Key Points
-
For more than 25 years, type 1 diabetes (T1D) has been recognized as an autoimmune disease. The mechanisms involve innate and adaptive immune responses that appear to be triggered by environmental factors on the appropriate genetic background.
-
Genetic studies have suggested that immunological pathways may contribute to disease development. These pathways involve genes that are involved in the acquisition and maintenance of immune tolerance.
-
Immune mechanisms that may contribute to the pathogenesis of T1D include failures in central tolerance, dysregulation of tolerance in the periphery as well as active immune responses against pathogens.
-
Preclinical studies in rodent models have shed light on pathological immunological mechanisms and have begun to address environmental factors that may modify the disease. These models have also been useful for identifying the autoantigens that are recognized by T cells in patients with T1D.
-
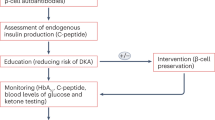
Some clinical studies have successfully modified the chronic loss of C peptide that is characteristic of T1D. Not all therapeutic approaches have worked, and the duration of the efficacy is limited. These findings suggest that future trials should explore other options, including combinations of therapeutic agents and earlier intervention at the pre-diabetic period.
-
Not all of the findings in preclinical studies have been successfully translated into the clinic. Some investigators have criticized the validity of the preclinical models, but a careful analysis of the preclinical studies suggests that their ability to predict outcomes is strong. Details concerning a broad number of variables, including dosage and timing of agents, may not have been fully considered in the design of clinical trials.
Abstract
Type 1 diabetes (T1D) remains an important health problem, particularly in western countries, where the incidence has been increasing in younger children. In 1986, Eisenbarth described T1D as a chronic autoimmune disease. Work over the past three-and-a-half decades has identified many of the genetic, immunological and environmental factors that are involved in the disease and have led to hypotheses concerning its pathogenesis. Clinical trials have been conducted to test these hypotheses but have had mixed results. Here, we discuss the findings that have led to our current concepts of the disease mechanisms involved in T1D and the clinical studies promoted by these studies. The findings from preclinical and clinical studies support the original proposed model for how T1D develops but have also suggested that this disease is more complex than was originally thought and will require broader treatment approaches.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Eisenbarth, G. S. Type I diabetes mellitus. A chronic autoimmune disease. N. Engl. J. Med. 314, 1360–1368 (1986).
Bottazzo, G. F., Florin-Christensen, A. & Doniach, D. Islet-cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. Lancet 2, 1279–1283 (1974).
Sibley, R. K., Sutherland, D. E., Goetz, F. & Michael, A. F. Recurrent diabetes mellitus in the pancreas iso- and allograft. A light and electron microscopic and immunohistochemical analysis of four cases. Lab. Invest. 53, 132–144 (1985).
Eisenbarth, G. S. et al. Anti-thymocyte globulin and prednisone immunotherapy of recent onset type 1 diabetes mellitus. Diabetes Res. 2, 271–276 (1985).
Stiller, C. R. et al. Effects of cyclosporine immunosuppression in insulin-dependent diabetes mellitus of recent onset. Science 223, 1362–1367 (1984).
Faber, O. K. & Binder, C. B-cell function and blood glucose control in insulin dependent diabetics within the first month of insulin treatment. Diabetologia 13, 263–268 (1977).
Palmer, J. P., et al. C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve β-cell function: report of an ADA workshop, 21–22 October 2001. Diabetes 53, 250–264 (2004).
Steffes, M. W. Sibley, S., Jackson, M. & Thomas, W. β-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care 26, 832–836 (2003).
Ziegler, A. G. et al. Accelerated progression from islet autoimmunity to diabetes is causing the escalating incidence of type 1 diabetes in young children. J. Autoimmun. 37, 3–7 (2011).
Dunne, D. W. & Cooke, A. A worm's eye view of the immune system: consequences for evolution of human autoimmune disease. Nature Rev. Immunol. 5, 420–426 (2005).
Atkinson, M. A. et al. Cellular immunity to a determinant common to glutamate decarboxylase and coxsackie virus in insulin-dependent diabetes. J. Clin. Invest. 94, 2125–2129 (1994).
Strachan, D. P. Hay fever, hygiene, and household size. BMJ 299, 1259–1260 (1989).
Bach, J. F. & Chatenoud, L. The hygiene hypothesis: an explanation for the increased frequency of insulin-dependent diabetes. Cold Spring Harb. Perspect. Med. 2, a007799 (2012).
Cooke, A., Zaccone, P., Raine, T., Phillips, J. M. & Dunne, D. W. Infection and autoimmunity: are we winning the war, only to lose the peace? Trends Parasitol. 20, 316–321 (2004).
David, T., Thomas, C., Zaccone, P., Dunne, D. W. & Cooke, A. The impact of infection on the incidence of autoimmune disease. Curr. Top. Med. Chem. 4, 521–529 (2004).
Dotta, F. et al. Coxsackie B4 virus infection of β cells and natural killer cell insulitis in recent-onset type 1 diabetic patients. Proc. Natl Acad. Sci. USA 104, 5115–5120 (2007).
Yoon, J. W., Austin, M., Onodera, T. & Notkins, A. L. Isolation of a virus from the pancreas of a child with diabetic ketoacidosis. N. Engl. J. Med. 300, 1173–1179 (1979).
