Key Points
-
Chronic kidney diseases are common in ageing populations and, if they progress to the end stage, are an economic burden because of the need for dialysis or transplantation. Immune mechanisms are centrally involved in the progression of kidney disease and their elucidation is an important research goal.
-
Recent studies have defined the immune mechanisms responsible for some previously unexplained kidney diseases. These may also be relevant to diseases affecting other organs.
-
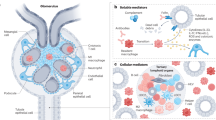
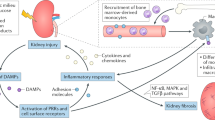
Greater knowledge of immune homeostasis in the kidneys has identified features that render this organ susceptible to distinct types of immune-mediated damage; for example, kidney-specific damage-associated molecular patterns (DAMPs) and a predisposition to crystal formation.
-
The availability of well-characterized animal models and of biopsy samples from patients with renal disease makes the kidney an attractive model organ to analyse novel pathways of host defence, autoimmune injury, sterile inflammation and fibrosis.
-
The kidney has a central role in electrolyte homeostasis and the removal of toxins and so, when its function is compromised, normal immune effector cell function and intestinal microbial homeostasis are disturbed.
-
Kidney failure increases susceptibility to infection and promotes exaggerated inflammatory responses, both of which increase mortality. The underlying mechanisms responsible remain to be clarified.
Abstract
The kidneys are frequently targeted by pathogenic immune responses against renal autoantigens or by local manifestations of systemic autoimmunity. Recent studies in rodent models and humans have uncovered several underlying mechanisms that can be used to explain the previously enigmatic immunopathology of many kidney diseases. These mechanisms include kidney-specific damage-associated molecular patterns that cause sterile inflammation, the crosstalk between renal dendritic cells and T cells, the development of kidney-targeting autoantibodies and molecular mimicry with microbial pathogens. Conversely, kidney failure affects general immunity, causing intestinal barrier dysfunction, systemic inflammation and immunodeficiency that contribute to the morbidity and mortality of patients with kidney disease. In this Review, we summarize the recent findings regarding the interactions between the kidneys and the immune system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kaissling, B. & Le Hir, M. Characterization and distribution of interstitial cell types in the renal cortex of rats. Kidney Int. 45, 709–720 (1994).
Kruger, T. et al. Identification and functional characterization of dendritic cells in the healthy murine kidney and in experimental glomerulonephritis. J. Am. Soc. Nephrol. 15, 613–621 (2004).
Soos, T. J. et al. CX3CR1+ interstitial dendritic cells form a contiguous network throughout the entire kidney. Kidney Int. 70, 591–596 (2006).
Woltman, A. M. et al. Quantification of dendritic cell subsets in human renal tissue under normal and pathological conditions. Kidney Int. 71, 1001–1008 (2007).
Guilliams, M. et al. From skin dendritic cells to a simplified classification of human and mouse dendritic cell subsets. Eur. J. Immunol. 40, 2089–2094 (2010).
Miller, J. C. et al. Deciphering the transcriptional network of the dendritic cell lineage. Nature Immunol. 13, 888–899 (2012).
Schraml, B. U. et al. Genetic tracing via expression history of DNGR-1 defines dendritic cells as a hematopoietic lineage. Cell 154, 843–858 (2013).
Tittel, A. P. et al. Functionally relevant neutrophilia in CD11c diphtheria toxin receptor transgenic mice. Nature Methods 9, 385–390 (2012).
Hochheiser, K. et al. Exclusive CX3CR1-dependence of kidney dendritic cells impacts glomerulonephritis progression. J. Clin. Invest. http://dx.doi.org/10.1172/JCI70143 (2013).
Kim, K. W. et al. In vivo structure/function and expression analysis of the CX3C chemokine fractalkine. Blood 118, e156–e167 (2011).
Dong, X. et al. Resident dendritic cells are the predominant TNF-secreting cell in early renal ischemia-reperfusion injury. Kidney Int. 71, 619–628 (2007).
Pindjakova, J. et al. Interleukin-1 accounts for intrarenal Th17 cell activation during ureteral obstruction. Kidney Int. 81, 379–390 (2012).
Merad, M., Ginhoux, F. & Collin, M. Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells. Nature Rev. Immunol. 8, 935–947 (2008).
Nelson, P. J. et al. The renal mononuclear phagocytic system. J. Am. Soc. Nephrol. 23, 194–203 (2012). This paper summarizes the phenotypical range of mononuclear phagocytes in healthy and diseased kidneys.
Timoshanko, J. R., Kitching, R., Semple, T. J., Tipping, P. G. & Holdsworth, S. R. A pathogenetic role for mast cells in experimental crescentic glomerulonephritis. J. Am. Soc. Nephrol. 17, 150–159 (2006).
Scandiuzzi, L. et al. Mouse mast cell protease-4 deteriorates renal function by contributing to inflammation and fibrosis in immune complex-mediated glomerulonephritis. J. Immunol. 185, 624–633 (2010).
