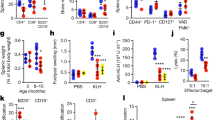
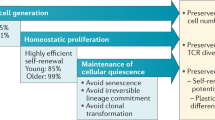
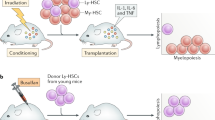
Key Points
-
The ageing of the immune system is initiated at the very top of the haematopoietic hierarchy, and the ageing of haematopoietic stem cells (HSCs) directly contributes to changes in the immune system.
-
In this Review, the authors summarize the phenotypes of ageing HSCs and discuss how the cell-intrinsic and cell-extrinsic mechanisms of HSC ageing might promote immunosenescence. Loss of polarity in HSCs mediated by increased activity of the RHO GTPase cell division control protein 42 (CDC42) is a novel ageing-associated phenotype in HSCs.
-
The relevance of epigenetic stability in stem cell ageing is only now beginning to emerge. Changes in the epigenetic landscape of HSCs upon ageing might be involved in the ageing of stem cells.
-
Moreover, new research supports the idea that the ageing of the stem cell niche contributes to the ageing of HSCs.
-
The ageing of HSCs can be ameliorated, for example, by pharmacological inhibition of CDC42 or by targeting the mammalian target of rapamycin (mTOR) pathway.
-
Several questions and controversies remain in stem cell ageing research. Dissecting the causes and the consequences of HSC ageing remains a major challenge.
Abstract
Stem cell ageing underlies the ageing of tissues, especially those with a high cellular turnover. There is growing evidence that the ageing of the immune system is initiated at the very top of the haematopoietic hierarchy and that the ageing of haematopoietic stem cells (HSCs) directly contributes to changes in the immune system, referred to as immunosenescence. In this Review, we summarize the phenotypes of ageing HSCs and discuss how the cell-intrinsic and cell-extrinsic mechanisms of HSC ageing might promote immunosenescence. Stem cell ageing has long been considered to be irreversible. However, recent findings indicate that several molecular pathways could be targeted to rejuvenate HSCs and thus to reverse some aspects of immunosenescence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rossi, D. J., Jamieson, C. H. & Weissman, I. L. Stems cells and the pathways to aging and cancer. Cell 132, 681–696 (2008).
Henry, C. J., Marusyk, A. & DeGregori, J. Aging-associated changes in hematopoiesis and leukemogenesis: what's the connection? Aging 3, 643–656 (2011).
Weiskopf, D., Weinberger, B. & Grubeck-Loebenstein, B. The aging of the immune system. Transplant Int. 22, 1041–1050 (2009).
Frasca, D. & Blomberg, B. B. Aging affects human B cell responses. J. Clin. Immunol. 31, 430–435 (2011).
Henry, C. J., Marusyk, A., Zaberezhnyy, V., Adane, B. & DeGregori, J. Declining lymphoid progenitor fitness promotes aging-associated leukemogenesis. Proc. Natl Acad. Sci. USA 107, 21713–21718 (2010).
Linton, P. J. & Dorshkind, K. Age-related changes in lymphocyte development and function. Nature Immunol. 5, 133–139 (2004).
Geiger, H. & Rudolph, K. L. Aging in the lympho-hematopoietic stem cell compartment. Trends Immunol. 30, 360–365 (2009).
Goronzy, J. J. & Weyand, C. M. T cell development and receptor diversity during aging. Curr. Opin. Immunol. 17, 468–475 (2005).
Saurwein-Teissl, M. et al. Lack of antibody production following immunization in old age: association with CD8+CD28− T cell clonal expansions and an imbalance in the production of Th1 and Th2 cytokines. J. Immunol. 168, 5893–5899 (2002).
Gibson, K. L. et al. B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 8, 18–25 (2009).
Cancro, M. P. et al. B cells and aging: molecules and mechanisms. Trends Immunol. 30, 313–318 (2009).
Kuranda, K. et al. Age-related changes in human hematopoietic stem/progenitor cells. Aging Cell 10, 542–546 (2011).
Plowden, J., Renshaw-Hoelscher, M., Engleman, C., Katz, J. & Sambhara, S. Innate immunity in aging: impact on macrophage function. Aging Cell 3, 161–167 (2004).
Franceschi, C. et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. NY Acad. Sci. 908, 244–254 (2000).
Miller, J. P. & Allman, D. Linking age-related defects in B lymphopoiesis to the aging of hematopoietic stem cells. Semin. Immunol. 17, 321–329 (2005).
Szilvassy, S. J., Nicolini, F. E., Eaves, C. J. & Miller, C. L. Quantitation of murine and human hematopoietic stem cells by limiting-dilution analysis in competitively repopulated hosts. Methods Mol. Med. 63, 167–187 (2002).
