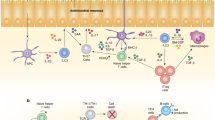
Key Points
-
TNF superfamily members have diverse functions in mammalian biology and are often expressed by cells of the immune system. TNF superfamily members are important therapeutic targets for the treatment of autoimmune disease and for encouraging tumour clearance.
-
Dendritic cells (DCs) are the most potent antigen-presenting cells in the immune system. DC-intrinsic signals can dictate whether T cells respond or show tolerance to an antigen.
-
The expression of TNF superfamily receptors and ligands by T cells and DCs suggests an important role for this molecular superfamily in T cell–DC crosstalk. TNF superfamily receptors on T cells can provide co-stimulation for the T cell response when they interact with their corresponding TNF superfamily ligands that have been upregulated on DCs.
-
TNF superfamily receptors that are expressed directly on DCs can provide homeostatic signals to the DCs in the steady state and can function to 'license' the DCs during an immune response by virtue of T cell–DC crosstalk. Licensed DCs secrete pro-inflammatory cytokines and express CD70.
-
Modulation of TNF superfamily receptor-derived signals in the gut can have a dramatic impact on mucosal inflammation and responses to mucosal pathogens. In the gut, T cell–DC crosstalk may be replaced by communication between DCs and innate lymphoid cells to provoke protective immunity and to maintain tolerance to the resident microbial communities in this location.
Abstract
Members of the tumour necrosis factor (TNF) superfamily have been implicated in a wide range of biological functions, and their expression by cells of the immune system makes them appealing targets for immunomodulation. One common theme for TNF superfamily members is their coordinated expression at the interface between antigen-specific T cells and antigen-presenting dendritic cells and, by virtue of this expression pattern, TNF superfamily members can shape T cell immune responses. Understanding how to manipulate such functions of the TNF superfamily may allow us to tip the balance between immunity and tolerance in the context of human disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Aggarwal, B. B. Signalling pathways of the TNF superfamily: a double-edged sword. Nature Rev. Immunol. 3, 745–756 (2003).
Mackay, F. & Schneider, P. Cracking the BAFF code. Nature Rev. Immunol. 9, 491–502 (2009).
Drayton, D. L., Liao, S., Mounzer, R. H. & Ruddle, N. H. Lymphoid organ development: from ontogeny to neogenesis. Nature Immunol. 7, 344–353 (2006).
van de Pavert, S. A. & Mebius, R. E. New insights into the development of lymphoid tissues. Nature Rev. Immunol. 10, 664–674 (2010).
Fritz, J. H. & Gommerman, J. L. Cytokine/stromal cell networks and lymphoid tissue environments. J. Interferon Cytokine Res. 31, 277–289 (2010).
Phan, T. G., Green, J. A., Gray, E. E., Xu, Y. & Cyster, J. G. Immune complex relay by subcapsular sinus macrophages and noncognate B cells drives antibody affinity maturation. Nature Immunol. 10, 786–793 (2009).
Kumar, V. et al. Global lymphoid tissue remodeling during a viral infection is orchestrated by a B cell-lymphotoxin-dependent pathway. Blood 115, 4725–4733 (2010).
Browning, J. L. et al. Lymphotoxin-β receptor signaling is required for the homeostatic control of HEV differentiation and function. Immunity 23, 539–550 (2005).
Moussion, C. & Girard, J. P. Dendritic cells control lymphocyte entry to lymph nodes through high endothelial venules. Nature 479, 542–546 (2011). This study shows that the expression of LTαβ on DCs is important for HEV maturation.
De Togni, P. et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science 264, 703–707 (1994).
Bekiaris, V. et al. CD30 is required for CCL21 expression and CD4 T cell recruitment in the absence of lymphotoxin signals. J. Immunol. 182, 4771–4775 (2009).
Zhu, M., Yang, Y., Wang, Y., Wang, Z. & Fu, Y. X. LIGHT regulates inflamed draining lymph node hypertrophy. J. Immunol. 186, 7156–7163 (2011).
Chyou, S. et al. Fibroblast-type reticular stromal cells regulate the lymph node vasculature. J. Immunol. 181, 3887–3896 (2008).