Akatsuka, H. et al. A case of fulminant type 1 diabetes with coxsackie B4 virus infection diagnosed by elevated serum levels of neutralizing antibody. Diabetes Res. Clin. Pract. 84, e50–e52 (2009).
Berg, A. K., Korsgren, O. & Frisk, G. Induction of the chemokine interferon-γ-inducible protein-10 in human pancreatic islets during enterovirus infection. Diabetologia 49, 2697–2703 (2006).
Christen, U., McGavern, D. B., Luster, A. D., von Herrath, M. G. & Oldstone, M. B. Among CXCR3 chemokines, IFN-γ-inducible protein of 10 kDa (CXC chemokine ligand (CXCL) 10) but not monokine induced by IFN-γ (CXCL9) imprints a pattern for the subsequent development of autoimmune disease. J. Immunol. 171, 6838–6845 (2003).
Schulte, B. M. et al. Phagocytosis of enterovirus-infected pancreatic β-cells triggers innate immune responses in human dendritic cells. Diabetes 59, 1182–1191 (2010).
Chung, H. et al. Gut immune maturation depends on colonization with a host-specific microbiota. Cell 149, 1578–1593 (2012).
Mathis, D. & Benoist, C. The influence of the microbiota on type-1 diabetes: on the threshold of a leap forward in our understanding. Immunol. Rev. 245, 239–249 (2012).
Round, J. L. & Mazmanian, S. K. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl Acad. Sci. USA 107, 12204–12209 (2010).
Giongo, A. et al. Toward defining the autoimmune microbiome for type 1 diabetes. ISME J. 5, 82–91 (2011).
Perez-Cano, F. J., Dong, H. & Yaqoob, P. In vitro immunomodulatory activity of Lactobacillus fermentum CECT5716 and Lactobacillus salivarius CECT5713: two probiotic strains isolated from human breast milk. Immunobiology 215, 996–1004 (2010).
Lee, Y. K., Menezes, J. S., Umesaki, Y. & Mazmanian, S. K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4615–4622 (2011).
Ivanov, I. I. et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349 (2008).
Geuking, M. B. et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity 34, 794–806 (2011).
Kriegel, M. A. et al. Naturally transmitted segmented filamentous bacteria segregate with diabetes protection in nonobese diabetic mice. Proc. Natl Acad. Sci. USA 108, 11548–11553 (2011).
Wen, L. et al. Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature 455, 1109–1113 (2008). The interaction between innate immune signalling, the gut microflora and development of T1D were studied. In an SPF facility, diabetes did not develop in NOD mice because of the role of innate signalling in modifying the gut microflora.
Knip, M., Virtanen, S. M. & Akerblom, H. K. Infant feeding and the risk of type 1 diabetes. Am J. Clin Nutr 91, 1506S–1513S (2010).
Ziegler, A. G., Schmid, S., Huber, D., Hummel, M. & Bonifacio, E. Early infant feeding and risk of developing type 1 diabetes-associated autoantibodies. JAMA 290, 1721–1728 (2003).
Norris, J. M. et al. Timing of initial cereal exposure in infancy and risk of islet autoimmunity. JAMA 290, 1713–1720 (2003).
Akerblom, H. K. et al. Dietary manipulation of β cell autoimmunity in infants at increased risk of type 1 diabetes: a pilot study. Diabetologia 48, 829–837 (2005).
Knip, M. et al. Dietary intervention in infancy and later signs of β-cell autoimmunity. N. Engl. J. Med. 363, 1900–1908 (2010).
Wong, F. S. et al. Investigation of the role of B-cells in type 1 diabetes in the NOD mouse. Diabetes 53, 2581–2587 (2004).
Fiorina, P. et al. Targeting CD22 reprograms B-cells and reverses autoimmune diabetes. Diabetes 57, 3013–3024 (2008).
Hu, C. Y. et al. Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. J. Clin. Invest. 117, 3857–3867 (2007).
Wenzlau, J. M. et al. The cation efflux transporter ZNT8 (SLC30A8) is a major autoantigen in human type 1 diabetes. Proc. Natl Acad. Sci. USA 104, 17040–17045 (2007).
Krishnamurthy, B. et al. Responses against islet antigens in NOD mice are prevented by tolerance to proinsulin but not IGRP. J. Clin. Invest. 116, 3258–3265 (2006).
Nakayama, M. et al. Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 435, 220–223 (2005). In this study, in which Ins1 and Ins2 were knocked out of a NOD mouse and replaced by a mutated proinsulin transgene, the investigators establish the primacy of an insulin epitope in the development of type 1 diabetes in NOD mice.
Bot, A. et al. Plasmid vaccination with insulin B chain prevents autoimmune diabetes in nonobese diabetic mice. J. Immunol. 167, 2950–2955 (2001).
von Herrath, M., Sanda, S. & Herold, K. Type 1 diabetes as a relapsing-remitting disease? Nature Rev. Immunol. 7, 988–994 (2007).
Yu, L. et al. Early expression of antiinsulin autoantibodies of humans and the NOD mouse: evidence for early determination of subsequent diabetes. Proc. Natl Acad. Sci. USA 97, 1701–1706 (2000).
