Key Points
-
There are two fundamentally distinct T cell lineages: αβ and γδ T cells. Individual lineage specification occurs in the thymus. This Review discusses recent findings that shed light on the factors that regulate this process, focusing on evidence that supports a central role for T cell receptor (TCR)-derived signals and thymus microenvironmental cues in mediating γδ T cell lineage choice and development.
-
Over the past two decades, two models have been proposed to account for the differences seen in γδ and αβ T cell commitment, namely lineage 'instruction' by cognate TCR expression and TCR-independent lineage 'pre-commitment' followed by selection. Rigid adherence to either of these models is not supported by the available data.
-
We present recent evidence that supports a 'TCR signal strength' model in which quantitatively larger TCR-mediated signals are proposed to favour γδ T cell lineage development. Specifically, the induction of inhibitor of DNA binding 3 (ID3) by strong TCR signals downstream of the extracellular signal-regulated kinase (ERK)–early growth response (EGR) pathway seems to be a crucial mechanism by which the γδ T cell lineage is instructed. Interestingly, ID3 is required for both the development of a normal repertoire of γδ T cells and for limiting the development of γδ T cell subsets that express high-affinity, autoreactive TCRs.
-
Although TCR-mediated signals seem to have the greatest impact on γδ versus αβ T cell specification, signals from the thymus microenvironment — for example, Notch ligands, lymphotoxin-mediated trans-conditioning and TCR ligands — also contribute to lineage and/or functional diversification.
-
Given the emerging understanding of the important roles that γδ T cells have in immune regulation, maintenance of tissue integrity and inflammation, future studies examining the acquisition of these functions will be of great interest.
Abstract
The thymus produces several types of functionally distinct T cell subsets. However, at a more fundamental level only two genetically distinct T cell lineages exist: the γδ and αβ T cell lineages. Precisely how these two T cell lineages are generated from common thymocyte progenitor cells remains to be fully elucidated and is under intense investigation. Here, we highlight recent findings that have helped to provide important clues to the mechanisms that underpin the generation of γδ T cells in the mouse thymus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Petrie, H. T. & Zúñiga-Pflücker, J. C. Zoned out: functional mapping of stromal signaling microenvironments in the thymus. Annu. Rev. Immunol. 25, 649–679 (2007).
Porritt, H. E. et al. Heterogeneity among DN1 prothymocytes reveals multiple progenitors with different capacities to generate T cell and non-T cell lineages. Immunity 20, 735–745 (2004).
Taghon, T., Yui, M. A., Pant, R., Diamond, R. A. & Rothenberg, E. V. Developmental and molecular characterization of emerging β- and γδ-selected pre-T cells in the adult mouse thymus. Immunity 24, 53–64 (2006).
Hoffman, E. S. et al. Productive T-cell receptor β-chain gene rearrangement: coincident regulation of cell cycle and clonality during development in vivo. Genes Dev. 10, 948–962 (1996).
Livak, F., Tourigny, M., Schatz, D. G. & Petrie, H. T. Characterization of TCR gene rearrangements during adult murine T cell development. J. Immunol. 162, 2575–2580 (1999).
Kruisbeek, A. M. et al. Branching out to gain control: how the pre-TCR is linked to multiple functions. Immunol. Today 21, 637–644 (2000).
Michie, A. M. & Zúñiga-Pflücker, J. C. Regulation of thymocyte differentiation: pre-TCR signals and beta-selection. Semin. Immunol. 14, 311–323 (2002).
Starr, T. K., Jameson, S. C. & Hogquist, K. A. Positive and negative selection of T cells. Annu. Rev. Immunol. 21, 139–176 (2003).
Taghon, T. & Rothenberg, E. V. Molecular mechanisms that control mouse and human TCR-αβ and TCR-γδ T cell development. Semin. Immunopathol. 30, 383–398 (2008).
Nanno, M., Shiohara, T., Yamamoto, H., Kawakami, K. & Ishikawa, H. γδ T cells: firefighters or fire boosters in the front lines of inflammatory responses. Immunol. Rev. 215, 103–113 (2007).
Hayday, A. & Tigelaar, R. Immunoregulation in the tissues by γδ T cells. Nature Rev. Immunol. 3, 233–242 (2003).
Ciofani, M., Knowles, G. C., Wiest, D. L., von Boehmer, H. & Zúñiga-Pflücker, J. C. Stage-specific and differential notch dependency at the αβ and γδ T lineage bifurcation. Immunity 25, 105–116 (2006). This paper delineates the developmental point(s) at which γδ and αβ T cell lineages diverge, and also establishes that Notch signals are required only for further αβ T cell lineage differentiation.
