Key Points
-
B cells express both a clonally rearranged, antigen-specific B cell receptor (BCR) and germline-encoded, pattern-recognizing Toll-like receptors (TLRs). Dual BCR and TLR engagement is responsible for fine-tuning functional B cell responses, directly linking innate and adaptive immune functions.
-
Consistent with the stronger in vitro responses of innate-like B-1 and marginal zone B cells to TLR ligands, T-independent B cell activation and antigen-specific antibody production is promoted by B cell-intrinsic TLR signalling that is dependent on myeloid differentiation primary-response protein 88 (MYD88).
-
Although not absolutely required for T-dependent B cell responses, B cell-intrinsic MYD88 signalling can enhance T-dependent antibody responses by driving class-switch recombination, promoting the differentiation of germinal centre and memory B cells into antibody-secreting plasma cells, and supporting effector T cell differentiation through cytokine secretion. The requirement for MYD88 signalling in B cells depends on both the nature of the protein antigen and the TLR ligand(s).
-
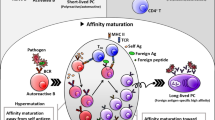
In addition to recognizing pathogens, B cell TLRs can recognize self ligands, in particular nuclear antigens derived from apoptotic cells. Activation of autoreactive B cells by dual BCR and TLR engagement has a crucial role in autoimmune pathogenesis and probably explains the restricted autoantibody repertoire of many human autoimmune diseases.
-
B cell-intrinsic TLR activation promotes the differentiation and expansion of interleukin-10-producing regulatory B cell populations that are able to suppress autoimmunity and limit immune responses to infectious challenges.
-
Activating mutations in signalling adaptors downstream of the BCR (including CARMA1, CD79a and CD79b) or of TLRs (such as MYD88) implicate dual BCR and TLR activation as a key survival signal in refractory human B cell lymphomas.
-
Although most of the above findings are derived from mouse models, emerging data from MYD88- or IL-1R-associated kinase 4 (IRAK4)-deficient patients and from human genome-wide association studies of autoimmunity and malignancy have begun to highlight the importance of BCR and TLR integration in human B cell tolerance and immune responses.
Abstract
Unlike other immune cells, B cells express both an antigen-specific B cell receptor (BCR) and Toll-like receptors (TLRs). Dual BCR and TLR engagement can fine-tune functional B cell responses, directly linking cell-intrinsic innate and adaptive immune programmes. Although most data regarding B cell-specific functions of the TLR signalling pathway have been obtained in mice, the discovery of patients with a deficiency in this pathway has recently provided an insight into human B cell responses. Here, we highlight the importance of the integration of signalling pathways downstream of BCRs and TLRs in modulating B cell function, focusing when possible on B cell-intrinsic roles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gavin, A. L. et al. Adjuvant-enhanced antibody responses in the absence of Toll-like receptor signaling. Science 314, 1936–1938 (2006).
Isnardi, I. et al. IRAK-4- and MyD88-dependent pathways are essential for the removal of developing autoreactive B cells in humans. Immunity 29, 746–757 (2008).
Meyer-Bahlburg, A., Bandaranayake, A. D., Andrews, S. F. & Rawlings, D. J. Reduced c-myc expression levels limit follicular mature B cell cycling in response to TLR signals. J. Immunol. 182, 4065–4075 (2009).
Gururajan, M., Jacob, J. & Pulendran, B. Toll-like receptor expression and responsiveness of distinct murine splenic and mucosal B-cell subsets. PLoS ONE 2, e863 (2007).
O'Neill, L. A. J. & Bowie, A. G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nature Rev. Immunol. 7, 353–364 (2007).
Mackay, F. & Schneider, P. Cracking the BAFF code. Nature Rev. Immunol. 9, 491–502 (2009).
Katsenelson, N. et al. Synthetic CpG oligodeoxynucleotides augment BAFF- and APRIL-mediated immunoglobulin secretion. Eur. J. Immunol. 37, 1785–1795 (2007).
Treml, L. S. et al. TLR stimulation modifies BLyS receptor expression in follicular and marginal zone B cells. J. Immunol. 178, 7531–7539 (2007).