Chyou, S. et al. Coordinated regulation of lymph node vascular-stromal growth first by CD11c+ cells and then by T and B cells. J. Immunol. 187, 5558–5567 (2011).
Mounzer, R. H. et al. Lymphotoxin-α contributes to lymphangiogenesis. Blood 116, 2173–2182 (2010).
Martín-Fontecha, A. et al. Regulation of dendritic cell migration to the draining lymph node: impact on T lymphocyte traffic and priming. J. Exp. Med. 198, 615–621 (2003).
Scandella, E. et al. Restoration of lymphoid organ integrity through the interaction of lymphoid tissue-inducer cells with stroma of the T cell zone. Nature Immunol. 9, 667–675 (2008).
Caux, C. et al. CD34+ hematopoietic progenitors from human cord blood differentiate along two independent dendritic cell pathways in response to GM-CSF+TNFα. J. Exp. Med. 184, 695–706 (1996).
Flores-Romo, L. et al. CD40 ligation on human cord blood CD34+ hematopoietic progenitors induces their proliferation and differentiation into functional dendritic cells. J. Exp. Med. 185, 341–349 (1997).
Ashcroft, A. J. et al. Colonic dendritic cells, intestinal inflammation, and T cell-mediated bone destruction are modulated by recombinant osteoprotegerin. Immunity 19, 849–861 (2003).
Josien, R. et al. TRANCE, a tumor necrosis factor family member, enhances the longevity and adjuvant properties of dendritic cells in vivo. J. Exp. Med. 191, 495–502 (2000).
Matthews, K. E. et al. Increasing the survival of dendritic cells in vivo does not replace the requirement for CD4+ T cell help during primary CD8+ T cell responses. J. Immunol. 179, 5738–5747 (2007).
Kong, Y. Y. et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 397, 315–323 (1999).
Cella, M. et al. Ligation of CD40 on dendritic cells triggers production of high levels of interleukin-12 and enhances T cell stimulatory capacity: T–T help via APC activation. J. Exp. Med. 184, 747–752 (1996).
Choi, B. K. et al. 4-1BB functions as a survival factor in dendritic cells. J. Immunol. 182, 4107–4115 (2009).
Lee, S.-W. et al. Identification of regulatory functions for 4-1BB and 4-1BBL in myelopoiesis and the development of dendritic cells. Nature Immunol. 9, 917–926 (2008).
Wu, Q. et al. The requirement of membrane lymphotoxin for the presence of dendritic cells in lymphoid tissues. J. Exp. Med. 190, 629–638 (1999).
Ngo, V. N. et al. Lymphotoxin α/β and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J. Exp. Med. 189, 403–412 (1999).
Kabashima, K. et al. Intrinsic lymphotoxin-β receptor requirement for homeostasis of lymphoid tissue dendritic cells. Immunity 22, 439–450 (2005).
Wang, Y.-G., Kim, K. D., Wang, J., Yu, P. & Fu, Y.-X. Stimulating lymphotoxin β receptor on the dendritic cells is critical for their homeostasis and expansion. J. Immunol. 175, 6997–7002 (2005).
Lewis, K. L. et al. Notch2 receptor signaling controls functional differentiation of dendritic cells in the spleen and intestine. Immunity 35, 780–791 (2011).
De Trez, C. et al. The inhibitory HVEM–BTLA pathway counter regulates lymphotoxin receptor signaling to achieve homeostasis of dendritic cells. J. Immunol. 180, 238–248 (2008). References 29 and 32 demonstrate that LTβR signalling is a positive regulator of DC homeostasis, whereas HVEM–BTLA signalling is a negative regulator of DC homeostasis. This was an important hint that direct LTβR signalling in DCs has a crucial role in immune responses beyond the well-described function of LTβR signalling in inducing chemokine production by the lymphoid stroma.
Summers-DeLuca, L. E. et al. Expression of lymphotoxin-αβ on antigen-specific T cells is required for DC function. J. Exp. Med. 204, 1071–1081 (2007). This paper demonstrated that in response to a protein antigen the expression of LTαβ on antigen-specific T H cells was required for DC function and was not redundant with the expression of CD40L.
