell differentiation through regulation of goblet cell silencer inhibitor. Gastroenterology 120, 1372–1380 (2001).
Hahm, K. B. et al. Loss of TGF-β signaling contributes to autoimmune pancreatitis. J. Clin. Invest. 105, 1057–1065 (2000).
Ebert, M. P. et al. Induction of TFF1 gene expression in pancreas overexpressing transforming growth factor α. Gut 45, 105–111 (1999).
Gum, J. R. Jr, Hicks, J. W. & Kim, Y. S. Identification and characterization of the MUC2 (human intestinal mucin) gene 5′-flanking region: promoter activity in cultured cells Biochem. J. 325, 259–267 (1997).
Tytgat, K. M. et al. Biosynthesis of human colonic mucin: Muc2 is the prominent secretory mucin. Gastroenterology 107, 1352–1363 (1994).
Van Seuningen, I., Pigny, P., Perrais, M., Porchet, N. & Aubert, J. P. Transcriptional regulation of the 11p15 mucin genes. Towards new biological tools in human therapy, in inflammatory diseases and cancer? Front. Biosci. 6, D1216–D1234 (2001).
Bulitta, C. J. et al. Autoinduction of the trefoil factor 2 (TFF2) promoter requires an upstream cis-acting element. Biochem. Biophys. Res. Commun. 293, 366–374 (2002).
Ip, Y. T., Poon, D., Stone, D., Granner, D. K. & Chalkley, R. Interaction of a liver-specific factor with an enhancer 4.8 kilobases upstream of the phosphoenolpyruvate carboxykinase gene. Mol. Cell. Biol. 10, 3770–3781 (1990).
Zaret, K. Developmental competence of the gut endoderm: genetic potentiation by GATA and HNF3/forkhead proteins. Dev. Biol. 209, 1–10 (1999).
Beck, S., Sommer, P., Blin, N. & Gott, P. 5′-flanking motifs control cell-specific expression of trefoil factor genes (TFF). Int. J. Mol. Med. 2, 353–361 (1998).
Beck, S., Sommer, P., dos Santos Silva, E., Blin, N. & Gott, P. Hepatocyte nuclear factor 3 (winged helix domain) activates trefoil factor gene TFF1 through a binding motif adjacent to the TATAA box. DNA Cell. Biol. 18, 157–164 (1999).
Ribieras, S., Lefebvre, O., Tomasetto, C. & Rio, M. -C. Mouse trefoil factor genes: genomic organization, sequences and methylation analyses. Gene 266, 67–75 (2001).
Morrisey, E. E., Ip, H. S., Lu, M. M. & Parmacek, M. S. GATA-6: a zinc finger transcription factor that is expressed in multiple cell lineages derived from lateral mesoderm. Dev. Biol. 177, 309–322 (1996).
Molkentin, J. D. The zinc finger-containing transcription factors GATA-4, -5, and -6. Ubiquitously expressed regulators of tissue-specific gene expression. J. Biol. Chem. 275, 38949–38952 (2000).
Al-Azzeh, E. D., Fegert, P., Blin, N. & Gott P. Transcription factor GATA-6 activates expression of gastroprotective trefoil genes TFF1 and TFF2. Biochim. Biophys. Acta 1490, 324–332 (2000).
Sawadogo, M. & Roeder, R. G. Interaction of a gene-specific transcription factor with the adenovirus major late promoter upstream of the TATA box region. Cell 43, 165–175 (1985).
Sirito, M., Lin, Q., Maity, T. & Sawadogo, M. Ubiquitous expression of the 43- and 44-kDa forms of transcription factor USF in mammalian cells. Nucl. Acids Res. 22, 427–433 (1994).
Al-Azzeh, E. et al. Gastroprotective peptide trefoil family 2 gene is activated by upstream stimulating factor but not by c-myc in gastrointestinal cancer cells. Gut 51, 685–690 (2002).
