Key Points
-
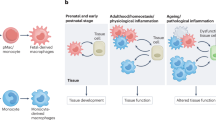
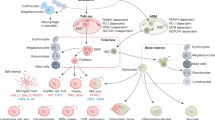
In steady-state conditions, blood monocyte subsets form in a developmental sequence with mouse LY6Chi monocytes giving rise to LY6Clow monocytes.
-
LY6Clow monocytes act within the vasculature by surveying the vessel wall for injury and LY6Chi monocytes are recruited to sites of inflammation and, after extravasation, differentiate in the tissue into cells with dendritic cell and macrophage activities.
-
Intestinal macrophages are continuously renewed by LY6Chi monocytes and thus differ from most other embryonic-derived tissue macrophages. LY6Chi monocytes are probably recruited in response to the tonic low inflammatory signals that are provided by the commensal gut microbiota. Other tissue macrophages that are derived from monocytes include dermal and heart macrophages.
-
Specific tissue-resident macrophage populations in mice are seeded before birth. At a very early stage, embryonic precursors — such as yolk sac-derived macrophages and fetal liver-derived monocytes — give rise to tissue macrophages that persist and maintain the macrophage pool into adulthood, without being superseded by adult bone marrow-derived or blood monocyte-derived cells.
-
Both yolk sac-derived macrophages and fetal liver-derived monocytes give rise to fetal macrophages. Their relative contribution to adult tissue macrophage populations varies between tissues and remains to be fully elucidated.
-
In adults, tissue macrophages maintain themselves by self-renewal at low levels in the steady state. Importantly, the ability of tissue macrophages to proliferate is enhanced during inflammation.
Abstract
Monocytes and macrophages have crucial and distinct roles in tissue homeostasis and immunity, but they also contribute to a broad spectrum of pathologies and are thus attractive therapeutic targets. Potential intervention strategies that aim to manipulate these cells will require an in-depth understanding of their origins and the mechanisms that ensure their homeostasis. Recent evidence shows that monocytes do not substantially contribute to most tissue macrophage populations in the steady state or during certain types of inflammation. Rather, most tissue macrophage populations in mice are derived from embryonic precursors, are seeded before birth and can maintain themselves in adults by self-renewal. In this Review, we discuss the evidence that has dramatically changed our understanding of monocyte and macrophage development, and the maintenance of these cells in the steady state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
van Furth, R. & Cohn, Z. A. The origin and kinetics of mononuclear phagocytes. J. Exp. Med. 128, 415–435 (1968).
Wynn, T. A., Chawla, A. & Pollard, J. W. Macrophage biology in development, homeostasis and disease. Nature 496, 445–455 (2013).
Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nature Immunol. 14, 986–995 (2013).
Williams, M. J. Drosophila hemopoiesis and cellular immunity. J. Immunol. 178, 4711–4716 (2007).
Cros, J. et al. Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity 33, 375–386 (2010).
Etzrodt, M. et al. Regulation of monocyte functional heterogeneity by miR-146a and Relb. Cell Rep. 1, 317–324 (2012).
Ingersoll, M. A. et al. Comparison of gene expression profiles between human and mouse monocyte subsets. Blood 115, e10–e19 (2010).
Mildner, A. et al. Mononuclear phagocyte miRNome analysis identifies miR-142 as critical regulator of murine dendritic cell homeostasis. Blood 121, 1016–1027 (2013).
van Furth, R. & Sluiter, W. Distribution of blood monocytes between a marginating and a circulating pool. J. Exp. Med. 163, 474–479 (1986).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013). References 11 and 12 establish that most adult tissue-resident macrophages do not rely on adult monocyte input.
Liu, K. et al. In vivo analysis of dendritic cell development and homeostasis. Science 324, 392–397 (2009).
Varol, C. et al. Intestinal lamina propria dendritic cell subsets have different origin and functions. Immunity 31, 502–512 (2009).
Schlitzer, A. et al. IRF4 transcription factor-dependent CD11b+ dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 38, 970–983 (2013).
Avraham-Davidi, I. et al. On-site education of VEGF-recruited monocytes improves their performance as angiogenic and arteriogenic accessory cells. J. Exp. Med. 210, 2611–2625 (2013).