Ng, L. G. et al. BAFF costimulation of Toll-like receptor-activated B-1 cells. Eur. J. Immunol. 36, 1837–1846 (2006).
Martin, F., Oliver, A. M. & Kearney, J. F. Marginal zone and B1 B cells unite in the early response against T-independent blood-borne particulate antigens. Immunity 14, 617–629 (2001). This is one of the first reports to clearly delineate the unique function of B-1 and marginal zone B cells in T-independent immune responses.
Haas, K. M., Poe, J. C., Steeber, D. A. & Tedder, T. F. B-1a and B-1b cells exhibit distinct developmental requirements and have unique functional roles in innate and adaptive immunity to S. pneumoniae. Immunity 23, 7–18 (2005).
Van der Hilst, J. C., Smits, B. W. & van der Meer, J. W. Hypogammaglobulinaemia: cumulative experience in 49 patients in a tertiary care institution. Neth. J. Med. 60, 140–147 (2002).
Rijkers, G. T., Sanders, L. A. & Zegers, B. J. Anti-capsular polysaccharide antibody deficiency states. Immunodeficiency 5, 1–21 (1993).
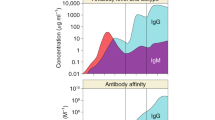
Kruetzmann, S. et al. Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen. J. Exp. Med. 197, 939–945 (2003).
Genestier, L. et al. TLR agonists selectively promote terminal plasma cell differentiation of B cell subsets specialized in thymus-independent responses. J. Immunol. 178, 7779–7786 (2007).
Meyer-Bahlburg, A., Khim, S. & Rawlings, D. J. B cell intrinsic TLR signals amplify but are not required for humoral immunity. J. Exp. Med. 204, 3095–3101 (2007). This study, together with reference 1, demonstrates that multiple protein immunization strategies can efficiently elicit specific antibody responses in the absence of MYD88 expression in B cells.
Oliver, A. M., Martin, F., Gartland, G. L., Carter, R. H. & Kearney, J. F. Marginal zone B cells exhibit unique activation, proliferative and immunoglobulin secretory responses. Eur. J. Immunol. 27, 2366–2374 (1997).
Oliver, A. M., Martin, F. & Kearney, J. F. IgMhighCD21high lymphocytes enriched in the splenic marginal zone generate effector cells more rapidly than the bulk of follicular B cells. J. Immunol. 162, 7198–7207 (1999).
Sindhava, V., Woodman, M. E., Stevenson, B. & Bondada, S. Interleukin-10 mediated autoregulation of murine B-1 B-cells and its role in Borrelia hermsii infection. PLoS ONE 5, e11445 (2010).
Blair, P. A. et al. Selective targeting of B cells with agonistic anti-CD40 is an efficacious strategy for the generation of induced regulatory T2-like B cells and for the suppression of lupus in MRL/lpr mice. J. Immunol. 182, 3492–3502 (2009).
Alugupalli, K. R., Akira, S., Lien, E. & Leong, J. M. MyD88- and Bruton's tyrosine kinase-mediated signals are essential for T cell-independent pathogen-specific IgM responses. J. Immunol. 178, 3740–3749 (2007).
Barr, T. A, Brown, S., Mastroeni, P. & Gray, D. B cell intrinsic MyD88 signals drive IFN-γ production from T cells and control switching to IgG2c. J. Immunol. 183, 1005–1012 (2009).
Neves, P. et al. Signaling via the MyD88 adaptor protein in B cells suppresses protective immunity during Salmonella typhimurium infection. Immunity 33, 777–790 (2010). This study demonstrated that regulatory B cell activity requires B cell-intrinsic MYD88 signalling in mice infected with S. Typhimurium.
Groeneveld, P. H., Erich, T. & Kraal, G. In vivo effects of LPS on B lymphocyte subpopulations. Migration of marginal zone-lymphocytes and IgD-blast formation in the mouse spleen. Immunobiology 170, 402–411 (1985).