Taupin, D. et al. The trefoil gene family are coordinately expressed immediate-early genes: EGF receptor- and MAP kinase-dependent interregulation. J. Clin. Invest. 103, 31–38 (1999).
Tebbutt, N. C. et al. Reciprocal regulation of gastrointestinal homeostasis by SHP2 and STAT-mediated trefoil gene activation in gp130 mutant mice. Nature Med. 8, 1089–1097 (2002).
Walia, H., Chen, H. Y., Sun, J. M., Holth, L. T. & Davie, J. R. Histone acetylation is required to maintain the unfolded nucleosome structure associated with transcribing DNA. J. Biol. Chem. 273, 14516–14522 (1998).
Tycko, B. Epigenetic gene silencing in cancer. J. Clin. Invest. 105, 401–407 (2000).
Boffa, L. C., Vidali, G., Mann, R. S. & Allfrey, V. G. Suppression of histone deacetylation in vivo and in vitro by sodium butyrate. J. Biol. Chem. 253, 3364–3366 (1978).
Camarero, N., Nadal, A., Barrero, M. J., Haro, D. & Marrero P. F. Histone deacetylase inhibitors stimulate mitochondrial HMG-CoA synthase gene expression via a promoter proximal Sp1 site. Nucl. Acids Res. 31, 693–703 (2003).
Hoshikawa, Y., Kwon, H. J., Yoshida, M., Horinouchi, S. & Beppu, T. Trichostatin A induces morphological changes and gelsolin expression by inhibiting histone deacetylase in human carcinoma cell lines. Exp. Cell. Res. 214, 189–197 (1994).
Tran, C. P., Familari, M., Parker, L. M., Whitehead, R. H. & Giraud, A. S. Short-chain fatty acids inhibit intestinal trefoil factor gene expression in colon cancer cells. Am. J. Physiol. 275, G85–G94 (1998).
Jaenisch, R. & Bird, A. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nature Genet. 33 (Suppl.), 245–254 (2003).
Bird, A. P. & Wolffe, A. P. Methylation-induced repression — belts, braces, and chromatin. Cell 99, 451–454 (1999).
Shiota, K. et al. Epigenetic marks by DNA methylation specific to stem, germ and somatic cells in mice. Genes Cells 7, 961–969 (2002).
Brown, C. B., Boyer, A. S., Runyan, R. B. & Barnett, J. V. Requirement of type III TGF-β receptor for endocardial cell transformation in the heart. Science 283, 2080–2082 (1999).
Wrana, J. L., Attisano, L., Wieser, R., Ventura, F. & Massague, J. Mechanism of activation of the TGF-β receptor. Nature 370, 341–347 (1994).
Kretzschmar, M. & Massague, J. SMADs: mediators and regulators of TGF-β signaling. Curr. Opin. Genet. Dev. 8, 103–111 (1998).
Takahashi, M. et al. Hepatocyte growth factor is the most potent endogenous stimulant of rabbit gastric epithelial cell proliferation and migration in primary culture. J. Clin. Invest. 95, 1994–2003 (1995).
Jiang, W., Hiscox, S., Matsumoto, K. & Nakamura, T. Hepatocyte growth factor/scatter factor, its molecular, cellular and clinical implications in cancer. Crit. Rev. Oncol. Hematol. 29, 209–248 (1999).
Polk, D. B. Epidermal growth factor receptor-stimulated intestinal epithelial cell migration requires phospholipase C activity. Gastroenterology 114, 493–502 (1998).
Wilson, A. J. & Gibson, P. R. Role of epidermal growth factor receptor in basal and stimulated colonic epithelial cell migration in vitro. Exp. Cell. Res. 250, 187–196 (1999).
Klemke, R. L. et al. Regulation of cell motility by mitogen-activated protein kinase. J. Cell. Biol. 137, 481–492 (1997).