Harrison, D. E., Jordan, C. T., Zhong, R. K. & Astle, C. M. Primitive hemopoietic stem cells: direct assay of most productive populations by competitive repopulation with simple binomial, correlation and covariance calculations. Exp. Hematol. 21, 206–219 (1993).
Kent, D. G. et al. Prospective isolation and molecular characterization of hematopoietic stem cells with durable self-renewal potential. Blood 113, 6342–6350 (2009).
Rossi, D. J. et al. Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc. Natl Acad. Sci. USA 102, 9194–9199 (2005). This article demonstrates that the changes in stem and progenitor cell number upon ageing are primarily regulated by stem cell-intrinsic mechanisms.
de Haan, G., Nijhof, W. & Van Zant, G. Mouse strain-dependent changes in frequency and proliferation of hematopoietic stem cells during aging: correlation between lifespan and cycling activity. Blood 89, 1543–1550 (1997).
Morrison, S. J., Wandycz, A. M., Akashi, K., Globerson, A. & Weissman, I. L. The aging of hematopoietic stem cells. Nature Med. 2, 1011–1016 (1996).
Chambers, S. M. & Goodell, M. A. Hematopoietic stem cell aging: wrinkles in stem cell potential. Stem Cell Rev. 3, 201–211 (2007).
Beerman, I., Maloney, W. J., Weissmann, I. L. & Rossi, D. J. Stem cells and the aging hematopoietic system. Curr. Opin. Immunol. 22, 500–506 (2010).
Beerman, I. et al. Functionally distinct hematopoietic stem cells modulate hematopoietic lineage potential during aging by a mechanism of clonal expansion. Proc. Natl Acad. Sci. USA 107, 5465–5470 (2010).
Florian, M. C. et al. Cdc42 activity regulates hematopoietic stem cell aging and rejuvenation. Cell Stem Cell 10, 520–530 (2012). This paper identifies a crucial role for CDC42-regulated stem cell polarity after the ageing of HSCs and shows that pharmacological inhibition of CDC42 activity functionally rejuvenates aged HSCs.
Chen, J., Astle, C. M. & Harrison, D. E. Development and aging of primitive hematopoietic stem cells in BALB/cBy mice. Exp. Hematol. 27, 928–935 (1999).
Sudo, K., Ema, H., Morita, Y. & Nakauchi, H. Age-associated characteristics of murine hematopoietic stem cells. J. Exp. Med. 192, 1273–1280 (2000).
Dykstra, B., Olthof, S., Schreuder, J., Ritsema, M. & de Haan, G. Clonal analysis reveals multiple functional defects of aged murine hematopoietic stem cells. J. Exp. Med. 208, 2691–2703 (2011).
Wagner, W., Horn, P., Bork, S. & Ho, A. D. Aging of hematopoietic stem cells is regulated by the stem cell niche. Exp. Gerontol. 43, 974–980 (2008).
Mercier, F. E., Ragu, C. & Scadden, D. T. The bone marrow at the crossroads of blood and immunity. Nature Rev. Immunol. 12, 49–60 (2011).
Larbi, A. et al. Aging of the immune system as a prognostic factor for human longevity. Physiol. 23, 64–74 (2008).
Signer, R. A., Montecino-Rodriguez, E., Witte, O. N., McLaughlin, J. & Dorshkind, K. Age-related defects in B lymphopoiesis underlie the myeloid dominance of adult leukemia. Blood 110, 1831–1839 (2007).
Luc, S., Buza-Vidas, N. & Jacobsen, S. E. Biological and molecular evidence for existence of lymphoid-primed multipotent progenitors. Ann. NY Acad. Sci. 1106, 89–94 (2007).
Pang, W. W. et al. Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc. Natl Acad. Sci. USA 108, 20012–20017 (2011). Along with reference 12, this article describes the ageing-associated phenotypes of human haematopoeitic stem and progenitor cells in great detail.
Muller-Sieburg, C. E., Cho, R. H., Thoman, M., Adkins, B. & Sieburg, H. B. Deterministic regulation of hematopoietic stem cell self-renewal and differentiation. Blood 100, 1302–1309 (2002).
Muller-Sieburg, C. E. & Sieburg, H. B. Clonal diversity of the stem cell compartment. Curr. Opin. Hematol. 13, 243–248 (2006).
Dykstra, B. et al. Long-term propagation of distinct hematopoietic differentiation programs in vivo. Cell Stem Cell 1, 218–229 (2007).
Benz, C. et al. Hematopoietic stem cell subtypes expand differentially during development and display distinct lymphopoietic programs. Cell Stem Cell 10, 273–283 (2012).
Muller-Sieburg, C. E., Cho, R. H., Karlsson, L., Huang, J. F. & Sieburg, H. B. Myeloid-biased hematopoietic stem cells have extensive self-renewal capacity but generate diminished lymphoid progeny with impaired IL-7 responsiveness. Blood 103, 4111–4118 (2004).