Kreslavsky, T., Garbe, A. I., Krueger, A. & von Boehmer, H. T cell receptor-instructed αβ versus γδ lineage commitment revealed by single-cell analysis. J. Exp. Med. 205, 1173–1186 (2008).
Prinz, I. et al. Visualization of the earliest steps of γδ T cell development in the adult thymus. Nature Immunol. 7, 995–1003 (2006). This paper makes use of a new reporter mouse model that identifies the earliest γδ T lineage cells and establishes that a single γδ TCR selection checkpoint occurs at the γδ T cell lineage bifurcation point.
Yamasaki, S. et al. Mechanistic basis of pre-T cell receptor-mediated autonomous signaling critical for thymocyte development. Nature Immunol. 7, 67–75 (2006).
Jensen, K. D. et al. Thymic selection determines γδ T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon γ. Immunity 29, 90–100 (2008). This paper reveals that the generation of IFNγ- and IL-17-producing γδ T cells is determined during thymic selection on the basis of potential TCR–ligand engagements.
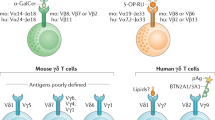
Chien, Y. H. & Konigshofer, Y. Antigen recognition by γδ T cells. Immunol. Rev. 215, 46–58 (2007).
Boyden, L. M. et al. Skint1, the prototype of a newly identified immunoglobulin superfamily gene cluster, positively selects epidermal γδ T cells. Nature Genet. 40, 656–662 (2008).
Hayday, A. C., Barber, D. F., Douglas, N. & Hoffman, E. S. Signals involved in γδ T cell versus αβ T cell lineage commitment. Semin. Immunol. 11, 239–249 (1999).
Narayan, K. & Kang, J. Molecular events that regulate αβ versus γδ T cell lineage commitment: old suspects, new players and different game plans. Curr. Opin. Immunol. 19, 169–175 (2007).
Lauritsen, J. P., Haks, M. C., Lefebvre, J. M., Kappes, D. J. & Wiest, D. L. Recent insights into the signals that control αβ/γδ-lineage fate. Immunol. Rev. 209, 176–190 (2006).
Hayes, S. M. & Love, P. E. A retrospective on the requirements for γδ T-cell development. Immunol. Rev. 215, 8–14 (2007).
Melichar, H. J. et al. Regulation of γδ versus αβ T lymphocyte differentiation by the transcription factor SOX13. Science 315, 230–233 (2007).
Marfil, V. et al. Interaction between Hhex and SOX13 modulates Wnt/TCF activity. J. Biol. Chem. 285, 5726–5737 (2010).
Lauritsen, J. P. et al. Marked induction of the helix-loop-helix protein Id3 promotes the γδ T cell fate and renders their functional maturation Notch independent. Immunity 31, 565–575 (2009).
Kang, J., Volkmann, A. & Raulet, D. H. Evidence that γδ versus αβ T cell fate determination is initiated independently of T cell receptor signaling. J. Exp. Med. 193, 689–698 (2001). Along with reference 23, this paper provides evidence for γδ T cell lineage determination prior to the provision of TCR-dependent signals.
Schmitt, T. M. & Zúñiga-Pflücker, J. C. Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity 17, 749–756 (2002).
Zúñiga-Pflücker, J. C. T-cell development made simple. Nature Rev. Immunol. 4, 67–72 (2004).
Schlissel, M. S., Durum, S. D. & Muegge, K. The interleukin 7 receptor is required for T cell receptor γ locus accessibility to the V(D)J recombinase. J. Exp. Med. 191, 1045–1050 (2000).
Hayes, S. M., Shores, E. W. & Love, P. E. An architectural perspective on signaling by the pre-, αβ and γδ T cell receptors. Immunol. Rev. 191, 28–37 (2003).
Haks, M. C. et al. Attenuation of γδTCR signaling efficiently diverts thymocytes to the αβ lineage. Immunity 22, 595–606 (2005).
Hayes, S. M., Li, L. & Love, P. E. TCR signal strength influences αβ/γδ lineage fate. Immunity 22, 583–593 (2005). References 31 and 32 provide evidence in support of the signal strength model, in which TCR-dependent signals determine γδ versus αβ T cell lineage outcomes.
Irving, B. A., Alt, F. W. & Killeen, N. Thymocyte development in the absence of pre-T cell receptor extracellular immunoglobulin domains. Science 280, 905–908 (1998).
