Key Points
-
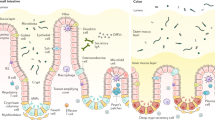
Intestinal epithelial cells (IECs) provide a physical and biochemical barrier that segregates host tissue and commensal bacteria to maintain intestinal homeostasis. Secretory IECs support this function through the secretion of mucins and antimicrobial peptides.
-
IECs maintain specialized pathways for the delivery of luminal antigens and bacteria to lamina propria-resident antigen-presenting cells.
-
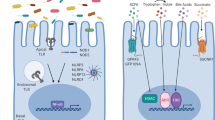
Microbial signals are recognized by IECs to promote intestinal homeostasis. Host–commensal microorganism interactions not only support tissue repair in the setting of injury or acute inflammation but also promote the development of intestinal cancers during chronic inflammation.
-
IECs possess mechanisms for maintaining altered responsiveness to microbial signals that allow for tolerance to continuous exposure to commensal bacteria.
-
IECs convey microbial signals to mucosal immune cells and promote the coordination of appropriate immune responses against commensal bacteria and enteric pathogens. Interactions between antigen-presenting cells and IECs regulate B cell and T cell responses that act on the intestinal barrier.
-
Both innate and adaptive effector cell function and homeostasis are influenced by IEC-derived signals. This allows IECs to locally regulate immune response at the intestinal barrier and promote the maintenance of intestinal homeostasis.
Abstract
The abundance of innate and adaptive immune cells that reside together with trillions of beneficial commensal microorganisms in the mammalian gastrointestinal tract requires barrier and regulatory mechanisms that conserve host–microbial interactions and tissue homeostasis. This homeostasis depends on the diverse functions of intestinal epithelial cells (IECs), which include the physical segregation of commensal bacteria and the integration of microbial signals. Hence, IECs are crucial mediators of intestinal homeostasis that enable the establishment of an immunological environment permissive to colonization by commensal bacteria. In this Review, we provide a comprehensive overview of how IECs maintain host–commensal microbial relationships and immune cell homeostasis in the intestine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mankertz, J. & Schulzke, J.-D. Altered permeability in inflammatory bowel disease: pathophysiology and clinical implications. Curr. Opin. Gastroenterol. 23, 379–383 (2007).
Brenchley, J. M. et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nature Med. 12, 1365–1371 (2006).
Sandler, N. G. et al. Host response to translocated microbial products predicts outcomes of patients with HBV or HCV infection. Gastroenterology 141, 1220–1230 (2011).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Amar, J. et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: molecular mechanisms and probiotic treatment. EMBO Mol. Med. 3, 559–572 (2011).
Wen, L. et al. Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature 455, 1109–1113 (2008).
Lee, Y. K., Menezes, J. S., Umesaki, Y. & Mazmanian, S. K. Colloquium paper: Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 108, 4615–4622 (2010).
Berer, K. et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 479, 538–541 (2011).
Wu, H.-J. et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827 (2010).
Kamada, N., Seo, S.-U., Chen, G. Y. & Núñez, G. Role of the gut microbiota in immunity and inflammatory disease. Nature Rev. Immunol. 13, 321–335 (2013).
Hooper, L. V., Littman, D. R. & Macpherson, A. J. Interactions between the microbiota and the immune system. Science 336, 1268–1273 (2012).
Honda, K. & Littman, D. R. The microbiome in infectious disease and inflammation. Annu. Rev. Immunol. 30, 759–795 (2012).
Maynard, C. L., Elson, C. O., Hatton, R. D. & Weaver, C. T. Reciprocal interactions of the intestinal microbiota and immune system. Nature 489, 231–241 (2012).
Moon, C. & Stappenbeck, T. S. Viral interactions with the host and microbiota in the intestine. Curr. Opin. Immunol. 24, 405–410 (2012).
Crosnier, C., Stamataki, D. & Lewis, J. Organizing cell renewal in the intestine: stem cells, signals and combinatorial control. Nature Rev. Genet. 7, 349–359 (2006).
Van der Flier, L. G. & Clevers, H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu. Rev. Physiol. 71, 241–260 (2009).
Kim, Y. S. & Ho, S. B. Intestinal goblet cells and mucins in health and disease: recent insights and progress. Curr. Gastroenterol. Rep. 12, 319–330 (2010).
Gallo, R. L. & Hooper, L. V. Epithelial antimicrobial defence of the skin and intestine. Nature Rev. Immunol. 12, 503–516 (2012).
Johansson, M. E. V. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
Velcich, A. et al. Colorectal cancer in mice genetically deficient in the mucin Muc2. Science 295, 1726–1729 (2002).
Van der Sluis, M. et al. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131, 117–129 (2006).
Taupin, D. R., Kinoshita, K. & Podolsky, D. K. Intestinal trefoil factor confers colonic epithelial resistance to apoptosis. Proc. Natl Acad. Sci. USA 97, 799–804 (2000).
Dignass, A., Lynch-Devaney, K., Kindon, H., Thim, L. & Podolsky, D. K. Trefoil peptides promote epithelial migration through a transforming growth factor β-independent pathway. J. Clin. Invest. 94, 376–383 (1994).
Artis, D. et al. RELMβ/FIZZ2 is a goblet cell-specific immune-effector molecule in the gastrointestinal tract. Proc. Natl Acad. Sci. USA 101, 13596–13600 (2004).