Challen, G. A., Boles, N. C., Chambers, S. M. & Goodell, M. A. Distinct hematopoietic stem cell subtypes are differentially regulated by TGF-β1. Cell Stem Cell 6, 265–278 (2010). Data presented in this paper and in reference 24 suggest a clonal diversity model for the ageing of the pool of HSCs rather than a clonal succession or cellular ageing model for HSC ageing.
Morita, Y., Ema, H. & Nakauchi, H. Heterogeneity and hierarchy within the most primitive hematopoietic stem cell compartment. J. Exp. Med. 207, 1173–1182 (2010).
Ergen, A. V., Boles, N. C. & Goodell, M. A. Rantes/Ccl5 influences hematopoietic stem cell subtypes and causes myeloid skewing. Blood 119, 2500–2509 (2012).
Vas, V., Senger, K., Dorr, K., Niebel, A. & Geiger, H. Aging of the microenvironment influences clonality in hematopoiesis. PLoS ONE 7, e42080 (2012).
Adams, G. B. & Scadden, D. T. The hematopoietic stem cell in its place. Nature Immunol. 7, 333–337 (2006).
Kiel, M. J. & Morrison, S. J. Uncertainty in the niches that maintain haematopoietic stem cells. Nature Rev. Immunol. 8, 290–301 (2008).
Lo Celso, C. & Scadden, D. T. The haematopoietic stem cell niche at a glance. J. Cell Sci. 124, 3529–3535 (2011).
Kohler, A. et al. Altered cellular dynamics and endosteal location of aged early hematopoietic progenitor cells revealed by time-lapse intravital imaging in long bones. Blood 114, 290–298 (2009).
Xing, Z. et al. Increased hematopoietic stem cell mobilization in aged mice. Blood 108, 2190–2197 (2006).
Geiger, H., Koehler, A. & Gunzer, M. Stem cells, aging, niche, adhesion and Cdc42: a model for changes in cell–cell interactions and hematopoietic stem cell aging. Cell Cycle 6, 884–887 (2007).
Liang, Y., Van Zant, G. & Szilvassy, S. J. Effects of aging on the homing and engraftment of murine hematopoietic stem and progenitor cells. Blood 106, 1479–1487 (2005). In this article the homing defect of aged HSCs was quantified and added to the emerging list of ageing-asscoiated phenotypes.
Chambers, S. M. et al. Aging hematopoietic stem cells decline in function and exhibit epigenetic dysregulation. PLoS Biol. 5, e201 (2007). This article correlates the ageing of HSCs with epigenetic dysregulation within HSCs.
Baraibar, M. A. & Friguet, B. Oxidative proteome modifications target specific cellular pathways during oxidative stress, cellular senescence and aging. Exp.Gerontol. 2 Nov 2012 (doi:10.1016/j.exger.2012.10.007).
Larsson, N. G. Somatic mitochondrial DNA mutations in mammalian aging. Annu. Rev. Biochem. 79, 683–706 (2010).
Trifunovic, A. et al. Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429, 417–423 (2004).
Ito, K. et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nature Med. 12, 446–451 (2006). This article identifies ROS as an important contributor to the HSC ageing phenotype.
Jang, Y. Y. & Sharkis, S. J. A low level of reactive oxygen species selects for primitive hematopoietic stem cells that may reside in the low-oxygenic niche. Blood 110, 3056–3063 (2007).
Miyamoto, K. et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell 1, 101–112 (2007).
Tothova, Z. et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell 128, 325–339 (2007).
Flachsbart, F. et al. Association of FOXO3A variation with human longevity confirmed in German centenarians. Proc. Natl Acad. Sci. USA 106, 2700–2705 (2009).
Norddahl, G. L. et al. Accumulating mitochondrial DNA mutations drive premature hematopoietic aging phenotypes distinct from physiological stem cell aging. Cell Stem Cell 8, 499–510 (2011). This crucial study separates regulatory mechanisms of premature stem cell ageing, in this case mitochondrial DNA mutations, from physiological stem cell ageing.
Armstrong, L. et al. Overexpression of telomerase confers growth advantage, stress resistance, and enhanced differentiation of ESCs toward the hematopoietic lineage. Stem Cells 23, 516–529 (2005).
Rossi, D. J. et al. Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature 447, 725–729 (2007).
Rossi, D. J. et al. Hematopoietic stem cell quiescence attenuates DNA damage response and permits DNA damage accumulation during aging. Cell Cycle 6, 2371–2376 (2007).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Zimmermann, S. & Martens, U. M. Telomeres, senescence, and hematopoietic stem cells. Cell Tissue Res. 331, 79–90 (2008).
Rube, C. E. et al. Accumulation of DNA damage in hematopoietic stem and progenitor cells during human aging. PLoS ONE 6, e17487 (2011).
Choudhury, A. R. et al. Cdkn1a deletion improves stem cell function and lifespan of mice with dysfunctional telomeres without accelerating cancer formation. Nature Genet. 39, 99–105 (2007).
