Key Points
-
Brown and beige adipocytes are thermogenic fat cells that are highly specialized in dissipating chemical energy in the form of heat. There is great hope that these cells can be targeted therapeutically to combat obesity, insulin resistance and type 2 diabetes.
-
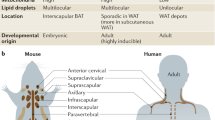
Brown adipocytes develop in distinctive developmental depots of brown adipose tissue (BAT) and have a relatively stable thermogenic phenotype. These cells are poised for heat production in response to various stimuli, including catecholamines that are secreted by sympathetic nerves in BAT on cold exposure.
-
Beige adipocytes are uncoupling protein 1 (UCP1)-expressing and thermogenically competent adipocytes that form in white adipose tissue (WAT) depots in response to various stimuli, including cold exposure or β3-adrenergic agonists. The beige phenotype of WAT is flexible, and the maintenance of beige cells requires ongoing stimulation.
-
Beige adipocytes can arise from adipogenic precursor cells in WAT through de novo differentiation or through the direct conversion of mature unilocular white-like adipocytes.
-
Brown and beige fat cells express certain transcription factors, such as early B-cell factor 2 (EBF2), PR domain zinc finger protein 16 (PRDM16), interferon regulatory factor 4 (IRF4) and zinc finger protein 516 (ZFP516), that cooperate with the general adipogenic factors peroxisome proliferator-activated receptor-γ (PPARγ) and the CCAAT/enhancer-binding proteins (C/EBPs) to drive brown adipocyte differentiation and thermogenic gene programming. ZFP423 acts in white adipocytes to suppress EBF2 and maintain white fat fate.
-
Type 2 cytokine signalling and alternative macrophage activation play a crucial part in regulating both brown fat thermogenesis and beige fat biogenesis. Alternatively activated macrophages secrete catecholamines in WAT to promote browning.
Abstract
Brown and beige adipocytes expend chemical energy to produce heat and are therefore important in regulating body temperature and body weight. Brown adipocytes develop in discrete and relatively homogenous depots of brown adipose tissue, whereas beige adipocytes are induced to develop in white adipose tissue in response to certain stimuli — notably, exposure to cold. Fate-mapping analyses have identified progenitor populations that give rise to brown and beige fat cells, and have revealed unanticipated cell-lineage relationships between vascular smooth muscle cells and beige adipocytes, and between skeletal muscle cells and brown fat. In addition, non-adipocyte cells in adipose tissue, including neurons, blood vessel-associated cells and immune cells, have crucial roles in regulating the differentiation and function of brown and beige fat.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fedorenko, A., Lishko, P. V. & Kirichok, Y. Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell 151, 400–413 (2012).
Cannon, B. & Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 (2004).
Cao, Y. Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat. Rev. Drug Discov. 9, 107–115 (2010).
Harms, M. & Seale, P. Brown and beige fat: development, function and therapeutic potential. Nat. Med. 19, 1252–1263 (2013).
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
Saito, M. et al. High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 58, 1526–1531 (2009).
Virtanen, K. A. et al. Functional brown adipose tissue in healthy adults. N. Engl. J. Med. 360, 1518–1525 (2009).
Nedergaard, J., Bengtsson, T. & Cannon, B. Unexpected evidence for active brown adipose tissue in adult humans. Am. J. Physiol. Endocrinol. Metab. 293, E444–E452 (2007).
Cypess, A. M. et al. Anatomical localization, gene expression profiling and functional characterization of adult human neck brown fat. Nat. Med. 19, 635–639 (2013).
Jespersen, N. Z. et al. A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans. Cell Metab. 17, 798–805 (2013).
Sharp, L. Z. et al. Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS ONE 7, e49452 (2012).
Wu, J. et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 150, 366–376 (2012). In this work, the authors demonstrate that beige adipocytes are a distinctive cell type that has a different molecular signature from classic brown fat or white fat cells. Importantly, human BAT depots are identified as having a beige rather than a brown fat profile.
Enerback, S. et al. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature 387, 90–94 (1997). Here, the authors show that UCP1 is genetically required for cold-induced adaptive thermogenesis and cold-tolerance in mice. The Ucp1 -null mice generated in this work have been widely studied in many laboratories around the world.
Rothwell, N. J. & Stock, M. J. A role for brown adipose tissue in diet-induced thermogenesis. Nature 281, 31–35 (1979).