Quezada, S. A., Jarvinen, L. Z., Lind, E. F. & Noelle, R. J. CD40/CD154 interactions at the interface of tolerance and immunity. Annu. Rev. Immunol. 22, 307–328 (2004).
Lefrancois, L., Altman, J. D., Williams, K. & Olson, S. Soluble antigen and CD40 triggering are sufficient to induce primary and memory cytotoxic T cells. J. Immunol. 164, 725–732 (2000).
Summers deLuca, L. et al. LTβR signaling in dendritic cells induces a type I IFN response that is required for optimal clonal expansion of CD8+ T cells. Proc. Natl Acad. Sci. USA 108, 2046–2051 (2011).
Hernandez, M. G., Shen, L. & Rock, K. L. CD40 on APCs is needed for optimal programming, maintenance, and recall of CD8+ T cell memory even in the absence of CD4+ T cell help. J. Immunol. 180, 4382–4390 (2008).
Taraban, V. Y., Rowley, T. F. & Al-Shamkhani, A. Cutting edge: a critical role for CD70 in CD8 T cell priming by CD40-licensed APCs. J. Immunol. 173, 6542–6546 (2004).
Xiao, Z., Casey, K. A., Jameson, S. C., Curtsinger, J. M. & Mescher, M. F. Programming for CD8 T cell memory development requires IL-12 or type I IFN. J. Immunol. 182, 2786–2794 (2009).
Bachmann, M. F. et al. Cutting edge: distinct roles for T help and CD40/CD40 ligand in regulating differentiation of proliferation-competent memory CD8+ T cells. J. Immunol. 173, 2217–2221 (2004).
Guo, Z. et al. Cutting edge: membrane lymphotoxin regulates CD8+ T cell-mediated intestinal allograft rejection. J. Immunol. 167, 4796–4800 (2001).
Puglielli, M. T. et al. Reversal of virus-induced systemic shock and respiratory failure by blockade of the lymphotoxin pathway. Nature Med. 5, 1370–1374 (1999).
Berger, D. P. et al. Lymphotoxin-β-deficient mice show defective antiviral immunity. Virology 260, 136–147 (1999).
Suresh, M. et al. Role of lymphotoxin α in T-cell responses during an acute viral infection. J. Virol. 76, 3943–3951 (2002).
Benedict, C. A. et al. Lymphotoxins and cytomegalovirus cooperatively induce interferon-β, establishing host–virus detente. Immunity 15, 617–626 (2001).
Schneider, K. et al. Lymphotoxin-mediated crosstalk between B cells and splenic stroma promotes the initial type I interferon response to cytomegalovirus. Cell Host Microbe 3, 67–76 (2008). References 36 and 46 demonstrate that LTβR signalling is required for type I IFN production. Reference 46 showed this to be the case in stromal cells, whereas reference 36 demonstrated LTβR-dependent type I IFN production in DCs. The LTβR-dependent production of type I IFNs in DCs was shown to influence the clonal expansion of CD8+ T cells.
Kolumam, G. A., Thomas, S., Thompson, L. J., Sprent, J. & Murali-Krishna, K. Type I interferons act directly on CD8 T cells to allow clonal expansion and memory formation in response to viral infection. J. Exp. Med. 202, 637–650 (2005).
Aichele, P. et al. CD8 T cells specific for lymphocytic choriomeningitis virus require type I IFN receptor for clonal expansion. J. Immunol. 176, 4525–4529 (2006).
Le Bon, A. et al. Cross-priming of CD8+ T cells stimulated by virus-induced type I interferon. Nature Immunol. 4, 1009–1015 (2003).
Grabner, R. et al. Lymphotoxin β receptor signaling promotes tertiary lymphoid organogenesis in the aorta adventitia of aged ApoE-/- mice. J. Exp. Med. 206, 233–248 (2009).
Seshasayee, D. et al. In vivo blockade of OX40 ligand inhibits thymic stromal lymphopoietin driven atopic inflammation. J. Clin. Invest. 117, 3868–3878 (2007).