Nair, M. G. et al. Goblet cell-derived resistin-like molecule β augments CD4+ T cell production of IFN-γ and infection-induced intestinal inflammation. J. Immunol. 181, 4709–4715 (2008).
Bevins, C. L. & Salzman, N. H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nature Rev. Microbiol. 9, 356–368 (2011).
Mukherjee, S. et al. Antibacterial membrane attack by a pore-forming intestinal C-type lectin. Nature 505, 103–107 (2014).
Darmoul, D. & Ouellette, A. J. Positional specificity of defensin gene expression reveals Paneth cell heterogeneity in mouse small intestine. Am. J. Physiol. 271, G68–G74 (1996).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011). In this study, the antimicrobial protein REGIII γ is identified as being necessary for the physical separation of commensal bacteria from the surface of the small intestinal epithelium, thus limiting the activation of the intestinal immune response.
Meyer-Hoffert, U. et al. Secreted enteric antimicrobial activity localises to the mucus surface layer. Gut 57, 764–771 (2008).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nature Genet. 39, 207–211 (2007).
Rioux, J. D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nature Genet. 39, 596–604 (2007). References 31 and 32 report the association between genetic variants in the gene for the autophagy protein ATG16L1 and Crohn's disease susceptibility, establishing a Crohn's disease-specific genetic link between IBD and the autophagy pathway.
Cadwell, K. et al. A key role for autophagy and the autophagy gene Atg16l1 in mouse and human intestinal Paneth cells. Nature 456, 259–263 (2008).
Kaser, A. et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134, 743–756 (2008).
Brandl, K. et al. Enhanced sensitivity to DSS colitis caused by a hypomorphic Mbtps1 mutation disrupting the ATF6-driven unfolded protein response. Proc. Natl Acad. Sci. USA 106, 3300–3305 (2009).
Cadwell, K. et al. Virus-plus-susceptibility gene interaction determines Crohn's disease gene Atg16L1 phenotypes in intestine. Cell 141, 1135–1145 (2010). In this study, the interaction between environmental exposures and genetic susceptibility is shown to determine the penetrance of disease in mouse models of intestinal inflammation.
Khor, B., Gardet, A. & Xavier, R. J. Genetics and pathogenesis of inflammatory bowel disease. Nature 474, 307–317 (2011).
Benjamin, J. L., Sumpter, R. Jr, Levine, B. & Hooper, L. V. Intestinal epithelial autophagy is essential for host defense against invasive bacteria. Cell Host Microbe 13, 723–734 (2013).
Adolph, T. E. et al. Paneth cells as a site of origin for intestinal inflammation. Nature 503, 272–276 (2013). This study demonstrates interactions between the regulation of the UPR and the autophagy pathway in Paneth cells and supports a model in which alterations in these two responses regulate the development of Crohn's disease.
Johansen, F.-E. & Kaetzel, C. S. Regulation of the polymeric immunoglobulin receptor and IgA transport: new advances in environmental factors that stimulate pIgR expression and its role in mucosal immunity. Mucosal Immunol. 4, 598–602 (2011).
Johansen, F.-E. et al. Absence of epithelial immunoglobulin a transport, with increased mucosal leakiness, in polymeric immunoglobulin receptor/secretory component–deficient mice. J. Exp. Med. 190, 915–922 (1999).
Shulzhenko, N. et al. Crosstalk between B lymphocytes, microbiota and the intestinal epithelium governs immunity versus metabolism in the gut. Nature Med. 17, 1585–1593 (2011). In this study, a compensatory response of IECs in the absence of adaptive IgA directed against commensal bacteria is characterized by the engagement of immune pathways and dysregulation of lipid metabolism.
Suzuki, K. et al. Aberrant expansion of segmented filamentous bacteria in IgA-deficient gut. Proc. Natl Acad. Sci. USA 101, 1981–1986 (2004).
Mabbott, N. A., Donaldson, D. S., Ohno, H., Williams, I. R. & Mahajan, A. Microfold (M) cells: important immunosurveillance posts in the intestinal epithelium. Mucosal Immunol. 6, 666–677 (2013).
Mowat, A. M. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Rev. Immunol. 3, 331–341 (2003).
Hase, K. et al. Uptake through glycoprotein 2 of FimH+ bacteria by M cells initiates mucosal immune response. Nature 462, 226–230 (2009).
McDole, J. R. et al. Goblet cells deliver luminal antigen to CD103+ dendritic cells in the small intestine. Nature 483, 345–349 (2012). This report shows that goblet cells can transport soluble luminal antigens to lamina propria DCs, implicating a pathway by which IECs directly mediate antigen delivery to immune cells in addition to M cell-mediated transport.
Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunol. 2, 361–367 (2001).
Chieppa, M., Rescigno, M., Huang, A. Y. C. & Germain, R. N. Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J. Exp. Med. 203, 2841–2852 (2006). This paper demonstrates a role for epithelial cell TLR signalling in promoting DC sampling of antigens from the intestinal lumen.
Shan, M. et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science 342, 447–453 (2013).
Abreu, M. T. Toll-like receptor signalling in the intestinal epithelium: how bacterial recognition shapes intestinal function. Nature Rev. Immunol. 10, 131–144 (2010).
Chen, G. Y. & Núñez, G. Inflammasomes in intestinal inflammation and cancer. Gastroenterology 141, 1986–1999 (2011).
