Key Points
-
Mounting an immune response is a metabolically costly endeavour and cannot operate well under conditions of energy deficit. Energy surplus (for example, in individuals that are obese or suffer from metabolic syndrome) can also impair immune responses and induce chronic inflammation. The integrated and proper functioning of each system is paramount to the health of the other.
-
Chronic inflammation, particularly when it occurs in metabolically important organs such as the liver and adipose tissue, has a crucial role in the emergence of many chronic metabolic diseases, including diabetes, fatty liver disease and cardiovascular disease. It is important to recognize that this is not a response that resembles classic inflammation and perhaps could be considered as an aberrant form of immunity that is triggered by nutrients or other intrinsic cues.
-
During most conditions of stress and inflammation, changes in metabolism occur, and insulin signalling is impaired through protein modifications, such as serine phosphorylation of insulin-receptor substrate proteins. These modifications resemble the events that occur during conditions of obesity, insulin resistance and type 2 diabetes. Nutrient overload and/or lack of protection from the inflammatory response lead to deterioration of metabolic health.
-
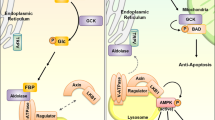
Recent studies have given rise to the concept that metabolic stress might be sensed by organelles, particularly the endoplasmic reticulum (ER) and mitochondria, and the resulting organelle dysfunction could trigger a network of stress signalling that could disrupt metabolic homeostasis. The ER can also serve as a convergence point of nutrient and pathogen responses.
-
Intriguingly, many of the nutrient-sensing pathways are subverted by pathogens for their proliferation and survival, and revisiting some of these links may provide a basis for the emergence of metabolic diseases and present new platforms for research in the fields of metabolism and infection.
Abstract
The proper functioning of the pathways that are involved in the sensing and management of nutrients is central to metabolic homeostasis and is therefore among the most fundamental requirements for survival. Metabolic systems are integrated with pathogen-sensing and immune responses, and these pathways are evolutionarily conserved. This close functional and molecular integration of the immune and metabolic systems is emerging as a crucial homeostatic mechanism, the dysfunction of which underlies many chronic metabolic diseases, including type 2 diabetes and atherosclerosis. In this Review we provide an overview of several important networks that sense and manage nutrients and discuss how they integrate with immune and inflammatory pathways to influence the physiological and pathological metabolic states in the body.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Demas, G. E., Chefer, V., Talan, M. I. & Nelson, R. J. Metabolic costs of mounting an antigen-stimulated immune response in adult and aged C57BL/56J mice. Am. J. Physiol. 273, R1631–R1637 (1997).
Marti, A., Marcos, A. & Martinez, J. A. Obesity and immune function relationships. Obes. Rev. 2, 131–140 (2001).
Romanyukha, A. A., Rudnev, S. G. & Sidorov, I. A. Energy cost of infection burden: an approach to understanding the dynamics of host-pathogen interactions. J. Theor. Biol. 241, 1–13 (2006).
Browning, R. C. & Kram, R. Energetic cost and preferred speed of walking in obese vs. normal weight women. Obes. Res. 13, 891–899 (2005).
Maier, S. F., Watkins, L. R. & Fleshner, M. Psychoneuroimmunology. The interface between behavior, brain, and immunity. Am. Psychol. 49, 1004–1017 (1994).
Demas, G. E., Drazen, D. L. & Nelson, R. J. Reductions in total body fat decrease humoral immunity. Proc. Biol. Sci. 270, 905–911 (2003).
Moret, Y. & Schmid-Hempel, P. Survival for immunity: the price of immune system activation for bumblebee workers. Science 290, 1166–1168 (2000). This study shows that immune activation is costly and subject to an energy trade-off during conditions of starvation, leading to the reduced survival of insects.
Leclerc, V. & Reichhart, J. M. The immune response of Drosophila melanogaster. Immunol. Rev. 198, 59–71 (2004).
Pond, C. M. & Mattacks, C. A. The source of fatty acids incorporated into proliferating lymphoid cells in immune-stimulated lymph nodes. Br. J. Nutr. 89, 375–383 (2003).
Knight, S. C. Specialized perinodal fat fuels and fashions immunity. Immunity 28, 135–138 (2008).