Kraal, G. Cells in the marginal zone of the spleen. Int. Rev. Cytol. 132, 31–74 (1992).
Martin, F. & Kearney, J. F. Marginal-zone B cells. Nature Rev. Immunol. 2, 323–335 (2002).
Cinamon, G. et al. Sphingosine 1-phosphate receptor 1 promotes B cell localization in the splenic marginal zone. Nature Immunol. 5, 713–720 (2004).
Rubtsov, A. V. et al. TLR agonists promote marginal zone B cell activation and facilitate T-dependent IgM responses. J. Immunol. 180, 3882–3888 (2008).
Ha, S. A. et al. Regulation of B1 cell migration by signals through Toll-like receptors. J. Exp. Med. 203, 2541–2550 (2006).
von Bernuth, H. et al. Pyogenic bacterial infections in humans with MyD88 deficiency. Science 321, 691–696 (2008).
Picard, C. et al. Clinical features and outcome of patients with IRAK-4 and MyD88 deficiency. Medicine 89, 403–425 (2010).
Picard, C. et al. Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science 299, 2076–2079 (2003). References 30 and 32 provided the first descriptions of mutations in MYD88 and IRAK4 , respectively, in immunodeficient patients.
He, B. et al. The transmembrane activator TACI triggers immunoglobulin class switching by activating B cells through the adaptor MyD88. Nature Immunol. 11, 836–845 (2010). This study identifies MYD88 as an adaptor molecule involved in TACI-driven NF-κB signalling, thereby providing a role for MYD88 in T-independent class switching.
Castigli, E. et al. TACI and BAFF-R mediate isotype switching in B cells. J. Exp. Med. 201, 35–39 (2005).
Salzer, U. et al. Mutations in TNFRSF13B encoding TACI are associated with common variable immunodeficiency in humans. Nature Genet. 37, 820–828 (2005).
Barr, T. A., Brown, S., Mastroeni, P. & Gray, D. TLR and B cell receptor signals to B cells differentially program primary and memory Th1 responses to Salmonella enterica. J. Immunol. 185, 2783–2789 (2010).
Hwang, I.-Y., Park, C., Harrison, K. & Kehrl, J. H. TLR4 signaling augments B lymphocyte migration and overcomes the restriction that limits access to germinal center dark zones. J. Exp. Med. 206, 2641–2657 (2009).
Bernasconi, N. L., Traggiai, E. & Lanzavecchia, A. Maintenance of serological memory by polyclonal activation of human memory B cells. Science 298, 2199–2202 (2002).
Ruprecht, C. R. & Lanzavecchia, A. Toll-like receptor stimulation as a third signal required for activation of human naive B cells. Eur. J. Immunol. 36, 810–816 (2006).
Pasare, C. & Medzhitov, R. Control of B-cell responses by Toll-like receptors. Nature 438, 364–368 (2005). This study examined the antibody response to protein immunizations and concluded that protein-specific, T-dependent B cell immune responses require B cell-intrinsic TLR signalling.
Lanzavecchia, A. & Sallusto, F. Toll-like receptors and innate immunity in B-cell activation and antibody responses. Curr. Opin. Immunol. 19, 268–274 (2007).
Palm, N. W. & Medzhitov, R. Immunostimulatory activity of haptenated proteins. Proc. Natl Acad. Sci. USA 106, 4782–4787 (2009).
Hou, B. et al. Selective utilization of Toll-like receptor and MyD88 signaling in B cells for enhancement of the antiviral germinal center response. Immunity 34, 375–384 (2011). In this study, the response to TLR ligands in various physical forms was evaluated in mice with a DC- or B cell-specific MYD88 deficiency. MYD88 signals in B cells were essential for optimal antibody responses to virus-like particles.
Yang, R. et al. B lymphocyte activation by human papillomavirus-like particles directly induces Ig class switch recombination via TLR4–MyD88. J. Immunol. 174, 7912–7919 (2005).
Jegerlehner, A. et al. TLR9 signaling in B cells determines class switch recombination to IgG2a. J. Immunol. 178, 2415–2420 (2007).