Skowera, A. et al. CTLs are targeted to kill β cells in patients with type 1 diabetes through recognition of a glucose-regulated preproinsulin epitope. J. Clin. Invest. 118, 3390–3402 (2008). This study shows that autoreactive CTLs are present in the circulation of patients with type 1 diabetes and that they can kill human β-cells.
Jaeckel, E., Lipes, M. A. & von Boehmer, H. Recessive tolerance to preproinsulin 2 reduces but does not abolish type 1 diabetes. Nature Immunol. 5, 1028–1035 (2004).
Thebault-Baumont, K. et al. Acceleration of type 1 diabetes mellitus in proinsulin 2-deficient NOD mice. J. Clin. Invest. 111, 851–857 (2003).
Mathis, D. & Benoist, C. Back to central tolerance. Immunity 20, 509–516 (2004).
Anderson, M. S. et al. The cellular mechanism of AIRE control of T cell tolerance. Immunity 23, 227–239 (2005).
Moriyama, H. et al. Evidence for a primary islet autoantigen (preproinsulin 1) for insulitis and diabetes in the nonobese diabetic mouse. Proc. Natl Acad. Sci. USA 100, 10376–10381 (2003).
Chentoufi, A. A. & Polychronakos, C. Insulin expression levels in the thymus modulate insulin-specific autoreactive T-cell tolerance: the mechanism by which the IDDM2 locus may predispose to diabetes. Diabetes 51, 1383–1390 (2002).
Mohan, J. F. et al. Unique autoreactive T cells recognize insulin peptides generated within the islets of Langerhans in autoimmune diabetes. Nature Immunol. 11, 350–354 (2010).
Mohan, J. F., Petzold, S. J. & Unanue, E. R. Register shifting of an insulin peptide–MHC complex allows diabetogenic T cells to escape thymic deletion. J. Exp. Med. 208, 2375–2383 (2011). This study describes a mechanism whereby autoreactive T cells that escape thymic selection can be activated by antigen-presenting cells that bear antigenic insulin peptides.
Stadinski, B., Kappler, J. & Eisenbarth, G. S. Molecular targeting of islet autoantigens. Immunity 32, 446–456 (2010). This report suggests that binding of an insulin peptide in an alternative register allowed T cells to escape negative selection in the thymus but responded to peptide presented in the islets of Langerhans.
Crawford, F. et al. Specificity and detection of insulin-reactive CD4+ T cells in type 1 diabetes in the nonobese diabetic (NOD) mouse. Proc. Natl Acad. Sci. USA 108, 16729–16734 (2011).
Stadinski, B. D. et al. Diabetogenic T cells recognize insulin bound to IAg7 in an unexpected, weakly binding register. Proc. Natl Acad. Sci. USA 107, 10978–10983 (2010).
Menard, L. et al. The PTPN22 allele encoding an R620W variant interferes with the removal of developing autoreactive B cells in humans. J. Clin. Invest. 121, 3635–3644 (2011).
Concannon, P., Rich, S. S. & Nepom, G. T. Genetics of type 1A diabetes. N. Engl. J. Med. 360, 1646–1654 (2009).
Christianson, S. W., Shultz, L. D. & Leiter, E. H. Adoptive transfer of diabetes into immunodeficient NOD-scid/scid mice. Relative contributions of CD4+ and CD8+ T-cells from diabetic versus prediabetic NOD.NON-Thy-1a donors. Diabetes 42, 44–55 (1993).
Miller, B. J., Appel, M. C., O'Neil, J. J. & Wicker, L. S. Both the Lyt-2+ and L3T4+ T cell subsets are required for the transfer of diabetes in nonobese diabetic mice. J. Immunol. 140, 52–58 (1988).
Andre, I. et al. Checkpoints in the progression of autoimmune disease: lessons from diabetes models. Proc. Natl Acad. Sci. USA 93, 2260–2263 (1996).
Katz, J. D., Wang, B., Haskins, K., Benoist, C. & Mathis, D. Following a diabetogenic T cell from genesis through pathogenesis. Cell 74, 1089–1100 (1993).
Verdaguer, J. et al. Acceleration of spontaneous diabetes in TCR-β-transgenic nonobese diabetic mice by β-cell cytotoxic CD8+ T cells expressing identical endogenous TCR-α chains. J. Immunol. 157, 4726–4735 (1996).
Lennon, G. P. et al. T cell islet accumulation in type 1 diabetes is a tightly regulated, cell-autonomous event. Immunity 31, 643–653 (2009).
van den Elzen, P. et al. Limited clonality in autoimmunity: drivers and regulators. Autoimmun. Rev. 3, 524–529 (2004).
Baker, F. J., Lee, M., Chien, Y. H. & Davis, M. M. Restricted islet-cell reactive T cell repertoire of early pancreatic islet infiltrates in NOD mice. Proc. Natl Acad. Sci. USA 99, 9374–9379 (2002).
Codina-Busqueta, E. et al. TCR bias of in vivo expanded T cells in pancreatic islets and spleen at the onset in human type 1 diabetes. J. Immunol. 186, 3787–3797 (2011).
Nakayama, M. et al. Germline TRAV5D-4 T-cell receptor sequence targets a primary insulin peptide of NOD mice. Diabetes 61, 857–865 (2012).
Arnold, P. Y., Burton, A. R. & Vignali, D. A. Diabetes incidence is unaltered in glutamate decarboxylase 65-specific TCR retrogenic nonobese diabetic mice: generation by retroviral-mediated stem cell gene transfer. J. Immunol. 173, 3103–3111 (2004).