Gan, P. Y. et al. Mast cells contribute to peripheral tolerance and attenuate autoimmune vasculitis. J. Am. Soc. Nephrol. 23, 1955–1966 (2012).
Dong, X. et al. Antigen presentation by dendritic cells in renal lymph nodes is linked to systemic and local injury to the kidney. Kidney Int. 68, 1096–1108 (2005).
Heymann, F. et al. Kidney dendritic cell activation is required for progression of renal disease in a mouse model of glomerular injury. J. Clin. Invest. 119, 1286–1297 (2009). This study shows that T cells can induce glomerular damage and that DC maturation drives glomerulonephritis progression.
Lukacs-Kornek, V. et al. The kidney-renal lymph node-system contributes to cross-tolerance against innocuous circulating antigen. J. Immunol. 180, 706–715 (2008).
Gottschalk, C. et al. Batf3-dependent dendritic cells in the renal lymph node induce tolerance against circulating antigens. J. Am. Soc. Nephrol. 24, 543–549 (2013).
Bohle, A., Kressel, G., Muller, C. A. & Muller, G. A. The pathogenesis of chronic renal failure. Pathol. Res. Pract. 185, 421–440 (1989).
Markovic-Lipkovski, J., Muller, C. A., Risler, T., Bohle, A. & Muller, G. A. Association of glomerular and interstitial mononuclear leukocytes with different forms of glomerulonephritis. Nephrol. Dial. Transplant. 5, 10–17 (1990).
Risdon, R. A., Sloper, J. C. & De Wardener, H. E. Relationship between renal function and histological changes found in renal-biopsy specimens from patients with persistent glomerular nephritis. Lancet 2, 363–366 (1968).
Kriz, W. & LeHir, M. Pathways to nephron loss starting from glomerular diseases-insights from animal models. Kidney Int. 67, 404–419 (2005).
Bohle, A., Mackensen-Haen, S. & Wehrmann, M. Significance of postglomerular capillaries in the pathogenesis of chronic renal failure. Kidney Blood Press. Res. 19, 191–195 (1996).
Floege, J. & Grone, H. J. Progression of renal failure: what is the role of cytokines? Nephrol. Dial. Transplant. 10, 1575–1586 (1995).
Abbate, M., Zoja, C. & Remuzzi, G. How does proteinuria cause progressive renal damage? J. Am. Soc. Nephrol. 17, 2974–2984 (2006).
Niedermeier, M. et al. CD4+ T cells control the differentiation of Gr1+ monocytes into fibrocytes. Proc. Natl Acad. Sci. USA 106, 17892–17897 (2009).
Rock, K. L., Latz, E., Ontiveros, F. & Kono, H. The sterile inflammatory response. Annu. Rev. Immunol. 28, 321–342 (2010).
Yamanishi, Y. et al. TIM1 is an endogenous ligand for LMIR5/CD300b: LMIR5 deficiency ameliorates mouse kidney ischemia/reperfusion injury. J. Exp. Med. 207, 1501–1511 (2010).
Anders, H. J. Toll-like receptors and danger signaling in kidney injury. J. Am. Soc. Nephrol. 21, 1270–1274 (2010).
Rosin, D. L. & Okusa, M. D. Dangers within: DAMP responses to damage and cell death in kidney disease. J. Am. Soc. Nephrol. 22, 416–425 (2011).
Mulay, S. R. et al. Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1β secretion. J. Clin. Invest. 123, 236–246 (2013). This paper identifies the role of the NLRP3 inflammasome in crystal nephropathy.
Leemans, J. C. et al. Renal-associated TLR2 mediates ischemia/reperfusion injury in the kidney. J. Clin. Invest. 115, 2894–2903 (2005).
Wu, H. et al. TLR4 activation mediates kidney ischemia/reperfusion injury. J. Clin. Invest. 117, 2847–2859 (2007).
Allam, R. et al. Histones from dying renal cells aggravate kidney injury via TLR2 and TLR4. J. Am. Soc. Nephrol. 23, 1375–1388 (2012).
McDonald, B. et al. Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330, 362–366 (2010).
Li, L. et al. Dendritic cells tolerized with adenosine A2AR agonist attenuate acute kidney injury. J. Clin. Invest. 122, 3931–3942 (2012).
Bonventre, J. V. & Yang, L. Cellular pathophysiology of ischemic acute kidney injury. J. Clin. Invest. 121, 4210–4221 (2011).
Zhang, B., Ramesh, G., Uematsu, S., Akira, S. & Reeves, W. B. TLR4 signaling mediates inflammation and tissue injury in nephrotoxicity. J. Am. Soc. Nephrol. 19, 923–932 (2008).
Babelova, A. et al. Biglycan, a danger signal that activates the NLRP3 inflammasome via toll-like and P2X receptors. J. Biol. Chem. 284, 24035–24048 (2009).