Tumanov, A. V. et al. Lymphotoxin controls the IL-22 protection pathway in gut innate lymphoid cells during mucosal pathogen challenge. Cell Host Microbe 10, 44–53 (2011).
Tsuji, M. et al. Requirement for lymphoid tissue-inducer cells in isolated follicle formation and T cell-independent immunoglobulin A generation in the gut. Immunity 29, 261–271 (2008).
Ota, N. et al. IL-22 bridges the lymphotoxin pathway with the maintenance of colonic lymphoid structures during infection with Citrobacter rodentium. Nature Immunol. 12, 941–948 (2011). References 52 and 54 provide mechanistic insight into the requirement for LTβR signalling in mounting an effective host response to Citrobacter rodentium . In these studies, the expression of LTαβ by innate lymphoid cells provoked LTβR signalling in DCs, resulting in the production of IL-23. This was essential for an IL-22 response, which provided host protection against the pathogen, and it provides a nice parallel in the mucosal immune system to the described function of LTβR-mediated DC–T cell crosstalk in the periphery.
Spahn, T. W. et al. The lymphotoxin-β receptor is critical for control of murine Citrobacter rodentium-induced colitis. Gastroenterology 127, 1463–1473 (2004).
Mackay, F. et al. Both the lymphotoxin and tumor necrosis factor pathways are involved in experimental murine models of colitis. Gastroenterology 115, 1464–1475 (1998).
Uhlig, H. H. et al. Differential activity of IL-12 and IL-23 in mucosal and systemic innate immune pathology. Immunity 25, 309–318 (2006).
De Jong, Y. P. et al. Chronic murine colitis is dependent on the CD154/CD40 pathway and can be attenuated by anti-CD154 administration. Gastroenterology 119, 715–723 (2000).
Cong, Y., Weaver, C. T., Lazenby, A. & Elson, C. O. Colitis induced by enteric bacterial antigen-specific CD4+ T cells requires CD40–CD40 ligand interactions for a sustained increase in mucosal IL-12. J. Immunol. 165, 2173–2182 (2000).
Steinberg, M. W. et al. A crucial role for HVEM and BTLA in preventing intestinal inflammation. J. Exp. Med. 205, 1463–1476 (2008).
Williamson, E., Bilsborough, J. M. & Viney, J. L. Regulation of mucosal dendritic cell function by receptor activator of NF-κB (RANK)/RANK ligand interactions: impact on tolerance induction. J. Immunol. 169, 3606–3612 (2002).
Totsuka, T. et al. RANK–RANKL signaling pathway is critically involved in the function of CD4+CD25+ regulatory T cells in chronic colitis. J. Immunol. 182, 6079–6087 (2009).
Salek-Ardakani, S. et al. OX40 (CD134) controls memory T helper 2 cells that drive lung inflammation. J. Exp. Med. 198, 315–324 (2003).
Doherty, T. A. et al. The tumor necrosis factor family member LIGHT is a target for asthmatic airway remodeling. Nature Med. 17, 596–603 (2011). This paper showed that the activity of the TNF superfamily member LIGHT in the airways after chronic antigen exposure was important for driving fibrosis and airway remodelling. This highlights the importance of understanding how TNF superfamily members may influence the parenchymal tissue in the context of a complex mixture of infiltrating inflammatory cells.
GeurtsvanKessel, C. H. et al. Dendritic cells are crucial for maintenance of tertiary lymphoid structures in the lung of influenza virus-infected mice. J. Exp. Med. 206, 2339–2349 (2009). Although lymphocytes, by virtue of their expression of membrane-bound LTαβ, are often the initiator cells in establishing follicle-like structures, this study shows that under certain conditions DCs may have a role in establishing ectopic lymphoid tissue. Intriguingly, LTαβ, which is normally expressed by lymphocytes, was found to be expressed on the DCs themselves, suggesting a novel mechanism for the delivery of LTβR signals to the underlying stroma.
Rangel-Moreno, J. et al. The development of inducible bronchus-associated lymphoid tissue depends on IL-17. Nature Immunol. 12, 639–646 (2011).