Karagiannides, I. & Pothoulakis, C. Obesity, innate immunity and gut inflammation. Curr. Opin. Gastroenterol. 23, 661–666 (2007).
Lago, R., Gomez, R., Lago, F., Gomez-Reino, J. & Gualillo, O. Leptin beyond body weight regulation — current concepts concerning its role in immune function and inflammation. Cell. Immunol. 252, 139–145 (2008).
Chung, S. et al. Preadipocytes mediate lipopolysaccharide-induced inflammation and insulin resistance in primary cultures of newly differentiated human adipocytes. Endocrinology 147, 5340–5351 (2006).
Charriere, G. et al. Preadipocyte conversion to macrophage. Evidence of plasticity. J. Biol. Chem. 278, 9850–9855 (2003).
Khazen, W. et al. Expression of macrophage-selective markers in human and rodent adipocytes. FEBS Lett. 579, 5631–5634 (2005).
Wellen, K. E. & Hotamisligil, G. S. Inflammation, stress, and diabetes. J. Clin. Invest. 115, 1111–1119 (2005).
Uysal, K. T., Wiesbrock, S. M., Marino, M. W. & Hotamisligil, G. S. Protection from obesity-induced insulin resistance in mice lacking TNF-α function. Nature 389, 610–614 (1997). Using genetic models, this study showed for the first time that inflammation and increased cytokine levels in conditions of obesity have a causal role in the development of insulin resistance.
Medzhitov, R. Origin and physiological roles of inflammation. Nature 454, 428–435 (2008).
Chandra, R. K. Immune response in overnutrition. Cancer Res. 41, 3795–3796 (1981).
Taniguchi C. M., Emanuelli, B. & Kahn, C.R. Critical nodes in signalling pathways: insights into insulin action. Nature Rev. Mol. Cell Biol. 7, 85–96 (2006).
Zick, Y. Ser/Thr phosphorylation of IRS proteins: a molecular basis for insulin resistance. Sci. STKE 2005, pe4 (2005).
Gao, Z. et al. Serine phosphorylation of insulin receptor substrate 1 by inhibitor κB kinase complex. J. Biol. Chem. 277, 48115–48121 (2002).
Um, S. H. et al. Absence of S6K1 protects against age- and diet-induced obesity while enhancing insulin sensitivity. Nature 431, 200–205 (2004).
Hotamisligil, G. S. et al. IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNFα- and obesity-induced insulin resistance. Science 271, 665–668 (1996). This study showed that the pro-inflammatory cytokine TNF is induced in adipose tissue in conditions of obesity and that TNF contributes to metabolic disease.
Xu, H. et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 112, 1821–1830 (2003).
Hotamisligil, G. S., Shargill, N. S. & Spiegelman, B. M. Adipose expression of tumor necrosis factor-α: direct role in obesity-linked insulin resistance. Science 259, 87–91 (1993).
Hirosumi, J. et al. A central role for JNK in obesity and insulin resistance. Nature 420, 333–336 (2002).
Arkan, M. C. et al. IKK-β links inflammation to obesity-induced insulin resistance. Nature Med. 11, 191–198 (2005). References 28 and 29 show that well-known inflammatory pathways are linked with the development of insulin resistance in obesity.
Wellen, K. E. et al. Coordinated regulation of nutrient and inflammatory responses by STAMP2 is essential for metabolic homeostasis. Cell 129, 537–548 (2007). This study shows that proper regulation of metabolic and immune pathways by proteins such as STAMP2 promotes metabolic health.
Bensinger, S. J. et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111 (2008).
Ozcan, U. et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306, 457–461 (2004).
Kaneto, H. et al. Possible novel therapy for diabetes with cell-permeable JNK-inhibitory peptide. Nature Med. 10, 1128–1132 (2004).
Nakatani, Y. et al. Modulation of the JNK pathway in liver affects insulin resistance status. J. Biol. Chem. 279, 45803–45809 (2004).
Ozcan, U. et al. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 313, 1137–1140 (2006).
Solinas, G. et al. JNK1 in hematopoietically derived cells contributes to diet-induced inflammation and insulin resistance without affecting obesity. Cell. Metab. 6, 386–397 (2007).