Manning, E. L., Crossland, J., Dewey, M. J. & Van Zant, G. Influences of inbreeding and genetics on telomere length in mice. Mammal. Genome 13, 234–238 (2002).
Martin-Ruiz, C. M., Gussekloo, J., van Heemst, D., von Zglinicki, T. & Westendorp, R. G. Telomere length in white blood cells is not associated with morbidity or mortality in the oldest old: a population-based study. Aging Cell 4, 287–290 (2005).
Allsopp, R. C., Morin, G. B., DePinho, R., Harley, C. B. & Weissman, I. L. Telomerase is required to slow telomere shortening and extend replicative lifespan of HSCs during serial transplantation. Blood 102, 517–520 (2003).
Wang, J. et al. A differentiation checkpoint limits hematopoietic stem cell self-renewal in response to DNA damage. Cell 148, 1001–1014 (2012).
Mandal, P. K. & Rossi, D. J. DNA-damage-induced differentiation in hematopoietic stem cells. Cell 148, 847–848 (2012).
Donehower, L. A. Using mice to examine p53 functions in cancer, aging, and longevity. Cold Spring Harb. Perspect. Biol. 1, a001081 (2009).
Nitta, E. et al. Telomerase reverse transcriptase protects ATM-deficient hematopoietic stem cells from ROS-induced apoptosis through a telomere-independent mechanism. Blood 117, 4169–4180 (2011).
Janzen, V. et al. Stem-cell ageing modified by the cyclin-dependent kinase inhibitor p16INK4a. Nature 443, 421–426 (2006).
Attema, J. L., Pronk, C. J., Norddahl, G. L., Nygren, J. M. & Bryder, D. Hematopoietic stem cell ageing is uncoupled from p16 INK4A-mediated senescence. Oncogene 28, 2238–2243 (2009).
Freitas, A. A. & de Magalhaes, J. P. A review and appraisal of the DNA damage theory of ageing. Mut. Res. 728, 12–22 (2011).
Sperka, T., Wang, J. & Rudolph, K. L. DNA damage checkpoints in stem cells, ageing and cancer. Nature Rev. Mol. Cell Biol. 13, 579–590 (2012).
Beerman, I. et al. Proliferation-dependent alterations of the DNA methylation landscape underlie hematopoietic stem cell aging. Cell Stem Cell 14 Feb 2013 (doi:10.1016/j.stem.2013.01.017).
Bocker, M. T. et al. Genome-wide promoter DNA methylation dynamics of human hematopoietic progenitor cells during differentiation and aging. Blood 117, e182–e189 (2011).
Hogart, A. et al. Genome-wide DNA methylation profiles in hematopoietic stem and progenitor cells reveal overrepresentation of ETS transcription factor binding sites. Genome Res. 22, 1407–1418 (2012).
Rando, T. A. & Chang, H. Y. Aging, rejuvenation, and epigenetic reprogramming: resetting the aging clock. Cell 148, 46–57 (2012).
Lansdorp, P. M. Immortal strands? Give me a break. Cell 129, 1244–1247 (2007).
Klauke, K. & de Haan, G. Polycomb group proteins in hematopoietic stem cell aging and malignancies. Int. J. Hematol. 94, 11–23 (2011).
Valk-Lingbeek, M. E., Bruggeman, S. W. & van Lohuizen, M. Stem cells and cancer; the polycomb connection. Cell 118, 409–418 (2004).
Greer, E. L. et al. Members of the H3K4 trimethylation complex regulate lifespan in a germline-dependent manner in C. elegans. Nature 466, 383–387 (2010). Although not directly discussing the ageing of HSCs, this paper demonstrates a causative role for epigenetic regulatory pathways in ageing and longevity.
Busque, L. et al. Recurrent somatic TET2 mutations in normal elderly individuals with clonal hematopoiesis. Nature Genet. 44, 1179–1181 (2012).
Tadokoro, Y., Ema, H., Okano, M., Li, E. & Nakauchi, H. De novo DNA methyltransferase is essential for self-renewal, but not for differentiation, in hematopoietic stem cells. J. Exp. Med. 204, 715–722 (2007).
Challen, G. A. et al. Dnmt3a is essential for hematopoietic stem cell differentiation. Nature Genet. 44, 23–31 (2011).
Mohrin, M. et al. Hematopoietic stem cell quiescence promotes error-prone DNA repair and mutagenesis. Cell Stem Cell 7, 174–185 (2010).
Yahata, T. et al. Accumulation of oxidative DNA damage restricts the self-renewal capacity of human hematopoietic stem cells. Blood 118, 2941–2950 (2011).
Noda, S., Ichikawa, H. & Miyoshi, H. Hematopoietic stem cell aging is associated with functional decline and delayed cell cycle progression. Biochem. Biophys. Res. Commun. 383, 210–215 (2009).
Pollina, E. A. & Brunet, A. Epigenetic regulation of aging stem cells. Oncogene 30, 3105–3126 (2011).