Feldmann, M. & Maini, R. N. Lasker Clinical Medical Research Award. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases. Nature Med. 9, 1245–1250 (2003).
Garber, K. Beyond ipilimumab: new approaches target the immunological synapse. J. Natl Cancer Inst. 103, 1079–1082 (2011).
Keller, A. M., Schildknecht, A., Xiao, Y., van den Broek, M. & Borst, J. Expression of costimulatory ligand CD70 on steady-state dendritic cells breaks CD8+ T cell tolerance and permits effective immunity. Immunity 29, 934–946 (2008). References 38 and 69 demonstrate that the expression of CD70 by DCs is required for overcoming T cell tolerance and can be induced by CD40 stimulation on DCs, illustrating an important link between CD40-mediated DC licensing and CD27-directed co-stimulation.
Piconese, S., Valzasina, B. & Colombo, M. P. OX40 triggering blocks suppression by regulatory T cells and facilitates tumor rejection. J. Exp. Med. 205, 825–839 (2008).
Pruitt, S. K. et al. Enhancement of anti-tumor immunity through local modulation of CTLA-4 and GITR by dendritic cells. Eur. J. Immunol. 41, 3553–3563 (2011).
Zaini, J. et al. OX40 ligand expressed by DCs costimulates NKT and CD4+ Th cell antitumor immunity in mice. J. Clin. Invest. 117, 3330–3338 (2007).
Dannull, J. et al. Enhancing the immunostimulatory function of dendritic cells by transfection with mRNA encoding OX40 ligand. Blood 105, 3206–3213 (2005).
Wiethe, C., Dittmar, K., Doan, T., Lindenmaier, W. & Tindle, R. Provision of 4-1BB ligand enhances effector and memory CTL responses generated by immunization with dendritic cells expressing a human tumor-associated antigen. J. Immunol. 170, 2912–2922 (2003).
Ohl, L. et al. CCR7 governs skin dendritic cell migration under inflammatory and steady-state conditions. Immunity 21, 279–288 (2004).
Aoshi, T. et al. Bacterial entry to the splenic white pulp initiates antigen presentation to CD8+ T cells. Immunity 29, 476–486 (2008).
Castellino, F. et al. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell–dendritic cell interaction. Nature 440, 890–895 (2006).
Hildebrand, J. M. et al. Roles of tumor necrosis factor receptor associated factor 3 (TRAF3) and TRAF5 in immune cell functions. Immunol. Rev. 244, 55–74 (2011).
Elgueta, R. et al. Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol. Rev. 229, 152–172 (2009).
Sanjo, H., Zajonc, D. M., Braden, R., Norris, P. S. & Ware, C. F. Allosteric regulation of the ubiquitin:NIK and ubiquitin:TRAF3 E3 ligases by the lymphotoxin-β receptor. J. Biol. Chem. 285, 17148–17155 (2010).
Bista, P. et al. TRAF3 controls activation of the canonical and alternative NFκB by the lymphotoxin β receptor. J. Biol. Chem. 285, 12971–12978 (2010).
Lo, J. C. et al. Coordination between NF-κB family members p50 and p52 is essential for mediating LTβR signals in the development and organization of secondary lymphoid tissues. Blood 107, 1048–1055 (2006).
Nocentini, G. & Riccardi, C. GITR: a multifaceted regulator of immunity belonging to the tumor necrosis factor receptor superfamily. Eur. J. Immunol. 35, 1016–1022 (2005).
Leibbrandt, A. & Penninger, J. M. Novel functions of RANKL signaling in the immune system. Adv. Exp. Med. Biol. 658, 77–94 (2010).
Summers Deluca, L. & Gommerman, J. L. The lymphotoxin pathway as a novel regulator of dendritic cell function. Adv. Exp. Med. Biol. 691, 363–374 (2011).
Peters, A. L., Stunz, L. L. & Bishop, G. A. CD40 and autoimmunity: the dark side of a great activator. Semin. Immunol. 21, 293–300 (2009).
Merad, M., Ginhoux, F. & Collin, M. Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells. Nature Rev. Immunol. 8, 935–947 (2008).