Dignass, A., Lynch-Devaney, K., Kindon, H., Thim, L. & Podolsky, D. K. Trefoil peptides promote epithelial migration through a transforming growth factor β-independent pathway. J. Clin. Invest. 94, 376–383 (1994). A robust and reproducible in vitro wound-healing model was used to provide the first demonstration of a motogenic action for TFF (see also reference 120).
Frandsen, E. K. Receptor binding of pancreatic spasmolytic polypeptide in intestinal mucosal cells and membranes. Regul. Pept. 20, 45–52 (1988).
Chinery, R. & Cox, H. M. Immunoprecipitation and characterization of a binding protein specific for the peptide, intestinal trefoil factor. Peptides 16, 49–55 (1995).
Tomasetto, C. et al. pS2/TFF1 interacts directly with the VWFC cysteine-rich domains of mucins. Gastroenterology 118, 70–80 (2000).
Thim, L. & Mortz, E. Isolation and characterization of putative trefoil peptide receptors. Regul. Pept. 90, 61–68 (2000).
Wong, W. M., Poulsom, R. & Wright, N. A. Trefoil peptides. Gut 44, 890–895 (1999).
Thim, L., Madsen, F. & Poulsen, S. S. Effect of trefoil factors on the viscoelastic properties of mucus gels. Eur. J. Clin. Invest. 32, 519–527 (2002).
Andoh, A., Kinoshita, K., Rosenberg, I. & Podolsky, D. K. Intestinal trefoil factor induces decay-accelerating factor expression and enhances the protective activities against complement activation in intestinal epithelial cells. J. Immunol. 167, 3887–3893 (2001).
Liu, D. et al. Phosphorylation of β-catenin and epidermal growth factor receptor by intestinal trefoil factor. Lab. Invest. 77, 557–563 (1997). The first demonstration of rapid signalling (here, transactivation of EGF-receptor and β-catenin) initiated by trefoil factors.
Taupin, D. R., Kinoshita, K. & Podolsky, D. K. Intestinal trefoil factor confers colonic epithelial resistance to apoptosis. Proc. Natl Acad. Sci. USA. 97, 799–804 (2000).
McKenzie, C. et al. Pancreatic spasmolytic polypeptide protects the gastric mucosa but does not inhibit acid secretion or motility. Am. J. Physiol. 273, G112–G117 (1997).
Nobes, C. D., Hall, A. Rho, rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell 81, 53–62 (1995).
Machesky, L. M. & Hall, A. Role of actin polymerization and adhesion to extracellular matrix in Rac- and Rho-induced cytoskeletal reorganization. J. Cell. Biol. 138, 913–926 (1997).
Frisch, S. M. & Screaton, R. A. Anoikis mechanisms. Curr. Opin. Cell. Biol. 13, 555–562 (2001).
Roura, S., Miravet, S., Piedra, J., Garcia de Herreros, A. & Dunach, M. Regulation of E-cadherin/Catenin association by tyrosine phosphorylation. J. Biol. Chem. 274, 36734–36740 (1999).
Behrens, J. et al. Loss of epithelial differentiation and gain of invasiveness correlates with tyrosine phosphorylation of the E-cadherin/β-catenin complex in cells transformed with a temperature-sensitive v-SRC gene. J. Cell. Biol. 120, 757–766 (1993).
Chen, H. C., Appeddu, P. A., Isoda, H. & Guan, J. L. Phosphorylation of tyrosine 397 in focal adhesion kinase is required for binding phosphatidylinositol 3-kinase. J. Biol. Chem. 271, 26329–26334 (1996).
Giancotti, F. G. & Ruoslahti, E. Integrin signaling. Science 285, 1028–1032 (1999).
Emami, S. et al. Induction of scattering and cellular invasion by trefoil peptides in src- and RhoA-transformed kidney and colonic epithelial cells. FASEB J. 15, 351–361 (2001).