Feldmann, H. M., Golozoubova, V., Cannon, B. & Nedergaard, J. UCP1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Cell Metab. 9, 203–209 (2009).
Lowell, B. B. et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature 366, 740–742 (1993).
Cederberg, A. et al. FOXC2 is a winged helix gene that counteracts obesity, hypertriglyceridemia, and diet-induced insulin resistance. Cell 106, 563–573 (2001).
Dempersmier, J. et al. Cold-inducible Zfp516 activates UCP1 transcription to promote browning of white fat and development of brown fat. Mol. Cell 57, 235–246 (2015).
Kopecky, J., Clarke, G., Enerback, S., Spiegelman, B. & Kozak, L. P. Expression of the mitochondrial uncoupling protein gene from the aP2 gene promoter prevents genetic obesity. J. Clin. Invest. 96, 2914–2923 (1995).
Qiang, L. et al. Brown remodeling of white adipose tissue by SirT1-dependent deacetylation of Pparγ. Cell 150, 620–632 (2012).
Seale, P. et al. Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J. Clin. Invest. 121, 96–105 (2011).
Stanford, K. I. et al. Brown adipose tissue regulates glucose homeostasis and insulin sensitivity. J. Clin. Invest. 123, 215–223 (2013).
van Marken Lichtenbelt, W. D. et al. Cold-activated brown adipose tissue in healthy men. N. Engl. J. Med. 360, 1500–1508 (2009).
Cypess, A. M. et al. Activation of human brown adipose tissue by a β3-adrenergic receptor agonist. Cell Metab. 21, 33–38 (2015). This study reports that human BAT thermogenesis can be pharmacologically stimulated by a β3-andrenergic agonist and that this increases energy expenditure. This is an important proof-of- concept that BAT-targeted therapies could be a promising approach for reducing metabolic disease.
Yoneshiro, T. et al. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Invest. 123, 3404–3408 (2013).
Claussnitzer, M. et al. FTO obesity variant circuitry and adipocyte browning in humans. N. Engl. J. Med. 373, 895–907 (2015).
Inagaki, T., Sakai, J. & Kajimura, S. Transcriptional and epigenetic control of brown and beige adipose cell fate and function. Nat. Rev. Mol. Cell Biol. 17, 480–495 (2016).
Klaus, S., Ely, M., Encke, D. & Heldmaier, G. Functional assessment of white and brown adipocyte development and energy metabolism in cell culture. Dissociation of terminal differentiation and thermogenesis in brown adipocytes. J. Cell Sci. 108, 3171–3180 (1995).
Ohno, H., Shinoda, K., Spiegelman, B. M. & Kajimura, S. PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab. 15, 395–404 (2012).
Petrovic, N., Shabalina, I. G., Timmons, J. A., Cannon, B. & Nedergaard, J. Thermogenically competent nonadrenergic recruitment in brown preadipocytes by a PPARγ agonist. Am. J. Physiol. Endocrinol. Metab. 295, E287–E296 (2008).
Bartesaghi, S. et al. Thermogenic activity of UCP1 in human white fat-derived beige adipocytes. Mol. Endocrinol. 29, 130–139 (2015).
Elabd, C. et al. Human multipotent adipose-derived stem cells differentiate into functional brown adipocytes. Stem Cells 27, 2753–2760 (2009).
Xue, B. et al. Genetic variability affects the development of brown adipocytes in white fat but not in interscapular brown fat. J. Lipid Res. 48, 41–51 (2007).
Li, Y., Bolze, F., Fromme, T. & Klingenspor, M. Intrinsic differences in BRITE adipogenesis of primary adipocytes from two different mouse strains. Biochim. Biophys. Acta 1841, 1345–1352 (2014).
Guerra, C., Koza, R. A., Yamashita, H., Walsh, K. & Kozak, L. P. Emergence of brown adipocytes in white fat in mice is under genetic control. Effects on body weight and adiposity. J. Clin. Invest. 102, 412–420 (1998). An important study showing that there is large mouse-strain-dependent variation in the induction of beige adipocytes with relatively little effect on classical BAT. In addition, beige fat levels among inbred and recombinant strains were highly correlated with the ability of β3-andrenergic agonists to decrease body weight.
Kazak, L. et al. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 163, 643–655 (2015). This study identifies a novel mechanism for UCP1-independent thermogenesis in beige adipocytes.