Heer, A. K. et al. TLR signaling fine-tunes anti-influenza B cell responses without regulating effector T cell responses. J. Immunol. 178, 2182–2191 (2007).
Guay, H. M., Andreyeva, T. A., Garcea, R. L., Welsh, R. M. & Szomolanyi-Tsuda, E. MyD88 is required for the formation of long-term humoral immunity to virus infection. J. Immunol. 178, 5124–5131 (2007).
Delgado, M. F. et al. Lack of antibody affinity maturation due to poor Toll-like receptor stimulation leads to enhanced respiratory syncytial virus disease. Nature Med. 15, 34–41 (2009).
Becker-Herman, S. et al. WASp-deficient B cells play a critical, cell-intrinsic role in triggering autoimmunity. J. Exp. Med. 208, 2033–2042 (2011). This study demonstrated that WASP-deficient B cells exhibit signalling abnormalities sufficient to promote a cell-intrinsic, MYD88-dependent break in T cell tolerance that triggers lethal humoral autoimmunity.
Shlomchik, M. J. Activating systemic autoimmunity: B's, T's, and tolls. Curr. Opin. Immunol. 21, 626–633 (2009).
Green, N. M. & Marshak-Rothstein, A. Toll-like receptor driven B cell activation in the induction of systemic autoimmunity. Semin. Immunol. 23, 106–112 (2011).
Leadbetter, E. A. et al. Chromatin–IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416, 603–607 (2002). This study provided the first demonstration that dual BCR and TLR signals can markedly promote the activation of self-reactive B cells by uptake of antibody–DNA complexes via a rheumatoid factor-specific BCR.
Lau, C. M. et al. RNA-associated autoantigens activate B cells by combined B cell antigen receptor/Toll-like receptor 7 engagement. J. Exp. Med. 202, 1171–1177 (2005). This study was the first to demonstrate that Myd88 deletion is sufficient to prevent humoral autoimmune disease in vivo . Multiple additional studies have confirmed these findings and expanded on the distinct roles for TLR7 versus TLR9 signals in these events.
Nickerson, K. M. et al. TLR9 regulates TLR7- and MyD88-dependent autoantibody production and disease in a murine model of lupus. J. Immunol. 184, 1840–1848 (2010).
Yasuda, K. et al. Murine dendritic cell type I IFN production induced by human IgG–RNA immune complexes is IFN regulatory factor (IRF)5 and IRF7 dependent and is required for IL-6 production. J. Immunol. 178, 6876–6885 (2007).
Groom, J. R. et al. BAFF and MyD88 signals promote a lupuslike disease independent of T cells. J. Exp. Med. 204, 1959–1971 (2007).
Ehlers, M., Fukuyama, H., McGaha, T. L., Aderem, A. & Ravetch, J. V. TLR9/MyD88 signaling is required for class switching to pathogenic IgG2a and 2b autoantibodies in SLE. J. Exp. Med. 203, 553–561 (2006).
Christensen, S. R. et al. Toll-like receptor 7 and TLR9 dictate autoantibody specificity and have opposing inflammatory and regulatory roles in a murine model of lupus. Immunity 25, 417–428 (2006).
Christensen, S. R. et al. Toll-like receptor 9 controls anti-DNA autoantibody production in murine lupus. J. Exp. Med. 202, 321–331 (2005).
Santiago-Raber, M.-L. et al. Critical role of TLR7 in the acceleration of systemic lupus erythematosus in TLR9-deficient mice. J. Autoimmun. 34, 339–348 (2010).
Zhang, L. et al. Transmembrane activator and calcium-modulating cyclophilin ligand interactor mutations in common variable immunodeficiency: clinical and immunologic outcomes in heterozygotes. J. Allergy Clin. Immunol. 120, 1178–1185 (2007).
Graham, R. R. et al. A common haplotype of interferon regulatory factor 5 (IRF5) regulates splicing and expression and is associated with increased risk of systemic lupus erythematosus. Nature Genet. 38, 550–555 (2006).