Timpson, P., Jones, G. E., Frame, M. C. & Brunton, V. G. Coordination of cell polarization and migration by the Rho family GTPases requires Src tyrosine kinase activity. Curr. Biol. 11, 1836–1846 (2001).
Rath, H. C. et al. Varying cecal bacterial loads influences colitis and gastritis in HLA-B27 transgenic rats. Gastroenterology 116, 310–319 (1999).
Duchmann, R. et al. Tolerance exists towards resident intestinal flora but is broken in active inflammatory bowel disease (IBD). Clin. Exp. Immunol. 102, 448–455 (1995).
Inohara, N., Ogura, Y. & Nunez, G. Nods: a family of cytosolic proteins that regulate the host response to pathogens. Curr. Opin. Microbiol. 5, 76–80 (2002).
Cook, G. A., Familari, M., Thim, L. & Giraud, A. S. The trefoil peptides TFF2 and TFF3 are expressed in rat lymphoid tissues and participate in the immune response. FEBS Lett. 456, 155–159 (1999).
Tran, C. P., Cook, G. A., Yeomans, N. D., Thim, L. & Giraud, A. S. Trefoil peptide TFF2 (spasmolytic polypeptide) potently accelerates healing and reduces inflammation in a rat model of colitis. Gut 44, 636–642 (1999). One of several studies showing that recombinant TFFs (in this case, TFF2) are active as anti-inflammatory and healing drugs.
McCormack, G. et al. Tissue cytokine and chemokine expression in inflammatory bowel disease. Inflamm. Res. 50, 491–495 (2001).
Hurst, S. M. et al. Il-6 and its soluble receptor orchestrate a temporal switch in the pattern of leukocyte recruitment seen during acute inflammation. Immunity 14, 705–714 (2001).
Jones, S. A., Horiuchi, S., Topley, N., Yamamoto, N. & Fuller, G. M. The soluble interleukin 6 receptor: mechanisms of production and implications in disease. FASEB J. 15, 43–58 (2001).
Graness, A., Chwieralski, C. E., Reinhold, D., Thim, L. & Hoffmann W. Protein kinase C and ERK activation are required for TFF-peptide-stimulated bronchial epithelial cell migration and tumor necrosis factor-α-induced interleukin-6 (IL-6) and IL-8 secretion. J. Biol. Chem. 277, 18440–18446 (2002).
Pierce, J. W. et al. Novel inhibitors of cytokine-induced IB α phosphorylation and endothelial cell adhesion molecule expression show anti-inflammatory effects in vivo. J. Biol. Chem. 272, 21096–21103 (1997).
Wang, T. C. et al. Synergistic interaction between hypergastrinemia and Helicobacter infection in a mouse model of gastric carcinoma. Gastroenterology 118, 36–47 (2000).
Schmidt, P. H. et al. Identification of a metaplastic cell lineage associated with human gastric adenocarcinoma. Lab. Invest. 79, 639–646 (1999).
Fujimoto, J. et al. DNA hypermethylation at the pS2 promoter region is associated with early stage of stomach carcinogenesis. Cancer Lett. 149, 125–134 (2000).
Bossenmeyer-Pourie, C. et al. The trefoil factor 1 participates in gastrointestinal cell differentiation by delaying G1–S phase transition and reducing apoptosis. J. Cell. Biol. 157, 761–770 (2002).
Kayademir, T. et al. A novel 25 bp tandem repeat within the human trefoil peptide gene TFF2 in 21q22. 3: polymorphism and mammalian evolution. Eur. J. Hum. Genet. 6, 121–128 (1998).
dos Santos Silva, E. et al. Variable distribution of TFF2 (Spasmolysin) alleles in Europeans does not indicate predisposition to gastric cancer. Hum. Hered. 49, 45–47 (1999).
Uchino, H., Kataoka, H., Itoh, H. & Koono, M. Expression of intestinal trefoil factor mRNA is downregulated during progression of colorectal carcinomas. J. Clin. Pathol. 50, 932–934 (1997).
