Key Points
-
The cancer-immunoediting hypothesis emphasizes that extrinsic immune pressure either can block tumour growth, development and survival or can facilitate tumour outgrowth (by sculpting tumour immunogenicity or by inhibiting host-protective antitumour immune responses). The acquired capacity of developing tumours to escape immune control is a seventh hallmark of cancer.
-
The interferons (IFNs) — both type I IFNs (IFNα and IFNβ) and type II IFN (IFNγ) — have long been recognized as pivotal antiviral and antimicrobial molecules. However, considerable evidence from physiologically relevant tumour models reveals that these molecules also have important roles in protecting the host against tumour development.
-
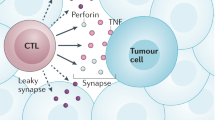
Endogenously produced IFNγ affects tumour cells directly during the antitumour immune response. IFNγ promotes tumour recognition by increasing the activity of the MHC class I pathway and by modulating the expression of ligands of NKG2D (natural-killer group 2, member D). IFNγ also affects the host immune system by polarization of T helper (TH) cells towards a TH1-cell phenotype, by induction of cytotoxic T lymphocytes and by inhibition of CD4+CD25+ regulatory T-cell development.
-
By contrast, endogenously produced type I IFNs do not act on tumour cells but, instead, on host haematopoietic cells, resulting in inhibition of tumour growth. Similar to tumours that are generated in mice that lack lymphocytes (recombination-activating gene 2 (RAG2)-deficient mice), tumours from mice that lack the IFNAR1 subunit of the type I IFN receptor are highly immunogenic and are therefore 'unedited'.
-
At present, type I IFNs are the only IFNs that are routinely used in a clinical setting for the treatment of cancer patients. Future work focused on controlling local delivery, identifying physiologically relevant IFN-producing cells and identifying IFN-dependent antitumour mechanisms will be vital to translate the well-established antitumour properties of the IFNs from animal modelsto the clinic.
Abstract
A clear picture of the dynamic relationship between the host immune system and cancer is emerging as the cells and molecules that participate in naturally occurring antitumour immune responses are being identified. The interferons (IFNs) — that is, the type I IFNs (IFNα and IFNβ) and type II IFN (IFNγ) — have emerged as central coordinators of tumour–immune-system interactions. Indeed, the decade-old finding that IFNγ has a pivotal role in promoting antitumour responses became the focus for a renewed interest in the largely abandoned concept of cancer immunosurveillance. More recently, type I IFNs have been found to have distinct functions in this process. In this Review, we discuss the roles of the IFNs, not only in cancer immunosurveillance but also in the broader process of cancer immunoediting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Burnet, F. M. Cancer: a biological approach. III. Viruses associated with neoplastic conditions. IV. Practical applications. Br. Med. J. 1, 841–847 (1957).
Thomas, L. in Cellular and Humoral Aspects of the Hypersensitive States (ed. Lawrence, H. S.) 529–532 (Hoeber–Harper, New York, 1959).
Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J. & Schreiber, R. D. Cancer immunoediting: from immunosurveillance to tumor escape. Nature Immunol. 3, 991–998 (2002). The first review of the concept of cancer immunoediting, with a focus on the historical evolution of the concept.
Dunn, G. P., Old, L. J. & Schreiber, R. D. The three Es of cancer immunoediting. Annu. Rev. Immunol. 22, 329–360 (2004). A comprehensive review of the concept of cancer immunoediting, including extensive discussion of evidence that cancer immunoediting also occurs in humans.
Dunn, G. P., Old, L. J. & Schreiber, R. D. The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21, 137–148 (2004).
Shankaran, V. et al. IFNγ and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410, 1107–1111 (2001). Reveals the dual function — both host protective and tumour sculpting — of cancer immunoediting.
Smyth, M. J., Dunn, G. P. & Schreiber, R. D. Cancer immunosurveillance and immunoediting: the roles of immunity in suppressing tumor development and shaping tumor immunogenicity. Adv. Immunol. 90, 1–50 (2006).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000). A seminal review that discusses the essential hallmarks of cancer.
Dunn, G. P. et al. A critical function for type I interferons in cancer immunoediting. Nature Immunol. 6, 722–729 (2005). Describes the host-protective and tumour-sculpting activities of type I IFNs in cancer immunoediting.
Takeda, K. et al. Critical role for tumor necrosis factor-related apoptosis-inducing ligand in immune surveillance against tumor development. J. Exp. Med. 195, 161–169 (2002). Reveals an overlapping role for TRAIL and IFNγ in cancer immunoediting.
