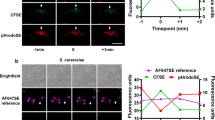
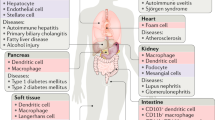
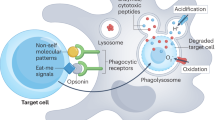
Abstract
Professional phagocytes ingest particulate material to fulfil a diverse array of functions in a multicellular organism. The ancestral function of phagosomes is digestion; however, through evolution this degradative capacity has become pivotal to the adaptive immune response by processing antigens to be presented to lymphocytes. Moreover, phagocytes have also acquired an active role in microbial killing. This Innovation article describes new assays that probe the biological activities which occur within phagosomes. These assays provide functional insights into how the phagosome fulfils its diverse roles in homeostasis and in innate and adaptive immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Chen, G., Zhuchenko, O. & Kuspa, A. Immune-like phagocyte activity in the social amoeba. Science 317, 678–681 (2007).
Hagedorn, M., Rohde, K. H., Russell, D. G. & Soldati, T. Infection by tubercular mycobacteria is spread by nonlytic ejection from their amoeba hosts. Science 323, 1729–1733 (2009).
Gazzinelli, R. T. & Denkers, E. Y. Protozoan encounters with Toll-like receptor signalling pathways: implications for host parasitism. Nature Rev. Immunol. 6, 895–906 (2006).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc. Natl Acad. Sci. USA 97, 13766–13771 (2000).
Trinchieri, G. & Sher, A. Cooperation of Toll-like receptor signals in innate immune defence. Nature Rev. Immunol. 7, 179–190 (2007).
Akira, S. TLR signaling. Curr. Top. Microbiol. Immunol. 311, 1–16 (2006).
DeLeo, F. R., Allen, L. A., Apicella, M. & Nauseef, W. M. NADPH oxidase activation and assembly during phagocytosis. J. Immunol. 163, 6732–6740 (1999).
MacMicking, J. D. et al. Identification of nitric oxide synthase as a protective locus against tuberculosis. Proc. Natl Acad. Sci. USA 94, 5243–5248 (1997).
MacMicking, J. D., Taylor, G. A. & McKinney, J. D. Immune control of tuberculosis by IFN-γ-inducible LRG-47. Science 302, 654–659 (2003).
Mosser, D. M. The many faces of macrophage activation. J. Leukoc. Biol. 73, 209–212 (2003).
Desjardins, M. & Griffiths, G. Phagocytosis: latex leads the way. Curr. Opin. Cell Biol. 15, 498–503 (2003).
Vieira, O. V., Botelho, R. J. & Grinstein, S. Phagosome maturation: aging gracefully. Biochem. J. 366, 689–704 (2002).
Estrada, S. et al. The role of Gly-4 of human cystatin A (stefin A) in the binding of target proteinases. Characterization by kinetic and equilibrium methods of the interactions of cystatin A Gly-4 mutants with papain, cathepsin B, and cathepsin L. Biochemistry 37, 7551–7560 (1998).
Yeung, T. & Grinstein, S. Lipid signaling and the modulation of surface charge during phagocytosis. Immunol. Rev. 219, 17–36 (2007).
Steinberg, B. E., Touret, N., Vargas-Caballero, M. & Grinstein, S. In situ measurement of the electrical potential across the phagosomal membrane using FRET and its contribution to the proton-motive force. Proc. Natl Acad. Sci. USA 104, 9523–9528 (2007).
Yeung, T. et al. Receptor activation alters inner surface potential during phagocytosis. Science 313, 347–351 (2006).
Yates, R. M., Hermetter, A. & Russell, D. G. The kinetics of phagosome maturation as a function of phagosome/lysosome fusion and acquisition of hydrolytic activity. Traffic 6, 413–420 (2005).
Yates, R. M. & Russell, D. G. Real-time spectrofluorometric assays for the lumenal environment of the maturing phagosome. Methods Mol. Biol. 445, 311–325 (2008).
Yates, R. M., Hermetter, A. & Russell, D. G. Recording phagosome maturation through the real-time, spectrofluorometric measurement of hydrolytic activities. Methods Mol. Biol. 531, 157–171 (2009).
Yates, R. M., Hermetter, A., Taylor, G. A. & Russell, D. G. Macrophage activation downregulates the degradative capacity of the phagosome. Traffic 8, 241–250 (2007).
Minakami, R. & Sumimotoa, H. Phagocytosis-coupled activation of the superoxide-producing phagocyte oxidase, a member of the NADPH oxidase (nox) family. Int. J. Hematol. 84, 193–198 (2006).