Taupin, D., Ooi, K., Yeomans, N. & Giraud, A. Conserved expression of intestinal trefoil factor in the human colonic adenoma-carcinoma sequence. Lab. Invest. 75, 25–32 (1996).
Uchino, H., Kataoka, H., Itoh, H., Hamasuna, R. & Koono, M. Overexpression of intestinal trefoil factor in human colon carcinoma cells reduces cellular growth in vitro and in vivo. Gastroenterology 118, 60–69 (2000).
Babyatsky, M. W., deBeaumont, M., Thim, L. & Podolsky, D. K. Oral trefoil peptides protect against ethanol- and indomethacin-induced gastric injury in rats. Gastroenterology 110, 489–497 (1996).
Playford, R. J. et al. Human spasmolytic polypeptide is a cytoprotective agent that stimulates cell migration. Gastroenterology 108, 108–116 (1995).
Potten, C. S., Merritt, A., Hickman, J., Hall, P. & Faranda, A. Characterisation of radiation-induced apoptosis in the small intestine and its biological implications. Int. J. Radiat. Biol. 65, 71–78 (1994).
Farrell, C. L. et al. Keratinocyte growth factor protects mice from chemotherapy and radiation-induced gastrointestinal injury and mortality. Cancer Res. 58, 933–939 (1998).
Farrell, C. L. et al. The effects of keratinocyte growth factor in preclinical models of mucositis. Cell Prolif. 35 (Suppl. 1), 78–85 (2002).
Xian, C. J. et al. Temporal changes in TFF3 expression and jejunal morphology during methotrexate-induced damage and repair. Am. J. Physiol. 277, G785–G795 (1999).
Carr, M. D. 1H NMR-based determination of the secondary structure of porcine pancreatic spasmolytic polypeptide: one of a new family of “trefoil” motif containing cell growth factors. Biochemistry 31, 1998–2004 (1992).
De, A. et al. Crystal structure of a disulfide-linked “trefoil” motif found in a large family of putative growth factors. Proc. Natl Acad. Sci. USA 91, 1084–1088 (1994). References 125 and 126 confirmed the compact molecular TFF structure that was predicted by Thim and co-workers.
Poulsen, S. S., Thulesen, J., Christensen, L., Nexo, E. & Thim, L. Metabolism of oral trefoil factor 2 (TFF2) and the effect of oral and parenteral TFF2 on gastric and duodenal ulcer healing in the rat. Gut 45, 516–522 (1999).
Marchbank, T. et al. Effect of ectopic expression of rat trefoil factor family 3 (intestinal trefoil factor) in the jejunum of transgenic mice. J. Biol. Chem. 276, 24088–24096 (2001).
Marchbank, T., Westley, B. R., May, F. E., Calnan, D. P. & Playford, R. J. Dimerization of human pS2 (TFF1) plays a key role in its protective/healing effects. J. Pathol. 185, 153–158 (1998).
Chinery, R. & Playford, R. J. Combined intestinal trefoil factor and epidermal growth factor is prophylactic against indomethacin-induced gastric damage in the rat. Clin. Sci. 88, 401–403 (1995).
Konturek, P. C. et al. Role of spasmolytic polypeptide in healing of stress-induced gastric lesions in rats. Regul. Pept. 68, 71–79 (1997).
Cook, G. A., Thim, L., Yeomans, N. D. & Giraud, A. S. Oral human spasmolytic polypeptide protects against aspirin-induced gastric injury in rats. J. Gastroenterol. Hepatol. 13, 363–370 (1998).
Efstathiou, J. A. et al. Intestinal trefoil factor controls the expression of the adenomatous polyposis coli–catenin and the E-cadherin–catenin complexes in human colon carcinoma cells. Proc. Natl Acad. Sci. USA 95, 3122–3127 (1998).