Iwasaki, A. Mucosal dendritic cells. Annu. Rev. Immunol. 25, 381–418 (2007).
Villadangos, J. A. & Schnorrer, P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nature Rev. Immunol. 7, 543–555 (2007).
Pabst, O. & Bernhardt, G. The puzzle of intestinal lamina propria dendritic cells and macrophages. Eur. J. Immunol. 40, 2107–2111 (2010).
Chen, A. I. et al. Ox40-ligand has a critical costimulatory role in dendritic cell:T cell interactions. Immunity 11, 689–698 (1999).
Jenkins, S. J., Perona-Wright, G., Worsley, A. G. F., Ishii, N. & MacDonald, A. S. Dendritic cell expression of OX40 ligand acts as a costimulatory, not polarizing, signal for optimal Th2 priming and memory induction in vivo. J. Immunol. 179, 3515–3523 (2007).
Salek-Ardakani, S. et al. The TNFR family members OX40 and CD27 link viral virulence to protective T cell vaccines in mice. J. Clin. Invest. 121, 296–307 (2011).
Dawicki, W. & Watts, T. H. Expression and function of 4-1BB during CD4 versus CD8 T cell responses in vivo. Eur. J. Immunol. 34, 743–751 (2004).
Lin, G. H. et al. Endogenous 4-1BB ligand plays a critical role in protection from influenza-induced disease. J. Immunol. 182, 934–947 (2009). This paper is a nice illustration of how the extent to which TNF superfamily receptor-mediated co-stimulation is required for effective T cell immunity is context dependent. In this case, the virulence of the virus strain was a major determinant in the dependence of CD8+ T cell responses on 4-1BB.
De Keersmaecker, B. et al. The combination of 4-1BBL and CD40L strongly enhances the capacity of dendritic cells to stimulate HIV-specific T cell responses. J. Leukoc. Biol. 89, 989–999 (2011).
Shao, Z. & Schwarz, H. CD137 ligand, a member of the tumor necrosis factor family, regulates immune responses via reverse signal transduction. J. Leukoc. Biol. 89, 21–29 (2011).
Hendriks, J., Xiao, Y. & Borst, J. CD27 promotes survival of activated T cells and complements CD28 in generation and establishment of the effector T cell pool. J. Exp. Med. 198, 1369–1380 (2003).
Schildknecht, A., Miescher, I., Yagita, H. & van den Broek, M. Priming of CD8+ T cell responses by pathogens typically depends on CD70-mediated interactions with dendritic cells. Eur. J. Immunol. 37, 716–728 (2007).
Stephens, G. L. et al. Engagement of glucocorticoid-induced TNFR family-related receptor on effector T cells by its ligand mediates resistance to suppression by CD4+CD25+ T cells. J. Immunol. 173, 5008–5020 (2004).
Tone, M. et al. Mouse glucocorticoid-induced tumor necrosis factor receptor ligand is costimulatory for T cells. Proc. Natl Acad. Sci. USA 100, 15059–15064 (2003).
Snell, L. M. et al. CD8 T cell-intrinsic GITR is required for T cell clonal expansion and mouse survival following severe influenza infection. J. Immunol. 185, 7223–7234 (2010).
Webster, B. et al. Regulation of lymph node vascular growth by dendritic cells. J. Exp. Med. 203, 1903–1913 (2006).
Zhu, M. & Fu, Y. X. The role of core TNF/LIGHT family members in lymph node homeostasis and remodeling. Immunol. Rev. 244, 75–84 (2011).
McLachlan, J. B. et al. Mast cell-derived tumor necrosis factor induces hypertrophy of draining lymph nodes during infection. Nature Immunol. 4, 1199–1205 (2003).
Agarwal, P. et al. Gene regulation and chromatin remodeling by IL-12 and type I IFN in programming for CD8 T cell effector function and memory. J. Immunol. 183, 1695–1704 (2009).
Tamada, K. et al. Blockade of LIGHT/LTβ and CD40 signaling induces allospecific T cell anergy, preventing graft-versus-host disease. J. Clin. Invest. 109, 549–557 (2002).