Elinav, E., Henao-Mejia, J. & Flavell, R. A. Integrative inflammasome activity in the regulation of intestinal mucosal immune responses. Mucosal Immunol. 6, 4–13 (2013).
Li, X.-D. et al. Mitochondrial antiviral signaling protein (MAVS) monitors commensal bacteria and induces an immune response that prevents experimental colitis. Proc. Natl Acad. Sci. USA 108, 17390–17395 (2011).
Broquet, A. H., Hirata, Y., McAllister, C. S. & Kagnoff, M. F. RIG-I/MDA5/MAVS are required to signal a protective IFN response in rotavirus-infected intestinal epithelium. J. Immunol. 186, 1618–1626 (2011).
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by Toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004). In this study, mice deficient in TLR signalling or greatly depleted of commensal bacteria exhibited increased susceptibility to experimentally induced intestinal inflammation, implicating commensal microorganism-dependent signals in the regulation of intestinal homeostasis and response to injury.
Brandl, K. et al. MyD88 signaling in nonhematopoietic cells protects mice against induced colitis by regulating specific EGF receptor ligands. Proc. Natl Acad. Sci. USA 107, 19967–19972 (2010).
Podolsky, D. K., Gerken, G., Eyking, A. & Cario, E. Colitis-associated variant of TLR2 causes impaired mucosal repair because of TFF3 deficiency. Gastroenterology 137, 209–220 (2009).
Cario, E., Gerken, G. & Podolsky, D. K. Toll-like receptor 2 enhances ZO-1-associated intestinal epithelial barrier integrity via protein kinase C. Gastroenterology 127, 224–238 (2004).
Greten, F. R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004). This study provides evidence that the NF-κB signalling pathway in IECs regulates the development of inflammation-induced cancer through the regulation of apoptosis.
Nenci, A. et al. Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature 446, 557–561 (2007).
Hugot, J.-P. et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 411, 599–603 (2001).
Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603–606 (2001). References 62 and 63 provide the first examples of genetic variants linking bacterial recognition with susceptibility to human IBD.
Zaki, M. H., Lamkanfi, M. & Kanneganti, T.-D. The Nlrp3 inflammasome: contributions to intestinal homeostasis. Trends Immunol. 32, 171–179 (2011).
Swanson, P. A. et al. Enteric commensal bacteria potentiate epithelial restitution via reactive oxygen species-mediated inactivation of focal adhesion kinase phosphatases. Proc. Natl Acad. Sci. USA 108, 8803–8808 (2011).
Leoni, G. et al. Annexin A1, formyl peptide receptor, and NOX1 orchestrate epithelial repair. J. Clin. Invest. 123, 443–454 (2012).
Lee, W.-J. Bacterial-modulated host immunity and stem cell activation for gut homeostasis. Genes Dev. 23, 2260–2265 (2009).
Hochmuth, C. E., Biteau, B., Bohmann, D. & Jasper, H. Redox regulation by Keap1 and Nrf2 controls intestinal stem cell proliferation in Drosophila. Cell Stem Cell 8, 188–199 (2011).
Jones, R. M. et al. Symbiotic lactobacilli stimulate gut epithelial proliferation via Nox-mediated generation of reactive oxygen species. EMBO J. 32, 3017–3028 (2013).
Rakoff-Nahoum, S. & Medzhitov, R. Regulation of spontaneous intestinal tumorigenesis through the adaptor protein MyD88. Science 317, 124–127 (2007).
Lee, S. H. et al. ERK activation drives intestinal tumorigenesis in Apcmin/+ mice. Nature Med. 16, 665–670 (2010).
Vlantis, K. et al. Constitutive IKK2 activation in intestinal epithelial cells induces intestinal tumors in mice. J. Clin. Invest. 121, 2781–2793 (2011).
Schwitalla, S. et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 152, 25–38 (2013).
Fukata, M. et al. Toll-Like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology 133, 1869–1869 (2007).
Xiao, H. et al. The Toll–interleukin-1 receptor member SIGIRR regulates colonic epithelial homeostasis, inflammation, and tumorigenesis. Immunity 26, 461–475 (2007).
Dupaul-Chicoine, J. et al. Control of intestinal homeostasis, colitis, and colitis-associated colorectal cancer by the inflammatory caspases. Immunity 32, 367–378 (2010).
Hu, B. et al. Inflammation-induced tumorigenesis in the colon is regulated by caspase-1 and NLRC4. PNAS 107, 21635–21640 (2010).
Zaki, M. H. et al. The NOD-like receptor NLRP12 attenuates colon inflammation and tumorigenesis. Cancer Cell 20, 649–660 (2011).
Normand, S. et al. Nod-like receptor pyrin domain-containing protein 6 (NLRP6) controls epithelial self-renewal and colorectal carcinogenesis upon injury. Proc. Natl Acad. Sci. USA 108, 9601–9606 (2011).
Allen, I. C. et al. NLRP12 suppresses colon inflammation and tumorigenesis through the negative regulation of noncanonical NF-κB signaling. Immunity 36, 742–754 (2012).
Salcedo, R. et al. MyD88-mediated signaling prevents development of adenocarcinomas of the colon: role of interleukin 18. J. Exp. Med. 207, 1625–1636 (2010).
Huber, S. et al. IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature 491, 259–263 (2012).