Vallerie, S. N., Furuhashi, M., Fucho, R. & Hotamisligil, G. S. A predominant role for parenchymal c-Jun amino terminal kinase (JNK) in the regulation of systemic insulin sensitivity. PLoS ONE 3, e3151 (2008).
Schroder, M. & Kaufman, R. J. The mammalian unfolded protein response. Annu. Rev. Biochem. 74, 739–789 (2005).
Todd, D. J., Lee, A. H. & Glimcher, L. H. The endoplasmic reticulum stress response in immunity and autoimmunity. Nature Rev. Immunol. 8, 663–674 (2008).
Cox, J. S., Shamu, C. E. & Walter, P. Transcriptional induction of genes encoding endoplasmic reticulum resident proteins requires a transmembrane protein kinase. Cell 73, 1197–1206 (1993).
Mori, K., Ma, W., Gething, M. J. & Sambrook, J. A transmembrane protein with a CDC2+/CDC28-related kinase activity is required for signaling from the ER to the nucleus. Cell 74, 743–756 (1993).
Urano, F. et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287, 664–666 (2000). This paper demonstrated that ER stress is coupled to the activation of the kinase JNK.
Yoshida, H., Matsui, T., Yamamoto, A., Okada, T. & Mori, K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell 107, 881–891 (2001).
Ron, D. & Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nature Rev. Mol. Cell Biol. 8, 519–529 (2007).
Gregor, M. F. & Hotamisligil, G. S. Thematic review series: adipocyte biology. Adipocyte stress: the endoplasmic reticulum and metabolic disease. J. Lipid Res. 48, 1905–1914 (2007).
Deng, J. et al. Translational repression mediates activation of nuclear factor κB by phosphorylated translation initiation factor 2. Mol. Cell Biol. 24, 10161–10168 (2004).
Hu, P., Han, Z., Couvillon, A. D., Kaufman, R. J. & Exton, J. H. Autocrine tumor necrosis factor α links endoplasmic reticulum stress to the membrane death receptor pathway through IRE1α-mediated NF-κB activation and down-regulation of TRAF2 expression. Mol. Cell Biol. 26, 3071–3084 (2006).
Zhang, K. et al. Endoplasmic reticulum stress activates cleavage of CREBH to induce a systemic inflammatory response. Cell 124, 587–599 (2006).
Xue, X. et al. Tumor necrosis factor α (TNFα) induces the unfolded protein response (UPR) in a reactive oxygen species (ROS)-dependent fashion, and the UPR counteracts ROS accumulation by TNFα. J. Biol. Chem. 280, 33917–33925 (2005).
Houstis, N., Rosen, E. D. & Lander, E. S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 440, 944–948 (2006).
Levine, T. & Loewen, C. Inter-organelle membrane contact sites: through a glass, darkly. Curr. Opin. Cell Biol. 18, 371–378 (2006).
Pouyssegur, J., Shiu, R. P. & Pastan, I. Induction of two transformation-sensitive membrane polypeptides in normal fibroblasts by a block in glycoprotein synthesis or glucose deprivation. Cell 11, 941–947 (1977).
Scheuner, D. et al. Translational control is required for the unfolded protein response and in vivo glucose homeostasis. Mol. Cell 7, 1165–1176 (2001).
Harding, H. P. et al. Diabetes mellitus and exocrine pancreatic dysfunction in perk−/− mice reveals a role for translational control in secretory cell survival. Mol. Cell 7, 1153–1163 (2001).
Lipson, K. L. et al. Regulation of insulin biosynthesis in pancreatic β cells by an endoplasmic reticulum-resident protein kinase IRE1. Cell. Metab. 4, 245–254 (2006).
Ota, T., Gayet, C. & Ginsberg, H. N. Inhibition of apolipoprotein B100 secretion by lipid-induced hepatic endoplasmic reticulum stress in rodents. J. Clin. Invest. 118, 316–332 (2008).
Borradaile, N. M. et al. A critical role for eukaryotic elongation factor 1A-1 in lipotoxic cell death. Mol. Biol. Cell 17, 770–778 (2006).
Ozcan, U. et al. Loss of the tuberous sclerosis complex tumor suppressors triggers the unfolded protein response to regulate insulin signaling and apoptosis. Mol. Cell 29, 541–551 (2008).