Holst, J. et al. Generation of T-cell receptor retrogenic mice. Nature Protoc. 1, 406–417 (2006).
Holst, J., Vignali, K. M., Burton, A. R. & Vignali, D. A. Rapid analysis of T-cell selection in vivo using T cell-receptor retrogenic mice. Nature Methods 3, 191–197 (2006).
Bettini, M. L., Bettini, M. & Vignali, D. A. T-cell receptor retrogenic mice: a rapid, flexible alternative to T-cell receptor transgenic mice. Immunology 136, 265–272 (2012).
Campbell-Thompson, M. et al. Network for Pancreatic Organ Donors with Diabetes (nPOD): developing a tissue biobank for type 1 diabetes. Diabetes Metab. Res. Rev. 28, 608–617 (2012).
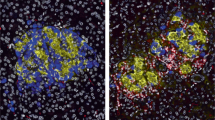
Coppieters, K. T. et al. Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. J. Exp. Med. 209, 51–60 (2012). Human insulitis is characterized in this analysis of autopsy material from patients with T1D. Diabetes-antigen-specific T cells could be identified within the insulitis.
Sherr, J., Sosenko, J., Skyler, J. S. & Herold, K. C. Prevention of type 1 diabetes: the time has come. Nature Clin. Pract. Endocrinol. Metab. 4, 334–343 (2008).
Orban, T. et al. Pancreatic islet autoantibodies as predictors of type 1 diabetes in the Diabetes Prevention Trial-Type 1. Diabetes Care 32, 2269–2274 (2009). This clinical study shows the effects of CTLA4–Ig on attenuation of the decline in C peptide in the first 2 years of T1D.
Krischer, J. P. et al. Screening strategies for the identification of multiple antibody-positive relatives of individuals with type 1 diabetes. J. Clin. Endocrinol. Metab. 88, 103–108 (2003).
Waldmann, H. & Cobbold, S. How do monoclonal antibodies induce tolerance? A role for infectious tolerance? Annu. Rev. Immunol. 16, 619–644 (1998).
Fourlanos, S. et al. Evidence that nasal insulin induces immune tolerance to insulin in adults with autoimmune diabetes. Diabetes 60, 1237–1245 (2011).
Chaillous, L. et al. Oral insulin administration and residual β-cell function in recent-onset type 1 diabetes: a multicentre randomised controlled trial. Lancet 356, 545–549 (2000).
Diabetes Prevention Trial—Type 1 Diabetes Study Group. Effects of insulin in relatives of patients with type 1 diabetes mellitus. N. Engl. J. Med. 346, 1685–1691 (2002).
Skyler, J. S. et al. Effects of oral insulin in relatives of patients with type 1 diabetes: The Diabetes Prevention Trial—Type 1. Diabetes Care 28, 1068–1076 (2005).
Ludvigsson, J. et al. GAD treatment and insulin secretion in recent-onset type 1 diabetes. N. Engl. J. Med. 359, 1909–1920 (2008).
Ludvigsson, J. et al. GAD65 antigen therapy in recently diagnosed type 1 diabetes mellitus. N. Engl. J. Med. 366, 433–442 (2012).
Wherrett, D. K. et al. Antigen-based therapy with glutamic acid decarboxylase (GAD) vaccine in patients with recent-onset type 1 diabetes: a randomised double-blind trial. Lancet 378, 319–327 (2011).
Gottlieb, P. A. et al. Failure to preserve β-cell function with mycophenolate mofetil and daclizumab combined therapy in patients with new- onset type 1 diabetes. Diabetes Care 33, 826–832 (2010).
Pescovitz, M. D. et al. Rituximab, B-lymphocyte depletion, and preservation of β-cell function. N. Engl. J. Med. 361, 2143–2152 (2009).
Herold, K. C. et al. Increased T cell proliferative responses to islet antigens identify clinical responders to anti-CD20 monoclonal antibody (rituximab) therapy in type 1 diabetes. J. Immunol. 187, 1998–2005 (2011).
Yu, L. et al. Rituximab selectively suppresses specific islet antibodies. Diabetes 60, 2560–2565 (2011).
Lenschow, D. J. et al. Differential effects of anti-B7-1 and anti-B7-2 monoclonal antibody treatment on the development of diabetes in the nonobese diabetic mouse. J. Exp. Med. 181, 1145–1155 (1995).
Lenschow, D. J. et al. CD28/B7 regulation of Th1 and Th2 subsets in the development of autoimmune diabetes. Immunity 5, 285–293 (1996).
Orban, T. et al. Co-stimulation modulation with abatacept in patients with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled trial. Lancet 378, 412–419 (2011).
Itoh, N. et al. Mononuclear cell infiltration and its relation to the expression of major histocompatibility complex antigens and adhesion molecules in pancreas biopsy specimens from newly diagnosed insulin-dependent diabetes mellitus patients. J. Clin. Invest. 92, 2313–2322 (1993).
Ogasawara, K. et al. NKG2D blockade prevents autoimmune diabetes in NOD mice. Immunity 20, 757–767 (2004).
Beilke, J. N. et al. NK cells are not required for spontaneous autoimmune diabetes in NOD mice. PLoS ONE 7, e36011 (2012).