Jacob, C. O. et al. Identification of IRAK1 as a risk gene with critical role in the pathogenesis of systemic lupus erythematosus. Proc. Natl Acad. Sci. USA 106, 6256–6261 (2009).
Graham, R. R. et al. Genetic variants near TNFAIP3 on 6q23 are associated with systemic lupus erythematosus. Nature Genet. 40, 1059–1061 (2008).
Han, J. W. et al. Genome-wide association study in a Chinese Han population identifies nine new susceptibility loci for systemic lupus erythematosus. Nature Genet. 41, 1234–1237 (2009).
Tavares, R. M. et al. The ubiquitin modifying enzyme A20 restricts B cell survival and prevents autoimmunity. Immunity 33, 181–191 (2010).
Nanda, S. K. et al. Polyubiquitin binding to ABIN1 is required to prevent autoimmunity. J. Exp. Med. 208, 1215–1228 (2011).
Shen, N. et al. Sex-specific association of X-linked Toll-like receptor 7 (TLR7) with male systemic lupus erythematosus. Proc. Natl Acad. Sci. USA 107, 15838–15843 (2010).
Huang, C. M. et al. Association of Toll-like receptor 9 gene polymorphism in Chinese patients with systemic lupus erythematosus in Taiwan. Rheumatol. Int. 17 Apr 2011 (doi:10.1007/s00296-011-1925-8).
Tao, K. et al. Genetic variations of Toll-like receptor 9 predispose to systemic lupus erythematosus in Japanese population. Ann. Rheum. Dis. 66, 905–909 (2007).
O'Garra, A. et al. Production of cytokines by mouse B cells: B lymphomas and normal B cells produce interleukin 10. Int. Immunol. 2, 821–832 (1990).
Burdin, N., Péronne, C., Banchereau, J. & Rousset, F. Epstein-Barr virus transformation induces B lymphocytes to produce human interleukin 10. J. Exp. Med. 177, 295–304 (1993).
Evans, J. G. et al. Novel suppressive function of transitional 2 B cells in experimental arthritis. J. Immunol. 178, 7868–7878 (2007).
Yanaba, K., Bouaziz, J. D., Matsushita, T., Tsubata, T. & Tedder, T. F. The development and function of regulatory B cells expressing IL-10 (B10 cells) requires antigen receptor diversity and TLR signals. J. Immunol. 182, 7459–7472 (2009).
Mizoguchi, A., Mizoguchi, E., Takedatsu, H., Blumberg, R. S. & Bhan, A. K. Chronic intestinal inflammatory condition generates IL-10-producing regulatory B cell subset characterized by CD1d upregulation. Immunity 16, 219–230 (2002).
Mauri, C. & Ehrenstein, M. R. The “short” history of regulatory B cells. Trends Immunol. 29, 34–40 (2008).
Tian, J. et al. Lipopolysaccharide-activated B cells down-regulate Th1 immunity and prevent autoimmune diabetes in nonobese diabetic mice. J. Immunol. 167, 1081–1089 (2001).
Parekh, V. V. et al. B cells activated by lipopolysaccharide, but not by anti-Ig and anti-CD40 antibody, induce anergy in CD8+ T cells: role of TGFβ1. J. Immunol. 170, 5897–5911 (2003).
Fillatreau, S., Sweenie, C. H., McGeachy, M. J., Gray, D. & Anderton, S. M. B cells regulate autoimmunity by provision of IL-10. Nature Immunol. 3, 944–950 (2002).
Ronet, C. et al. Regulatory B cells shape the development of Th2 immune responses in BALB/c mice infected with Leishmania major through IL-10 production. J. Immunol. 184, 886–894 (2010).
Hernandez, H. J., Wang, Y. & Stadecker, M. J. In infection with Schistosoma mansoni, B cells are required for T helper type 2 cell responses but not for granuloma formation. J. Immunol. 158, 4832–4837 (1997).
Gillan, V., Lawrence, R. A. & Devaney, E. B cells play a regulatory role in mice infected with the L3 of Brugia pahangi. Int. Immunol. 17, 373–382 (2005).