Jakubzick, C. et al. Minimal differentiation of classical monocytes as they survey steady-state tissues and transport antigen to lymph nodes. Immunity 39, 599–610 (2013).
Cecchini, M. G. et al. Role of colony stimulating factor-1 in the establishment and regulation of tissue macrophages during postnatal development of the mouse. Development 120, 1357–1372 (1994).
Dai, X.-M. et al. Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 99, 111–120 (2002).
Wiktor-Jedrzejczak, W. & Gordon, S. Cytokine regulation of the macrophage (M phi) system studied using the colony stimulating factor-1-deficient op/op mouse. Physiol. Rev. 76, 927–947 (1996).
Robbins, C. S. et al. Extramedullary hematopoiesis generates Ly-6Chigh monocytes that infiltrate atherosclerotic lesions. Circulation 125, 364–374 (2012).
Fogg, D. K. et al. A clonogenic bone marrow progenitor specific for macrophages and dendritic cells. Science 311, 83–87 (2006).
Varol, C. et al. Monocytes give rise to mucosal, but not splenic, conventional dendritic cells. J. Exp. Med. 204, 171–180 (2007).
Ginhoux, F. et al. The origin and development of nonlymphoid tissue CD103+ DCs. J. Exp. Med. 206, 3115–3130 (2009).
Hettinger, J. et al. Origin of monocytes and macrophages in a committed progenitor. Nature Immunol. 14, 821–830 (2013). References 22, 23 and 25 define MDPs and their potential to give rise to monocytes. Reference 25 establishes monocyte ontogeny in vivo.
Naik, S. H. et al. Diverse and heritable lineage imprinting of early haematopoietic progenitors. Nature 496, 229–232 (2013).
Jaitin, D. A. et al. Massively parallel single-cell RNA-seq for marker-free decomposition of tissues into cell types. Science 343, 776–779 (2014).
Passlick, B., Flieger, D. & Ziegler-Heitbrock, H. W. Identification and characterization of a novel monocyte subpopulation in human peripheral blood. Blood 74, 2527–2534 (1989). This is the first report that defines human monocyte subsets according to their expression of CD14 and CD16, thus establishing the concept of monocyte heterogeneity.
Ziegler-Heitbrock, L. & Hofer, T. P. J. Toward a refined definition of monocyte subsets. Front. Immunol. 4, 23 (2013).
Jung, S. et al. Analysis of fractalkine receptor CX3CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Geissmann, F., Jung, S. & Littman, D. R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003).
Palframan, R. T. et al. Inflammatory chemokine transport and presentation in HEV: a remote control mechanism for monocyte recruitment to lymph nodes in inflamed tissues. J. Exp. Med. 194, 1361–1373 (2001). References 31 and 32 highlight, for the first time, monocyte heterogeneity in mouse blood and pioneer the study of monocyte functions in vivo by introducing CX 3 CR1GFP reporter mice.
Auffray, C. et al. Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior. Science 317, 666–670 (2007). This hallmark study represents a breakthrough in our understanding of LY6Clow monocytes and the functional differences between monocyte subsets.
Carlin, L. M. et al. Nr4a1-dependent Ly6Clow monocytes monitor endothelial cells and orchestrate their disposal. Cell 153, 362–375 (2013).
Nguyen, K. D. et al. Circadian gene Bmal1 regulates diurnal oscillations of Ly6Chi inflammatory monocytes. Science 341, 1483–1488 (2013).
Shechter, R. et al. Infiltrating blood-derived macrophages are vital cells playing an anti-inflammatory role in recovery from spinal cord injury in mice. PLoS Med. 6, e1000113 (2009).
Serbina, N. V., Salazar-Mather, T. P., Biron, C. A., Kuziel, W. A. & Pamer, E. G. TNF/iNOS-producing dendritic cells mediate innate immune defense against bacterial infection. Immunity 19, 59–70 (2003).
Drevets, D. A. et al. The Ly-6Chigh monocyte subpopulation transports Listeria monocytogenes into the brain during systemic infection of mice. J. Immunol. 172, 4418–4424 (2004).