Feng, B. et al. The endoplasmic reticulum is the site of cholesterol-induced cytotoxicity in macrophages. Nature Cell Biol. 5, 781–792 (2003). In this study, the authors show that ER stress has an important role in macrophage cell death that is caused by free-cholesterol loading.
Tabas, I. Consequences and therapeutic implications of macrophage apoptosis in atherosclerosis: the importance of lesion stage and phagocytic efficiency. Arterioscler. Thromb. Vasc. Biol. 25, 2255–2264 (2005).
Han, S. et al. Macrophage insulin receptor deficiency increases ER stress-induced apoptosis and necrotic core formation in advanced atherosclerotic lesions. Cell. Metab. 3, 257–266 (2006).
Hosoi, T. et al. Endoplasmic reticulum stress induces leptin resistance. Mol. Pharmacol. 28 August 2008 (doi:10.1124/mol.108.050070).
Myoishi, M. et al. Increased endoplasmic reticulum stress in atherosclerotic plaques associated with acute coronary syndrome. Circulation 116, 1226–1233 (2007).
Ozawa, K. et al. The endoplasmic reticulum chaperone improves insulin resistance in type 2 diabetes. Diabetes 54, 657–663 (2005).
Reiling, J. H. & Sabatini, D. M. Stress and mTORture signaling. Oncogene 25, 6373–6383 (2006).
Manning, B. D. & Cantley, L. C. AKT/PKB signaling: navigating downstream. Cell 129, 1261–1274 (2007).
Harrington, L. S. et al. The TSC1–2 tumor suppressor controls insulin–PI3K signaling via regulation of IRS proteins. J. Cell Biol. 166, 213–223 (2004).
Pende, M. et al. Hypoinsulinaemia, glucose intolerance and diminished β-cell size in S6K1-deficient mice. Nature 408, 994–997 (2000).
Colombani, J. et al. A nutrient sensor mechanism controls Drosophila growth. Cell 114, 739–749 (2003).
Cota, D. et al. Hypothalamic mTOR signaling regulates food intake. Science 312, 927–930 (2006).
Lee, D. F. et al. IKKβ suppression of TSC1 links inflammation and tumorangiogenesis via the mTOR pathway. Cell 130, 440–455 (2007). This is an interesting paper showing that a well-known inflammatory pathway can directly intercept an important nutrient pathway. This study documents a molecular link between amino-acid monitoring and regulation of lipid metabolism in the liver.
Mellor, A. L. & Munn, D. H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nature Rev. Immunol. 4, 762–774 (2004).
Rodriguez, P. C. & Ochoa, A. C. Arginine regulation by myeloid derived suppressor cells and tolerance in cancer: mechanisms and therapeutic perspectives. Immunol. Rev. 222, 180–191 (2008). References 72 and 73 both provide an insightful review of how the metabolism of a single amino acid ( L -arginine) can directly affect immunity.
Guo, F. & Cavener, D. R. The GCN2 eIF2α kinase regulates fatty-acid homeostasis in the liver during deprivation of an essential amino acid. Cell. Metab. 5, 103–114 (2007).
Gingras, A. C., Svitkin, Y., Belsham, G. J., Pause, A. & Sonenberg, N. Activation of the translational suppressor 4E-BP1 following infection with encephalomyocarditis virus and poliovirus. Proc. Natl Acad. Sci. USA 93, 5578–5583 (1996).
Connor, J. H. & Lyles, D. S. Vesicular stomatitis virus infection alters the eIF4F translation initiation complex and causes dephosphorylation of the eIF4E binding protein 4E-BP1. J. Virol. 76, 10177–10187 (2002).
Shi, H. et al. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Invest. 116, 3015–3025 (2006).
Medzhitov, R. Toll-like receptors and innate immunity. Nature Rev. Immunol. 1, 135–145 (2001).
Tsukumo, D. M. et al. Loss-of-function mutation in Toll-like receptor 4 prevents diet-induced obesity and insulin resistance. Diabetes 56, 1986–1998 (2007).
Davis, J. E., Gabler, N. K., Walker-Daniels, J. & Spurlock, M. E. Tlr-4 deficiency selectively protects against obesity induced by diets high in saturated fat. Obesity (Silver Spring) 16, 1248–1255 (2008).
