Key Points
-
Lymphotoxin is best known for its roles in promoting lymphoid tissue development, but this cytokine also has other important immunological functions.
-
Lymphotoxin produced by dendritic cells has steady-state immunological functions, including roles in maintaining the cellularity of secondary lymphoid organs and in IgA production.
-
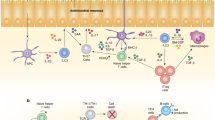
The coordination of the multicellular interactions that contribute to T helper cell and germinal centre responses depends on the lymphotoxin pathway.
-
Antiviral immunity requires the lymphotoxin pathway, both for the regulation of type I interferon production and for innate aspects of the B cell response.
-
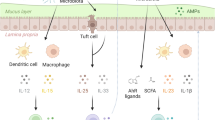
Innate lymphoid cells produce lymphotoxin and this promotes interleukin-23 (IL-23)- and IL-22-dependent immune responses at mucosal surfaces. Lymphotoxin production at mucosal surfaces also regulates the microbiota and can contribute to metabolic disease.
-
The lymphotoxin pathway has a crucial role in active immune responses that occur in adult hosts, and a better understanding of these roles may help to guide the development of a new class of therapeutic agents to treat inflammatory diseases.
Abstract
Lymphotoxin (LT) is a member of the tumour necrosis factor (TNF) superfamily that was originally thought to be functionally redundant to TNF, but these proteins were later found to have independent roles in driving lymphoid organogenesis. More recently, LT-mediated signalling has been shown to actively contribute to effector immune responses. LT regulates dendritic cell and CD4+ T cell homeostasis in the steady state and determines the functions of these cells during pathogenic challenges. The LT receptor pathway is essential for controlling pathogens and even contributes to the regulation of the intestinal microbiota, with recent data suggesting that LT-induced changes in the microbiota promote metabolic disease. In this Review, we discuss these newly defined roles for LT, with a particular focus on how the LT receptor pathway regulates innate and adaptive immune responses to microorganisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eberl, G. A new vision of immunity: homeostasis of the superorganism. Mucosal Immunol. 3, 450–460 (2010).
Ware, C. F., VanArsdale, T. L., Crowe, P. D. & Browning, J. L. The ligands and receptors of the lymphotoxin system. Curr. Top. Microbiol. Immunol. 198, 175–218 (1995).
Josefowicz, S. Z. et al. Extrathymically generated regulatory T cells control mucosal TH2 inflammation. Nature 482, 395–399 (2012).
Lathrop, S. K. et al. Peripheral education of the immune system by colonic commensal microbiota. Nature 478, 250–254 (2011).
Geuking, M. B. et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity 34, 794–806 (2011).
De Togni, P. et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science 264, 703–707 (1994).
Fu, Y.-X. & Chaplin, D. D. Development and maturation of secondary lymphoid tissues. Annu. Rev. Immunol. 17, 399–433 (1999).
Zhu, M., Brown, N. K. & Fu, Y.-X. Direct and indirect roles of the LTβR pathway in central tolerance induction. Trends Immunol. 31, 325–331 (2010).
Chin, R. K. et al. Lymphotoxin pathway directs thymic Aire expression. Nature Immunol. 4, 1121–1127 (2003).
Iizuka, K. et al. Requirement for membrane lymphotoxin in natural killer cell development. Proc. Natl Acad. Sci. USA 96, 6336–6340 (1999).
Fütterer, A., Mink, K., Luz, A., Kosco-Vilbois, M. H. & Pfeffer, K. The lymphotoxin β receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity 9, 59–70 (1998).
Crowe, P. D. et al. A lymphotoxin-β-specific receptor. Science 264, 707–710 (1994).
Samstein, R. M., Josefowicz, S. Z., Arvey, A., Treuting, P. M. & Rudensky, A. Y. Extrathymic generation of regulatory T cells in placental mammals mitigates maternal-fetal conflict. Cell 150, 29–38 (2012).
van de Pavert, S. A. & Mebius, R. E. New insights into the development of lymphoid tissues. Nature Rev. Immunol. 10, 664–674 (2010).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl Acad. Sci. USA 105, 16731–16736 (2008).