Sayi, A. et al. TLR-2-activated B cells suppress Helicobacter-induced preneoplastic gastric immunopathology by inducing T regulatory-1 cells. J. Immunol. 186, 878–890 (2011).
Lenert, P., Brummel, R., Field, E. H. & Ashman, R. F. TLR-9 activation of marginal zone B cells in lupus mice regulates immunity through increased IL-10 production. J. Clin. Immunol. 25, 29–40 (2005).
Lampropoulou, V. et al. TLR-activated B cells suppress T cell-mediated autoimmunity. J. Immunol. 180, 4763–4773 (2008).
Iwata, Y. et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood 117, 530–541 (2011).
Blair, P. A. et al. CD19+CD24hiCD38hi B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic lupus erythematosus patients. Immunity 32, 129–140 (2010).
Staudt, L. M. Oncogenic activation of NF-κB. Cold Spring Harb. Perspect. Biol. 2, a000109 (2010).
Davis, R. E., Brown, K. D., Siebenlist, U. & Staudt, L. M. Constitutive nuclear factor κB activity is required for survival of activated B cell-like diffuse large B cell lymphoma cells. J. Exp. Med. 194, 1861–1874 (2001).
Rawlings, D. J., Sommer, K. & Moreno-García, M. E. The CARMA1 signalosome links the signalling machinery of adaptive and innate immunity in lymphocytes. Nature Rev. Immunol. 6, 799–812 (2006).
Sommer, K. et al. Phosphorylation of the CARMA1 linker controls NF-κB activation. Immunity 23, 561–574 (2005).
Ngo, V. N. et al. A loss-of-function RNA interference screen for molecular targets in cancer. Nature 441, 106–110 (2006).
Lenz, G. et al. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science 319, 1676–1679 (2008).
Davis, R. E. et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature 463, 88–92 (2010).
Ngo, V. N. et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 470, 115–119 (2011). This study identified activating mutations in MYD88 in poor-prognosis human B cell lymphomas and showed that many such tumours also have activating mutations in genes associated with the BCR signalling cascade.
Lam, L. T. et al. Cooperative signaling through the signal transducer and activator of transcription 3 and nuclear factor-κB pathways in subtypes of diffuse large B-cell lymphoma. Blood 111, 3701–3713 (2008).
Ding, B. B. et al. Constitutively activated STAT3 promotes cell proliferation and survival in the activated B-cell subtype of diffuse large B-cell lymphomas. Blood 111, 1515–1523 (2008).
Puente, X. S. et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 475, 101–105 (2011).
Michelsen, K. S. et al. Lack of Toll-like receptor 4 or myeloid differentiation factor 88 reduces atherosclerosis and alters plaque phenotype in mice deficient in apolipoprotein E. Proc. Natl Acad. Sci. USA 101, 10679–10684 (2004).
Bjorkbacka, H. et al. Reduced atherosclerosis in MyD88-null mice links elevated serum cholesterol levels to activation of innate immunity signaling pathways. Nature Med. 10, 416–421 (2004).
Kiechl, S. et al. Toll-like receptor 4 polymorphisms and atherogenesis. N. Engl. J. Med. 347, 185–192 (2002).
Lewis, M. J. et al. Immunoglobulin M is required for protection against atherosclerosis in low-density lipoprotein receptor-deficient mice. Circulation 120, 417–426 (2009).
Caligiuri, G., Nicoletti, A., Poirier, B. & Hansson, G. K. Protective immunity against atherosclerosis carried by B cells of hypercholesterolemic mice. J. Clin. Invest. 109, 745–753 (2002).
Zhao, M. et al. FcγRIIB inhibits the development of atherosclerosis in low-density lipoprotein receptor-deficient mice. J. Immunol. 184, 2253–2260 (2010).
Kyaw, T. et al. Conventional B2 B cell depletion ameliorates whereas its adoptive transfer aggravates atherosclerosis. J. Immunol. 185, 4410–4419 (2010).
Ait-Oufella, H. et al. B cell depletion reduces the development of atherosclerosis in mice. J. Exp. Med. 207, 1579–1587 (2010).