Wang, W. et al. Ebf2 is a selective marker of brown and beige adipogenic precursor cells. Proc. Natl Acad. Sci. USA 111, 14466–14471 (2014). The helix–loop–helix transcription factor EBF2 is identified as a specific marker gene and protein, and a functional regulator of brown and beige fat precursor cells.
Lidell, M. E. et al. Evidence for two types of brown adipose tissue in humans. Nat. Med. 19, 631–634 (2013).
Atit, R. et al. β-Catenin activation is necessary and sufficient to specify the dorsal dermal fate in the mouse. Dev. Biol. 296, 164–176 (2006).
Seale, P. et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature 454, 961–967 (2008). This study shows that brown adipocytes and muscle have a common or similar developmental origin. PRDM16 was identified as a transcriptional factor that drives brown fat differentiation and suppresses muscle differentiation.
Lepper, C. & Fan, C. M. Inducible lineage tracing of Pax7-descendant cells reveals embryonic origin of adult satellite cells. Genesis 48, 424–436 (2010).
Sanchez-Gurmaches, J. et al. PTEN loss in the Myf5 lineage redistributes body fat and reveals subsets of white adipocytes that arise from Myf5 precursors. Cell Metab. 16, 348–362 (2012).
Walden, T. B., Timmons, J. A., Keller, P., Nedergaard, J. & Cannon, B. Distinct expression of muscle-specific microRNAs (myomirs) in brown adipocytes. J. Cell. Physiol. 218, 444–449 (2009).
Timmons, J. A. et al. Myogenic gene expression signature establishes that brown and white adipocytes originate from distinct cell lineages. Proc. Natl Acad. Sci. USA 104, 4401–4406 (2007).
Forner, F. et al. Proteome differences between brown and white fat mitochondria reveal specialized metabolic functions. Cell Metab. 10, 324–335 (2009).
Ohno, H., Shinoda, K., Ohyama, K., Sharp, L. Z. & Kajimura, S. EHMT1 controls brown adipose cell fate and thermogenesis through the PRDM16 complex. Nature 504, 163–167 (2013).
Park, J. H. et al. A multifunctional protein, EWS, is essential for early brown fat lineage determination. Dev. Cell 26, 393–404 (2013).
Trajkovski, M., Ahmed, K., Esau, C. C. & Stoffel, M. MyomiR-133 regulates brown fat differentiation through Prdm16. Nat. Cell Biol. 14, 1330–1335 (2012).
Yin, H. et al. MicroRNA-133 controls brown adipose determination in skeletal muscle satellite cells by targeting Prdm16. Cell Metab. 17, 210–224 (2013).
Sun, L. et al. Mir193b-365 is essential for brown fat differentiation. Nat. Cell Biol. 13, 958–965 (2011).
Harms, M. J. et al. Prdm16 is required for the maintenance of brown adipocyte identity and function in adult mice. Cell Metab. 19, 593–604 (2014).
Tseng, Y. H. et al. New role of bone morphogenetic protein 7 in brown adipogenesis and energy expenditure. Nature 454, 1000–1004 (2008).
Nabeshima, Y. et al. Myogenin gene disruption results in perinatal lethality because of severe muscle defect. Nature 364, 532–535 (1993).
Rajakumari, S. et al. EBF2 determines and maintains brown adipocyte identity. Cell Metab. 17, 562–574 (2013).
Shao, M. et al. Zfp423 maintains white adipocyte identity through suppression of the beige cell thermogenic gene program. Cell Metab. 23, 1167–1184 (2016).
Bukowiecki, L., Collet, A. J., Follea, N., Guay, G. & Jahjah, L. Brown adipose tissue hyperplasia: a fundamental mechanism of adaptation to cold and hyperphagia. Am. J. Physiol. 242, E353–E359 (1982).
Bronnikov, G., Houstek, J. & Nedergaard, J. β-Adrenergic, cAMP-mediated stimulation of proliferation of brown fat cells in primary culture. Mediation via β1 but not via β3 adrenoceptors. J. Biol. Chem. 267, 2006–2013 (1992).
Lee, M. W. et al. Activated type 2 innate lymphoid cells regulate beige fat biogenesis. Cell 160, 74–87 (2015).
Lee, Y. H., Petkova, A. P., Konkar, A. A. & Granneman, J. G. Cellular origins of cold-induced brown adipocytes in adult mice. FASEB J. 29, 286–299 (2015).