Zindl, C. L. et al. The lymphotoxin LT1β2 controls postnatal and adult spleen marginal sinus vascular structure and function. Immunity 30, 408–420 (2009).
Mebius, R. E. Organogenesis of lymphoid tissues. Nature Rev. Immunol. 3, 292–303 (2003).
Moussion, C. & Girard, J.-P. Dendritic cells control lymphocyte entry to lymph nodes through high endothelial venules. Nature 479, 542–546 (2011). This is the first demonstration of LT being produced by a non-lymphoid cell type.
Schneider, K., Potter, K. G. & Ware, C. F. Lymphotoxin and LIGHT signaling pathways and target genes. Immunol. Rev. 202, 49–66 (2004).
Tamada, K. et al. LIGHT, a TNF-like molecule, costimulates T cell proliferation and is required for dendritic cell-mediated allogeneic T cell response. J. Immunol. 164, 4105–4110 (2000).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Steinman, R. M. Decisions about dendritic cells: past, present, and future. Annu. Rev. Immunol. 30, 1–22 (2012).
MartIn-Fontecha, A. et al. Regulation of dendritic cell migration to the draining lymph node: impact on T lymphocyte traffic and priming. J. Exp. Med. 198, 615–621 (2003).
Girard, J.-P., Moussion, C. & Förster, R. HEVs, lymphatics and homeostatic immune cell trafficking in lymph nodes. Nature Rev. Immunol. 12, 762–773 (2012).
Uchimura, K. et al. A major class of L-selectin ligands is eliminated in mice deficient in two sulfotransferases expressed in high endothelial venules. Nature Immunol. 6, 1105–1113 (2005).
Kawashima, H. et al. N-acetylglucosamine-6-O-sulfotransferases 1 and 2 cooperatively control lymphocyte homing through L-selectin ligand biosynthesis in high endothelial venules. Nature Immunol. 6, 1096–1104 (2005).
Browning, J. L. et al. Lymphotoxin-β receptor signaling is required for the homeostatic control of HEV differentiation and function. Immunity 23, 539–550 (2005).
León, B. et al. Regulation of TH2 development by CXCR5+ dendritic cells and lymphotoxin-expressing B cells. Nature Immunol. 13, 681–690 (2012). This article provides a very clear demonstration of the ability of the LT pathway to coordinate key multicellular interactions that influence T H 2 cell development.
Kabashima, K. et al. Intrinsic lymphotoxin-β receptor requirement for homeostasis of lymphoid tissue dendritic cells. Immunity 22, 439–450 (2005).
Lewis, K. L. et al. Notch2 receptor signaling controls functional differentiation of dendritic cells in the spleen and intestine. Immunity 35, 780–791 (2011).
Ngo, V. N. et al. Lymphotoxin α/β and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J. Exp. Med. 189, 403–412 (1999).
Chow, A., Toomre, D., Garrett, W. & Mellman, I. Dendritic cell maturation triggers retrograde MHC class II transport from lysosomes to the plasma membrane. Nature 418, 988–994 (2002).
Cella, M., Engering, A., Pinet, V., Pieters, J. & Lanzavecchia, A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 388, 782–787 (1997).
Kaisho, T. & Akira, S. Dendritic-cell function in Toll-like receptor- and MyD88-knockout mice. Trends Immunol. 22, 78–83 (2001).
Suzuki, K. & Fagarasan, S. How host-bacterial interactions lead to IgA synthesis in the gut. Trends Immunol. 29, 523–531 (2008).
Fritz, J. H. et al. Acquisition of a multifunctional IgA+ plasma cell phenotype in the gut. Nature 481, 199–203 (2011). This paper further defines the mechanism by which LT controls IgA production through the LT-dependent regulation of iNOS+ DCs.
Kang, H.-S. et al. Signaling via LTβR on the lamina propria stromal cells of the gut is required for IgA production. Nature Immunol. 3, 576–582 (2002).
Macpherson, A. J. S. et al. IgA production without μ or δ chain expression in developing B cells. Nature Immunol. 2, 625–631 (2001).
Tsuji, M. et al. Requirement for lymphoid tissue-inducer cells in isolated follicle formation and T cell-independent immunoglobulin A generation in the gut. Immunity 29, 261–271 (2008).