Ajuwon, K. M. & Spurlock, M. E. Palmitate activates the NF-κB transcription factor and induces IL-6 and TNFα expression in 3T3-L1 adipocytes. J. Nutr. 135, 1841–1846 (2005).
Ghanim, H. et al. Acute modulation of Toll-like receptors by insulin. Diabetes Care 31, 1827–1831 (2008).
Vitseva, O. I. et al. Inducible Toll-like receptor and NF-κB regulatory pathway expression in human adipose tissue. Obesity (Silver Spring) 16, 932–937 (2008).
Song, M. J., Kim, K. H., Yoon, J. M. & Kim, J. B. Activation of Toll-like receptor 4 is associated with insulin resistance in adipocytes. Biochem. Biophys. Res. Commun. 346, 739–745 (2006).
Suganami, T. et al. Attenuation of obesity-induced adipose tissue inflammation in C3H/HeJ mice carrying a Toll-like receptor 4 mutation. Biochem. Biophys. Res. Commun. 354, 45–49 (2007).
Xu, X. H. et al. Toll-like receptor-4 is expressed by macrophages in murine and human lipid-rich atherosclerotic plaques and upregulated by oxidized LDL. Circulation 104, 3103–3108 (2001).
Michelsen, K. S. et al. Lack of Toll-like receptor 4 or myeloid differentiation factor 88 reduces atherosclerosis and alters plaque phenotype in mice deficient in apolipoprotein E. Proc. Natl Acad. Sci. USA 101, 10679–10684 (2004).
Kiechl, S. et al. Toll-like receptor 4 polymorphisms and atherogenesis. N. Engl. J. Med. 347, 185–192 (2002).
Erbay, E., Cao, H. & Hotamisligil, G. S. Adipocyte/macrophage fatty acid binding proteins in metabolic syndrome. Curr. Atheroscler. Rep. 9, 222–229 (2007).
Tuncman, G. et al. A genetic variant at the fatty acid-binding protein aP2 locus reduces the risk for hypertriglyceridemia, type 2 diabetes, and cardiovascular disease. Proc. Natl Acad. Sci. USA 103, 6970–6975 (2006).
Tan, N. S. et al. Selective cooperation between fatty acid binding proteins and peroxisome proliferator-activated receptors in regulating transcription. Mol. Cell. Biol. 22, 5114–5127 (2002).
Ayers, S. D., Nedrow, K. L., Gillilan, R. E. & Noy, N. Continuous nucleocytoplasmic shuttling underlies transcriptional activation of PPARγ by FABP4. Biochemistry 46, 6744–6752 (2007).
Schug, T. T., Berry, D. C., Shaw, N. S., Travis, S. N. & Noy, N. Opposing effects of retinoic acid on cell growth result from alternate activation of two different nuclear receptors. Cell 129, 723–733 (2007).
Makowski, L., Brittingham, K. C., Reynolds, J. M., Suttles, J. & Hotamisligil, G. S. The fatty acid-binding protein, aP2, coordinates macrophage cholesterol trafficking and inflammatory activity. Macrophage expression of aP2 impacts peroxisome proliferator-activated receptor γ and IκB kinase activities. J. Biol. Chem. 280, 12888–12895 (2005).
Knutson, M. D. Steap proteins: implications for iron and copper metabolism. Nutr. Rev. 65, 335–340 (2007).
Arner, P. et al. Expression of six transmembrane protein of prostate 2 in human adipose tissue associates with adiposity and insulin resistance. J. Clin. Endocrinol. Metab. 93, 2249–2254 (2008).
Bloom, J. D. Glucose intolerance in pulmonary tuberculosis. Am. Rev. Respir. Dis. 100, 38–41 (1969).
Neuschwander-Tetri, B. A. Hepatitis C virus-induced insulin resistance: not all genotypes are the same. Gastroenterology 134, 619–622 (2008).
Manel, N. et al. The ubiquitous glucose transporter GLUT-1 is a receptor for HTLV. Cell 115, 449–459 (2003).
Calder, P. C., Dimitriadis, G. & Newsholme, P. Glucose metabolism in lymphoid and inflammatory cells and tissues. Curr. Opin. Clin. Nutr. Metab. Care 10, 531–540 (2007).
Roy, C. R., Salcedo, S. P. & Gorvel, J. P. Pathogen–endoplasmic-reticulum interactions: in through the out door. Nature Rev. Immunol. 6, 136–147 (2006).