Schulz, T. J. et al. Identification of inducible brown adipocyte progenitors residing in skeletal muscle and white fat. Proc. Natl Acad. Sci. USA 108, 143–148 (2011).
Petrovic, N. et al. Chronic peroxisome proliferator-activated receptor γ (PPARγ) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J. Biol. Chem. 285, 7153–7164 (2010).
Ussar, S. et al. ASC-1, PAT2, and P2RX5 are cell surface markers for white, beige, and brown adipocytes. Sci. Transl Med. 6, 247ra103 (2014).
Stine, R. R. et al. EBF2 promotes the recruitment of beige adipocytes in white adipose tissue. Mol. Metab. 5, 57–65 (2016).
Long, J. Z. et al. A smooth muscle-like origin for beige adipocytes. Cell Metab. 19, 810–820 (2014).
Berry, D. C., Jiang, Y. & Graff, J. M. Mouse strains to study cold-inducible beige progenitors and beige adipocyte formation and function. Nat. Commun. 7, 10184 (2016).
Chang, L. et al. Loss of perivascular adipose tissue on peroxisome proliferator-activated receptor-γ deletion in smooth muscle cells impairs intravascular thermoregulation and enhances atherosclerosis. Circulation 126, 1067–1078 (2012).
Jiang, Y., Berry, D. C., Tang, W. & Graff, J. M. Independent stem cell lineages regulate adipose organogenesis and adipose homeostasis. Cell Rep. 9, 1007–1022 (2014).
Vishvanath, L. et al. Pdgfrβ+ mural preadipocytes contribute to adipocyte hyperplasia induced by high-fat-diet feeding and prolonged cold exposure in adult mice. Cell Metab. 23, 350–359 (2016).
McDonald, M. E. et al. Myocardin-related transcription factor A regulates conversion of progenitors to beige adipocytes. Cell 160, 105–118 (2015). This study reports that BMP7 promotes beige adipogenesis and suppresses smooth muscle programming in mesenchymal stem cells by regulating RHO-associated protein kinase (ROCK) signalling and reducing the activity of MRTFA, a transcription factor. Genetic loss of Mrtfa in mice enhances beige adipocyte differentiation.
Yadav, H. et al. Protection from obesity and diabetes by blockade of TGF-β/Smad3 signaling. Cell Metab. 14, 67–79 (2011).
Ghorbani, M. & Himms-Hagen, J. Appearance of brown adipocytes in white adipose tissue during CL 316,243-induced reversal of obesity and diabetes in Zucker fa/fa rats. Int. J. Obes Relat. Metab. Disord. 21, 465–475 (1997).
Himms-Hagen, J. et al. Multilocular fat cells in WAT of CL-316243-treated rats derive directly from white adipocytes. Am. J. Physiol. Cell Physiol. 279, C670–C681 (2000).
Vitali, A. et al. The adipose organ of obesity-prone C57BL/6J mice is composed of mixed white and brown adipocytes. J. Lipid Res. 53, 619–629 (2012).
Barbatelli, G. et al. The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am. J. Physiol. Endocrinol. Metab. 298, E1244–E1253 (2010).
Wang, Q. A., Tao, C., Gupta, R. K. & Scherer, P. E. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat. Med. 19, 1338–1344 (2013). This study reports the development of a genetic system called AdipoChaser in mice to examine the fate of mature adipocytes in vivo . A key result in this paper is that most cold-induced beige adipocytes in inguinal WAT do not arise from pre-existing mature fat cells in the tissue. This suggests that de novo differentiation of resident precursor cells is the main mechanism of beige fat formation.
Rosenwald, M., Perdikari, A., Rulicke, T. & Wolfrum, C. Bi-directional interconversion of brite and white adipocytes. Nat. Cell Biol. 15, 659–667 (2013). This study reports that beige adipocytes lose Ucp1 expression and become lipid-replete white adipocyte-like cells after warm adaptation. These cells can be reactivated to regain their multilocular beige phenotype and UCP1 expression after another round of cold stimulation, demonstrating the phenotypic plasticity of beige adipocytes.
Cao, W. et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell. Biol. 24, 3057–3067 (2004).
Puigserver, P. et al. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92, 829–839 (1998).
Uldry, M. et al. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 3, 333–341 (2006).
Kong, X. et al. IRF4 is a key thermogenic transcriptional partner of PGC-1α. Cell 158, 69–83 (2014).