Cheng, J. et al. Centrosome misorientation reduces stem cell division during ageing. Nature 456, 599–604 (2008).
Kerber, R. A., O'Brien, E. & Cawthon, R. M. Gene expression profiles associated with aging and mortality in humans. Aging Cell 8, 239–250 (2009).
Yang, L. & Zheng, Y. Cdc42: a signal coordinator in hematopoietic stem cell maintenance. Cell Cycle 6, 1445–1450 (2007).
Florian, M. C. & Geiger, H. Concise review: polarity in stem cells, disease, and aging. Stem Cells 28, 1623–1629 (2010).
Macara, I. G. & Mili, S. Polarity and differential inheritance — universal attributes of life? Cell 135, 801–812 (2008).
Nakamura, Y. et al. Isolation and characterization of endosteal niche cell populations that regulate hematopoietic stem cells. Blood 116, 1422–1432 (2010).
Scadden, D. T. The stem-cell niche as an entity of action. Nature 441, 1075–1079 (2006).
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Katayama, Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124, 407–421 (2006).
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Bellantuono, I., Aldahmash, A. & Kassem, M. Aging of marrow stromal (skeletal) stem cells and their contribution to age-related bone loss. Biochim. Biophys. Acta 1792, 364–370 (2009).
Freemont, A. J. & Hoyland, J. A. Morphology, mechanisms and pathology of musculoskeletal ageing. J. Pathol. 211, 252–259 (2007).
Vas, V., Wandhoff, C., Dorr, K., Niebel, A. & Geiger, H. Contribution of an aged microenvironment to aging-associated myeloproliferative disease. PLoS ONE 7, e31523 (2012).
Tuljapurkar, S. R. et al. Changes in human bone marrow fat content associated with changes in hematopoietic stem cell numbers and cytokine levels with aging. J. Anat. 219, 574–581 (2011).
Kubo, M. et al. Hypoxic preconditioning enhances angiogenic potential of bone marrow cells with aging-related functional impairment. Circ. J. 76, 986–994 (2012).
Gonzalez-Nieto, D. et al. Connexin-43 in the osteogenic BM niche regulates its cellular composition and the bidirectional traffic of hematopoietic stem cells and progenitors. Blood 119, 5144–5154 (2012).
Cancelas, J. A. et al. Connexin-43 gap junctions are involved in multiconnexin-expressing stromal support of hemopoietic progenitors and stem cells. Blood 96, 498–505 (2000).
Taniguchi Ishikawa, E. et al. Connexin-43 prevents hematopoietic stem cell senescence through transfer of reactive oxygen species to bone marrow stromal cells. Proc. Natl Acad. Sci. USA 109, 9071–9076 (2012).
Norddahl, G. L., Wahlestedt, M., Gisler, S., Sigvardsson, M. & Bryder, D. Reduced repression of cytokine signaling ameliorates age-induced decline in hematopoietic stem cell function. Aging Cell 11, 1128–1131 (2012).
Brack, A. S. et al. Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis. Science 317, 807–810 (2007).
Rando, T. A. Stem cells, ageing and the quest for immortality. Nature 441, 1080–1086 (2006).
Adams, G. B. et al. Therapeutic targeting of a stem cell niche. Nature Biotech. 25, 238–243 (2007).
Mendez-Ferrer, S. et al. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 466, 829–834 (2010).
Togari, A. Adrenergic regulation of bone metabolism: possible involvement of sympathetic innervation of osteoblastic and osteoclastic cells. Microsc. Res. Technique 58, 77–84 (2002).
Villeda, S. A. et al. The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 477, 90–94 (2011).
Chen, C. Liu, Y. & Zheng, P. mTOR regulation and therapeutic rejuvenation of aging hematopoietic stem cells. Sci. Signal. 2, ra75 (2009). This article demonstrates a role for mTOR signalling in the ageing and rejuvenation of HSCs.
Conboy, I. M. et al. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 433, 760–764 (2005). This paper shows that a young microenvironment can revert stem cell aging (in this case the ageing of muscle stem cells), and thus demonstrates that the ageing of stem cells is reversible.
Carrillo-Garcia, C. & Janzen, V. Restoring cell polarity: an HSC fountain of youth. Cell Stem Cell 10, 481–482 (2012).
Hall, M. N. mTOR — what does it do? Transplant. Proc. 40, S5–S8 (2008).
Mionnet, C., Bogliolo, S. & Arkowitz, R. A. Oligomerization regulates the localization of Cdc24, the Cdc42 activator in Saccharomyces cerevisiae. J. Biol. Chem. 283, 17515–17530 (2008).
Etienne-Manneville, S. Cdc42 — the centre of polarity. J. Cell Sci. 117, 1291–1300 (2004).