Haks, M. C. et al. Low activation threshold as a mechanism for ligand-independent signaling in pre-T cells. J. Immunol. 170, 2853–2861 (2003).
Murre, C. Helix-loop-helix proteins and lymphocyte development. Nature Immunol. 6, 1079–1086 (2005).
Carleton, M. et al. Early growth response transcription factors are required for development of CD4−CD8− thymocytes to the CD4+CD8+ stage. J. Immunol. 168, 1649–1658 (2002).
Xi, H., Schwartz, R., Engel, I., Murre, C. & Kersh, G. J. Interplay between RORγt, Egr3, and E proteins controls proliferation in response to pre-TCR signals. Immunity 24, 813–826 (2006).
Ueda-Hayakawa, I., Mahlios, J. & Zhuang, Y. Id3 restricts the developmental potential of γδ lineage during thymopoiesis. J. Immunol. 182, 5306–5316 (2009).
Alonzo, E. S. et al. Development of promyelocytic zinc finger and ThPOK-expressing innate γδ T cells is controlled by strength of TCR signaling and Id3. J. Immunol. 184, 1268–1279 (2010).
He, X. et al. The zinc finger transcription factor Th-POK regulates CD4 versus CD8 T-cell lineage commitment. Nature 433, 826–833 (2005).
Sun, G. et al. The zinc finger protein cKrox directs CD4 lineage differentiation during intrathymic T cell positive selection. Nature Immunol. 6, 373–381 (2005).
Park, K. et al. TCR-mediated ThPOK induction promotes development of mature (CD24−) γδ thymocytes. EMBO J. 29, 2329–2341 (2010).
Ciofani, M. & Zúñiga-Pflücker, J. C. The thymus as an inductive site for T lymphopoiesis. Annu. Rev. Cell Dev. Biol. 23, 463–493 (2007).
Rothenberg, E. V. Negotiation of the T lineage fate decision by transcription-factor interplay and microenvironmental signals. Immunity 26, 690–702 (2007).
Hayday, A. C. & Pennington, D. J. Key factors in the organized chaos of early T cell development. Nature Immunol. 8, 137–144 (2007).
Radtke, F. et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 10, 547–558 (1999).
Sambandam, A. et al. Notch signaling controls the generation and differentiation of early T lineage progenitors. Nature Immunol. 6, 663–670 (2005).
Schmitt, T. M., Ciofani, M., Petrie, H. T. & Zúñiga-Pflücker, J. C. Maintenance of T cell specification and differentiation requires recurrent notch receptor-ligand interactions. J. Exp. Med. 200, 469–479 (2004).
Tan, J. B., Visan, I., Yuan, J. S. & Guidos, C. J. Requirement for Notch1 signals at sequential early stages of intrathymic T cell development. Nature Immunol. 6, 671–679 (2005).
Ciofani, M. & Zúñiga-Pflücker, J. C. Notch promotes survival of pre-T cells at the β-selection checkpoint by regulating cellular metabolism. Nature Immunol. 6, 881–888 (2005).
Palomero, T. et al. NOTCH1 directly regulates c-MYC and activates a feed-forward-loop transcriptional network promoting leukemic cell growth. Proc. Natl Acad. Sci. USA 103, 18261–18266 (2006).
Weng, A. P. et al. c-Myc is an important direct target of Notch1 in T-cell acute lymphoblastic leukemia/lymphoma. Genes Dev. 20, 2096–2109 (2006).
Janas, M. L. et al. Thymic development beyond β-selection requires phosphatidylinositol 3-kinase activation by CXCR4. J. Exp. Med. 207, 247–261 (2010).
Trampont, P. C. et al. CXCR4 acts as a costimulator during thymic β-selection. Nature Immunol. 11, 162–170 (2010).
Garbe, A. I., Krueger, A., Gounari, F., Zúñiga-Pflücker, J. C. & von Boehmer, H. Differential synergy of Notch and T cell receptor signaling determines αβ versus γδ lineage fate. J. Exp. Med. 203, 1579–1590 (2006).
Van de Walle, I. et al. An early decrease in Notch activation is required for human TCR-αβ lineage differentiation at the expense of TCR-γδ T cells. Blood 113, 2988–2998 (2009).
Taghon, T. et al. Notch signaling is required for proliferation but not for differentiation at a well-defined β-selection checkpoint during human T-cell development. Blood 113, 3254–3263 (2009).
Pennington, D. J. et al. Early events in the thymus affect the balance of effector and regulatory T cells. Nature 444, 1073–1077 (2006).