Fagarasan, S., Kawamoto, S., Kanagawa, O. & Suzuki, K. Adaptive immune regulation in the gut: T cell-dependent and T cell-independent IgA synthesis. Annu. Rev. Immunol. 28, 243–273 (2010).
Lee, M.-R., Seo, G.-Y., Kim, Y.-M. & Kim, P.-H. iNOS potentiates mouse Ig isotype switching through AID expression. Biochem. Biophys. Res. Commun. 410, 602–607 (2011).
Tezuka, H. et al. Regulation of IgA production by naturally occurring TNF/iNOS-producing dendritic cells. Nature 448, 929–933 (2007).
Cerutti, A. The regulation of IgA class switching. Nature Rev. Immunol. 8, 421–434 (2008).
Fagarasan, S. et al. Critical roles of activation-induced cytidine deaminase in the homeostasis of gut flora. Science 298, 1424–1427 (2002).
Kang, H.-S. et al. Lymphotoxin is required for maintaining physiological levels of serum IgE that minimizes Th1-mediated airway inflammation. J. Exp. Med. 198, 1643–1652 (2003).
Spahn, T. W. et al. The lymphotoxin-β receptor is critical for control of murine Citrobacter rodentium-induced colitis. Gastroenterology 127, 1463–1473 (2004).
Ehrchen, J. M. et al. The absence of cutaneous lymph nodes results in a Th2 response and increased susceptibility to Leishmania major infection in mice. Infect. Immun. 76, 4241–4250 (2008).
Dohi, T. et al. Elimination of colonic patches with lymphotoxin β receptor-Ig prevents Th2 cell-type colitis. J. Immunol. 167, 2781–2790 (2001).
Gramaglia, I., Mauri, D. N., Miner, K. T., Ware, C. F. & Croft, M. Lymphotoxin αβ is expressed on recently activated naive and Th1-like CD4 cells but is down-regulated by IL-4 during Th2 differentiation. J. Immunol. 162, 1333–1338 (1999).
Morel, P. A. & Oriss, T. B. Crossregulation between Th1 and Th2 cells. Crit. Rev. Immunol. 18, 275–303 (1998).
Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nature Rev. Immunol. 3, 133–146 (2003).
Zhu, J. & Paul, W. E. Heterogeneity and plasticity of T helper cells. Cell Res. 20, 4–12 (2009).
Else, K. J. & Finkelman, F. D. Intestinal nematode parasites, cytokines and effector mechanisms. Int. J. Parasitol. 28, 1145–1158 (1998).
Matsumoto, M. et al. Affinity maturation without germinal centres in lymphotoxin-α-deficient mice. Nature 382, 462–466 (1996).
Thai, T.-H. et al. Regulation of the germinal center response by microRNA-155. Science 316, 604–608 (2007). A demonstration that a miRNA-dependent programme regulates LT expression to contribute to germinal centre formation.
Vu, F., Dianzani, U., Ware, C. F., Mak, T. & Gommerman, J. L. ICOS, CD40, and lymphotoxin β receptors signal sequentially and interdependently to initiate a germinal center reaction. J. Immunol. 180, 2284–2293 (2008).
Junt, T. et al. Subcapsular sinus macrophages in lymph nodes clear lymph-borne viruses and present them to antiviral B cells. Nature 450, 110–114 (2007).
Phan, T. G., Green, J. A., Gray, E. E., Xu, Y. & Cyster, J. G. Immune complex relay by subcapsular sinus macrophages and noncognate B cells drives antibody affinity maturation. Nature Immunol. 10, 786–793 (2009).
Phan, T. G., Grigorova, I., Okada, T. & Cyster, J. G. Subcapsular encounter and complement-dependent transport of immune complexes by lymph node B cells. Nature Immunol. 8, 992–1000 (2007).
Moseman, E. A. et al. B cell maintenance of subcapsular sinus macrophages protects against a fatal viral infection independent of adaptive immunity. Immunity 36, 415–426 (2012). A mechanistic demonstration that LT from B cells maintains the sentinel function of SCS macrophages and prevents the dissemination of lethal pathogens.