Kleiner, S. et al. Development of insulin resistance in mice lacking PGC-1α in adipose tissues. Proc. Natl Acad. Sci. USA 109, 9635–9640 (2012).
Lee, Y. H., Petkova, A. P., Mottillo, E. P. & Granneman, J. G. In vivo identification of bipotential adipocyte progenitors recruited by β3-adrenoceptor activation and high-fat feeding. Cell Metab. 15, 480–491 (2012).
Bartness, T. J., Shrestha, Y. B., Vaughan, C. H., Schwartz, G. J. & Song, C. K. Sensory and sympathetic nervous system control of white adipose tissue lipolysis. Mol. Cell. Endocrinol. 318, 34–43 (2010).
Morrison, S. F., Madden, C. J. & Tupone, D. Central control of brown adipose tissue thermogenesis. Front. Endocrinol. (Lausanne) 3, 00005 (2012).
Ryu, V., Garretson, J. T., Liu, Y., Vaughan, C. H. & Bartness, T. J. Brown adipose tissue has sympathetic-sensory feedback circuits. J. Neurosci. 35, 2181–2190 (2015).
Vaughan, C. H. & Bartness, T. J. Anterograde transneuronal viral tract tracing reveals central sensory circuits from brown fat and sensory denervation alters its thermogenic responses. Am. J. Physiol. Regul. Integr. Comp. Physiol. 302, R1049–R1058 (2012).
Rothwell, N. J. & Stock, M. J. Effects of denervating brown adipose tissue on the responses to cold, hyperphagia and noradrenaline treatment in the rat. J. Physiol. 355, 457–463 (1984).
Silva, J. E. & Larsen, P. R. Adrenergic activation of triiodothyronine production in brown adipose tissue. Nature 305, 712–713 (1983).
Takahashi, A., Shimazu, T. & Maruyama, Y. Importance of sympathetic nerves for the stimulatory effect of cold exposure on glucose utilization in brown adipose tissue. Jpn J. Physiol. 42, 653–664 (1992).
Murano, I., Barbatelli, G., Giordano, A. & Cinti, S. Noradrenergic parenchymal nerve fiber branching after cold acclimatisation correlates with brown adipocyte density in mouse adipose organ. J. Anat. 214, 171–178 (2009).
Nisoli, E., Tonello, C., Benarese, M., Liberini, P. & Carruba, M. O. Expression of nerve growth factor in brown adipose tissue: implications for thermogenesis and obesity. Endocrinology 137, 495–503 (1996).
Sornelli, F., Fiore, M., Chaldakov, G. N. & Aloe, L. Adipose tissue-derived nerve growth factor and brain-derived neurotrophic factor: results from experimental stress and diabetes. Gen. Physiol. Biophys. 28, 179–183 (2009).
Rosell, M. et al. Brown and white adipose tissues: intrinsic differences in gene expression and response to cold exposure in mice. Am. J. Physiol. Endocrinol. Metab. 306, E945–E964 (2014).
Nguyen, K. D. et al. Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis. Nature 480, 104–108 (2011). These authors discover a novel and crucial role for macrophage-derived catecholamines in mediating BAT thermogenesis.
Asano, A., Kimura, K. & Saito, M. Cold-induced mRNA expression of angiogenic factors in rat brown adipose tissue. J. Vet. Med. Sci. 61, 403–409 (1999).
Asano, A., Morimatsu, M., Nikami, H., Yoshida, T. & Saito, M. Adrenergic activation of vascular endothelial growth factor mRNA expression in rat brown adipose tissue: implication in cold-induced angiogenesis. Biochem. J. 328, 179–183 (1997).
Fredriksson, J. M., Nikami, H. & Nedergaard, J. Cold-induced expression of the VEGF gene in brown adipose tissue is independent of thermogenic oxygen consumption. FEBS Lett. 579, 5680–5684 (2005).
Xue, Y. et al. Hypoxia-independent angiogenesis in adipose tissues during cold acclimation. Cell Metab. 9, 99–109 (2009).
Bagchi, M. et al. Vascular endothelial growth factor is important for brown adipose tissue development and maintenance. FASEB J. 27, 3257–3271 (2013).
Elias, I. et al. Adipose tissue overexpression of vascular endothelial growth factor protects against diet-induced obesity and insulin resistance. Diabetes 61, 1801–1813 (2012).