Bernatsky, S. et al. Mortality in systemic lupus erythematosus. Arthritis Rheum. 54, 2550–2557 (2006).
Svenungsson, E. et al. Risk factors for cardiovascular disease in systemic lupus erythematosus. Circulation 104, 1887–1893 (2001).
Bendelac, A., Bonneville, M. & Kearney, J. F. Autoreactivity by design: innate B and T lymphocytes. Nature Rev. Immunol. 1, 177–186 (2001).
Godin, I. E., Garcia-Porrero, J. A., Coutinho, A., Dieterlen-Lievre, F. & Marcos, M. A. Para-aortic splanchnopleura from early mouse embryos contains B1a cell progenitors. Nature 364, 67–70 (1993).
Montecino-Rodriguez, E., Leathers, H. & Dorshkind, K. Identification of a B-1 B cell-specified progenitor. Nature Immunol. 7, 293–301 (2006).
Griffin, D. O., Holodick, N. E. & Rothstein, T. L. Human B1 cells in umbilical cord and adult peripheral blood express the novel phenotype CD20+CD27+CD43+CD70−. J. Exp. Med. 208, 67–80 (2011).
Ehrenstein, M. R. & Notley, C. A. The importance of natural IgM: scavenger, protector and regulator. Nature Rev. Immunol. 10, 778–786 (2010).
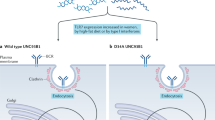
Casrouge, A. et al. Herpes simplex virus encephalitis in human UNC-93B deficiency. Science 314, 308–312 (2006).
Zhang, S. Y. et al. TLR3 deficiency in patients with herpes simplex encephalitis. Science 317, 1522–1527 (2007).
Shlomchik, M. J. Sites and stages of autoreactive B cell activation and regulation. Immunity 28, 18–28 (2008).
Acknowledgements
This work was supported by US National Institutes of Health grants HD037091, HL075453, AI084457 and AI071163 (to D.J.R.); a Cancer Research Institute Predoctoral Training Grant (to M.A.S.); Rheumatology T32 Postdoctoral Training Grant 5T32AR007108 (to S.W.J.) and German Research Foundation (Deutsche Forschungsgemeinschaft) grant ME2709/2-1 (to A.M.B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- B-1 cells
-
IgMhiIgDlowMAC1+B220lowCD23− cells that are dominant in the peritoneal and pleural cavities. The size of the B-1 cell population is kept constant owing to the self-renewing capacity of these cells. B-1 cells recognize self components, as well as common bacterial antigens, and they secrete antibodies that tend to have low affinity and broad specificity.
- Marginal zone B cells
-
Mature B cells that are enriched mainly in the spleen marginal zone, which is located at the border of the white pulp.
- Follicular B cells
-
A recirculating mature B cell subset that populates the follicles of the spleen and lymph nodes.
- Germinal centre
-
A lymphoid structure that arises in follicles after immunization with, or exposure to, a T-dependent antigen. This structure is specialized for facilitating the development of high-affinity, long-lived plasma cells and memory B cells.
- Class-switch recombination
-
(CSR). The process by which a heavy-chain variable-region gene segment that is attached to one heavy-chain constant-region gene segment in the expressed heavy-chain gene is recombined with a downstream constant-region gene segment, leading to the expression of a new antibody class.
- Systemic lupus erythematosus
-
(SLE). An autoimmune disease in which autoantibodies that are specific for DNA, RNA or proteins associated with nucleic acids form immune complexes that damage small blood vessels, especially in the kidney. Patients with SLE generally have abnormal B and T cell function.
- Wiskott–Aldrich syndrome
-
A life-threatening X-linked immunodeficiency caused by mutations in the gene encoding Wiskott–Aldrich syndrome protein. It is characterized by thrombocytopenia with small platelets, eczema, recurrent infections caused by immunodeficiency and an increased incidence of autoimmune manifestations and malignancies.