Platanias, L. C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nature Rev. Immunol. 5, 375–386 (2005).
Schneider, K. et al. Lymphotoxin-mediated crosstalk between B cells and splenic stroma promotes the initial type I interferon response to cytomegalovirus. Cell Host Microbe 3, 67–76 (2008).
Banks, T. A. et al. A lymphotoxin-IFN-β axis essential for lymphocyte survival revealed during cytomegalovirus infection. J. Immunol. 174, 7217–7225 (2005).
Kumar, V. et al. Global lymphoid tissue remodeling during a viral infection is orchestrated by a B cell-lymphotoxin-dependent pathway. Blood 115, 4725–4733 (2010).
Louten, J., van Rooijen, N. & Biron, C. A. Type 1 IFN deficiency in the absence of normal splenic architecture during lymphocytic choriomeningitis virus infection. J. Immunol. 177, 3266–3272 (2006).
Heath, W. R. & Carbone, F. R. Cross-presentation in viral immunity and self-tolerance. Nature Rev. Immunol. 1, 126–135 (2001).
Banks, T. A., Rickert, S. & Ware, C. F. Restoring immune defenses via lymphotoxin signaling: lessons from cytomegalovirus. Immunol. Res. 34, 243–254 (2006).
Summers deLuca, L. et al. LTβR signaling in dendritic cells induces a type I IFN response that is required for optimal clonal expansion of CD8+ T cells. Proc. Natl Acad. Sci. USA 108, 2046–2051 (2011).
Roozendaal, R. et al. Conduits mediate transport of low-molecular-weight antigen to lymph node follicles. Immunity 30, 264–276 (2009).
Iannacone, M. et al. Subcapsular sinus macrophages prevent CNS invasion on peripheral infection with a neurotropic virus. Nature 465, 1079–1083 (2010).
Moyron-Quiroz, J. E. et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nature Med. 10, 927–934 (2004).
Ehlers, S. et al. The lymphotoxin β receptor is critically involved in controlling infections with the intracellular pathogens Mycobacterium tuberculosis and Listeria monocytogenes. J. Immunol. 170, 5210–5218 (2003).
Barthel, M. et al. Pretreatment of mice with streptomycin provides a Salmonella enterica serovar typhimurium colitis model that allows analysis of both pathogen and host. Infect. Immun. 71, 2839–2858 (2003).
Hapfelmeier, S. et al. The Salmonella pathogenicity island (SPI)-2 and SPI-1 type III secretion systems allow Salmonella serovar typhimurium to trigger colitis via MyD88-dependent and MyD88-independent mechanisms. J. Immunol. 174, 1675–1685 (2005).
Wang, Y. et al. Lymphotoxin β receptor signaling in intestinal epithelial cells orchestrates innate immune responses against mucosal bacterial infection. Immunity 32, 403–413 (2010).
Ota, N. et al. IL-22 bridges the lymphotoxin pathway with the maintenance of colonic lymphoid structures during infection with Citrobacter rodentium. Nature Immunol. 12, 941–948 (2011).
Tumanov, A. V. et al. Lymphotoxin controls the IL-22 protection pathway in gut innate lymphoid cells during mucosal pathogen challenge. Cell Host Microbe 10, 44–53 (2011). References 76 and 77 demonstrate the importance of the LT pathway in the regulation of the innate IL-23–IL-22 axis.
Schlüter, D. et al. Both lymphotoxin-α and TNF are crucial for control of Toxoplasma gondii in the central nervous system. J. Immunol. 170, 6172–6182 (2003).
Eberl, G. et al. An essential function for the nuclear receptor RORγt in the generation of fetal lymphoid tissue inducer cells. Nature Immunol. 5, 64–73 (2003).
Eberl, G. & Lochner, M. The development of intestinal lymphoid tissues at the interface of self and microbiota. Mucosal Immunol. 2, 478–485 (2009).
Eberl, G. & Littman, D. R. Thymic origin of intestinal αβT cells revealed by fate mapping of RORγt+ cells. Sci. Signal. 305, 248–251 (2004).
Langrish, C. L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
Ivanov, I. I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Takatori, H. et al. Lymphoid tissue inducer-like cells are an innate source of IL-17 and IL-22. J. Exp. Med. 206, 35–41 (2009).