Adams, D. O. The structure of mononuclear phagocytes differentiating in vivo. I. Sequential fine and histologic studies of the effect of Bacillus Calmette-Guerin (BCG). Am. J. Pathol. 76, 17–48 (1974).
Russell, D. G. Phagosomes, fatty acids and tuberculosis. Nature Cell Biol. 5, 776–778 (2003).
Beatty, W. L. et al. Trafficking and release of mycobacterial lipids from infected macrophages. Traffic 1, 235–247 (2000).
Shiratori, Y., Okwu, A. K. & Tabas, I. Free cholesterol loading of macrophages stimulates phosphatidylcholine biosynthesis and up-regulation of CTP: phosphocholine cytidylyltransferase. J. Biol. Chem. 269, 11337–11348 (1994).
Elased, K. M. et al. Reversal of type 2 diabetes in mice by products of malaria parasites. II. Role of inositol phosphoglycans (IPGs). Mol. Genet. Metab. 73, 248–258 (2001).
Schneider, J. G. et al. ATM-dependent suppression of stress signaling reduces vascular disease in metabolic syndrome. Cell. Metab. 4, 377–389 (2006).
Clemens, M. J. Translational control in virus-infected cells: models for cellular stress responses. Semin. Cell Dev. Biol. 16, 13–20 (2005).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Backhed, F. et al. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl Acad. Sci. USA 101, 15718–15723 (2004). This study examines the potential effect of microbial flora on energy stores.
Ley, R. E. et al. Obesity alters gut microbial ecology. Proc. Natl Acad. Sci. USA 102, 11070–11075 (2005).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Wolowczuk, I. et al. Feeding our immune system: impact on metabolism. Clin. Dev. Immunol. 2008, 639–803 (2008).
Cho, H. J. et al. Induction of dendritic cell-like phenotype in macrophages during foam cell formation. Physiol. Genomics 29, 149–160 (2007).
Ross, S. E. et al. Microarray analyses during adipogenesis: understanding the effects of Wnt signaling on adipogenesis and the roles of liver X receptor α in adipocyte metabolism. Mol. Cell. Biol. 22, 5989–5999 (2002).
Zhang, X. et al. Hypothalamic IKKβ/ NFκB and ER stress link overnutrition to energy imbalance and obesity. Cell 135, 61–73 (2008).
Yang, L. & Hotamisligil, G. S. Stressing the brain, fattening the body. Cell 135, 20–22 (2008).
Sinclair, L. V. et al. Phospatidylinositol-3-OH kinase and nutrients sensing mTOR pathways control T lymphocyte trafficking. Nature Immunol. 9, 513–521 (2008).
Acknowledgements
This work is supported in part by grants from National Institutes of Health (DK52539, DK64360, HL65405), and American Diabetes Association (I03RA41) to G.S.H. E.E. is supported by a Ruth Kirschstein National Research Award. We thank the members of the Hotamisligil laboratory for discussions, comments and research contributions that lead to maturation of this review. We are especially grateful to A. Onur for help with the figures, S. Hummasti for discussion and editing, H. Xu for assistance with analysis of microarray data and R. Foote for technical assistance with the manuscript. We regret that we could not cite or review the scientific contributions of many others owing to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr. Hotamisligil is the founder of Syndexa Pharmaceuticals and is on its scientific advisory board. He is also on the scientific advisory board of Lipomics, Inc. Dr. Hotamisligil receives research funding from Syndexa Pharmaceuticals.
Related links
Glossary
- Sepsis
-
A systemic response to severe bacterial infections that are generally caused by Gram-negative bacterial endotoxins. Sepsis induces a hyperactive and out-of-balance network of pro-inflammatory cytokines, affecting vascular permeability, cardiac function and metabolic balance, and ultimately leads to tissue necrosis, multiple-organ failure and death.
- Metabolic syndrome
-
A cluster of conditions, such as hypertension, hyperinsulinaemia, hypercholesteraemia and abdominal obesity, that occur together, increasing the risk of heart disease, stroke and diabetes.
- Adipose tissue
-
Connective tissue with a network of blood vessels in which fat is stored and the cells are distended by droplets of fat.