Silva-Santos, B., Pennington, D. J. & Hayday, A. C. Lymphotoxin-mediated regulation of γδ cell differentiation by αβ T cell progenitors. Science 307, 925–928 (2005). This paper shows that γδ T cell differentiation in the thymus is affected by αβ T lineage cells through LT-dependent signals, demonstrating the molecular basis for the trans -conditioning phenomenon.
Pennington, D. J. et al. The inter-relatedness and interdependence of mouse T cell receptor γδ+ and αβ+ cells. Nature Immunol. 4, 991–998 (2003).
Ribot, J. C. et al. CD27 is a thymic determinant of the balance between interferon-γ- and interleukin 17-producing γδ T cell subsets. Nature Immunol. 10, 427–436 (2009).
Kreslavsky, T. et al. TCR-inducible PLZF transcription factor required for innate phenotype of a subset of γδ T cells with restricted TCR diversity. Proc. Natl Acad. Sci. USA 106, 12453–12458 (2009).
Acknowledgements
This work was supported by a grant from the Canadian Institutes of Health Research (CIHR-MOP42387). J.C.Z.-P. is supported by a Canada Research Chair in Developmental Immunology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Gene rearrangement
-
The ordered rearrangement of variable regions of genes encoding antigen receptors. This rearrangement contributes to increased receptor diversity.
- Pre-T cell receptor
-
(Pre-TCR). A receptor that is expressed by pre-T cells. It is formed by a T cell receptor-β (TCRβ)-chain paired with a surrogate TCRα-chain (known as the invariant pre-Tα protein). The receptor complex includes CD3 proteins and transduces signals that allow further T cell development.
- β-selection
-
The controlled developmental transition beyond the double-negative 3 (DN3) stage to the double-positive (DP) stage that is limited to T cells that have successfully rearranged their T cell receptor-β (TCRβ)-chain genes to express a functional cell surface pre-TCR. The conditional developmental arrest encountered at the DN3 stage is termed the β-selection checkpoint.
- Signal strength
-
The overall amount of signal emanating from the T cell receptor (TCR), which is typically assessed by monitoring phosphorylation events in signalling cascades downstream of the TCR. This is affected by the concentration of antigen, the extent of signal amplification by co-stimulatory molecules and the duration of the antigen-presenting cell–T cell immune synapse. These parameters can vary widely and in a stochastic manner.
- Notch
-
A signalling system comprised of highly conserved transmembrane receptors that regulate cell-fate choice in the development of many cell lineages and so are vital in the regulation of embryonic differentiation and development.
- Recombinase-activating genes
-
Genes that are expressed by developing lymphocytes. Mice that lack either recombinase-activation gene 1 (Rag1) or Rag2 fail to produce B or T cells owing to a developmental block in the gene rearrangement that is necessary for receptor expression.
- KN6 γδ TCR-transgenic mice
-
Mice that express the γδ T cell receptor (TCR) transgene (Vγ4–Vδ5), which is positively selected on the MHC class Ib molecule T10d, but is deleted by the higher affinity T10b and/or T22b ligands.
- Dendritic epidermal T cells
-
(DETCs). γδ T cell receptor (TCR)+ cells that specifically localize in the epidermis and are present in rodents and cattle but not in humans. In mice, essentially all DETCs express the same Vγ5–Vδ1 TCR, forming a prototype lymphocyte repertoire of limited diversity.
- Lymphotoxin
-
(LT). A protein that belongs to the tumour necrosis factor family and that can be produced as a secreted homotrimer, lymphotoxin-α3 (LTα3), or as a membrane-bound heterotrimer, LTα1β2. The heterotrimer LTα1β2 binds to the lymphotoxin-β receptor (LTβR).
Rights and permissions
About this article
Cite this article
Ciofani, M., Zúñiga-Pflücker, J. Determining γδ versus αβ T cell development. Nat Rev Immunol 10, 657–663 (2010). https://doi.org/10.1038/nri2820
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2820
This article is cited by
-
γδ T cells: origin and fate, subsets, diseases and immunotherapy
Signal Transduction and Targeted Therapy (2023)
-
Dietary glucosamine overcomes the defects in αβ-T cell ontogeny caused by the loss of de novo hexosamine biosynthesis
Nature Communications (2022)
-
Distinct and temporary-restricted epigenetic mechanisms regulate human αβ and γδ T cell development
Nature Immunology (2020)
-
Peripheral T cell lymphoma, NOS with aberrant αβ and γδ T cell receptor expression in a post-heart transplant patient
Journal of Hematopathology (2020)
-
Casein kinase 2 controls the survival of normal thymic and leukemic γδ T cells via promotion of AKT signaling
Leukemia (2017)