Sawa, S. et al. Lineage relationship analysis of RORγt+ innate lymphoid cells. Science 330, 665–669 (2010).
Cherrier, M., Sawa, S. & Eberl, G. Notch, Id2, and RORγt sequentially orchestrate the fetal development of lymphoid tissue inducer cells. J. Exp. Med. 209, 729–740 (2012).
Vonarbourg, C. et al. Regulated expression of nuclear receptor RORγt confers distinct functional fates to NK cell receptor-expressing RORγt+ innate lymphocytes. Immunity 33, 736–751 (2010).
Satoh-Takayama, N. et al. Lymphotoxin-β receptor-independent development of intestinal IL-22-producing NKp46+ innate lymphoid cells. Eur. J. Immunol. 41, 780–786 (2011).
Tumanov, A. V. et al. Dissecting the role of lymphotoxin in lymphoid organs by conditional targeting. Immunol. Rev. 195, 106–116 (2003).
Mahajan, A. et al. Obesity-dependent association of TNF-LTA locus with type 2 diabetes in North Indians. J. Mol. Med. 88, 515–522 (2010).
Lo, J. C. et al. Lymphotoxin β receptor-dependent control of lipid homeostasis. Science 316, 285–288 (2007).
Lotzer, K. et al. Mouse aorta smooth muscle cells differentiate into lymphoid tissue organizer-like cells on combined tumor necrosis factor receptor-1/Lymphotoxin β-receptor NF-κB signaling. Arterioscler. Thromb. Vasc. Biol. 30, 395–402 (2010).
Tumanov, A. V., Christiansen, P. A. & Fu, Y.-X. The role of lymphotoxin receptor signaling in diseases. Curr. Mol. Med. 7, 567–578 (2007).
Norman, R., Bogardus, C. & Ravussin, E. Linkage between obesity and a marker near the tumor necrosis factor-α locus in Pima Indians. J. Clin. Invest. 96, 158–162 (1995).
Hamid, Y. H. et al. The common T60N polymorphism of the lymphotoxin-α gene is associated with type 2 diabetes and other phenotypes of the metabolic syndrome. Diabetologia 48, 445–451 (2005).
Upadhyay, V. et al. Lymphotoxin regulates commensal responses to enable diet-induced obesity. Nature Immunol. 13, 947–953 (2012). This paper demonstrates the ability of the LT pathway to regulate the commensal microbiota.
Pamir, N., McMillen, T. S., Edgel, K. A., Kim, F. & LeBoeuf, R. C. Deficiency of Lymphotoxin-α does not exacerbate high fat diet induced obesity but does enhance inflammation in mice. Am. J. Physiol. Endocrinol. Metab. 302, e961–e971 (2012).
Spiegelman, B. M. & Flier, J. S. Obesity and the regulation of energy balance. Cell 104, 531–543 (2001).
Turnbaugh, P. J. et al. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci. Transl. Med. 1, 6ra14 (2009).
Bouskra, D. et al. Lymphoid tissue genesis induced by commensals through NOD1 regulates intestinal homeostasis. Nature 456, 507–510 (2008).
Fava, R. A., Browning, J. L., Gatumu, M., Skarstein, K. & Bolstad, A.-I. LTBR-pathway in Sjogren's syndrome: CXCL13 levels and B-cell-enriched ectopic lymphoid aggregates in NOD mouse lacrimal glands are dependent on LTBR. Adv. Exp. Med. Biol. 691, 383–390 (2011).
Wen, L. et al. Innate immunity and intestinal microbiota in the development of Type 1 diabetes. Nature 455, 1109–1113 (2008).
Berer, K. et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 479, 1–5 (2011).
Devkota, S. et al. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature 487, 104–108 (2012).
Dejardin, E. et al. The lymphotoxin-β receptor induces different patterns of gene expression via two NF-κB pathways. Immunity 17, 525–535 (2002).
Chang, Y.-H., Chao, Y., Hsieh, S.-L. & Lin, W.-W. Mechanism of LIGHT/interferon-γ-induced cell death in HT-29 cells. J. Cell. Biochem. 93, 1188–1202 (2004).