Wang, L., Yang, L., Debidda, M., Witte, D. & Zheng, Y. Cdc42 GTPase-activating protein deficiency promotes genomic instability and premature aging-like phenotypes. Proc. Natl Acad. Sci. USA 104, 1248–1253 (2007).
Wilkinson, J. E. et al. Rapamycin slows aging in mice. Aging Cell 11, 675–682 (2012).
Miller, R. A. et al. Rapamycin, but not resveratrol or simvastatin, extends life span of genetically heterogeneous mice. J. Gerontol. A Biol. Sci. Med. Sci. 66, 191–201 (2011).
Harrison, D. E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Powers, R. W., Kaeberlein, M., Caldwell, S. D., Kennedy, B. K. & Fields, S. Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev. 20, 174–184 (2006).
Katewa, S. D. & Kapahi, P. Role of TOR signaling in aging and related biological processes in Drosophila melanogaster. Exp. Gerontol. 46, 382–390 (2011).
Kapahi, P. et al. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr. Biol. 14, 885–890 (2004).
Liu, L. & Rando, T. A. Manifestations and mechanisms of stem cell aging. J. Cell Biol. 193, 257–266 (2011).
Conboy, I. M. & Rando, T. A. Heterochronic parabiosis for the study of the effects of aging on stem cells and their niches. Cell Cycle 11, 2260–2267 (2012).
Geiger, H., True, J. M., de Haan, G. & Van Zant, G. Age- and stage-specific regulation patterns in the hematopoietic stem cell hierarchy. Blood 98, 2966–2972 (2001).
Purton, L. E. & Scadden, D. T. The hematopoietic stem cell niche. StemBook [online], (2008).
Hoggatt, J. & Scadden, D. T. The stem cell niche: tissue physiology at a single cell level. J. Clin. Invest. 122, 3029–3034 (2012).
Park, D., Sykes, D. B. & Scadden, D. T. The hematopoietic stem cell niche. Front. Biosci. 17, 30–39 (2012).
Gillette, J. M., Larochelle, A., Dunbar, C. E. & Lippincott-Schwartz, J. Intercellular transfer to signalling endosomes regulates an ex vivo bone marrow niche. Nature Cell Biol. 11, 303–311 (2009).
Yamazaki, S. et al. Nonmyelinating Schwann cells maintain hematopoietic stem cell hibernation in the bone marrow niche. Cell 147, 1146–1158 (2011).
Shen, Y. & Nilsson, S. K. Bone, microenvironment and hematopoiesis. Curr. Opin. Hematol. 19, 250–255 (2012).
Reik, W. Stability and flexibility of epigenetic gene regulation in mammalian development. Nature 447, 425–432 (2007)
Lister, R. et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 462, 315–322 (2009).
Lister, R. et al. Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature 471, 68–73 (2011).
Berdasco, M. & Esteller, M. Hot topics in epigenetic mechanisms of aging: 2011. Aging Cell 11, 181–186 (2012).
Handy, D. E., Castro, R. & Loscalzo, J. Epigenetic modifications: basic mechanisms and role in cardiovascular disease. Circulation 123, 2145–2156 (2011).
O'Sullivan, R. J. & Karlseder, J. The great unravelling: chromatin as a modulator of the aging process. Trends Biochem. Sci. 37, 466–476 (2012).
Burgess, R. C., Misteli, T. & Oberdoerffer, P. DNA damage, chromatin, and transcription: the trinity of aging. Curr. Opin. Cell Biol. 24, 724–730 (2012).
Spangrude, G. J., Heimfeld, S. & Weissman, I. L. Purification and characterization of mouse hematopoietic stem cells. Science 241, 58–62 (1988).
Osawa, M., Hanada, K., Hamada, H. & Nakauchi, H. Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science 273, 242–245 (1996).
Goodell, M. A., Brose, K., Paradis, G., Conner, A. S. & Mulligan, R. C. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J. Exp. Med. 183, 1797–1806 (1996).
Matsuzaki, Y., Kinjo, K., Mulligan, R. C. & Okano, H. Unexpectedly efficient homing capacity of purified murine hematopoietic stem cells. Immunity 20, 87–93 (2004).
Yang, L. et al. Identification of Lin−Sca1+kit+CD34+Flt3− short-term hematopoietic stem cells capable of rapidly reconstituting and rescuing myeloablated transplant recipients. Blood 105, 2717–2723 (2005).
Kiel, M. J. et al. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005).
Wilson, A. et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 135, 1118–1129 (2008).
Doulatov, S. et al. Revised map of the human progenitor hierarchy shows the origin of macrophages and dendritic cells in early lymphoid development. Nature Immunol. 11, 585–593 (2010).
Notta, F. et al. Isolation of single human hematopoietic stem cells capable of long-term multilineage engraftment. Science 333, 218–221 (2011).