Tamoutounour, S. et al. Origins and functional specialization of macrophages and of conventional and monocyte-derived dendritic cells in mouse skin. Immunity 39, 925–938 (2013).
Leuschner, F. et al. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nature Biotech. 29, 1005–1010 (2011).
Liu, K. et al. Origin of dendritic cells in peripheral lymphoid organs of mice. Nature Immunol. 8, 578–583 (2007).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nature Neurosci. 10, 1538–1543 (2007). This study elegantly highlights the dynamics between resident microglial cells and infiltrating monocytes, and their distinct functional relevance.
Serbina, N. V. & Pamer, E. G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nature Immunol. 7, 311–317 (2006).
Leirião, P., del Fresno, C. & Ardavín, C. Monocytes as effector cells: activated Ly-6Chigh mouse monocytes migrate to the lymph nodes through the lymph and cross-present antigens to CD8+ T cells. Eur. J. Immunol. 42, 2042–2051 (2012).
Jaensson, E. et al. Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J. Exp. Med. 205, 2139–2149 (2008).
Bogunovic, M. et al. Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 (2009). Together with reference 14, this study establishes that intestinal macrophages are derived from LY6Chi monocytes and are thus ontogenetically distinct from most other tissue macrophage compartments.
Tamoutounour, S. et al. CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur. J. Immunol. 42, 3150–3166 (2012).
Rivollier, A., He, J., Kole, A., Valatas, V. & Kelsall, B. L. Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J. Exp. Med. 209, 139–155 (2012).
Zigmond, E. et al. Ly6Chi monocytes in the inflamed colon give rise to proinflammatory effector cells and migratory antigen-presenting cells. Immunity 37, 1076–1090 (2012).
A-Gonzalez, N. et al. The nuclear receptor LXRα controls the functional specialization of splenic macrophages. Nature Immunol. 14, 831–839 (2013).
Probst, H. C. et al. Histological analysis of CD11c–DTR/GFP mice after in vivo depletion of dendritic cells. Clin. Exp. Immunol. 141, 398–404 (2005).
Zigmond, E. & Jung, S. Intestinal macrophages: well educated exceptions from the rule. Trends Immunol. 34, 162–168 (2013).
Guilliams, M. et al. Alveolar macrophages develop from fetal monocytes that differentiate into long-lived cells in the first week of life via GM-CSF. J. Exp. Med. 210, 1977–1992 (2013).
Tagliani, E. et al. Coordinate regulation of tissue macrophage and dendritic cell population dynamics by CSF-1. J. Exp. Med. 208, 1901–1916 (2011).
Epelman, S. et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 40, 91–104 (2014).
Arnold, L. et al. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J. Exp. Med. 204, 1057–1069 (2007).
Mildner, A. et al. CCR2+Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132, 2487–2500 (2009).
Ajami, B., Bennett, J. L., Krieger, C., McNagny, K. M. & Rossi, F. M. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nature Neurosci. 14, 1142–1149 (2011).
Lessner, S. M., Prado, H. L., Waller, E. K. & Galis, Z. S. Atherosclerotic lesions grow through recruitment and proliferation of circulating monocytes in a murine model. Am. J. Pathol. 160, 2145–2155 (2002).
Swirski, F. K. et al. Monocyte accumulation in mouse atherogenesis is progressive and proportional to extent of disease. Proc. Natl Acad. Sci. USA 103, 10340–10345 (2006).
Landsman, L. et al. CX3CR1 is required for monocyte homeostasis and atherogenesis by promoting cell survival. Blood 113, 963–972 (2009).
Robbins, C. S. et al. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nature Med. 19, 1166–1172 (2013).
Orkin, S. H. & Zon, L. I. Hematopoiesis: an evolving paradigm for stem cell biology. Cell 132, 631–644 (2008).
Cumano, A. & Godin, I. Ontogeny of the hematopoietic system. Annu. Rev. Immunol. 25, 745–785 (2007).
Kumaravelu, P. et al. Quantitative developmental anatomy of definitive haematopoietic stem cells/long-term repopulating units (HSC/RUs): role of the aorta–gonad–mesonephros (AGM) region and the yolk sac in colonisation of the mouse embryonic liver. Development 129, 4891–4899 (2002).