Browning, J. L. et al. Signaling through the lymphotoxin β receptor induces the death of some adenocarcinoma tumor lines. J. Exp. Med. 183, 867–878 (1996).
Mackay, F., Majeau, G. R., Lawton, P., Hochman, P. S. & Browning, J. L. Lymphotoxin but not tumor necrosis factor functions to maintain splenic architecture and humoral responsiveness in adult mice. Eur. J. Immunol. 27, 2033–2042 (1997).
Lorenz, R. G., Chaplin, D. D., McDonald, K. G., McDonough, J. S. & Newberry, R. D. Isolated lymphoid follicle formation is inducible and dependent upon lymphotoxin-sufficient B lymphocytes, lymphotoxin β receptor, and TNF receptor I function. J. Immunol. 170, 5475–5482 (2003).
Lochner, M. et al. Microbiota-induced tertiary lymphoid tissues aggravate inflammatory disease in the absence of RORγt and LTi cells. J. Exp. Med. 208, 125–134 (2011). This article demonstrates the LT-dependent formation of aberrant lymphoid structures near an epithelial surface.
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1131 (2006).
Acknowledgements
The authors would like to thank A. Tumanov and M. Zhu for their contribution to the lymphotoxin project. This work is partially supported by DK080736 and CA141975 to Y.-X.F.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Innate lymphoid cells
-
(ILCs). Populations of lymphoid cells that lack lineage markers typically associated with other immune cell subsets, but that express transcription factors and cytokines that are usually associated with differentiated CD4+ T helper cell subsets.
- Cd11c–DTR system
-
A system in which mice express the diphtheria toxin receptor (DTR) under the control of the Cd11c gene promoter. This allows the conditional depletion of dendritic cells (and some other CD11c+ cells) in response to diphtheria toxin administration.
- μMT−/− mice
-
Mice that contain a targeted mutation of Ighm; these mice lack mature, conventional B cells.
- LTR-Ig and TNFR-Ig
-
LTR-Ig and TNFR-Ig are decoy receptors for the lymphotoxin receptor (LTR) and the tumour necrosis factor receptor (TNFR), respectively. Their administration prevents ligand–receptor interactions and circumvents signalling through these pathways. They block lymphoid organogenesis if delivered in utero.
- Subcapsular sinus (SCS) macrophages
-
CD11b+CD169+ macrophages that populate the subcapsular sinus of lymph nodes.
- Lymphoid tissue-inducer cells
-
A CD4+CD90+RORγt+CD3− subset of innate lymphoid cells that express lymphotoxin-α1β2 to enable the formation of lymph nodes and Peyer's patches during development.
- 16S rRNA
-
A component of the 30S subunit of the bacterial ribosome; a unique secondary structure characterized by stem and loop regions makes this target useful in the unbiased identification of currently uncultured commensal bacteria.
- Segmented filamentous bacteria
-
(SFB). A bacterial species that lives in the terminal ileum in direct contact with intestinal epithelial cells and that induces the expression of interleukin-17A (IL-17A), IL-22 and IgA in the host. Candidatus savagella has been proposed as a name for this uncultured species.
Rights and permissions
About this article
Cite this article
Upadhyay, V., Fu, YX. Lymphotoxin signalling in immune homeostasis and the control of microorganisms. Nat Rev Immunol 13, 270–279 (2013). https://doi.org/10.1038/nri3406
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3406
This article is cited by
-
Evaluation of lymphotoxin-alpha in pterygium and diagnostic value in active and inactive pterygium states
Scientific Reports (2024)
-
COVID-19 immune signatures in Uganda persist in HIV co-infection and diverge by pandemic phase
Nature Communications (2024)
-
Prognostic significance of the genetic variant of lymphotoxin alpha (p.Thr60Asn) in egyptian patients with advanced hepatocellular carcinoma
Molecular Biology Reports (2023)
-
Neonatal LTβR signaling is required for the accumulation of eosinophils in the inflamed adult mesenteric lymph node
Mucosal Immunology (2022)
-
Lymphotoxin: from the physiology to the regeneration of the thymic function
Cell Death & Differentiation (2021)