Shigeoka, A. A. et al. An inflammasome-independent role for epithelial-expressed Nlrp3 in renal ischemia-reperfusion injury. J. Immunol. 185, 6277–6285 (2010).
Vilaysane, A. et al. The NLRP3 inflammasome promotes renal inflammation and contributes to CKD. J. Am. Soc. Nephrol. 21, 1732–1744 (2010).
Wang, W. et al. Inflammasome-independent NLRP3 augments TGF-β signaling in kidney epithelium. J. Immunol. 190, 1239–1249 (2013). This paper shows that NLRP3 and the adaptor protein ASC are needed for TGFβR1 signalling, which might contribute to kidney fibrosis.
Eddy, A. A. Scraping fibrosis: UMODulating renal fibrosis. Nature Med. 17, 553–555 (2011).
Saemann, M. D. et al. Tamm-Horsfall glycoprotein links innate immune cell activation with adaptive immunity via a Toll-like receptor-4-dependent mechanism. J. Clin. Invest. 115, 468–475 (2005).
Darisipudi, M. N. et al. Uromodulin triggers IL-1β-dependent innate immunity via the NLRP3 Inflammasome. J. Am. Soc. Nephrol. 23, 1783–1789 (2012).
Sethi, S. & Fervenza, F. C. Membranoproliferative glomerulonephritis — a new look at an old entity. N. Engl. J. Med. 366, 1119–1131 (2012).
Fakhouri, F., Fremeaux-Bacchi, V., Noel, L. H., Cook, H. T. & Pickering, M. C. C3 glomerulopathy: a new classification. Nature Rev. Nephrol. 6, 494–499 (2010).
Chen, Q. et al. Combined C3b and factor B autoantibodies and MPGN type II. N. Engl. J. Med. 365, 2340–2342 (2011).
Martinez-Barricarte, R. et al. Human C3 mutation reveals a mechanism of dense deposit disease pathogenesis and provides insights into complement activation and regulation. J. Clin. Invest. 120, 3702–3712 (2010).
Gale, D. P. et al. Identification of a mutation in complement factor H-related protein 5 in patients of Cypriot origin with glomerulonephritis. Lancet 376, 794–801 (2010).
Bomback, A. S. & Appel, G. B. Pathogenesis of the C3 glomerulopathies and reclassification of MPGN. Nature Rev. Nephrol. 8, 634–642 (2012).
Vivarelli, M., Pasini, A. & Emma, F. Eculizumab for the treatment of dense-deposit disease. N. Engl. J. Med. 366, 1163–1165 (2012).
Daina, E., Noris, M. & Remuzzi, G. Eculizumab in a patient with dense-deposit disease. N. Engl. J. Med. 366, 1161–1163 (2012).
Trachtman, H., Austin, C., Lewinski, M. & Stahl, R. A. Renal and neurological involvement in typical Shiga toxin-associated HUS. Nature Rev. Nephrol. 8, 658–669 (2012).
Morigi, M. et al. Alternative pathway activation of complement by Shiga toxin promotes exuberant C3a formation that triggers microvascular thrombosis. J. Immunol. 187, 172–180 (2011).
Thurman, J. M. et al. Alternative pathway of complement in children with diarrhea-associated hemolytic uremic syndrome. Clin. J. Am. Soc. Nephrol. 4, 1920–1924 (2009).
Lapeyraque, A. L. et al. Eculizumab in severe Shiga-toxin-associated HUS. N. Engl. J. Med. 364, 2561–2563 (2011).
Noris, M., Mescia, F. & Remuzzi, G. STEC-HUS, atypical HUS and TTP are all diseases of complement activation. Nature Rev. Nephrol. 8, 622–633 (2012).
Legendre, C. M. et al. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N. Engl. J. Med. 368, 2169–2181 (2013). This is a groundbreaking study showing that terminal complement inhibition (using eculizumab) abolishes complement-mediated thrombotic microangiopathy and results in a time-dependent improvement in renal function in patients with aHUS.
Furlan, M. et al. von Willebrand factor-cleaving protease in thrombotic thrombocytopenic purpura and the hemolytic-uremic syndrome. N. Engl. J. Med. 339, 1578–1584 (1998).
Schreiber, A. et al. C5a receptor mediates neutrophil activation and ANCA-induced glomerulonephritis. J. Am. Soc. Nephrol. 20, 289–298 (2009).
Dixon, F. J. What are sensitized cells doing in glomerulonephritis? N. Engl. J. Med. 283, 536–537 (1970).
Couser, W. G. Sensitized cells come of age: a new era in renal immunology with important therapeutic implications. J. Am. Soc. Nephrol. 10, 664–665 (1999).
Bolton, W. K. What sensitized cells just might be doing in glomerulonephritis. J. Clin. Invest. 109, 713–714 (2002).