Crowe, N. Y., Smyth, M. J. & Godfrey, D. I. A critical role for natural killer T cells in immunosurveillance of methylcholanthrene-induced sarcomas. J. Exp. Med. 196, 119–127 (2002). Demonstrates the importance of NKT cells that express a semi-invariant T-cell receptor containing Jα281 and their IFNγ-producing capacity in cancer immunoediting.
Decker, T., Muller, M. & Stockinger, S. The Yin and Yang of type I interferon activity in bacterial infection. Nature Rev. Immunol. 5, 675–687 (2005).
Berenson, L. S., Ota, N. & Murphy, K. M. Issues in T-helper 1 development — resolved and unresolved. Immunol. Rev. 202, 157–174 (2004).
Bach, E. A., Aguet, M. & Schreiber, R. D. The IFNγ receptor: a paradigm for cytokine receptor signaling. Annu. Rev. Immunol. 15, 563–591 (1997).
Boehm, U., Klamp, T., Groot, M. & Howard, J. C. Cellular responses to interferon-γ. Annu. Rev. Immunol. 15, 749–795 (1997).
Stark, G. R., Kerr, I. M., Williams, B. R., Silverman, R. H. & Schreiber, R. D. How cells respond to interferons. Annu. Rev. Biochem. 67, 227–264 (1998).
Dighe, A. S., Richards, E., Old, L. J. & Schreiber, R. D. Enhanced in vivo growth and resistance to rejection of tumor cells expressing dominant negative IFNγ receptors. Immunity 1, 447–456 (1994).
Kaplan, D. H. et al. Demonstration of an interferon γ-dependent tumor surveillance system in immunocompetent mice. Proc. Natl Acad. Sci. USA 95, 7556–7561 (1998). Original description of the importance of IFNγ in cancer immunosurveillance.
Street, S. E., Cretney, E. & Smyth, M. J. Perforin and interferon-γ activities independently control tumor initiation, growth, and metastasis. Blood 97, 192–197 (2001).
Street, S. E., Trapani, J. A., MacGregor, D. & Smyth, M. J. Suppression of lymphoma and epithelial malignancies effected by interferon-γ. J. Exp. Med. 196, 129–134 (2002). Reveals that the functions of IFNγ and perforin in surveillance of spontaneous malignancies are not completely overlapping.
Enzler, T. et al. Deficiencies of GM-CSF and interferon-γ link inflammation and cancer. J. Exp. Med. 197, 1213–1219 (2003).
Gao, Y. et al. γδ T cells provide an early source of interferon-γ in tumor immunity. J. Exp. Med. 198, 433–442 (2003). Shows the importance of IFNγ production by γδ T cells in cancer immunosurveillance.
Tripp, C. S., Wolf, S. F. & Unanue, E. R. Interleukin 12 and tumor necrosis factor α are costimulators of interferon-γ production by natural killer cells in severe combined immunodeficiency mice with listeriosis, and interleukin 10 is a physiologic antagonist. Proc. Natl Acad. Sci. USA 90, 3725–3729 (1993).
Yang, J., Murphy, T. L., Ouyang, W. & Murphy, K. M. Induction of interferon-γ production in TH1 CD4+ T cells: evidence for two distinct pathways for promoter activation. Eur. J. Immunol. 29, 548–555 (1999).
Nastala, C. L. et al. Recombinant IL-12 administration induces tumor regression in association with IFN-γ production. J. Immunol. 153, 1697–1706 (1994).
Noguchi, Y., Jungbluth, A., Richards, E. & Old, L. J. Effect of interleukin 12 on tumor induction by 3-methylcholanthrene. Proc. Natl Acad. Sci. USA 93, 11798–11801 (1996).
Smyth, M. J., Crowe, N. Y. & Godfrey, D. I. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int. Immunol. 13, 459–463 (2001).
Gresser, I., Bourali, C., Levy, J. P., Fontaine-Brouty-Boye, D. & Thomas, M. T. Increased survival in mice inoculated with tumor cells and treated with interferon preparations. Proc. Natl Acad. Sci. USA 63, 51–57 (1969).
Gresser, I., Maury, C. & Brouty-Boye, D. Mechanism of the antitumour effect of interferon in mice. Nature 239, 167–168 (1972).
Belardelli, F., Ferrantini, M., Proietti, E. & Kirkwood, J. M. Interferon-α in tumor immunity and immunotherapy. Cytokine Growth Factor Rev. 13, 119–134 (2002).
Reid, L. M. et al. Influence of anti-mouse interferon serum on the growth and metastasis of tumor cells persistently infected with virus and of human prostatic tumors in athymic nude mice. Proc. Natl Acad. Sci. USA 78, 1171–1175 (1981).