Lalani, E. N. et al. Trefoil factor-2, human spasmolytic polypeptide, promotes branching morphogenesis in MCF-7 cells. Lab. Invest. 79, 537–546 (1999).
Kato, K. et al. Effects of growth factors and trefoil peptides on migration and replication in primary oxyntic cultures. Am. J. Physiol. 276, G1105–G1116 (1999).
Oertel, M. et al. Trefoil factor family-peptides promote migration of human bronchial epithelial cells: synergistic effect with epidermal growth factor. Am. J. Respir. Cell Mol. Biol. 25, 418–424 (2001).
Goke, M. N. et al. Trefoil peptides promote restitution of wounded corneal epithelial cells. Exp. Cell. Res. 264, 337–344 (2001).
Tanaka, S., Podolsky, D. K., Engel, E., Guth, P. H. & Kaunitz, J. D. Human spasmolytic polypeptide decreases proton permeation through gastric mucus in vivo and in vitro. Am. J. Physiol. 272, G1473–G1480 (1997).
Kindon, H., Pothoulakis, C., Thim, L., Lynch-Devaney, K. & Podolsky, D. K. Trefoil peptide protection of intestinal epithelial barrier function: cooperative interaction with mucin glycoprotein. Gastroenterology 109, 516–523 (1995).
Calnan, D. P. et al. The trefoil peptide TFF1 inhibits the growth of the human gastric adenocarcinoma cell line AGS. J. Pathol. 188, 312–317 (1999).
Rodrigues, S. et al. Trefoil peptides as proangiogenic factors in vivo and in vitro: implication of cyclooxygenase-2 and EGF receptor signaling. FASEB J. 17, 7–16 (2003).
Playford, R. J. et al. Transgenic mice that overexpress the human trefoil peptide pS2 have an increased resistance to intestinal damage. Proc. Natl Acad. Sci. USA 93, 2137–2142 (1996).
Acknowledgements
Studies in the authors laboratory are supported by the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Glossary
- RESTITUTION
-
The process that promotes epithelial-cell migration to reseal superficial wounds after injury. This involves the rapid disassembly of cell–cell and cell–substratum adhesion, de-differentiation and spreading of surface cells.
- REGENERATION
-
The process of epithelial repair, whereby the normal epithelia is recapitulated. It involves proliferation and differentiation of epithelial cells and restoration of specialized elements.
- BASEMENT MEMBRANE
-
A dense, sheet-like, laminated extracellular matrix that separates epithelia, muscle or other tissues that form connective tissue.
- CONTIG
-
Usually refers to an overlapping set of DNA fragments. Here, we use contig in the context of clusters of DNA in which contiguous series of genes can be found.
- METAPLASIA
-
The conversion of one cell or tissue type into another. Includes transdifferentiation and also conversion between undifferentiated stem cells of different tissues.
- PIT CELLS
-
Columnar mucous cells that line surface invaginations of the gastric mucosa, which are known as gastric pits.
- THECA
-
The apical storage compartment of the goblet cell. It is typically filled with granules of secreted mucins.
- GOBLET CELLS
-
Specialized secretory cells of the mucosa, which are named for their apparent flask-like shape (which is an artefact of tissue fixation). Goblet cells are highly polarized with a basal nucleus, mitochondria, endoplasmic reticulum and Golgi. The body and apex of the cell (theca) is filled with mucin-filled membrane-bound secretory granules.
- BASIC-HELIX–LOOP–HELIX
-
A transcription factor with a basic domain that binds to a hexanucleotide known as the E-box, and a hydrophobic domain (the helix–loop–helix) that allows the formation of homo- and heterodimers. They can also have leucine repeats known as a leucine-zipper motif.
- ULCERATIVE COLITIS
-
An inflammatory disorder of the large intestine that is characterized by loss of surface absorptive cells, often with ulceration and bleeding. This disorder is typically chronic and idiopathic.