Sung, S. S. & Bolton, W. K. T cells and dendritic cells in glomerular disease: the new glomerulotubular feedback loop. Kidney Int. 77, 393–399 (2010).
Tipping, P. G. & Holdsworth, S. R. T cells in crescentic glomerulonephritis. J. Am. Soc. Nephrol. 17, 1253–1263 (2006).
Turner, J. E. et al. IL-17A production by renal γδ T cells promotes kidney injury in crescentic GN. J. Am. Soc. Nephrol. 23, 1486–1495 (2012).
Summers, S. A. et al. Th1 and Th17 cells induce proliferative glomerulonephritis. J. Am. Soc. Nephrol. 20, 2518–2524 (2009).
Paust, H. J. et al. The IL-23/Th17 axis contributes to renal injury in experimental glomerulonephritis. J. Am. Soc. Nephrol. 20, 969–979 (2009).
Turner, J. E. et al. CCR6 recruits regulatory T cells and Th17 cells to the kidney in glomerulonephritis. J. Am. Soc. Nephrol. 21, 974–985 (2010).
Steinmetz, O. M. et al. The Th17-defining transcription factor RORγt promotes glomerulonephritis. J. Am. Soc. Nephrol. 22, 472–483 (2011).
Riedel, J. H. et al. Immature renal dendritic cells recruit regulatory CXCR6+ invariant natural killer T cells to attenuate crescentic GN. J. Am. Soc. Nephrol. 23, 1987–2000 (2012). This paper is one of the first descriptions of regulatory iNKT cells having an anti-inflammatory effect. It shows that immature renal DCs attract these cells via the CXCL16–CXCR6 axis to attenuate crescentic glomerulonephritis.
Panzer, U. et al. Chemokine receptor CXCR3 mediates T cell recruitment and tissue injury in nephrotoxic nephritis in mice. J. Am. Soc. Nephrol. 18, 2071–2084 (2007).
Menke, J. et al. CXCL9, but not CXCL10, promotes CXCR3-dependent immune-mediated kidney disease. J. Am. Soc. Nephrol. 19, 1177–1189 (2008).
Chavele, K. M. et al. Mannose receptor interacts with Fc receptors and is critical for the development of crescentic glomerulonephritis in mice. J. Clin. Invest. 120, 1469–1478 (2010).
Wolf, D. et al. CD4+CD25+ regulatory T cells inhibit experimental anti-glomerular basement membrane glomerulonephritis in mice. J. Am. Soc. Nephrol. 16, 1360–1370 (2005). This is the first study to describe a role for T Reg cells in kidney disease.
Paust, H. J. et al. Regulatory T cells control the Th1 immune response in murine crescentic glomerulonephritis. Kidney Int. 80, 154–164 (2011).
Ooi, J. D. et al. Endogenous foxp3+ T-regulatory cells suppress anti-glomerular basement membrane nephritis. Kidney Int. 79, 977–986 (2011).
Hochheiser, K. et al. Kidney dendritic cells become pathogenic during crescentic glomerulonephritis with proteinuria. J. Am. Soc. Nephrol. 22, 306–316 (2011).
Feng, L. et al. Prevention of crescentic glomerulonephritis by immunoneutralization of the fractalkine receptor CX3CR1 rapid communication. Kidney Int. 56, 612–620 (1999).
Goldwich, A. et al. Podocytes are nonhematopoietic professional antigen-presenting cells. J. Am. Soc. Nephrol. 24, 906–916 (2013).
Li, S., Kurts, C., Kontgen, F., Holdsworth, S. R. & Tipping, P. G. Major histocompatibility complex class II expression by intrinsic renal cells is required for crescentic glomerulonephritis. J. Exp. Med. 188, 597–602 (1998).
Ryu, M. et al. Plasma leakage through glomerular basement membrane ruptures triggers the proliferation of parietal epithelial cells and crescent formation in non-inflammatory glomerular injury. J. Pathol. 228, 448–494 (2012).
Neale, T. J., Tipping, P. G., Carson, S. D. & Holdsworth, S. R. Participation of cell-mediated immunity in deposition of fibrin in glomerulonephritis. Lancet 2, 421–424 (1988).
Zaferani, A. et al. Identification of tubular heparan sulfate as a docking platform for the alternative complement component properdin in proteinuric renal disease. J. Biol. Chem. 286, 5359–5367 (2011).
Macconi, D. et al. Proteasomal processing of albumin by renal dendritic cells generates antigenic peptides. J. Am. Soc. Nephrol. 20, 123–130 (2009).
Ooi, J. D., Holdsworth, S. R. & Kitching, A. R. Advances in the pathogenesis of Goodpasture's disease: From epitopes to autoantibodies to effector T cells. J. Autoimmun. 31, 295–300 (2008).
Reynolds, J. Strain differences and the genetic basis of experimental autoimmune anti-glomerular basement membrane glomerulonephritis. Int. J. Exp. Pathol. 92, 211–217 (2011).