Otte, J.-M., Cario, E. & Podolsky, D. K. Mechanisms of cross hyporesponsiveness to Toll-like receptor bacterial ligands in intestinal epithelial cells. Gastroenterology 126, 1054–1070 (2004).
Lotz, M. et al. Postnatal acquisition of endotoxin tolerance in intestinal epithelial cells. J. Exp. Med. 203, 973–984 (2006). This study demonstrates the inhibition of TLR signalling in IECs and the acquisition of tolerance to microbial colonization that occurs shortly after birth to establish host–microbial homeostasis in the intestine.
Vereecke, L. et al. Enterocyte-specific A20 deficiency sensitizes to tumor necrosis factor-induced toxicity and experimental colitis. J. Exp. Med. 207, 1513–1523 (2010).
Chassin, C. et al. miR-146a mediates protective innate immune tolerance in the neonate intestine. Cell Host Microbe 8, 358–368 (2010).
Guma, M. et al. Constitutive intestinal NF-κB does not trigger destructive inflammation unless accompanied by MAPK activation. J. Exp. Med. 208, 1889–1900 (2011).
Neish, A. S. et al. Prokaryotic regulation of epithelial responses by inhibition of IκB-α ubiquitination. Science 289, 1560–1563 (2000).
Kumar, A. et al. The bacterial fermentation product butyrate influences epithelial signaling via reactive oxygen species-mediated changes in cullin-1 neddylation. J. Immunol. 182, 538–546 (2009).
Kondo, T., Kawai, T. & Akira, S. Dissecting negative regulation of Toll-like receptor signaling. Trends Immunol. 33, 449–458 (2012).
Blander, J. M. & Sander, L. E. Beyond pattern recognition: five immune checkpoints for scaling the microbial threat. Nature Rev. Immunol. 12, 215–225 (2012).
Gewirtz, A. T., Navas, T. A., Lyons, S., Godowski, P. J. & Madara, J. L. Cutting edge: bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J. Immunol. 167, 1882–1885 (2001).
Rhee, S. H. et al. Pathophysiological role of Toll-like receptor 5 engagement by bacterial flagellin in colonic inflammation. Proc. Natl Acad. Sci. USA 102, 13610–13615 (2005).
Lee, J. et al. Maintenance of colonic homeostasis by distinctive apical TLR9 signalling in intestinal epithelial cells. Nature Cell Biol. 8, 1327–1336 (2006).
Barnich, N., Aguirre, J. E., Reinecker, H.-C., Xavier, R. & Podolsky, D. K. Membrane recruitment of NOD2 in intestinal epithelial cells is essential for nuclear factor-κB activation in muramyl dipeptide recognition. J. Cell Biol. 170, 21–26 (2005).
Lipinski, S. et al. RNAi screening identifies mediators of NOD2 signaling: Implications for spatial specificity of MDP recognition. Proc. Natl Acad. Sci. USA 109, 21426–21431 (2012).
Matzinger, P. The danger model: A renewed sense of self. Science 296, 301–305 (2002).
Sander, L. E. et al. Detection of prokaryotic mRNA signifies microbial viability and promotes immunity. Nature 474, 385–389 (2011). In this study, bacterial mRNA is identified as a signal that enables the gauging of infectious risk posed by live versus dead bacteria, exemplifying a vita-PAMP.
Hooper, L. V., Stappenbeck, T. S., Hong, C. V. & Gordon, J. I. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nature Immunol. 4, 269–273 (2003).
Kobayashi, K. S. et al. Nod2-dependent regulation of innate and adaptive immunity in the intestinal tract. Science 307, 731–734 (2005).
Vaishnava, S., Behrendt, C. L., Ismail, A. S., Eckmann, L. & Hooper, L. V. Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proc. Natl Acad. Sci. USA 105, 20858–20863 (2008).
Bruno, M. E. C., Frantz, A. L., Rogier, E. W., Johansen, F.-E. & Kaetzel, C. S. Regulation of the polymeric immunoglobulin receptor by the classical and alternative NF-κB pathways in intestinal epithelial cells. Mucosal Immunol. 4, 468–478 (2011).
Rimoldi, M. et al. Intestinal immune homeostasis is regulated by the crosstalk between epithelial cells and dendritic cells. Nature Immunol. 6, 507–514 (2005). This study describes the conditioning of mucosal DCs towards a non-inflammatory phenotype by interactions with IECs, representing a mechanism for the indirect IEC-mediated regulation of adaptive immune cell priming.
Zeuthen, L. H., Fink, L. N. & Frokiaer, H. Epithelial cells prime the immune response to an array of gut-derived commensals towards a tolerogenic phenotype through distinct actions of thymic stromal lymphopoietin and transforming growth factor-β. Immunology 123, 197–208 (2008).
Zaph, C. et al. Epithelial-cell-intrinsic IKKβ expression regulates intestinal immune homeostasis. Nature 446, 552–556 (2007). This study demonstrates a crucial role for IEC-intrinsic NF-κB signalling and the production of the cytokine TSLPthymic stromal lymphopoietin in regulating intestinal immune responses and coordinating anti-helminth immunity.
Atarashi, K. et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 331, 337–341 (2011).
Zaph, C. et al. Commensal-dependent expression of IL-25 regulates the IL-23–IL-17 axis in the intestine. J. Exp. Med. 205, 2191–2198 (2008).