Medvinsky, A. & Dzierzak, E. Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86, 897–906 (1996).
Tavian, M. & Peault, B. Embryonic development of the human hematopoietic system. Int. J. Dev. Biol. 49, 243–250 (2005).
Ginhoux, F., Lim, S., Hoeffel, G., Low, D. & Huber, T. Origin and differentiation of microglia. Front. Cell Neurosci. 7, 45 (2013).
Ginhoux, F. & Merad, M. Ontogeny and homeostasis of Langerhans cells. Immunol. Cell Biol. 88, 387–392 (2010).
Sorokin, S. P., Hoyt, R. F. J., Blunt, D. G. & McNelly, N. A. Macrophage development: II. Early ontogeny of macrophage populations in brain, liver, and lungs of rat embryos as revealed by a lectin marker. Anat. Rec. 232, 527–550 (1992).
Mizoguchi, S., Takahashi, K., Takeya, M., Naito, M. & Morioka, T. Development, differentiation, and proliferation of epidermal Langerhans cells in rat ontogeny studied by a novel monoclonal antibody against epidermal Langerhans cells, RED-1. J. Leukocyte Biol. 52, 52–61 (1992).
Enzan, H. Electron microscopic studies of macrophages in early human yolk sacs. Acta Pathol. Jpn 36, 49–64 (1986).
Migliaccio, G. et al. Human embryonic hemopoiesis. Kinetics of progenitors and precursors underlying the yolk sac–liver transition. J. Clin. Invest. 78, 51–60 (1986).
Takahashi, K., Yamamura, F. & Naito, M. Differentiation, maturation, and proliferation of macrophages in the mouse yolk sac: a light-microscopic, enzyme-cytochemical, immunohistochemical, and ultrastructural study. J. Leukoc. Biol. 45, 87–96 (1989).
Takahashi, K. & Naito, M. Development, differentiation, and proliferation of macrophages in the rat yolk sac. Tissue Cell 25, 351–362 (1993).
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
McGrath, K. E., Koniski, A. D., Malik, J. & Palis, J. Circulation is established in a stepwise pattern in the mammalian embryo. Blood 101, 1669–1676 (2003).
Naito, M., Takahashi, K. & Nishikawa, S. Development, differentiation, and maturation of macrophages in the fetal mouse liver. J. Leukoc. Biol. 48, 27–37 (1990).
Hoeffel, G. et al. Adult Langerhans cells derive predominantly from embryonic fetal liver monocytes with a minor contribution of yolk sac-derived macrophages. J. Exp. Med. 209, 1167–1181 (2012). References 76, 77 and 80 establish a contribution of embryonic yolk sac progenitors to adult tissue-resident macrophages.
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nature Neurosci. 16, 273–280 (2013).
Gomez Perdiguero, E. & Geissmann, F. Myb-independent macrophages: a family of cells that develops with their tissue of residence and is involved in its homeostasis. Cold Spring Harb. Symp. Quant. Biol. http://dx.doi.org/10.1101/sqb.2013.78.020032 (2013).
Bertrand, J. Y. et al. Three pathways to mature macrophages in the early mouse yolk sac. Blood 106, 3004–3011 (2005).
Palis, J., Robertson, S., Kennedy, M., Wall, C. & Keller, G. Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 126, 5073–5084 (1999).
Godin, I. E., Garcia-Porrero, J. A., Coutinho, A., Dieterlen-Lièvre, F. & Marcos, M. A. Para-aortic splanchnopleura from early mouse embryos contains B1a cell progenitors. Nature 364, 67–70 (1993).
Medvinsky, A. L., Samoylina, N. L., Müller, A. M. & Dzierzak, E. A. An early pre-liver intraembryonic source of CFU-S in the developing mouse. Nature 364, 64–67 (1993).
Swiers, G., Rode, C., Azzoni, E. & de Bruijn, M. F. A short history of hemogenic endothelium. Blood Cells Mol. Dis. 51, 206–212 (2013).