Amrani, A. et al. Interleukin-1 effect on glycemia in the non-obese diabetic mouse at the pre-diabetic stage. J. Endocrinol. 148, 139–148 (1996).
Rabinovitch, A., Baquerizo, H. & Sumoski, W. Cytotoxic effects of cytokines on islet β-cells: evidence for involvement of eicosanoids. Endocrinology 126, 67–71 (1990).
Thomas, H. E. et al. IL-1 receptor deficiency slows progression to diabetes in the NOD mouse. Diabetes 53, 113–121 (2004).
Cailleau, C., Diu-Hercend, A., Ruuth, E., Westwood, R. & Carnaud, C. Treatment with neutralizing antibodies specific for IL-1β prevents cyclophosphamide-induced diabetes in nonobese diabetic mice. Diabetes 46, 937–940 (1997).
Foulis, A. K., McGill, M. & Farquharson, M. A. Insulitis in type 1 (insulin-dependent) diabetes mellitus in man—macrophages, lymphocytes, and interferon-γ containing cells. J. Pathol. 165, 97–103 (1991).
Koulmanda, M. et al. Curative and β cell regenerative effects of α1-antitrypsin treatment in autoimmune diabetic NOD mice. Proc. Natl Acad. Sci. USA 105, 16242–16247 (2008).
Schilling, E. et al. Inhibition of nicotinamide phosphoribosyltransferase modifies LPS-induced inflammatory responses of human monocytes. Innate Immun. 18, 518–530 (2012).
Gale, E. A., Bingley, P. J., Emmett, C. L. & Collier, T. European Nicotinamide Diabetes Intervention Trial (ENDIT): a randomised controlled trial of intervention before the onset of type 1 diabetes. Lancet 363, 925–931 (2004).
Larsen, C. M. et al. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med. 356, 1517–1526 (2007).
Sanda, S. et al. Short-term IL-1β blockade reduces monocyte CD11b integrin expression in an IL-8 dependent fashion in patients with type 1 diabetes. Clin. Immunol. 136, 170–173 (2010).
Zanin-Zhorov, A. et al. Heat shock protein 60 enhances CD4+ CD25+ regulatory T cell function via innate TLR2 signaling. J. Clin. Invest. 116, 2022–2032 (2006).
Raz, I. et al. β-cell function in new-onset type 1 diabetes and immunomodulation with a heat-shock protein peptide (DiaPep277): a randomised, double-blind, phase II trial. Lancet 358, 1749–1753 (2001).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 203, 1701–1711 (2006).
Brusko, T. M., Putnam, A. L. & Bluestone, J. A. Human regulatory T cells: role in autoimmune disease and therapeutic opportunities. Immunol. Rev. 223, 371–390 (2008).
Zhou, X. et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nature Immunol. 10, 1000–1007 (2009).
Zheng, Y. et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature 463, 808–812 (2010).
Salomon, B. & Bluestone, J. A. Complexities of CD28/B7: CTLA-4 costimulatory pathways in autoimmunity and transplantation. Annu. Rev. Immunol. 19, 225–252 (2001).
Salomon, B. et al. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T cells that control autoimmune diabetes. Immunity 12, 431–440 (2000).
Ablamunits, V., Henegariu, O., Preston-Hurlburt, P. & Herold, K. C. NKG2A is a marker for acquisition of regulatory function by human CD8+ T cells activated with anti-CD3 antibody. Eur. J. Immunol. 41, 1832–1842 (2011).
Green, E. A., Gorelik, L., McGregor, C. M., Tran, E. H. & Flavell, R. A. CD4+CD25+ T regulatory cells control anti-islet CD8+ T cells through TGF-β–TGF-β receptor interactions in type 1 diabetes. Proc. Natl Acad. Sci. USA 100, 10878–10883 (2003).
You, S. et al. Adaptive TGF-β-dependent regulatory T cells control autoimmune diabetes and are a privileged target of anti-CD3 antibody treatment. Proc. Natl Acad. Sci. USA 104, 6335–6340 (2007).
Levings, M. K., Sangregorio, R. & Roncarolo, M. G. Human CD25+CD4+ T regulatory cells suppress naive and memory T cell proliferation and can be expanded in vitro without loss of function. J. Exp. Med. 193, 1295–1302 (2001).
Levings, M. K. et al. Human CD25+CD4+ T suppressor cell clones produce transforming growth factor β, but not interleukin 10, and are distinct from type 1 T regulatory cells. J. Exp. Med. 196, 1335–1346 (2002).
Roncarolo, M. G., Bacchetta, R., Bordignon, C., Narula, S. & Levings, M. K. Type 1 T regulatory cells. Immunol. Rev. 182, 68–79 (2001).
Heiber, J. F. & Geiger, T. L. Context and location dependence of adaptive Foxp3+regulatory T cell formation during immunopathological conditions. Cell. Immunol. 279, 60–65 (2012).
Bettini, M. et al. Cutting edge: accelerated autoimmune diabetes in the absence of LAG-3. J. Immunol. 187, 3493–3498 (2011).
Tivol, E. A. et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity 3, 541–547 (1995).
Tang, Q. et al. Distinct roles of CTLA-4 and TGF-β in CD4+CD25+ regulatory T cell function. Eur. J. Immunol. 34, 2996–3005 (2004).