Gresser, I., Belardelli, F., Maury, C., Maunoury, M. T. & Tovey, M. G. Injection of mice with antibody to interferon enhances the growth of transplantable murine tumors. J. Exp. Med. 158, 2095–2107 (1983).
Picaud, S., Bardot, B., De Maeyer, E. & Seif, I. Enhanced tumor development in mice lacking a functional type I interferon receptor. J. Interferon Cytokine Res. 22, 457–462 (2002).
Sheehan, K. C. F. et al. Blocking monoclonal antibodies specific for mouse IFN-α/β receptor subunit 1 (IFNAR1) from mice immunized by in vivo hydrodynamic transfection. J. Interferon Cytokine Res. 26, 809–819 (2006).
Hahn, W. C. & Weinberg, R. A. Rules for making human tumor cells. N. Engl. J. Med. 347, 1593–1603 (2002).
Vogelstein, B. & Kinzler, K. W. Cancer genes and the pathways they control. Nature Med. 10, 789–799 (2004).
Wheelock, E. F., Weinhold, K. J. & Levich, J. The tumor dormant state. Adv. Cancer Res. 34, 107–140 (1981).
Uhr, J. W., Tucker, T., May, R. D., Siu, H. & Vitetta, E. S. Cancer dormancy: studies of the murine BCL1 lymphoma. Cancer Res. 51, 5045s–5053s (1991).
Smyth, M. J. et al. NKG2D function protects the host from tumor initiation. J. Exp. Med. 202, 583–588 (2005). Provides evidence that supports the idea of perforin-dependent cancer immunoediting of NKG2D ligands.
Smyth, M. J. et al. Differential tumor surveillance by natural killer (NK) and NKT cells. J. Exp. Med. 191, 661–668 (2000).
Willimsky, G. & Blankenstein, T. Sporadic immunogenic tumours avoid destruction by inducing T-cell tolerance. Nature 437, 141–146 (2005).
Dunn, G. P., Sheehan, K. C., Old, L. J. & Schreiber, R. D. IFN unresponsiveness in LNCaP cells due to the lack of JAK1 gene expression. Cancer Res. 65, 3447–3453 (2005).
Sanda, M. G. et al. Molecular characterization of defective antigen processing in human prostate cancer. J. Natl Cancer Inst. 87, 280–285 (1995).
Wong, L. H. et al. Interferon-resistant human melanoma cells are deficient in ISGF3 components, STAT1, STAT2, and p48-ISGF3γ. J. Biol. Chem. 272, 28779–28785 (1997).
Xi, S. et al. Decreased STAT1 expression by promoter methylation in squamous cell carcinogenesis. J. Natl Cancer Inst. 98, 181–189 (2006).
Wellbrock, C. et al. STAT5 contributes to interferon resistance of melanoma cells. Curr. Biol. 15, 1629–1639 (2005).
Seliger, B., Maeurer, M. J. & Ferrone, S. Antigen-processing machinery breakdown and tumor growth. Immunol. Today 21, 455–464 (2000).
Algarra, I., Cabrera, T. & Garrido, F. The HLA crossroad in tumor immunology. Hum. Immunol. 61, 65–73 (2000).
Marincola, F. M., Jaffee, E. M., Hicklin, D. J. & Ferrone, S. Escape of human solid tumors from T-cell recognition: molecular mechanisms and functional significance. Adv. Immunol. 74, 181–273 (2000).
Li, Z. et al. Expression of SOCS-1, suppressor of cytokine signalling-1, in human melanoma. J. Invest. Dermatol. 123, 737–745 (2004).
Dighe, A. S., Farrar, M. A. & Schreiber, R. D. Inhibition of cellular responsiveness to interferon-γ (IFNγ) induced by overexpression of inactive forms of the IFNγ receptor. J. Biol. Chem. 268, 10645–10653 (1993).
Coughlin, C. M. et al. Tumor cell responses to IFNγ affect tumorigenicity and response to IL-12 therapy and antiangiogenesis. Immunity 9, 25–34 (1998).
Beatty, G. & Paterson, Y. IFN-γ-dependent inhibition of tumor angiogenesis by tumor-infiltrating CD4+ T cells requires tumor responsiveness to IFN-γ. J. Immunol. 166, 2276–2282 (2001).
Bui, J. D., Carayannopoulos, L. N., Lanier, L. L., Yokoyama, W. M. & Schreiber, R. D. IFN-dependent down-regulation of the NKG2D ligand H60 on tumors. J. Immunol. 176, 905–913 (2006).