Pedchenko, V. et al. Molecular architecture of the Goodpasture autoantigen in anti-GBM nephritis. N. Engl. J. Med. 363, 343–354 (2010). This paper clarifies the crucial importance of autoantigen conformation for autoantibody binding in anti-GBM disease.
Olaru, F. et al. Proteolysis breaks tolerance toward intact α345(IV) collagen, eliciting novel anti-glomerular basement membrane autoantibodies specific for α345NC1 hexamers. J. Immunol. 190, 1424–1432 (2013).
Phelps, R. G. & Rees, A. J. The HLA complex in Goodpasture's disease: A model for analyzing susceptibility to autoimmunity. Kidney Int. 56, 1638–1653 (1999).
Cairns, L. S. et al. The fine specificity and cytokine profile of T-helper cells responsive to the α3 chain of type IV collagen in Goodpasture´s disease. J. Am. Soc. Nephrol. 14, 2801–2812 (2003).
Zou, J. et al. Healthy individuals have Goodpasture autoantigen-reactive T cells. J. Am. Soc. Nephrol. 19, 396–404 (2008).
Ooi, J. D. et al. The HLA-DRB1*15: 01-restricted Goodpasture's T cell epitope induces GN. J. Am. Soc. Nephrol. 24, 419–431 (2013). This study shows the crucial role of HLA-DR*15:01 in the autoimmune response to the α3(IV)NC1, which is the target of autoimmunity in anti-GBM disease.
Phelps, R. G., Jones, V. L., Coughlan, M., Turner, A. N. & Rees, A. J. Presentation of the Goodpasture autoantigen to CD4 T cells Is influenced more by processing constraints than by HLA class II peptide binding preferences. J. Biol. Chem. 273, 11440–11447 (1998).
Kerjaschki, D. & Farquhar, M. G. Immunocytochemical localization of the Heymann nephritis antigen (GP330) in glomerular epithelial cells of normal Lewis rats. J. Exp. Med. 157, 667–686 (1983).
Beck, L. H. Jr et al. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N. Engl. J. Med. 361, 11–21 (2009). This seminal paper identifies PLA2R as the enigmatic autoantigen in membranous nephropathy.
Stahl, R., Hoxha, E. & Fechner, K. PLA2R autoantibodies and recurrent membranous nephropathy after transplantation. N. Engl. J. Med. 363, 496–498 (2010).
Stanescu, H. C. et al. Risk HLA-DQA1 and PLA2R1 alleles in idiopathic membranous nephropathy. N. Engl. J. Med. 364, 616–626 (2011).
Suzuki, H. et al. IgA1-secreting cell lines from patients with IgA nephropathy produce aberrantly glycosylated IgA1. J. Clin. Invest. 118, 629–639 (2008).
Suzuki, H. et al. Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J. Clin. Invest. 119, 1668–1677 (2009). This is a seminal paper indicating that glycan-specific autoantibodies are associated with IgA nephropathy and may represent a disease-specific marker and potential therapeutic target.
Berthoux, F. et al. Autoantibodies targeting galactose-deficient IgA1 associate with progression of IgA nephropathy. J. Am. Soc. Nephrol. 23, 1579–1587 (2012).
Goodnow, C. C. Multistep pathogenesis of autoimmune disease. Cell 130, 25–35 (2007).
Hakkim, A. et al. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc. Natl Acad. Sci. USA 107, 9813–9818 (2010).
Bosch, X. Systemic lupus erythematosus and the neutrophil. N. Engl. J. Med. 365, 758–760 (2011).
Marshak-Rothstein, A. & Rifkin, I. R. Immunologically active autoantigens: the role of toll-like receptors in the development of chronic inflammatory disease. Annu. Rev. Immunol. 25, 419–441 (2007).
Guiducci, C. et al. TLR recognition of self nucleic acids hampers glucocorticoid activity in lupus. Nature 465, 937–941 (2010). This study shows that TLR7 and TLR9 signalling drives lupus nephritis by rendering DCs resistant to steroids.
Pawar, R. D. et al. Inhibition of Toll-like receptor-7 (TLR-7) or TLR-7 plus TLR-9 attenuates glomerulonephritis and lung injury in experimental lupus. J. Am. Soc. Nephrol. 18, 1721–1731 (2007).
Savarese, E. et al. U1 small nuclear ribonucleoprotein immune complexes induce type I interferon in plasmacytoid dendritic cells through TLR7. Blood 107, 3229–3234 (2006).
Garcia-Romo, G. S. et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci. Transl. Med. 3, 73ra20 (2011).
Theofilopoulos, A. N., Baccala, R., Beutler, B. & Kono, D. H. Type I interferons (α/β) in immunity and autoimmunity. Annu. Rev. Immunol. 23, 307–336 (2005).
Migliorini, A. & Anders, H. J. A novel pathogenetic concept-antiviral immunity in lupus nephritis. Nature Rev. Nephrology 8, 183–189 (2012).