- Common variable immune deficiency syndrome
-
(CVID syndrome). The most common symptomatic primary antibody deficiency, characterized by decreased levels of serum immunoglobulins and a low or normal number of B cells. Most patients suffer from recurrent infections, predominantly of the respiratory and gastrointestinal tracts. The incidence of malignancies, such as gastric carcinoma or lymphoma, is increased in patients with CVID syndrome.
- μMT mice
-
These mice carry a stop codon in the first membrane exon of the immunoglobulin μ-chain constant region. They lack IgM+ B cells, and B cell development is arrested before the differentiation stage at which IgD can be expressed.
- Ribi adjuvant
-
An emulsion containing a metabolizable oil, a detergent and bacterial products, including the TLR4 ligand monophosphoryl lipid A.
- Hapten
-
A molecule that can bind to antibodies but cannot by itself elicit an immune response. Antibodies that are specific for a hapten can be generated when the hapten is chemically linked to a protein carrier that can elicit a T cell response.
- Virus-like particles
-
(VLPs). Virion-like structures that are formed from the self-assembly of viral envelope or capsid proteins in vitro. VLPs are not infectious because they do not contain a viral genome.
- Antinuclear antibodies
-
(ANAs). Heterogeneous autoantibodies that are specific for one or more antigens present in the nucleus, including chromatin, nucleosomes and ribonuclear proteins. ANAs are found in association with many different autoimmune diseases.
- Immune complexes
-
Complexes of antigen bound to antibody and, sometimes, components of the complement system. The levels of immune complexes are increased in many autoimmune disorders, in which they become deposited in tissues and cause tissue damage.
- MRL–lpr mice
-
A mouse strain that spontaneously develops glomerulonephritis and other symptoms of systemic lupus erythematosus. The lpr mutation causes a defect in CD95 (also known as FAS), preventing apoptosis of activated lymphocytes. The MRL strain contributes disease-associated mutations that have yet to be identified.
- MRL–gld mice
-
A mouse strain that has a naturally occurring mutation in CD95 ligand that causes a generalized lymphoproliferative disease, similar to that of MRL–lpr mice.
- T regulatory type 1 cells
-
A subset of CD4+ regulatory T cells that secrete high levels of IL-10 and that downregulate TH1 and TH2 cell responses in vitro and in vivo through one or more contact-independent mechanisms that are mediated by the secretion of soluble IL-10 and TGFβ1.
- Apolipoprotein E-deficient mice
-
(Apoe−/− mice). A widely used mouse model that is prone to develop atherosclerosis because the mice have high levels of remnant lipoproteins (a type of atherogenic lipoprotein). This lipoprotein abnormality is caused by the genetic absence of apolipoprotein E, which normally clears remnant lipoproteins from the bloodstream by interacting with hepatocytes.
- Low-density lipoprotein receptor-deficient mice
-
(Ldlr−/− mice). A mouse model of atherosclerosis caused by a targeted deletion of the gene encoding the low-density lipoprotein receptor (LDLR). In humans, homozygous mutations in LDLR cause familial hypercholesterolaemia, a disease characterized by pronounced hyperlipidaemia and accelerated atherosclerotic cardiovascular disease. Ldlr−/− mice fed a high-fat, high-cholesterol diet have a plasma lipoprotein profile resembling humans and develop aortic lesions that are morphologically similar to human atherosclerotic plaques.
Rights and permissions
About this article
Cite this article
Rawlings, D., Schwartz, M., Jackson, S. et al. Integration of B cell responses through Toll-like receptors and antigen receptors. Nat Rev Immunol 12, 282–294 (2012). https://doi.org/10.1038/nri3190
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3190
This article is cited by
-
T-independent B-cell effect of agents associated with swine grower-finisher diarrhea
Veterinary Research Communications (2023)
-
Inhibitors targeting Bruton’s tyrosine kinase in cancers: drug development advances
Leukemia (2021)
-
Targeting IRAK4 disrupts inflammatory pathways and delays tumor development in chronic lymphocytic leukemia
Leukemia (2020)
-
Homeostasis and regulation of autoreactive B cells
Cellular & Molecular Immunology (2020)
-
Immune metabolism regulation of the germinal center response
Experimental & Molecular Medicine (2020)