Böiers, C. et al. Lymphomyeloid contribution of an immune-restricted progenitor emerging prior to definitive hematopoietic stem cells. Cell Stem Cell 13, 535–548 (2013).
Jenkins, S. J. et al. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 332, 1284–1288 (2011). This is a seminal study that establishes the local proliferation capacity of terminally differentiated tissue macrophages upon inflammation.
Jenkins, S. J. et al. IL-4 directly signals tissue-resident macrophages to proliferate beyond homeostatic levels controlled by CSF-1. J. Exp. Med. 210, 2477–2491 (2013).
Ghigo, C. et al. Multicolor fate mapping of Langerhans cell homeostasis. J. Exp. Med. 210, 1657–1664 (2013).
Seré, K. et al. Two distinct types of Langerhans cells populate the skin during steady state and inflammation. Immunity 37, 905–916 (2012).
Merad, M. et al. Langerhans cells renew in the skin throughout life under steady-state conditions. Nature Immunol. 3, 1135–1141 (2002). This is a seminal study that establishes the unique homeostasis of epidermal Langerhans cells and shows that these cells self-maintain in situ independently of any blood input.
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nature Neurosci. 10, 1544–1553 (2007).
Amano, S. U. et al. Local proliferation of macrophages contributes to obesity-associated adipose tissue inflammation. Cell. Metab. 19, 162–171 (2014).
MacDonald, K. P. A. et al. An antibody against the colony-stimulating factor 1 receptor depletes the resident subset of monocytes and tissue- and tumor-associated macrophages but does not inhibit inflammation. Blood 116, 3955–3963 (2010).
Hashimoto, D. et al. Pretransplant CSF-1 therapy expands recipient macrophages and ameliorates GVHD after allogeneic hematopoietic cell transplantation. J. Exp. Med. 208, 1069–1082 (2011).
Hanna, R. N. et al. The transcription factor NR4A1 (Nur77) controls bone marrow differentiation and the survival of Ly6C− monocytes. Nature Immunol. 12, 778–785 (2011).
Hanna, R. N. et al. NR4A1 (Nur77) deletion polarizes macrophages toward an inflammatory phenotype and increases atherosclerosis. Circ. Res. 110, 416–427 (2012).
Randolph, G. J., Inaba, K., Robbiani, D. F., Steinman, R. M. & Muller, W. A. Differentiation of phagocytic monocytes into lymph node dendritic cells in vivo. Immunity 11, 753–761 (1999).
Rotta, G. et al. Lipopolysaccharide or whole bacteria block the conversion of inflammatory monocytes into dendritic cells in vivo. J. Exp. Med. 198, 1253–1263 (2003).
Kool, M. et al. Alum adjuvant boosts adaptive immunity by inducing uric acid and activating inflammatory dendritic cells. J. Exp. Med. 205, 869–882 (2008).
Hohl, T. M. et al. Inflammatory monocytes facilitate adaptive CD4 T cell responses during respiratory fungal infection. Cell Host Microbe 6, 470–481 (2009).
Plantinga, M. et al. Conventional and monocyte-derived CD11b+ dendritic cells initiate and maintain T helper 2 cell-mediated immunity to house dust mite allergen. Immunity 38, 322–335 (2013).
Schreiber, H. A. et al. Intestinal monocytes and macrophages are required for T cell polarization in response to Citrobacter rodentium. J. Exp. Med. 210, 2025–2039 (2013).
Nakano, H. et al. Blood-derived inflammatory dendritic cells in lymph nodes stimulate acute T helper type 1 immune responses. Nature Immunol. 10, 394–402 (2009).
Leon, B., Lopez-Bravo, M. & Ardavin, C. Monocyte-derived dendritic cells formed at the infection site control the induction of protective T helper 1 responses against Leishmania. Immunity 26, 519–531 (2007).
Samstein, M. et al. Essential yet limited role for CCR2+ inflammatory monocytes during Mycobacterium tuberculosis-specific T cell priming. Elife 2, e01086 (2013).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nature Neurosci. 16, 1618–1626 (2013). This is a recent study that demonstrates how investigation of the ontogeny of monocytes and tissue macrophages can be used to gain insights into the different functions of these cells.