Chin, Y. E. et al. Cell growth arrest and induction of cyclin-dependent kinase inhibitor p21WAF1/CIP1 mediated by STAT1. Science 272, 719–722 (1996).
Bromberg, J. F., Horvath, C. M., Wen, Z., Schreiber, R. D. & Darnell, J. E. Jr. Transcriptionally active Stat1 is required for the antiproliferative effects of both interferon α and interferon γ. Proc. Natl Acad. Sci. USA 93, 7673–7678 (1996).
Chin, Y. E., Kitagawa, M., Kuida, K., Flavell, R. A. & Fu, X. Y. Activation of the STAT signaling pathway can cause expression of caspase 1 and apoptosis. Mol. Cell. Biol. 17, 5328–5337 (1997).
Xu, X., Fu, X. Y., Plate, J. & Chong, A. S. IFN-γ induces cell growth inhibition by Fas-mediated apoptosis: requirement of STAT1 protein for up-regulation of Fas and FasL expression. Cancer Res. 58, 2832–2837 (1998).
Meng, R. D. & El-Deiry, W. S. p53-independent upregulation of KILLER/DR5 TRAIL receptor expression by glucocorticoids and interferon-γ. Exp. Cell Res. 262, 154–169 (2001).
Luster, A. D. & Leder, P. IP-10, a C-X-C-chemokine, elicits a potent thymus-dependent antitumor response in vivo. J. Exp. Med. 178, 1057–1065 (1993).
Mumberg, D. et al. CD4+ T cells eliminate MHC class II-negative cancer cells in vivo by indirect effects of IFN-γ. Proc. Natl Acad. Sci. USA 96, 8633–8638 (1999).
Fallarino, F. & Gajewski, T. F. Differentiation of antitumor CTL in vivo requires host expression of Stat1. J. Immunol. 163, 4109–4113 (1999).
Kacha, A. K., Fallarino, F., Markiewicz, M. A. & Gajewski, T. F. Spontaneous rejection of poorly immunogenic P1.HTR tumors by Stat6-deficient mice. J. Immunol. 165, 6024–6028 (2000).
Nishikawa, H. et al. IFN-γ controls the generation/activation of CD4+CD25+ regulatory T cells in antitumor immune response. J. Immunol. 175, 4433–4440 (2005). Illustrates the important influence of IFNγ on T Reg -cell-dependent suppression of cancer immunosurveillance.
Nishikawa, H. et al. CD4+ CD25+ T cells responding to serologically defined autoantigens suppress antitumor immune responses. Proc. Natl Acad. Sci. USA 100, 10902–10906 (2003).
de Visser, K. E., Eichten, A. & Coussens, L. M. Paradoxical roles of the immune system during cancer development. Nature Rev. Cancer 6, 24–37 (2006).
Kawanishi, S., Hiraku, Y., Pinlaor, S. & Ma, N. Oxidative and nitrative DNA damage in animals and patients with inflammatory diseases in relation to inflammation-related carcinogenesis. Biol. Chem. 387, 365–372 (2006).
Langowski, J. L. et al. IL-23 promotes tumour incidence and growth. Nature 442, 461–465 (2006). Details the importance of the IL-12 and IL-23 cytokine milieu in determining the balance between the tumour-promoting and tumour-suppressive actions of the antitumour immune response. And begins to synthesize, into one process, the seemingly contradictory processes of tumour-promoting inflammatory reactions and host-protective immune responses.
Moore, R. J. et al. Mice deficient in tumor necrosis factor-α are resistant to skin carcinogenesis. Nature Med. 5, 828–831 (1999).
Hanada, T. et al. IFNγ-dependent, spontaneous development of colorectal carcinomas in SOCS1-deficient mice. J. Exp. Med. 203, 1391–1397 (2006).
Brassard, D. L., Grace, M. J. & Bordens, R. W. Interferon-α as an immunotherapeutic protein. J. Leukoc. Biol. 71, 565–581 (2002).
Belardelli, F., Gresser, I., Maury, C. & Maunoury, M. T. Antitumor effects of interferon in mice injected with interferon-sensitive and interferon-resistant Friend leukemia cells. I. Int. J. Cancer 30, 813–820 (1982).
Lesinski, G. B. et al. The antitumor effects of IFN-α are abrogated in a STAT1-deficient mouse. J. Clin. Invest. 112, 170–180 (2003).
Bogdan, C., Mattner, J. & Schleicher, U. The role of type I interferons in non-viral infections. Immunol. Rev. 202, 33–48 (2004).
Kerbel, R. & Folkman, J. Clinical translation of angiogenesis inhibitors. Nature Rev. Cancer 2, 727–739 (2002).