Yung, S., Cheung, K. F., Zhang, Q. & Chan, T. M. Anti-dsDNA antibodies bind to mesangial annexin II in lupus nephritis. J. Am. Soc. Nephrol. 21, 1912–1927 (2010).
Mortensen, E. S. & Rekvig, O. P. Nephritogenic potential of anti-DNA antibodies against necrotic nucleosomes. J. Am. Soc. Nephrol. 20, 696–704 (2009).
Weening, J. J. et al. The classification of glomerulonephritis in systemic lupus erythematosus revisited. J. Am. Soc. Nephrol. 15, 241–250 (2004).
Chen, M. & Kallenberg, C. G. ANCA-associated vasculitides — advances in pathogenesis and treatment. Nature Rev. Rheumatol 6, 653–664 (2010).
Kain, R. et al. Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nature Med. 14, 1088–1096 (2008). This paper describes molecular mimicry of bacterial antigens underlying pauci-immune FNGN.
Kain, R. et al. High prevalence of autoantibodies to hLAMP-2 in anti-neutrophil cytoplasmic antibody-associated vasculitis. J. Am. Soc. Nephrol. 23, 556–566 (2012).
Roth, A. J. et al. Anti-LAMP-2 antibodies are not prevalent in patients with antineutrophil cytoplasmic autoantibody glomerulonephritis. J. Am. Soc. Nephrol. 23, 545–555 (2012).
Kessenbrock, K. et al. Netting neutrophils in autoimmune small-vessel vasculitis. Nature Med. 15, 623–625 (2009).
Gou, S.-J., Yuan, J., Chen, M., Yu, F. & Zhao, M.-H. Circulating complement activation in patients with anti-neutrophil cytoplasmic antibody-associated vasculitis. Kidney Int. 83, 129–137 (2013).
Schreiber, A. & Kettritz, R. The neutrophil in antineutrophil cytoplasmic autoantibody-associated vasculitis. J. Leukoc. Biol. http://dx.doi.org/10.1189/jlb.1012525 (2013).
Abdulahad, W. H., Lamprecht, P. & Kallenberg, C. G. T-helper cells as new players in ANCA-associated vasculitides. Arthritis Res. Ther. 13, 236 (2011).
Velden, J. et al. Renal IL-17 expression in human ANCA-associated glomerulonephritis. Am. J. Physiol. Renal Physiol. 302, F1663–F1673 (2012).
McKinney, E. F. et al. A CD8+ T cell transcription signature predicts prognosis in autoimmune disease. Nature Med. 16, 586–591 (2010).
Lyons, P. A. et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 367, 214–223 (2012).
Jennette, J. C., Falk, R. J., Hu, P. & Xiao, H. Pathogenesis of antineutrophil cytoplasmic autoantibody-associated small-vessel vasculitis. Annu. Rev. Pathol. 8, 139–160 (2013).
Heeringa, P. & Little, M. A. In vivo approaches to investigate ANCA-associated vasculitis: lessons and limitations. Arthritis Res. Ther. 13, 204 (2011).
Kuligowski, M. P. et al. Antimyeloperoxidase antibodies rapidly induce α4-integrin-dependent glomerular neutrophil adhesion. Blood 113, 6485–6494 (2009).
Little, M. A. et al. Anti-proteinase 3 anti-neutrophil cytoplasm autoantibodies recapitulate systemic vasculitis in mice with a humanized immune system. PLoS ONE 7, e28626 (2012).
Ruth, A. J. et al. Anti-neutrophil cytoplasmic antibodies and effector CD4+ cells play nonredundant roles in anti-myeloperoxidase crescentic glomerulonephritis. J. Am. Soc. Nephrol. 17, 1940–1949 (2006).
Tan, D. S. et al. Thymic deletion and regulatory T cells prevent antimyeloperoxidase GN. J. Am. Soc. Nephrol. 24, 573–585 (2013).
Ooi, J. D. et al. The immunodominant myeloperoxidase T-cell epitope induces local cell-mediated injury in antimyeloperoxidase glomerulonephritis. Proc. Natl Acad. Sci. USA 109, E2615–E2624 (2012).
Gan, P. Y. et al. Th17 cells promote autoimmune anti-myeloperoxidase glomerulonephritis. J. Am. Soc. Nephrol. 21, 925–931 (2010).
Tadema, H. et al. Bacterial DNA motifs trigger ANCA production in ANCA-associated vasculitis in remission. Rheumatology (Oxford) 50, 689–696 (2011).
Tadema, H., Kallenberg, C. G., Stegeman, C. A. & Heeringa, P. Reactivity against complementary proteinase-3 is not increased in patients with PR3-ANCA-associated vasculitis. PLoS ONE 6, e17972 (2011).
Vaziri, N. D. CKD impairs barrier function and alters microbial flora of the intestine: a major link to inflammation and uremic toxicity. Curr. Opin. Nephrol. Hypertens. 21, 587–592 (2012).