He, B. et al. Intestinal bacteria trigger T cell-independent immunoglobulin A2 class switching by inducing epithelial-cell secretion of the cytokine APRIL. Immunity 26, 812–826 (2007).
Xu, W. et al. Epithelial cells trigger frontline immunoglobulin class switching through a pathway regulated by the inhibitor SLPI. Nature Immunol. 8, 294–303 (2007). References 108 and 109 identify IEC production of APRIL and BAFF in response to microbial stimulation as important for the regulation of B cell CSR and the mucosal IgA response.
Taylor, B. C. et al. TSLP regulates intestinal immunity and inflammation in mouse models of helminth infection and colitis. J. Exp. Med. 206, 655–667 (2009).
Pabst, O. & Bernhardt, G. The puzzle of intestinal lamina propria dendritic cells and macrophages. Eur. J. Immunol. 40, 2107–2111 (2010).
Bogunovic, M. et al. Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 (2009).
Varol, C. et al. Intestinal lamina propria dendritic cell subsets have different origin and functions. Immunity 31, 502–512 (2009).
Rivollier, A., He, J., Kole, A., Valatas, V. & Kelsall, B. L. Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J. Exp. Med. 209, 139–155 (2012).
Zigmond, E. & Jung, S. Intestinal macrophages: well educated exceptions from the rule. Trends Immunol. 34, 162–168 (2013).
Schulz, O. et al. Intestinal CD103+, but not CX3CR1+, antigen sampling cells migrate in lymph and serve classical dendritic cell functions. J. Exp. Med. 206, 3101–3114 (2009).
Macpherson, A. J. & Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665 (2004).
Coombes, J. L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β– and retinoic acid–dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Sun, C.-M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
Mora, J. R. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature 424, 88–93 (2003).
Johansson-Lindbom, B. et al. Functional specialization of gut CD103+ dendritic cells in the regulation of tissue-selective T cell homing. J. Exp. Med. 202, 1063–1073 (2005).
Iwata, M. et al. Retinoic acid imprints gut-homing specificity on T cells. Immunity 21, 527–538 (2004).
Jaensson, E. et al. Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J. Exp. Med. 205, 2139–2149 (2008).
Mora, J. R. & von Andrian, U. H. Differentiation and homing of IgA-secreting cells. Mucosal Immunol. 1, 96–109 (2008).
Niess, J. H. et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 307, 254–258 (2005).
Hadis, U. et al. Intestinal tolerance requires gut homing and expansion of FoxP3+ regulatory T cells in the lamina propria. Immunity 34, 237–246 (2011).
Murai, M. et al. Interleukin 10 acts on regulatory T cells to maintain expression of the transcription factor Foxp3 and suppressive function in mice with colitis. Nature Immunol. 10, 1178–1184 (2009).
Kayama, H. et al. Intestinal CX3C chemokine receptor 1high (CX3CR1high) myeloid cells prevent T-cell-dependent colitis. Proc. Natl Acad. Sci. USA 109, 5010–5015 (2012).
Kang, S. et al. Intestinal epithelial cell-derived semaphorin 7A negatively regulates development of colitis via αvβ1 integrin. J. Immunol. 188, 1108–1116 (2012).
Saenz, S. A. et al. IL25 elicits a multipotent progenitor cell population that promotes TH2 cytokine responses. Nature 464, 1362–1366 (2010).
Siracusa, M. C. et al. TSLP promotes interleukin-3-independent basophil haematopoiesis and type 2 inflammation. Nature 477, 229–233 (2011).
Saenz, S. A. et al. IL-25 simultaneously elicits distinct populations of innate lymphoid cells and multipotent progenitor type 2 (MPPtype2) cells. J. Exp. Med. 210, 1823–1837 (2013).
Siracusa, M. C. et al. Thymic stromal lymphopoietin-mediated extramedullary hematopoiesis promotes allergic inflammation. Immunity 39, 1158–1170 (2013).
Spits, H. & Cupedo, T. Innate lymphoid cells: emerging insights in development, lineage relationships, and function. Annu. Rev. Immunol. 30, 647–675 (2012).
Monticelli, L. A., Sonnenberg, G. F. & Artis, D. Innate lymphoid cells: critical regulators of allergic inflammation and tissue repair in the lung. Curr. Opin. Immunol. 24, 284–289 (2012).
Kim, B. S. et al. TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci. Transl. Med. 5, 170ra16 (2013).
Tait Wojno, E. D. & Artis, D. Innate lymphoid cells: balancing immunity, inflammation, and tissue repair in the intestine. Cell Host Microbe 12, 445–457 (2012).
Walker, J. A., Barlow, J. L. & McKenzie, A. N. J. Innate lymphoid cells — how did we miss them? Nature Rev. Immunol. 13, 75–87 (2013).
Cherrier, M., Ohnmacht, C., Cording, S. & Eberl, G. Development and function of intestinal innate lymphoid cells. Curr. Opin. Immunol. 24, 277–283 (2012).
Spits, H. et al. Innate lymphoid cells — a proposal for uniform nomenclature. Nature Rev. Immunol. 13, 145–149 (2013).
Bernink, J. H. et al. Human type 1 innate lymphoid cells accumulate in inflamed mucosal tissues. Nature Immunol. 14, 221–229 (2013).
Fuchs, A. et al. Intraepithelial type 1 innate lymphoid cells are a unique subset of IL-12- and IL-15-responsive IFN-γ-producing cells. Immunity 38, 769–781 (2013).