- ENTEROCYTES
-
The columnar surface epithelial cells of the intestine. These cells mediate transport and barrier functions.
- PARACRINE
-
Describing, or relating to, a regulatory cell that secretes an agonist into intercellular spaces from which it diffuses to a target cell other than the one that produces it.
- AUTOCRINE
-
Induction of cellular effects by factors that are produced by the same cell in which the effect occurs.
- MOTOGENS
-
Compounds that induce cell motility.
- BASOLATERAL POLE
-
The area of an epithelial cell below the tight junction that seals the apical surface.
- TRANSACTIVATION
-
Cross-communication between signalling pathways, typically involving receptor tyrosine kinases. This is sometimes known as 'signal transactivation', to differentiate it from 'transcriptional transactivation', which results from binding of a transcriptional activator or repressor to a target gene.
- STRESS FIBRES
-
Axial bundles of F-actin that underlie the cell bodies, which are typically induced by the activity of the GTPase RhoA.
- LAMELLIPODIA
-
Flattened, sheet-like structures, which are composed of a crosslinked F-actin meshwork, that project from the surface of a cell. They are often associated with cell migration.
- ANOIKIS
-
Induction of programmed cell death by detachment of cells from the extracellular matrix.
- ADHERENS JUNCTION
-
A cell–cell adhesion complex that is composed of cadherins and catenins that are attached to cytoplasmic actin filaments.
- RHO-FAMILY GTPASES
-
Ras-related GTPases that are involved in controlling the polymerization of actin.
- DOMINANT-NEGATIVE PROTEIN
-
A defective protein that retains interaction abilities and so distorts or competes with normal proteins.
- COMMENSAL
-
Either of two species that live in close association with benefit to one partner but with little or no effect on the other partner.
- MONOCYTES
-
Large leukocytes with horseshoe-shaped nuclei. They derive from pluripotent stem cells and become phagocytic macrophages when they enter the tissues.
- COMPLEMENT
-
A family of plasma proteins that is essential for antimicrobial defence. They function either as enzymes or as binding proteins, and are activated by a cascade of cleavage events after binding of antibody to a foreign particle (classical pathway) or directly by invading micro-organisms (alternative pathway).
- INTEGRINS
-
A large family of heterodimeric transmembrane proteins that act as receptors for cell-adhesion molecules.
- ALLOGRAFT
-
A tissue or organ graft between individuals of the same species who are genetically dissimilar. Such grafts are invariably rejected unless the recipient is immunosuppressed, or the graft is to an immune-privileged site.
- MUCOSITIS
-
Injury and inflammation of gastrointestinal-tract mucosal surfaces that results from chemotherapy or radiotherapy.
- METHOTREXATE
-
A commonly used chemotherapy drug that acts through inhibition of DNA synthesis.
Rights and permissions
About this article
Cite this article
Taupin, D., Podolsky, D. Trefoil factors: initiators of mucosal healing. Nat Rev Mol Cell Biol 4, 721–732 (2003). https://doi.org/10.1038/nrm1203
Issue Date:
DOI: https://doi.org/10.1038/nrm1203
This article is cited by
-
Effect of In Ovo Trace Element Supplementation on Immune-Related Cells of the Small Intestine of Post-hatched Broiler Chicken
Biological Trace Element Research (2023)
-
Integrating TCGA and single-cell sequencing data for colorectal cancer: a 10-gene prognostic risk assessment model
Discover Oncology (2023)
-
Association of functional genetic variants in TFF1 and nephrolithiasis risk in a Chinese population
BMC Urology (2022)
-
Pathological and therapeutic roles of bioactive peptide trefoil factor 3 in diverse diseases: recent progress and perspective
Cell Death & Disease (2022)
-
IGFBP-1 and IGFBP-2 are associated with a decreased pulse-wave velocity in young, healthy adults
BMC Cardiovascular Disorders (2021)