Sun, K. et al. Brown adipose tissue derived VEGF-A modulates cold tolerance and energy expenditure. Mol. Metab. 3, 474–483 (2014).
Sung, H. K. et al. Adipose vascular endothelial growth factor regulates metabolic homeostasis through angiogenesis. Cell Metab. 17, 61–72 (2013).
Shimizu, I. et al. Vascular rarefaction mediates whitening of brown fat in obesity. J. Clin. Invest. 124, 2099–2112 (2014).
Fitzgibbons, T. P. et al. Similarity of mouse perivascular and brown adipose tissues and their resistance to diet-induced inflammation. Am. J. Physiol. Heart Circ. Physiol. 301, H1425–H1437 (2011).
Takaoka, M. et al. Endovascular injury induces rapid phenotypic changes in perivascular adipose tissue. Arterioscler Thromb. Vasc. Biol. 30, 1576–1582 (2010).
Lumeng, C. N. & Saltiel, A. R. Inflammatory links between obesity and metabolic disease. J. Clin. Invest. 121, 2111–2117 (2011).
Olefsky, J. M. & Glass, C. K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 72, 219–246 (2010).
Cawthorn, W. P., Heyd, F., Hegyi, K. & Sethi, J. K. Tumour necrosis factor-α inhibits adipogenesis via a β-catenin/TCF4(TCF7L2)-dependent pathway. Cell Death Differ. 14, 1361–1373 (2007).
Vidal, C. et al. Interferon γ inhibits adipogenesis in vitro and prevents marrow fat infiltration in oophorectomized mice. Stem Cells 30, 1042–1048 (2012).
Nisoli, E. et al. Tumor necrosis factor α mediates apoptosis of brown adipocytes and defective brown adipocyte function in obesity. Proc. Natl Acad. Sci. USA 97, 8033–8038 (2000).
Lumeng, C. N., DelProposto, J. B., Westcott, D. J. & Saltiel, A. R. Phenotypic switching of adipose tissue macrophages with obesity is generated by spatiotemporal differences in macrophage subtypes. Diabetes 57, 3239–3246 (2008).
Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 332, 243–247 (2011).
Qiu, Y. et al. Eosinophils and type 2 cytokine signaling in macrophages orchestrate development of functional beige fat. Cell 157, 1292–1308 (2014).
Rao, R. R. et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 157, 1279–1291 (2014).
Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Takatsu, K. & Nakajima, H. IL-5 and eosinophilia. Curr. Opin. Immunol. 20, 288–294 (2008).
Brestoff, J. R. et al. Group 2 innate lymphoid cells promote beiging of white adipose tissue and limit obesity. Nature 519, 242–246 (2015).
Cipolletta, D. et al. PPAR-γ is a major driver of the accumulation and phenotype of adipose tissue Treg cells. Nature 486, 549–553 (2012).
Medrikova, D. et al. Brown adipose tissue harbors a distinct sub-population of regulatory T cells. PLoS ONE 10, e0118534 (2015).
Hankir, M. K. et al. Differential effects of Roux-en-Y gastric bypass surgery on brown and beige adipose tissue thermogenesis. Metabolism 64, 1240–1249 (2015).
Neinast, M. D. et al. Activation of natriuretic peptides and the sympathetic nervous system following Roux-en-Y gastric bypass is associated with gonadal adipose tissues browning. Mol. Metab. 4, 427–436 (2015).
Nicholls, D. G. The physiological regulation of uncoupling proteins. Biochim. Biophys. Acta 1757, 459–466 (2006).
Ricquier, D. Uncoupling protein 1 of brown adipocytes, the only uncoupler: a historical perspective. Front. Endocrinol. (Lausanne) 2, 85 (2011).
Chouchani, E. T. et al. Mitochondrial ROS regulate thermogenic energy expenditure and sulfenylation of UCP1. Nature 532, 112–116 (2016).
Wang, G. X. et al. The brown fat-enriched secreted factor Nrg4 preserves metabolic homeostasis through attenuation of hepatic lipogenesis. Nat. Med. 20, 1436–1443 (2014).
Rahman, S. et al. Inducible brown adipose tissue, or beige fat, is anabolic for the skeleton. Endocrinology 154, 2687–2701 (2013).
Bartelt, A. et al. Brown adipose tissue activity controls triglyceride clearance. Nat. Med. 17, 200–205 (2011).