Moro, K. et al. Innate production of TH2 cytokines by adipose tissue-associated c-Kit+Sca-1+ lymphoid cells. Nature 463, 540–544 (2009).
Neill, D. R. et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464, 1367–1370 (2010). References 143 and 144 identify a population of innate lymphoid cells that mediate early T H 2 cytokine production in response to the cytokines IL-25 and IL-33 and during intestinal helminth infection, that promotes goblet cell hyperplasia and worm expulsion.
Price, A. E. et al. Systemically dispersed innate IL-13-expressing cells in type 2 immunity. Proc. Natl Acad. Sci. USA 107, 11489–11494 (2010).
Monticelli, L. A. et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nature Immunol. 12, 1045–1054 (2011).
Chang, Y.-J. et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nature Immunol. 12, 631–638 (2011).
Wilhelm, C. et al. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nature Immunol. 12, 1071–1077 (2011).
Halim, T. Y. F., Krauß, R. H., Sun, A. C. & Takei, F. Lung natural helper cells are a critical source of Th2 cell-type cytokines in protease allergen-induced airway inflammation. Immunity 36, 451–463 (2012).
Mjösberg, J. et al. The transcription factor GATA3 Is essential for the function of human type 2 innate lymphoid cells. Immunity 37, 649–659 (2012).
Mebius, R. E., Rennert, P. & Weissman, I. L. Developing lymph nodes collect, CD4+CD3− LTβ+ cells that can differentiate to APC, NK cells, and follicular cells but not T or B cells. Immunity 7, 493–504 (1997).
Zenewicz, L. A. et al. Innate and adaptive interleukin-22 protects mice from inflammatory bowel disease. Immunity 29, 947–957 (2008).
Sonnenberg, G. F., Monticelli, L. A., Elloso, M. M., Fouser, L. A. & Artis, D. CD4+ lymphoid tissue-inducer cells promote innate immunity in the gut. Immunity 34, 122–134 (2011).
Sonnenberg, G. F. et al. Innate lymphoid cells promote anatomical containment of lymphoid-resident commensal bacteria. Science 336, 1321–1325 (2012). In this study, innate lymphoid cell-derived IL-22 is demonstrated to mediate the containment of specialized intestinal lymphoid tissue-resident commensal bacteria to prevent systemic immune activation.
Hanash, A. M. et al. Interleukin-22 protects intestinal stem cells from immune-mediated tissue damage and regulates sensitivity to graft versus host disease. Immunity 37, 339–350 (2012).
Kirchberger, S. et al. Innate lymphoid cells sustain colon cancer through production of interleukin-22 in a mouse model. J. Exp. Med. 210, 917–931 (2013).
Muñoz, M. et al. Interleukin (IL)-23 mediates Toxoplasma gondii–induced immunopathology in the gut via matrixmetalloproteinase-2 and IL-22 but independent of IL-17. J. Exp. Med. 206, 3047–3059 (2009).
Buonocore, S. et al. Innate lymphoid cells drive interleukin-23-dependent innate intestinal pathology. Nature 464, 1371–1375 (2010).
Geremia, A. et al. IL-23–responsive innate lymphoid cells are increased in inflammatory bowel disease. J. Exp. Med. 208, 1127–1133 (2011).
Coccia, M. et al. IL-1β mediates chronic intestinal inflammation by promoting the accumulation of IL-17A secreting innate lymphoid cells and CD4+ Th17 cells. J. Exp. Med. 209, 1595–1609 (2012).
Sawa, S. et al. RORγt+ innate lymphoid cells regulate intestinal homeostasis by integrating negative signals from the symbiotic microbiota. Nature Immunol. 12, 320–326 (2011).
Vonarbourg, C. et al. Regulated expression of nuclear receptor RORγt confers distinct functional fates to NK cell receptor-expressing RORγt+ innate lymphocytes. Immunity 33, 736–751 (2010).
Satoh-Takayama, N. et al. Microbial flora drives interleukin 22 production in intestinal NKp46+ cells that provide innate mucosal immune defense. Immunity 29, 958–970 (2008).
Yu, Q. et al. MyD88-dependent signaling for IL-15 production plays an important role in maintenance of CD8αα TCRαβ and TCRγδ intestinal intraepithelial lymphocytes. J. Immunol. 176, 6180–6185 (2006).
Cheroutre, H., Lambolez, F. & Mucida, D. The light and dark sides of intestinal intraepithelial lymphocytes. Nature Rev. Immunol. 11, 445–456 (2011).
Edelblum, K. L. et al. Dynamic migration of γδ intraepithelial lymphocytes requires occludin. Proc. Natl Acad. Sci. USA 109, 7097–7102 (2012).
Ismail, A. S. et al. γδ intraepithelial lymphocytes are essential mediators of host-microbial homeostasis at the intestinal mucosal surface. Proc. Natl Acad. Sci. USA 108, 8743–8748 (2011).
Mucida, D. et al. Transcriptional reprogramming of mature CD4+ helper T cells generates distinct MHC class II-restricted cytotoxic T lymphocytes. Nature Immunol. 14, 281–289 (2013).
Gebhardt, T., Mueller, S. N., Heath, W. R. & Carbone, F. R. Peripheral tissue surveillance and residency by memory T cells. Trends Immunol. 34, 27–32 (2013).