Alyanakian, M. A. et al. Transforming growth factor-β and natural killer T-cells are involved in the protective effect of a bacterial extract on type 1 diabetes. Diabetes 55, 179–185 (2006).
Mellor, A. L. & Munn, D. H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nature Rev. Immunol. 4, 762–774 (2004).
Collison, L. W. et al. IL-35-mediated induction of a potent regulatory T cell population. Nature Immunol. 11, 1093–1101 (2010).
Maier, L. M. et al. IL2RA genetic heterogeneity in multiple sclerosis and type 1 diabetes susceptibility and soluble interleukin-2 receptor production. PLoS Genet. 5, e1000322 (2009).
Ueda, H. et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature 423, 506–511 (2003).
Long, S. A. et al. Defects in IL-2R signaling contribute to diminished maintenance of FOXP3 expression in CD4+CD25+ regulatory T-cells of type 1 diabetic subjects. Diabetes 59, 407–415 (2010).
Grinberg-Bleyer, Y. et al. IL-2 reverses established type 1 diabetes in NOD mice by a local effect on pancreatic regulatory T cells. J. Exp. Med. 207, 1871–1878 (2010).
McClymont, S. A. et al. Plasticity of human regulatory T cells in healthy subjects and patients with type 1 diabetes. J. Immunol. 186, 3918–3926 (2011).
Rabinovitch, A., Suarez-Pinzon, W. L., Shapiro, A. M., Rajotte, R. V. & Power, R. Combination therapy with sirolimus and interleukin-2 prevents spontaneous and recurrent autoimmune diabetes in NOD mice. Diabetes 51, 638–645 (2002).
Simon, G. et al. Murine antithymocyte globulin therapy alters disease progression in NOD mice by a time-dependent induction of immunoregulation. Diabetes 57, 405–414 (2008).
Belghith, M. et al. TGF-β-dependent mechanisms mediate restoration of self-tolerance induced by antibodies to CD3 in overt autoimmune diabetes. Nature Med. 9, 1202–1208 (2003).
Esplugues, E. et al. Control of TH17 cells occurs in the small intestine. Nature 475, 514–518 (2011).
Waldron-Lynch, F. et al. Teplizumab induces human gut-tropic regulatory cells in humanized mice and patients. Sci. Transl. Med. 4, 118ra112 (2012).
Herold, K. C. et al. A single course of anti-CD3 monoclonal antibody hOKT3γ1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes. Diabetes 54, 1763–1769 (2005).
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med. 346, 1692–1698 (2002).
Keymeulen, B. et al. Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N. Engl. J. Med. 352, 2598–2608 (2005). References 139 and 140 shows the efficacy of humanized CD3-specific antibodies in patients with new-onset T1D. They are based on initial studies by Chatenoud et al . demonstrating efficacy in hyperglycaemic NOD mice.
Keymeulen, B. et al. Four-year metabolic outcome of a randomised controlled CD3-antibody trial in recent-onset type 1 diabetic patients depends on their age and baseline residual β cell mass. Diabetologia 53, 614–623 (2010).
Bisikirska, B., Colgan, J., Luban, J., Bluestone, J. A. & Herold, K. C. TCR stimulation with modified anti-CD3 mAb expands CD8 T cell population and induces CD8CD25 TRegs . J. Clin. Invest. 115, 2904–2913 (2005).
Herold, K. C. et al. Activation of human T cells by FcR nonbinding anti-CD3 mAb, hOKT3γ1(Ala-Ala). J. Clin. Invest. 111, 409–418 (2003).
Sherry, N. et al. Teplizumab for treatment of type 1 diabetes (Protege study): 1-year results from a randomised, placebo-controlled trial. Lancet 378, 487–497 (2011).
Herold, K. C. et al. Teplizumab treatment may improve C-peptide responses in participants with type 1 diabetes after the new-onset period: a randomised controlled trial. Diabetologia 56, 391–400 (2012).
Long, S. A. et al. Rapamycin/IL-2 combination therapy in patients with type 1 diabetes augments Tregs yet transiently impairs β-cell function. Diabetes 61, 2340–2348 (2012).
Qin, S. et al. “Infectious” transplantation tolerance. Science 259, 974–977 (1993).
Ablamunits, V. et al. Synergistic reversal of type 1 diabetes in NOD mice with anti-CD3 and interleukin-1 blockade: evidence of improved immune regulation. Diabetes 61, 145–154 (2012).
Kwon, G., Corbett, J. A., Rodi, C. P., Sullivan, P. & McDaniel, M. L. Interleukin-1 β-induced nitric oxide synthase expression by rat pancreatic β-cells: evidence for the involvement of nuclear factor κ B in the signaling mechanism. Endocrinology 136, 4790–4795 (1995).
Gysemans, C. et al. Interferon regulatory factor-1 is a key transcription factor in murine β cells under immune attack. Diabetologia 52, 2374–2384 (2009).
Akirav, E., Kushner, J. A. & Herold, K. C. β-cell mass and type 1 diabetes: going, going, gone? Diabetes 57, 2883–2888 (2008).
Sherry, N. A. et al. Effects of autoimmunity and immune therapy on β-cell turnover in type 1 diabetes. Diabetes 55, 3238–3245 (2006).
Akirav, E. M. et al. Detection of β cell death in diabetes using differentially methylated circulating DNA. Proc. Natl Acad. Sci. USA 108, 19018–19023 (2011).