Lech, M., Rommele, C. & Anders, H. J. Pentraxins in nephrology: C-reactive protein, serum amyloid P and pentraxin-3. Nephrol. Dial. Transplant. 28, 803–811 (2013).
Carrero, J. J. & Stenvinkel, P. Inflammation in end-stage renal disease — what have we learned in 10 years? Semin. Dial. 23, 498–509 (2010).
Cohen, G. & Horl, W. H. Immune dysfunction in uremia — an update. Toxins 4, 962–990 (2012).
Weichhart, T. et al. Serum amyloid A in uremic HDL promotes inflammation. J. Am. Soc. Nephrol. 23, 934–947 (2012).
Anders, H. J., Andersen, K. & Stecher, B. The intestinal microbiota, a leaky gut, and abnormal immunity in kidney disease. Kidney Int. 83, 1010–1016 (2013).
Meyer, T. W. & Hostetter, T. H. Uremic solutes from colon microbes. Kidney Int. 81, 949–954 (2012).
McIntyre, C. W. et al. Circulating endotoxemia: a novel factor in systemic inflammation and cardiovascular disease in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 6, 133–141 (2011).
Hotchkiss, R. S., Coopersmith, C. M., McDunn, J. E. & Ferguson, T. A. The sepsis seesaw: tilting toward immunosuppression. Nature Med. 15, 496–497 (2009).
Stearns-Kurosawa, D. J., Osuchowski, M. F., Valentine, C., Kurosawa, S. & Remick, D. G. The pathogenesis of sepsis. Annu. Rev. Pathol. 6, 19–48 (2011).
Oppermann, M. et al. Elevated plasma levels of the immunosuppressive complement fragment Ba in renal failure. Kidney Int. 40, 939–947 (1991).
Pelajo, C. F., Lopez-Benitez, J. M. & Miller, L. C. Vitamin D and autoimmune rheumatologic disorders. Autoimmun Rev. 9, 507–510 (2010).
Vaziri, N. D. Oxidative stress in uremia: nature, mechanisms, and potential consequences. Semin. Nephrol. 24, 469–473 (2004).
Herrada, A. A. et al. Aldosterone promotes autoimmune damage by enhancing Th17-mediated immunity. J. Immunol. 184, 191–202 (2010).
Kleinewietfeld, M. et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 496, 518–522 (2013).
Wu, C. et al. Induction of pathogenic TH17 cells by inducible salt-sensing kinase SGK1. Nature 496, 513–517 (2013).
Madhur, M. S. et al. Interleukin 17 promotes angiotensin II-induced hypertension and vascular dysfunction. Hypertension 55, 500–507 (2010).
Machnik, A. et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nature Med. 15, 545–552 (2009).
Harrison, D. G., Marvar, P. J. & Titze, J. M. Vascular inflammatory cells in hypertension. Front. Physiol. 3, 128 (2012).
Acknowledgements
We apologize to all colleagues whose work could not be cited or discussed in greater detail due to space restrictions. The authors are supported by the German Research foundation (DFG Klinische Forschergruppe 228, SFB704 and TR57, Graduiertenkolleg 1202 and Excellence Cluster ImmunoSensation) and the EU Consortia INTRICATE and REDDSTAR.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
Animal models of immune-mediated kidney disease (PDF 281 kb)
Glossary
- Nephrons
-
Anatomically and functionally independent kidney units that each consist of one glomerulus and one tubule. The nephron delivers urine into collecting ducts that empty into the renal pelvis and, through the ureters, into the urinary bladder.
- Glomerulus
-
An anatomical structure that is located in the kidney cortex and that filters blood into the tubular system.
- Tubulointerstitium
-
The space between the tubuli and glomeruli, which contains capillaries, fibroblasts and dendritic cells, and thus is an important site for the progression of nephritis.
- Bacterial pyelonephritis
-
A bacterial infection of the kidney, mostly due to uropathogenic Escherichia coli that ascend through the urethra, bladder and ureter into the kidneys.
- Tubules
-
Hairpin-like structures that receive filtered blood. The tubular epithelium reabsorbs water, electrolytes, nutrients and proteins. Each nephron has a single tubule, which defines proximal and distal tubules as parts of the nephron.
- Chronic kidney disease
-
(CKD). Chronic (and often progressive) impairment of renal functions, such as blood purification, barrier function of the glomerular filter, water, electrolyte and acid–base homeostasis, endocrine functions such as vitamin D processing, erythropoietin production and blood pressure regulation.
- Uraemia
-
End-stage chronic kidney disease, the treatment of which requires dialysis or kidney transplantation.
- Glomerulonephritis
-
A heterogeneous group of immune-mediated kidney diseases that initiate in the glomeruli.
- Podocyte
-
A visceral epithelial cell that covers the glomerular capillaries in the Bowman's capsule. Podocytes are a component of the glomerular filtration barrier.