Sathaliyawala, T. et al. Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity 38, 187–197 (2013).
Schön, M. P. et al. Mucosal T lymphocyte numbers are selectively reduced in integrin αE (CD103)-deficient mice. J. Immunol. 162, 6641–6649 (1999).
El-Asady, R. et al. TGF-β-dependent CD103 expression by CD8+ T cells promotes selective destruction of the host intestinal epithelium during graft-versus-host disease. J. Exp. Med. 201, 1647–1657 (2005).
Huang, Y. et al. Mucosal memory CD8+ T cells are selected in the periphery by an MHC class I molecule. Nature Immunol. 12, 1086–1095 (2011).
Mackay, L. K. et al. Long-lived epithelial immunity by tissue-resident memory T (TRM) cells in the absence of persisting local antigen presentation. Proc. Natl Acad. Sci. USA 109, 7037–7042 (2012).
Shin, H. & Iwasaki, A. A vaccine strategy that protects against genital herpes by establishing local memory T cells. Nature 491, 463–467 (2012).
Hansen, S. G. et al. Profound early control of highly pathogenic SIV by an effector memory T-cell vaccine. Nature 473, 523–527 (2011).
Cerutti, A. The regulation of IgA class switching. Nature Rev. Immunol. 8, 421–434 (2008).
Mora, J. R. et al. Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells. Science 314, 1157–1160 (2006).
Litinskiy, M. B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nature Immunol. 3, 822–829 (2002).
Castigli, E. et al. TACI and BAFF-R mediate isotype switching in B cells. J. Exp. Med. 201, 35–39 (2005).
Salzer, U. et al. Mutations in TNFRSF13B encoding TACI are associated with common variable immunodeficiency in humans. Nature Genet. 37, 820–828 (2005).
Castigli, E. et al. TACI is mutant in common variable immunodeficiency and IgA deficiency. Nature Genet. 37, 829–834 (2005).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007).
Takeda, N. et al. Interconversion between intestinal stem cell populations in distinct niches. Science 334, 1420–1424 (2011).
Yan, K. S. et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl Acad. Sci. USA 109, 466–471 (2012).
Capaldo, C. T. et al. IFN-γ and TNF-α-induced GBP-1 inhibits epithelial cell proliferation through suppression of β-catenin/TCF signaling. Mucosal Immunol. 5, 681–690 (2012).
Nava, P. et al. Interferon-γ regulates intestinal epithelial homeostasis through converging β-catenin signaling pathways. Immunity 32, 392–402 (2010).
Alenghat, T. et al. Histone deacetylase 3 coordinates commensal-bacteria-dependent intestinal homeostasis. Nature 504, 153–157 (2013).
Blair, S. A., Kane, S. V., Clayburgh, D. R. & Turner, J. R. Epithelial myosin light chain kinase expression and activity are upregulated in inflammatory bowel disease. Lab. Invest. 86, 191–201 (2006).
Marchiando, A. M. et al. Caveolin-1–dependent occludin endocytosis is required for TNF-induced tight junction regulation in vivo. J. Cell Biol. 189, 111–126 (2010).
Wang, F. et al. Active deformation of apoptotic intestinal epithelial cells with adhesion-restricted polarity contributes to apoptotic clearance. Lab. Invest. 91, 462–471 (2011).
Eisenhoffer, G. T. et al. Crowding induces live cell extrusion to maintain homeostatic cell numbers in epithelia. Nature 484, 546–549 (2012).
D'Incà, R. et al. Intestinal permeability test as a predictor of clinical course in Crohn's disease. Am. J. Gastroenterol. 94, 2956–2960 (1999).
Wyatt, J. & Vogelsang, H. Intestinal permeability and the prediction of relapse in Crohn's disease. Lancet 341, 1437 (1993).
Khounlotham, M. et al. Compromised intestinal epithelial barrier induces adaptive immune compensation that protects from colitis. Immunity 37, 563–573 (2012).
Su, L. et al. Targeted epithelial tight junction dysfunction causes immune activation and contributes to development of experimental colitis. Gastroenterology 136, 551–563 (2009).
Acknowledgements
The authors thank all members of the Artis laboratory for discussions and critical reading of the manuscript. This work is supported by US National Institutes of Health grants (AI061570, AI095608, AI087990, AI074878, AI095466, AI106697, AI102942 and AI097333 to D.A.; T32AI00744 to L.W.P.), the Burroughs Wellcome Fund Investigator in Pathogenesis of Infectious Disease Award (D.A.) and the Crohn's and Colitis Foundation of America (D.A.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
The role of pattern recognition receptors (PRRs) in regulating intestinal epithelial cells (IECs) and tissue homeostasis. (PDF 713 kb)
Glossary
- Inflammatory bowel disease
-
(IBD). A chronic condition of the intestine characterized by severe inflammation and mucosal destruction. The most common forms of IBD in humans are ulcerative colitis and Crohn's disease, which have both distinct and overlapping pathological and clinical characteristics.
- Mucins
-
Heavily glycosylated proteins that are the major component of the mucus that coats epithelial barrier surfaces.
- Crypts
-
Tubular invaginations of the intestinal epithelium. Lining the base of the crypts are small intestinal Paneth cells, which produce numerous antimicrobial proteins, and stem cells, which continuously divide to give rise to the entire intestinal epithelium.