Talchai, C., Xuan, S., Lin, H. V., Sussel, L. & Accili, D. Pancreatic β cell dedifferentiation as a mechanism of diabetic β cell failure. Cell 150, 1223–1234 (2012).
Eizirik, D.L. & Darville, M.I. β-cell apoptosis and defense mechanisms: lessons from type 1 diabetes. Diabetes 50 (Suppl. 1), S64–69 (2001).
Melton, D. A. Using stem cells to study and possibly treat type 1 diabetes. Phil. Trans. R. Soc. B 366, 2307–2311 (2011).
Patterson, C. C., Dahlquist, G. G., Gyurus, E., Green, A. & Soltesz, G. Incidence trends for childhood type 1 diabetes in Europe during 1989–2003 and predicted new cases 2005–20: a multicentre prospective registration study. Lancet 373, 2027–2033 (2009).
Riley, W. J. et al. A prospective study of the development of diabetes in relatives of patients with insulin-dependent diabetes. N. Engl. J. Med. 323, 1167–1172 (1990).
Sabbah, E. et al. Diabetes-associated autoantibodies in relation to clinical characteristics and natural course in children with newly diagnosed type 1 diabetes. J. Clin. Endocrinol. Metab. 84, 1534–1539 (1999).
Greenbaum, C. J. et al. Fall in C-peptide during first 2 years from diagnosis: evidence of at least two distinct phases from composite TrialNet data. Diabetes 61, 2066–2073 (2012).
Goodnow, C. C., Sprent, J., Fazekas de St Groth, B. & Vinuesa, C. G. Cellular and genetic mechanisms of self tolerance and autoimmunity. Nature 435, 590–597 (2005).
Halverson, R., Torres, R. M. & Pelanda, R. Receptor editing is the main mechanism of B cell tolerance toward membrane antigens. Nature Immunol. 5, 645–650 (2004).
Hertz, M. & Nemazee, D. BCR ligation induces receptor editing in IgM+IgD− bone marrow B cells in vitro. Immunity 6, 429–436 (1997).
Bluestone, J. A. & Abbas, A. K. Natural versus adaptive regulatory T cells. Nature Rev. Immunol. 3, 253–257 (2003).
Harjutsalo, V., Podar, T. & Tuomilehto, J. Cumulative incidence of type 1 diabetes in 10,168 siblings of Finnish young-onset type 1 diabetic patients. Diabetes 54, 563–569 (2005).
Steck, A. K. & Rewers, M. J. Genetics of type 1 diabetes. Clin. Chem. 57, 176–185 (2011).
Redondo, M. J., Jeffrey, J., Fain, P. R., Eisenbarth, G. S. & Orban, T. Concordance for islet autoimmunity among monozygotic twins. N. Engl. J. Med. 359, 2849–2850 (2008).
Barrett, J. C. et al. Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nature Genet. 41, 703–707 (2009).
Fernando, M. M. et al. Defining the role of the MHC in autoimmunity: a review and pooled analysis. PLoS Genet. 4, e1000024 (2008).
Zhang, Z. J., Davidson, L., Eisenbarth, G. & Weiner, H. L. Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc. Natl Acad. Sci. USA 88, 10252–10256 (1991).
Tian, J. et al. Modulating autoimmune responses to GAD inhibits disease progression and prolongs islet graft survival in diabetes-prone mice. Nature Med. 2, 1348–1353 (1996).
Moran, A. et al. Interleukin-1 antagonism in type 1 diabetes of recent onset: two multicenter, randomized double-masked, placebo-controlled trials. Lancet (in the press).
Shoda, L. K. et al. A comprehensive review of interventions in the NOD mouse and implications for translation. Immunity 23, 115–126 (2005).
Stoffers, D. A., Desai, B. M., DeLeon, D. D. & Simmons, R. A. Neonatal exendin-4 prevents the development of diabetes in the intrauterine growth retarded rat. Diabetes 52, 734–740 (2003).
Mestas, J. & Hughes, C. C. Of mice and not men: differences between mouse and human immunology. J. Immunol. 172, 2731–2738 (2004).
Steele, C. et al. Insulin secretion in type 1 diabetes. Diabetes 53, 426–433 (2004).
Sosenko, J. M. et al. Patterns of metabolic progression to type 1 diabetes in the diabetes prevention trial-type 1. Diabetes Care 29, 643–649 (2006).
Voltarelli, J. C. et al. Autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA 297, 1568–1576 (2007).
Couri, C. E. et al. C-peptide levels and insulin independence following autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA 301, 1573–1539 (2009).
Mastrandrea, L. et al. Etanercept treatment in children with new-onset type 1 diabetes: pilot randomized, placebo-controlled, double-blind study. Diabetes Care 32, 1244–1249 (2009).