Sul, H. S. Minireview: Pref-1: role in adipogenesis and mesenchymal cell fate. Mol. Endocrinol. 23, 1717–1725 (2009).
Chau, Y. Y. et al. Visceral and subcutaneous fat have different origins and evidence supports a mesothelial source. Nat. Cell Biol. 16, 367–375 (2014).
Xue, R. et al. Clonal analyses and gene profiling identify genetic biomarkers of the thermogenic potential of human brown and white preadipocytes. Nat. Med. 21, 760–768 (2015).
Acknowledgements
This work was supported by an American Heart Association postdoctoral fellowship to W.W. and US National Institute of Diabetes and Digestive and Kidney Diseases grant 5R01DK10300802 to P.S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
Summary of lineage tracing studies. (PDF 2865 kb)
Glossary
- Sympathetic nervous system
-
(SNS). Regulates bodily function (unconsciously) by connecting the brain to internal organs through nerves in the spinal column. Presynaptic neurons in the spinal cord secrete acetylcholine to activate postsynaptic neurons that innervate target organs and tissues. The postsynaptic neurons secrete noradrenaline, which activates β-adrenergic receptors on various cell types, including adipocytes.
- Noradrenaline
-
A neurotransmitter in the catecholamine family that is secreted by sympathetic neurons to stimulate various responses, including adaptive thermogenesis in brown and beige fat.
- Adrenergic receptors
-
A class of G protein-coupled cell surface receptors that are activated by catecholamines.
- Adaptive thermogenesis
-
A facultative process by which animals produce heat only in response to stimuli, such as cold exposure or high-fat diet. Muscle shivering and uncoupled respiration in brown and beige fat are major mechanisms.
- Thiazolidinediones
-
A class of synthetic high-affinity agonists for the nuclear hormone receptor peroxisome proliferator-activated receptor-γ (PPARγ). Thiazolidinediones improve insulin action in mice and humans through activation of PPARγ in adipocytes and other cell types.
- Homeobox gene
-
A family of genes that encode proteins that are characterized by a DNA sequence called the homeobox. Members of this gene family have crucial roles in patterning and morphogenesis.
- Dermomyotome
-
The mesodermal domain of the somite that is fated to differentiate into the skeletal muscle (myotome) and dermis (dermatome).
- Helix–loop–helix transcription factor
-
A transcription factor family characterized by a structural motif. These factors are known to have important roles in various developmental processes.
- Mural cells
-
Cells that are closely associated with the vasculature, such as vascular smooth muscle cells or pericytes.
- Catecholamine
-
A class of naturally occurring chemicals, including noradrenaline and adrenaline, that act as neurotransmitters.
- M1-like macrophages
-
Macrophage populations that have a pro-inflammatory profile and are characterized by secretion of interferon-γ, tumour necrosis factor and interleukin-1.
- M2-like macrophages
-
Alternatively activated macrophage populations that are characterized by secretion of arginase and interleukin-10, and that have important roles in tissue repair and homeostasis.
- Eosinophils
-
Specialized white blood cells that are characterized by granules that contain histamine and other chemical mediators. They play an important part in anti-parasite immunity.
- Lipolysis
-
Hydrolysis of lipids into their component free fatty acids and glycerol.
- Met-encephalin
-
A type of encephalin, which is a five-amino-acid peptide that is classically known to regulate nociception by binding to opioid receptors. Met-encephalin contains methionine, whereas Leu-encephalin contains leucine.
- Adipokine
-
Cytokine or other protein secreted by adipocytes.
Rights and permissions
About this article
Cite this article
Wang, W., Seale, P. Control of brown and beige fat development. Nat Rev Mol Cell Biol 17, 691–702 (2016). https://doi.org/10.1038/nrm.2016.96
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm.2016.96
This article is cited by
-
Characteristic and fate determination of adipose precursors during adipose tissue remodeling
Cell Regeneration (2023)
-
Age-dependent Pdgfrβ signaling drives adipocyte progenitor dysfunction to alter the beige adipogenic niche in male mice
Nature Communications (2023)
-
Epitranscriptomics in metabolic disease
Nature Metabolism (2023)
-
Blockage of PPARγ T166 phosphorylation enhances the inducibility of beige adipocytes and improves metabolic dysfunctions
Cell Death & Differentiation (2023)
-
Obesity and prostate cancer — microenvironmental roles of adipose tissue
Nature Reviews Urology (2023)