- Fibrocytes
-
Monocyte-derived collagen-producing cells that have been suggested to contribute to kidney fibrosis.
- Kidney fibrosis
-
The end stage of chronic kidney disease, when functional renal tissue has been replaced by fibrotic scar tissue and is usually accompanied by uraemia.
- Inflammasome
-
An intracellular complex containing pattern recognition receptors that activate caspase 1. Caspase 1 activation induces pyroptotic cell death and interleukin-1β (IL-1β) and IL-18 secretion.
- Haemolytic uraemic syndrome
-
(HUS). A group of diseases, which are induced by infection with Shiga toxin-producing bacteria, or by genetic or acquired defects in complement regulators, that are characterized by microvascular injury and thrombosis, which results in haemolytic anaemia, thrombocytopenia and organ dysfunction (kidney and often brain).
- Thrombotic thrombocytopenic purpura
-
(TTP). A rare life-threatening disease, characterized by the development of platelet thrombi and microvascular injury, which results from either genetic or acquired defects of the enzyme a disintegrin and metalloproteinase with thrombospondin motifs 13 (ADAMTS13), which has a unique role in the homeostasis of the coagulation system.
- Anti-neutrophil cytoplasmic antibody
-
(ANCA). An autoantibody that is commonly found in pauci-immune focal necrotizing glomerulonephritis.
- Crescentic glomerulonephritis
-
A rapidly progressive form of glomerulonephritis characterized by the hyperproliferation of parietal epithelial cells, which is driven by T cell and macrophage infiltrates and by plasma components leaking through the glomerular filter.
- Delayed-type hypersensitivity
-
(DTH). An inappropriate T cell-initiated response to self or foreign antigens that is carried out by macrophages, eosinophils or cytotoxic T cells.
- Proteinuria
-
The urinary loss of protein, which has numerous clinical consequences. Proteinuria is also used as a biomarker for renal filter dysfunction.
- Anti-GBM disease
-
(Anti-glomerular basement membrane disease; also known as Goodpasture's disease). A severe form of crescentic glomerulonephritis caused by autoantibodies that are specific for the NC1 domain of the α3 chain of type IV collagen (α3(IV)NC1) in the GBM.
- Membranous nephropathy
-
A glomerulonephritis form characterized by the subepithelial deposition of secretory phospholipase A2 receptor (PLA2R)-specific antibodies, which leads to podocyte injury and heavy proteinuria. It is the most common cause of the nephrotic syndrome in adults.
- Nephrotic syndrome
-
A syndrome characterized by heavy proteinuria, hypoalbuminaemia and a loss of immunoglobulins, which results in humoral immunodeficiency, oedema, hyperlipidaemia and thrombosis. This syndrome results from damage to the glomerular filter, which causes the loss of proteins above 50 kDa in size from the circulation.
- IgA nephropathy
-
The most common form of glomerulonephritis worldwide. It is characterized by the deposition of IgA-containing immune complexes in the mesangial compartment of glomeruli, which leads to mesangial cell-proliferative lesions, haematuria and proteinuria.
- Pauci-immune focal necrotizing glomerulonephritis
-
(Pauci-immune FNGN). A highly inflammatory form of glomerulonephritis in which glomerular immune complex deposits are absent or scarce. It is commonly associated with small vessel vasculitis and with anti-neutrophil cytoplasmic antibodies.
- NETosis
-
The formation and the release of neutrophil extracellular traps (NETs) by activated neutrophils to ensnare invading microorganisms. NETs enhance neutrophil killing of extracellular pathogens while minimizing damage to the host cells.
- Humanized mice
-
Immunodeficient mice that are engrafted with human haematopoietic cells or tissues, or mice that transgenically express human genes.
- Endotoxin tolerance
-
A transient state of hyporesponsiveness of the host or of cultured macrophages and/or monocytes to lipopolysaccharide (LPS) following previous exposure to LPS.
Rights and permissions
About this article
Cite this article
Kurts, C., Panzer, U., Anders, HJ. et al. The immune system and kidney disease: basic concepts and clinical implications. Nat Rev Immunol 13, 738–753 (2013). https://doi.org/10.1038/nri3523
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3523
This article is cited by
-
The causal relationship between COVID-19 and estimated glomerular filtration rate: a bidirectional Mendelian randomization study
BMC Nephrology (2024)
-
Combined Plasma Olink Proteomics and Transcriptomics Identifies CXCL1 and TNFRSF12A as Potential Predictive and Diagnostic Inflammatory Markers for Acute Kidney Injury
Inflammation (2024)
-
Clinical and epidemiological factors causing longer SARS-CoV 2 viral shedding: the results from the CoviCamp cohort
Infection (2024)
-
Glomerulonephritis: immunopathogenesis and immunotherapy
Nature Reviews Immunology (2023)
-
Glomerular injury after trauma, burn, and sepsis
Journal of Nephrology (2023)