- Foam cells
-
Macrophages that localize to sites of early-stage inflammation in the vessel wall, which subsequently ingest oxidized low-density lipoprotein and slowly become overloaded with lipids. They are called foam cells because they have numerous cytoplasmic vesicles that contain cholesterol and other lipids. Foam cells eventually die and attract more macrophages, and further propagate inflammation in the vessel wall.
- Atherosclerosis
-
A chronic disorder of the arterial wall that is characterized by damage to the endothelium, which gradually induces deposits of cholesterol, cellular debris, calcium and other substances and ultimately triggers local inflammation. These deposits finally lead to plaque formation and arterial stiffness.
- Anabolic hormone
-
A hormone that is involved in the synthesis of macromolecules from simpler intermediates.
- Insulin resistance
-
The reduced sensitivity of the body's insulin-dependent processes (such as glucose uptake and lipolysis) to insulin. Insulin resistance is typical of type 2 diabetes but often occurs in the absence of diabetes.
- Mammalian target of rapamycin
-
(mTOR). A conserved serine/threonine protein kinase that regulates cell growth and metabolism, as well as cytokine and growth-factor expression, in response to environmental cues. mTOR receives stimulatory signals from RAS and phosphoinositide 3-kinase downstream of growth factors and nutrients, such as amino acids, glucose and oxygen.
- Peroxisome-proliferator-activated receptors
-
Nuclear receptors that participate in the regulation of cellular metabolism and differentiation.
- Unfolded-protein response
-
An adaptive response that increases the ability of the endoplasmic reticulum to fold and translocate proteins, decreases the synthesis of proteins, coordinates stress and antioxidant responses, and can result in the arrest of the cell cycle and apoptosis.
- Chaperone protein
-
A protein that assists the folding of newly synthesized proteins into a particular three-dimensional conformation by binding and stabilizing folding intermediates.
- Acute-phase proteins
-
A group of proteins, including Creactive protein, serum amyloid A, fibrinogen and α1-acid glycoprotein, that are secreted into the blood in increased or decreased quantities by hepatocytes in response to trauma, inflammation or disease. These proteins can be inhibitors or mediators of inflammatory processes.
- Plasma cells
-
Terminally differentiated quiescent B cells that develop from plasmablasts and are characterized by their capacity to secrete large amounts of antibodies.
- Atherosclerotic plaque
-
A lesion that consists of a fibrotic cap surrounding a lipid-rich core. The lesion is the site of inflammation, lipid accumulation and cell death.
- Leptin resistance
-
Reduced sensitivity to the effects of leptin, which is a hormone that is produced by fat cells and has a key role in regulating energy intake and energy expenditure.
- Rapamycin
-
An immunosuppressive drug that, in contrast to calcineurin inhibitors, does not prevent T-cell activation but blocks interleukin-2-mediated clonal expansion by blocking mTOR.
- Myeloid-derived suppressor cells
-
(MDSCs). A population of cells that consists of mature and immature myeloid cells. MDSCs are generated and/or activated during an inflammatory immune response and negatively affect T cells through direct interactions and secreted components, which leads to the impairment of T-cell function.
- Essential amino acid
-
An amino acid that cannot be synthesized by cells and must be obtained through the diet.
Rights and permissions
About this article
Cite this article
Hotamisligil, G., Erbay, E. Nutrient sensing and inflammation in metabolic diseases. Nat Rev Immunol 8, 923–934 (2008). https://doi.org/10.1038/nri2449
Issue Date:
DOI: https://doi.org/10.1038/nri2449
This article is cited by
-
Combined effect of adiposity and elevated inflammation on incident type 2 diabetes: a prospective cohort study
Cardiovascular Diabetology (2023)
-
D-mannose is a rapid inducer of ACSS2 to trigger rapid and long-lasting antidepressant responses through augmenting BDNF and TPH2 levels
Translational Psychiatry (2023)
-
IRE1α: from the function to the potential therapeutic target in atherosclerosis
Molecular and Cellular Biochemistry (2023)
-
Immunometabolic rewiring of tubular epithelial cells in kidney disease
Nature Reviews Nephrology (2022)
-
Interplay between Inflammaging, Frailty and Nutrition in Covid-19: Preventive and Adjuvant Treatment Perspectives
The Journal of nutrition, health and aging (2022)