Acknowledgements
The authors apologize to those researchers whose work could not be discussed or referenced owing to space limitations. The work in the laboratory of H. Geiger is supported by grants from the Deutsche Forschungsgemeinschaft, Germany (KFO 142, GE2063/1 and SFB 1074); the German Federal Ministry of Education and Research (SyStaR); the excellence programme of the Baden-Württemberg Foundation, Germany; the US National Institutes of Health (HL076604, DK077762 and AG040118); the Edward P. Evans foundation, USA; the European Commission (FP7 Marie Curie Initial Training Network MARRIAGE) and a “Bausteinprogramm” from the Department of Medicine, University of Ulm, Germany, awarded to M. C. Florian. Work in the laboratory of G. de Haan is supported by grants from the Netherlands Institute for Regenerative Medicine (NIRM), the European Commission (FP7 Marie Curie Initial Training Networks EuroCancerCSC, HaEM-ID and MARRIAGE) and the Mouse Clinic for Cancer and Ageing (MCCA) funded by the Netherlands Organization for Scientific Research. The authors thank N. Guidi for her contribution to the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- HSC niche
-
A specialized microenvironment that interacts with haematopoietic stem cells (HSCs) to regulate their fate.
- Self-renewal
-
The capacity to recreate stem cells with differentiation potentials that are similar to those of the mother stem cell. Self-renewal can result from either an asymmetric cell division that yields a daughter stem cell and a cell committed to differentiation, or from a symmetric cell division that yields two daughter stem cells. Alternatively, stem cell differentiation can result in the loss of self-renewal capacity.
- Serial transplantation assays
-
Assays used to test the ability of haematopoietic stem cells (HSCs) to undergo self-renewal. The number of serial transplantations that the original donor's bone marrow can perform successfully is a measure of the self-renewal capacity of the HSCs it contains. The serial transplantation of defined numbers of purified HSCs allows the normalization of differences in stem cell frequencies among mice of different ages and offers a more controlled 'per cell' readout than serial transplantation of unfractionated bone marrow cells.
- Symmetric cell divisions
-
Stem cell divisions in which the two daughter cells are identical with respect to function and differentiation potential.
- Common lymphoid progenitor
-
(CLP). A progenitor that is committed to the lymphoid lineage and can give rise to all lymphocyte subsets, including T cells, B cells and natural killer cells. CLP cells are defined as FLK2+IL-7Rα+KITlow/hi SCA1low/hiLIN− in mice, and LIN−CD34+CD38+CD127+ in humans.
- Common myeloid progenitor
-
(CMP). A progenitor that is committed to the myeloid lineage and can give rise to all myeloid subsets, including neutrophils, basophils, monocytes and platelets. CMP cells are defined as LIN−IL-7Rα−SCA1−KIT+FcRIlowCD34+ in mice and LIN−CD34+CD38+ CD123+CD45RA− in humans.
- Bone marrow homing
-
The process whereby haematopoietic stem cells (HSCs) migrate from the circulation to the HSC niche. Homing is distinct from lodgement, which is the ability of HSCs to enter the niche and stay there, as well as from engraftment, which is the ability of HSCs to respond to appropriate maintenance and differentiation signals when they are lodged.
- Quiescence
-
A non-cycling resting state in the G0 phase of the cell cycle that is important for long-term function.
- Reactive oxygen species
-
(ROS). Highly reactive molecules that consist of several diverse chemical species including the superoxide anion (O2•−), hydroxyl radical (OH•) and hydrogen peroxide (H2O2). Because of their potential to cause oxidative deterioration of DNA, proteins and lipids, ROS have been implicated as one of the causative factors of ageing. As ROS are generated mainly as by-products of mitochondrial respiration, mitochondria are thought to be the primary target of oxidative damage.
- Replicative senescence
-
An inability of most cell types to divide indefinitely owing to terminal cell cycle arrest after a defined number of cell cycles, probably as a result of telomere attrition.
- Mammalian target of rapamycin
-
(mTOR). A conserved serine/threonine kinase regulating metabolism and the expression of growth factors in response to environmental cues. mTOR activity is inhibited by the drug rapamycin.
- FOXO protein family
-
A subgroup of the forkhead box (FOX) family of transcription factors. FOXO proteins are regulated by the insulin–phosphoinositide 3-kinase–AKT signalling pathway.
- Telomere
-
A repetitive nucleotide sequence at the end of chromosomes that protects the ends from deterioration or from fusion with neighbouring chromosomes.
- Telomerase
-
An enzyme that is capable of extending the ends of telomeres after replication using an RNA template that is part of the enzyme complex. This counteracts telomere shortening after each cell division cycle.
- H2AX
-
Histone 2A family, member X; phosphorylated H2AX is a sensitive marker for DNA double-strand breaks. Almost every DNA double-strand break forms a H2AX focus, but whether every H2AX focus identifies a double-strand break remains controversial.
- p53
-
A tumour suppressor protein that responds to diverse cellular stresses by regulating target genes that induce cell cycle arrest, apoptosis, senescence, DNA repair or changes in metabolism.