Acknowledgements
K.C.H. is supported by the US National Institutes of Health (U19 AI082713, DK045735, DK057846 and UL1RR024139) and the JDRF (2011-248, 2007–1059). D.A.A.V. is supported by the National Institutes of Health (DK089125, AI039480, AI091977, AI052199), the St. Jude National Cancer Institute Comprehensive Cancer Center (CA21765) and the American Lebanese Syrian Associated Charities (ALSAC). J.A.B is supported by the National Institutes of Health (AI50834, AI046643, JDRF 4-2011-248, U19 AI056388). A.C. is supported by the UK Medical Research Council, the Wellcome Trust and the European Union.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Jeffrey A. Bluestone has a patent for teplizumab. Kevan C. Herold has received a grant from MacroGenic, Inc., and Islet Sciences, Inc. He has a patent pending for analysis of β-cell death and is on the Advisory Board for Islet Sciences, Inc. Dario A. A. Vignali and Anne Cooke declare no competing financial interests.
Related links
Glossary
- Anti-thymocyte globulin
-
Polyclonal antibodies against human T cells that are produced by immunizing rabbits or horses.
- Cyclosporin A
-
An immunosuppressive drug that inhibits calcineurin, a Ca2+-dependent serine/threonine phosphatase that is necessary for the nuclear translocation of the transcription factor nuclear factor of activated T cells (NFAT).
- C peptide
-
The connecting peptide that joins the A chain and B chain of insulin in the proinsulin molecule.
- Hygiene hypothesis
-
The theory that the lack of early childhood exposure to infectious agents, symbiotic microorganisms (for example, changes in gut microflora) and parasites increases susceptibility to allergic and autoimmune diseases by modulating immune system development.
- Insulitis
-
Inflammation of the islets of Langerhans in the pancreas that comprises a complex cellular infiltrate that invades and destroys the islets of Langerhans. The cellular composition includes CD4+ T cells, CD8+ T cells, regulatory T cells, B cells, dendritic cells, natural killer cells and macrophages.
- Gut microbiome
-
This is the collective community of bacteria in the small and large intestines.
- Forkhead box P3
-
(FOXP3). A forkhead/winged helix family transcription factor that is a crucial master regulator of regulatory T cell development and function.
- Regulatory T cells
-
(TReg cells). A rare subpopulation of CD4+ T cells that are endowed with potent suppressive capacity. They typically express the transcription factor FOXP3+. Both naturally occurring TReg cells (which develop in the thymus) and adaptive TReg cells (which acquire their regulatory activity in the periphery) have been described.
- Invariant natural killer T cells
-
(iNKT cells). Cells that share properties of T cells and natural killer (NK) cells and recognize the non-polymorphic CD1d molecule, which is an antigen-presenting molecule that binds self lipids and foreign lipids and glycolipids. They recognize α-galactosylceramide presented on CD1d molecules and have restricted T cell receptor (TCR) usage.
- T helper 17 cells
-
(TH17 cells). A subset of CD4+ T cells that is characterized by its expression of the transcription factors RORγ, RORα and signal transducer and activator of transcription 3 (STAT3). They are involved in inflammatory responses and normally have an important protective role at epithelial and mucosal surfaces. Their development involves a combination of TGFβ, interleukin 21 (IL-21), IL-23 and IL-1β, and they secrete IL-17, IL-22, IL-22 and in some circumstances granulocyte-macrophage colony-stimulating factor (GM-CSF) and/or interferon-γ (IFNγ).
- Specific-pathogen-free conditions
-
(SPF conditions). Mice raised under SPF conditions are guaranteed to be free of a defined list of mouse pathogens.
- Germ-free mice
-
Also known as gnotobiotic mice, these are mice that do not harbour any bacteria, viruses or parasites.
- 'Type B' T cells
-
A term that has been used to describe CD4+ T cells that recognize and respond to unstable peptide–MHC complexes, which arise when peptides are loaded onto MHC class II molecules in the absence of H2-DM-mediated editing.
- HEp-2
-
A human epithelial cell line that is commonly used as a target for immunofluorescent detection of a wide range of nuclear and cytoplasmic staining antibodies. Distinct staining patterns are associated with particular antibody specificities. For example, a homogeneous nuclear staining pattern is indicative of antibodies that react with double-stranded DNA or chromatin, whereas a speckled nuclear-staining pattern is indicative of antibodies that react with small nuclear ribonucleoproteins.
- CD28–B7 co-stimulation
-
A crucial receptor–ligand interaction that is required for maximal T cell activation and survival.
- Rapamycin
-
An immunosuppressive drug that, in contrast to calcineurin inhibitors (such as cyclosporin A and FK506), does not prevent T cell activation but blocks interleukin-2-mediated clonal expansion by blocking mammalian target of rapamycin (mTOR). It does not interfere with the function and expansion of naturally occurring regulatory T cells.
Rights and permissions
About this article
Cite this article
Herold, K., Vignali, D., Cooke, A. et al. Type 1 diabetes: translating mechanistic observations into effective clinical outcomes. Nat Rev Immunol 13, 243–256 (2013). https://doi.org/10.1038/nri3422
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3422
This article is cited by
-
I-Ag7 β56/57 polymorphisms regulate non-cognate negative selection to CD4+ T cell orchestrators of type 1 diabetes
Nature Immunology (2023)
-
Urine-derived stem cell therapy for diabetes mellitus and its complications: progress and challenges
Endocrine (2023)
-
An autoimmune stem-like CD8 T cell population drives type 1 diabetes
Nature (2022)
-
Anti-CD20 therapy ameliorates β cell function and rebalances Th17/Treg cells in NOD mice
Endocrine (2022)
-
The IGFBP3/TMEM219 pathway regulates beta cell homeostasis
Nature Communications (2022)