Nathan, C. Role of iNOS in human host defense. Science 312, 1874–1875 (2006).
VanderVen, B. C., Yates, R. M. & Russell, D. G. Intraphagosomal measurement of the magnitude and duration of the oxidative burst. Traffic 10, 372–378 (2009).
Underhill, D. M. et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811–815 (1999).
Blander, J. M. & Medzhitov, R. Regulation of phagosome maturation by signals from toll-like receptors. Science 304, 1014–1018 (2004).
Blander, J. M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Yates, R. M. & Russell, D. G. Phagosome maturation proceeds independently of stimulation of toll-like receptors 2 and 4. Immunity 23, 409–417 (2005).
Kobayashi, N. et al. TIM-1 and TIM-4 glycoproteins bind phosphatidylserine and mediate uptake of apoptotic cells. Immunity 27, 927–940 (2007).
Wu, Y., Tibrewal, N. & Birge, R. B. Phosphatidylserine recognition by phagocytes: a view to a kill. Trends Cell Biol. 16, 189–197 (2006).
Steinman, R. M. Dendritic cells: understanding immunogenicity. Eur. J. Immunol. 37, S53–S60 (2007).
Tsang, A. W., Oestergaard, K., Myers, J. T. & Swanson, J. A. Altered membrane trafficking in activated bone marrow-derived macrophages. J. Leukoc. Biol. 68, 487–494 (2000).
Desjardins, M. Biogenesis of phagolysosomes: the 'kiss and run' hypothesis. Trends Cell Biol. 5, 183–186 (1995).
Lennon-Dumenil, A. M. et al. Analysis of protease activity in live antigen-presenting cells shows regulation of the phagosomal proteolytic contents during dendritic cell activation. J. Exp. Med. 196, 529–540 (2002).
Fujiwara, N. & Kobayashi, K. Macrophages in inflammation. Curr. Drug Targets Inflamm. Allergy 4, 281–286 (2005).
Savina, A. et al. NOX2 controls phagosomal pH to regulate antigen processing during crosspresentation by dendritic cells. Cell 126, 205–218 (2006).
Depraetere, V. “Eat me” signals of apoptotic bodies. Nature Cell Biol. 2, E104 (2000).
Delamarre, L., Couture, R., Mellman, I. & Trombetta, E. S. Enhancing immunogenicity by limiting susceptibility to lysosomal proteolysis. J. Exp. Med. 203, 2049–2055 (2006).
Jutras, I. et al. Modulation of the phagosome proteome by interferon-γ. Mol. Cell. Proteomics 7, 697–715 (2008).
Trost, M. et al. The phagosomal proteome in interferon-γ-activated macrophages. Immunity 30, 143–154 (2009).
Dinauer, M. C. & Orkin, S. H. Chronic granulomatous disease. Annu. Rev. Med. 43, 117–124 (1992).
Koziel, H., Li, X., Armstrong, M. Y., Richards, F. F. & Rose, R. M. Alveolar macrophages from human immunodeficiency virus-infected persons demonstrate impaired oxidative burst response to Pneumocystis carinii in vitro. Am. J. Respir. Cell Mol. Biol. 23, 452–459 (2000).
Acknowledgements
We acknowledge funding from the National Institutes of Health, the Bill and Melinda Gates Foundation and the Wellcome Trust that supported the research behind this article. The authors thank R. Yates and D. Fullerton for their contributions to these studies.
Author information
Authors and Affiliations
Corresponding author
Related links
Rights and permissions
About this article
Cite this article
Russell, D., VanderVen, B., Glennie, S. et al. The macrophage marches on its phagosome: dynamic assays of phagosome function. Nat Rev Immunol 9, 594–600 (2009). https://doi.org/10.1038/nri2591
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2591
This article is cited by
-
The microbiome of a bacterivorous marine choanoflagellate contains a resource-demanding obligate bacterial associate
Nature Microbiology (2022)
-
Efferocytosis requires periphagosomal Ca2+-signaling and TRPM7-mediated electrical activity
Nature Communications (2022)
-
Engineered macrophages as near-infrared light activated drug vectors for chemo-photodynamic therapy of primary and bone metastatic breast cancer
Nature Communications (2021)
-
Identification of the general stress stimulon related to colonization in Enterococcus faecalis
Archives of Microbiology (2020)
-
Increased survival and proliferation of the epidemic strain Mycobacterium abscessus subsp. massiliense CRM0019 in alveolar epithelial cells
BMC Microbiology (2017)