- p16INK4A
-
A cyclin-dependent kinase inhibitor that stabilizes cell cycle arrest by activating the checkpoint activity of the retinoblastoma protein.
- BCL-2 family
-
A family of proteins containing at least one BCL-2 homology (BH) region. The family is divided into anti-apoptotic multidomain proteins (such as BCL-2 and BCL-XL), which contain four BH domains (BH1, BH2, BH3 and BH4), pro-apoptotic multidomain proteins (for example, BAX and BAK), which contain BH1, BH2 and BH3 domains, and the pro-apoptotic BH3-only family (such as BH3-interacting-domain death agonist (BID), BCL-2-interacting mediator of cell death (BIM) and p53-upregulated modulator of apoptosis (PUMA)).
- Epigenetic information
-
Non-genetic information that promotes alternative cell states and is regulated by developmental and environmental cues. Epigenetic information maintains distinct phenotypes among cells that share identical DNA sequences.
- DNA methyltransferases
-
(DNMTs). Enzymes that transfer methyl groups from S-adenosylmethionine to specific adenines or cytosines in DNA.
- Histone methyltransferases
-
Enzymes that catalyse the transfer of methyl groups to lysine and/or arginine residues on histones; the most well-studied histone methyltransferase is SUV39H1 (suppressor of variegation B9 homolog 1) and its mammalian homologues, which methylate histone H3 on lysine 9.
- SWI/SNF complex
-
An ATP-dependent chromatin-remodelling protein complex that was initially identified in yeast. Related complexes exist in mammals and are involved in the remodelling of chromatin in various genes.
- Polycomb repressive complexes
-
(PRCs). Groups of proteins that maintain gene expression states throughout development by regulating chromatin structure. In mammals there are two core Polycomb complexes: PRC1 and PRC2. PRC1 catalyses the monoubiquitylation of histone H2A. Both complexes contribute to chromatin compaction. PRC2 harbours the histone methyltransferases EZH1 (enhancer of zeste homolog 1) and EZH2, which catalyse the methylation of histone H3 at lysine 27. These two complexes are involved in differentiation, in the maintenance of cell identity and proliferation, and in stem cell plasticity.
- H3K4 trimethylation complex
-
A complex of proteins that are involved in the addition of three methyl groups to histone H3 on lysine 4.
- Cell polarity
-
The asymmetric distribution of proteins, lipids and/or their complexes within the cell. It is believed that polarity determines the mode of cell division and thus the fate of the two daughter cells.
- Centrosomes
-
Cytoplasmic organelles that organize the microtubules. Preceding mitosis, the centrosome doubles and is then involved in the generation of the mitotic spindle for subsequent chromosome segregation during mitosis. In many cell types the centrosome is directly located in the centre of the cell and is therefore assigned a polar distribution, in which case cells frequently undergo asymmetric divisions.
- Mode of stem cell division
-
An symmetric or asymmetric event, with respect to the potential of the daughter cells. An asymmetric division balances stem cell self-renewal and differentiation through the production of one stem cell and one differentiating cell, whereas a symmetric division might result in either two stem cells or two differentiated cells.
- Osteoblasts
-
Cells of mesenchymal origin that are responsible for the formation of bone. Osteoblasts are thought to be a crucial component of the haematopoietic stem cell niche.
- Mesenchymal stem cells
-
(MSCs). Multipotent progenitor cells originally identified in the bone marrow stroma. MSCs can be expanded in vitro and, under appropriate conditions, give rise to several cell types, including bone and fat precursors.
- Gap junction channels
-
Channels composed of integral membrane proteins called connexins. Gap junction channels connect the cytoplasms of adjacent cells, allowing for the diffusion of ions and small metabolites between cells.
- CCL11
-
CC-chemokine ligand 11; a member of the CC-chemokine family and selectively recruits eosinophils. Increased CCL11 levels in the blood are found in aged mice and humans.
Rights and permissions
About this article
Cite this article
Geiger, H., de Haan, G. & Florian, M. The ageing haematopoietic stem cell compartment. Nat Rev Immunol 13, 376–389 (2013). https://doi.org/10.1038/nri3433
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3433
This article is cited by
-
Neutrophil, lymphocyte count, and neutrophil to lymphocyte ratio predict multimorbidity and mortality—results from the Baltimore Longitudinal Study on Aging follow-up study
GeroScience (2024)
-
A state-of-the-art review on the MicroRNAs roles in hematopoietic stem cell aging and longevity
Cell Communication and Signaling (2023)
-
EVA1A regulates hematopoietic stem cell regeneration via ER-mitochondria mediated apoptosis
Cell Death & Disease (2023)
-
Induction of mitochondrial recycling reverts age-associated decline of the hematopoietic and immune systems
Nature Aging (2023)
-
Breast cancer remotely imposes a myeloid bias on haematopoietic stem cells by reprogramming the bone marrow niche
Nature Cell Biology (2023)