- Villi
-
Projections of the intestinal epithelium into the lumen of the small intestine that have an outer layer consisting of mature, absorptive enterocytes, mucus-secreting goblet cells and enteroendocrine cells.
- Pluripotent intestinal epithelial stem cells
-
(Pluripotent IESCs). Tissue-resident stem cells that undergo continuous self-renewal and are responsible for regenerating all lineages of mature intestinal epithelial cells, including enterocytes, enteroendocrine cells, goblet cells and Paneth cells.
- Autophagy
-
A cellular process by which cytoplasmic organelles and macromolecular complexes are engulfed by double membrane-bound vesicles for delivery to lysosomes and subsequent degradation. This process is involved in constitutive turnover of proteins and organelles and is central to cellular activities that maintain a balance between the synthesis and breakdown of various proteins.
- Unfolded protein response
-
(UPR). A response that increases the ability of the endoplasmic reticulum to fold and translocate proteins, decreases the synthesis of proteins, causes the arrest of the cell cycle and promotes apoptosis.
- Plasma cells
-
Terminally differentiated cells of the B cell lineage that secrete large amounts of antibodies.
- Lamina propria
-
Connective tissue that underlies the epithelium of the mucosa and contains stromal and haematopoietic cells.
- Peyer's patches
-
Groups of lymphoid aggregates located in the submucosa of the small intestine that contain many immune cells, including B cells, T cells and dendritic cells. They have a luminal barrier consisting of specialized epithelial cells, called microfold cells, which sample the lumen and transport antigens.
- Pattern-recognition receptors
-
(PRRs). Receptors that recognize structures shared by foreign microorganisms or endogenous molecules associated with pathogenesis. Signalling through these receptors promotes tissue-specific innate immune responses including the production of cytokines.
- Toll-like receptor
-
(TLR). An evolutionarily conserved pattern-recognition receptor located at the cell surface or at intracellular membranes. The natural ligands of TLRs are conserved molecular structures found in bacteria, viruses and fungi.
- NOD-like receptor
-
(NLR). A pattern-recognition receptor located in the cytosol. NLRs recognize a wide range of foreign structures and patterns associated with pathogenesis. Some members of this family form multiprotein complexes known as inflammasomes, which regulate the processing and secretion of pro-inflammatory cytokines.
- RIG-I-like receptor
-
(RLR). A pattern-recognition receptor located in the cytosol that responds to viral RNA.
- Dextran sodium sulphate
-
(DSS). A large polysaccharide that causes epithelial injury and inflammation in the intestinal tract and is commonly used in models of experimentally induced colitis for studying the response to intestinal injury.
- Nuclear factor-κB
-
(NF-κB). A family of transcription factors important for pro-inflammatory and anti-apoptotic responses that are activated by the ubiquitin-dependent degradation of their respective inhibitors, members of the inhibitor of NF-κB (IκB) family. This process is mediated by the kinases, IκB kinase 1 (IKK1) and IKK2.
- Inflammasomes
-
Multiprotein complexes that contain a member of the NOD-like receptor family, adaptor proteins and the protease caspase 1. These complexes regulate the catalytic processing and secretion of pro-inflammatory cytokines, including interleukin-1β (IL-1β) and IL-18.
- Reactive oxygen species
-
(ROS). Chemically reactive molecules containing oxygen that, when produced in large amounts, have pro-inflammatory and antimicrobial effects. Physiological levels of ROS have been shown to regulate cellular signalling pathways.
- Viability-associated PAMPs
-
(Vita-PAMPs). Members of a special class of pathogen-associated molecular patterns recognized by the innate immune system to signify microbial life. These patterns differentiate dead and living microorganisms to allow for scaling of appropriate immune responses based on the level of threat the microbial signals represents.
- Innate lymphoid cells
-
(ILCs). A group of innate immune cells that are lymphoid in morphology and developmental origin, but lack properties of adaptive B cells and T cells such as recombined antigen-specific receptors. They function in the regulation of immunity, tissue homeostasis and inflammation in response to cytokine stimulation.
- Natural killer cells
-
(NK cells). A subset of innate lymphoid cells originally defined on the basis of their cytolytic activity against tumour targets but now recognized to serve a broader role in host defence and inflammation through the production of cytokines.
- Class-switch recombination
-
(CSR). The process by which proliferating B cells rearrange their DNA to switch from expressing IgM (or another class of immunoglobulin) to expressing a different immunoglobulin heavy-chain constant region, thereby producing antibody with different effector functions.
Rights and permissions
About this article
Cite this article
Peterson, L., Artis, D. Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat Rev Immunol 14, 141–153 (2014). https://doi.org/10.1038/nri3608
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3608
This article is cited by
-
A novel avian intestinal epithelial cell line: its characterization and exploration as an in vitro infection culture model for Eimeria species
Parasites & Vectors (2024)
-
The gut-liver axis in hepatobiliary diseases
Inflammation and Regeneration (2024)
-
Death receptor 5 is required for intestinal stem cell activity during intestinal epithelial renewal at homoeostasis
Cell Death & Disease (2024)
-
Expression of E-cadherin by CD8+ T cells promotes their invasion into biliary epithelial cells
Nature Communications (2024)
-
Effect of diet supplemented with functional amino acids and polyphenols on gut health in broilers subjected to a corticosterone-induced stress
Scientific Reports (2024)