Acknowledgements
Funding for the research on how the lymphotoxin pathway supports T cell–DC crosstalk was provided by the Canadian Institutes of Health Research (MOP 67157). The authors wish to acknowledge D. Ng, L. Snell and O. Rojas for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Dendritic cell
-
(DC). A potent antigen-presenting cell that acts as a sentinel of the immune system. Located throughout peripheral tissues as well as in the lymphoid tissues of the periphery and the mucosae, these cells acquire and process antigens and present them to rare, antigen-specific T cells.
- TNF superfamily receptors
-
A molecular superfamily of receptors that trigger signalling events in a wide range of cell types, including cells of the immune system, such as T cells and DCs. Some TNF superfamily receptors (such as TNFR1) can activate cell death (in addition to other functions), whereas others (such as LTβR) can promote cell survival and inflammation.
- Secondary lymphoid organs
-
Highly organized anatomical structures, including the spleen and peripheral lymph nodes as well as the Peyer's patches and mucosa-associated lymphoid tissues (such as isolated lymphoid follicles and nasopharynx-associated lymphoid tissue). The cellular compartmentalization within these structures is actively maintained by constitutive LTβR signalling.
- Lymphoid-tissue inducer cells
-
A population of cells that is present in developing lymph nodes, Peyer's patches and nasopharynx-associated lymphoid tissue. Lymphoid-tissue inducer cells are required for the development of these lymphoid organs, and this function is mediated by their interactions with lymphoid-tissue organizer cells.
- Subcapsular sinus macrophages
-
Specialized macrophages that reside in the subcapsular sinuses of lymph nodes and are efficient at capturing antigens from the lymph.
- Microfold cells
-
Specialized antigen-sampling cells that are located in the follicle-associated epithelium of the organized mucosa-associated lymphoid tissues. Microfold cells deliver antigens by transepithelial vesicular transport from the aerodigestive lumen directly to subepithelial lymphoid tissues of the nasopharynx-associated lymphoid tissue and Peyer's patches.
- Tertiary lymphoid structures
-
(Also known as follicle-like structures). Collections of lymphocytes that occur in the context of pathogen-driven or autoimmune inflammation. The organization of such structures varies: some are clusters of lymphocytes and DCs, whereas others have ongoing germinal centre reactions. The organization of such structures is maintained by constitutive LTβR signalling, although the cellular source of LTαβ can vary.
- DC licensing
-
A concept that a DC must be converted to a functional state by an antigen-specific T helper cell in order to activate cytotoxic CD8+ T cells. T helper cells license DCs by expressing co-stimulatory molecules (such as the TNF superfamily ligands CD40L and LTαβ) or producing cytokines (such as IFNγ).
- Inducible co-stimulatory TNF superfamily receptors
-
Co-stimulation of the T cell response involves the integration of many signals, including those induced by the engagement of CD28 and other T cell-associated TNF superfamily receptors. T cell-expressed TNF superfamily receptors are typically upregulated in response to T cell receptor activation (hence the term 'inducible co-stimulators'). Likewise, the ligands for these inducible co-stimulators are TNF superfamily members, many of which are upregulated by activated DCs.
Rights and permissions
About this article
Cite this article
deLuca, L., Gommerman, J. Fine-tuning of dendritic cell biology by the TNF superfamily. Nat Rev Immunol 12, 339–351 (2012). https://doi.org/10.1038/nri3193
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3193
This article is cited by
-
Effects of an aged tissue niche on the immune potency of dendritic cells using simulated microgravity
npj Aging (2023)
-
Neonatal LTβR signaling is required for the accumulation of eosinophils in the inflamed adult mesenteric lymph node
Mucosal Immunology (2022)
-
Anti-inflammatory effect of naringin and sericin combination on human peripheral blood mononuclear cells (hPBMCs) from patient with psoriasis
BMC Complementary and Alternative Medicine (2019)
-
TNF-α enhances Th9 cell differentiation and antitumor immunity via TNFR2-dependent pathways
Journal for ImmunoTherapy of Cancer (2019)
-
The non-canonical NF-κB pathway in immunity and inflammation
Nature Reviews Immunology (2017)