Herbomel, P., Thisse, B. & Thisse, C. Ontogeny and behaviour of early macrophages in the zebrafish embryo. Development 126, 3735–3745 (1999).
Cuadros, M. A. & Navascues, J. Early origin and colonization of the developing central nervous system by microglial precursors. Prog. Brain Res. 132, 51–59 (2001).
Cuadros, M. A., Moujahid, A., Martin-Partido, G. & Navascues, J. Microglia in the mature and developing quail brain as revealed by a monoclonal antibody recognizing hemopoietic cells. Neurosci. Lett. 148, 11–14 (1992).
Herbomel, P., Thisse, B. & Thisse, C. Zebrafish early macrophages colonize cephalic mesenchyme and developing brain, retina, and epidermis through a M-CSF receptor-dependent invasive process. Dev. Biol. 238, 274–288 (2001).
Cuadros, M. A. & Navascues, J. The origin and differentiation of microglial cells during development. Prog. Neurobiol. 56, 173–189 (1998).
Kurz, H. & Christ, B. Embryonic CNS macrophages and microglia do not stem from circulating, but from extravascular precursors. Glia 22, 98–102 (1998).
Koushik, S. V. et al. Targeted inactivation of the sodium-calcium exchanger (Ncx1) results in the lack of a heartbeat and abnormal myofibrillar organization. FASEB J. 15, 1209–1211 (2001).
de Jong, J. L. O. & Zon, L. I. Use of the zebrafish system to study primitive and definitive hematopoiesis. Annu. Rev. Genet. 39, 481–501 (2005).
Daneman, R., Zhou, L., Kebede, A. A. & Barres, B. A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 468, 562–566 (2010).
Eidsmo, L. et al. Differential migration of epidermal and dermal dendritic cells during skin infection. J. Immunol. 182, 3165–3172 (2009).
Ginhoux, F. et al. Langerhans cells arise from monocytes in vivo. Nature Immunol. 7, 265–273 (2006).
Merad, M. et al. Depletion of host Langerhans cells before transplantation of donor alloreactive T cells prevents skin graft-versus-host disease. Nature Med. 10, 510–517 (2004).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Mononuclear phagocyte system
-
(MPS). A term introduced by Van Furth that encompasses myeloid immune cells other than polymorphonuclear granulocytes and initially included monocytes and macrophages, and then, following their discovery, dendritic cells.
- microRNA
-
(miRNA). A class of short endogenous non-coding RNAs that modulate the proteome through binding to complementary mRNAs and repressing translational initiation or by inducing mRNA degradation. This post-transcriptional regulation of gene expression depends on an imperfect match of short 'seed' sequences with their target mRNA and hence each miRNA has potential to suppress multiple targets.
- Classical dendritic cells
-
(cDCs). A term introduced by Ralph Steinman that should be reserved for short-lived, FLT3 ligand-dependent cells with migratory capability and a potential to efficiently stimulate naive T cells.
- Macrophage and DC precursor
-
(MDP). A bone marrow-resident LIN−CD117+CD135+ clonotypic precursor with plasmacytoid dendritic cell (pDC), classical DC (cDC) and monocyte potential.
- Common monocyte progenitor
-
(cMoP). A bone marrow-resident LIN−CD117+ precursor with monocyte potential that is the direct progeny of macrophage and dendritic cell precursors (MDPs).
- Intravital microscopy
-
A technique that is based on the combination of photonics, particularly multi-photon microscopy, and new molecular and genetic tools that enables imaging of tissues of living animals and is yielding insights into the mechanisms controlling immune cell motility and interactions in tissues.
- Necrosis
-
A form of uncontrolled cell death caused by factors that are external to the cell or tissue — such as infection, toxins or trauma — that result in the unregulated destruction of cellular components.
- Anticipatory inflammation
-
A recently coined term that describes the finding in mice that the abundance of circulating LY6Chi monocytes is under circadian control, which might have evolved to prepare the organism for insults.
- Cre–loxP
-
A site-specific recombination system in which two short DNA sequences (loxP sites) are engineered to flank the target DNA and the expression of Cre recombinase leads to excision of the intervening 'floxed' sequence. Depending on the type of promoter, Cre recombinase can be expressed at specific times during development or in specific sets of cells.
- Kupffer cells
-
Specialized macrophages of the liver that reside within the lumen of the liver sinusoids.
- Langerhans cells
-
These cells were classically considered as a dendritic cell (DC) population that resides in the skin epidermis and are now considered as a DC–macrophage hybrid, as they are unique among DC subsets in that they arise from embryonic progenitors and are radio-resistant, long-lived and independent of FLT3 ligand. They also show considerable transcriptome overlap with macrophages.
- Parabiotic mice
-
A model in which pairs of mice are surgically joined by cutaneous vascular anastomoses so that they have a common blood circulation while maintaining separate organs and tissues. A few weeks after the initiation of parabiosis, a steady state is achieved in these mice in which circulating leukocyte populations are stably comprised of cells that are derived from both animals.
- Microglial cells
-
This cell type constitutes a radio-resistant, highly ramified macrophage population of the central nervous system that is derived from primitive yolk sac macrophages and contributes to brain development and homeostasis by synaptic pruning and debris clearance.
- Primed homeostasis
-
The unique homeostatic 'alert state' of the intestinal mucosa, which is constantly exposed to microbial products.
- Blood–brain barrier
-
A semipermeable cellular structure that separates the blood from the cerebrospinal fluid, and constitutes a barrier to the passage of cells, particles and large molecules into the brain parenchyma.
- Experimental autoimmune encephalomyelitis
-
(EAE). A well-studied mouse model of the human disease multiple sclerosis, which is characterized by extensive infiltration of the central nervous system by inflammatory cells and involves the activation of pathogenic myelin-specific TH1 or TH17 cells.
- Primitive haematopoiesis
-
Blood cell production that originates from the yolk sac and constitutes the first wave of haematopoiesis. It is transient and starts from murine embryonic day 7.0 in the extra-embryonic yolk sac where, shortly after the onset of gastrulation, the mesodermal layer develops into structures that are referred to as blood islands that predominantly produce nucleated erythrocytes and macrophages, but not lymphocytes.
- Haematopoietic stem cells
-
(HSCs). Cells that have both the ability to generate all types of haematopoietic cell (multipotentiality) and to replace themselves (self-renewal) during the whole lifespan of an individual.
- Aorta–gonads–mesonephros
-
(AGM). The embryonic site where definitive haematopoietic stem cells (HSCs) are produced. It comprises the aorta, and the developing reproductive and excretory (mesonephros) systems. Within this haemogenic site, HSCs are concentrated in the aorta region.
- Definitive haematopoiesis
-
A process that occurs in the fetal liver in the embryo, during which blood cells are produced from haematopoietic stem cells (HSCs) that are generated initially in the splanchnopleura or aorta–gonads–mesonephros (AGM), and then in the bone marrow of adults. It is responsible for the generation of the multipotent HSCs with multi-lineage haematopoietic potential, which arise within the AGM from embryonic day 10.5.
- Induced pluripotent stem cells
-
Also known as iPS cells (or iPSCs), these are a type of pluripotent stem cell that can be generated directly from adult cells by specific genetic reprogramming.
Rights and permissions
About this article
Cite this article
Ginhoux, F., Jung, S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat Rev Immunol 14, 392–404 (2014). https://doi.org/10.1038/nri3671
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3671
This article is cited by
-
Restoration of aberrant gene expression of monocytes in systemic lupus erythematosus via a combined transcriptome-reversal and network-based drug repurposing strategy
BMC Genomics (2023)
-
The kynurenic acid analog SZR104 induces cytomorphological changes associated with the anti-inflammatory phenotype in cultured microglia
Scientific Reports (2023)
-
Molecular characterization of human cytomegalovirus infection with single-cell transcriptomics
Nature Microbiology (2023)
-
Fibrosis induced by resident macrophages has divergent roles in pancreas inflammatory injury and PDAC
Nature Immunology (2023)
-
Monocyte subsets and monocyte-related chemokines in Takayasu arteritis
Scientific Reports (2023)