Takaoka, A. et al. Integration of interferon-α/β signalling to p53 responses in tumour suppression and antiviral defence. Nature 424, 516–523 (2003).
Silverman, R. H. Implications for RNase L in prostate cancer biology. Biochemistry 42, 1805–1812 (2003).
Carpten, J. et al. Germline mutations in the ribonuclease L gene in families showing linkage with HPC1. Nature Genet. 30, 181–184 (2002).
Casey, G. et al. RNASEL Arg462Gln variant is implicated in up to 13% of prostate cancer cases. Nature Genet. 32, 581–583 (2002).
Urisman, A. et al. Identification of a novel γ retrovirus in prostate tumors of patients homozygous for R462Q RNASEL variant. PLoS Pathog. 2, e25 (2006).
Kirkwood, J. M. et al. Interferon α-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J. Clin. Oncol. 14, 7–17 (1996).
Gogas, H. et al. Prognostic significance of autoimmunity during treatment of melanoma with interferon. N. Engl. J. Med. 354, 709–718 (2006).
Gleave, M. E. et al. Interferon γ-1b compared with placebo in metastatic renal-cell carcinoma. Canadian Urologic Oncology Group. N. Engl. J. Med. 338, 1265–1271 (1998).
Wiesenfeld, M. et al. Controlled clinical trial of interferon-γ as postoperative surgical adjuvant therapy for colon cancer. J. Clin. Oncol. 13, 2324–2329 (1995).
Jett, J. R. et al. Phase III trial of recombinant interferon γ in complete responders with small-cell lung cancer. J. Clin. Oncol. 12, 2321–2326 (1994).
Windbichler, G. H. et al. Interferon-γ in the first-line therapy of ovarian cancer: a randomized phase III trial. Br. J. Cancer 82, 1138–1144 (2000).
Giannopoulos, A. et al. The immunomodulating effect of interferon-γ intravesical instillations in preventing bladder cancer recurrence. Clin. Cancer Res. 9, 5550–5558 (2003).
Lienard, D., Eggermont, A. M., Kroon, B. B., Schraffordt Koops, H. & Lejeune, F. J. Isolated limb perfusion in primary and recurrent melanoma: indications and results. Semin. Surg. Oncol. 14, 202–209 (1998).
Gasser, S. & Raulet, D. H. The DNA damage response arouses the immune system. Cancer Res. 66, 3959–3962 (2006).
Zhang, J. J. et al. Two contact regions between Stat1 and CBP/p300 in interferon γ signaling. Proc. Natl Acad. Sci. USA 93, 15092–15096 (1996).
DaFonseca, C. J., Shu, F. & Zhang, J. J. Identification of two residues in MCM5 critical for the assembly of MCM complexes and Stat1-mediated transcription activation in response to IFN-γ. Proc. Natl Acad. Sci. USA 98, 3034–3039 (2001).
Schreiber, G. H. & Schreiber, R. D. in The Cytokine Handbook 4th edn (eds Thomson, A. & Lotze, M.) 567–601 (Academic, London, 2003).
Alexander, W. S. et al. SOCS1 is a critical inhibitor of interferon γ signaling and prevents the potentially fatal neonatal actions of this cytokine. Cell 98, 597–608 (1999).
Marine, J. C. et al. SOCS1 deficiency causes a lymphocyte-dependent perinatal lethality. Cell 98, 609–616 (1999).
Liu, B. et al. PIAS1 selectively inhibits interferon-inducible genes and is important in innate immunity. Nature Immunol. 5, 891–898 (2004).
Platanias, L. C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nature Rev. Immunol. 5, 375–386 (2005).
Pestka, S., Krause, C. D. & Walter, M. R. Interferons, interferon-like cytokines, and their receptors. Immunol. Rev. 202, 8–32 (2004).
Karaghiosoff, M. et al. Partial impairment of cytokine responses in Tyk2-deficient mice. Immunity 13, 549–560 (2000).
Shimoda, K. et al. Tyk2 plays a restricted role in IFNα signaling, although it is required for IL-12-mediated T cell function. Immunity 13, 561–571 (2000).
Girardi, M. et al. Regulation of cutaneous malignancy by γδ T cells. Science 294, 605–609 (2001).
Girardi, M. et al. The distinct contributions of murine T cell receptor (TCR)γδ+ and TCRαβ+ T cells to different stages of chemically induced skin cancer. J. Exp. Med. 198, 747–755 (2003).
Smyth, M. J. et al. Perforin-mediated cytotoxicity is critical for surveillance of spontaneous lymphoma. J. Exp. Med. 192, 755–760 (2000).
Hayashi, T. & Faustman, D. L. Development of spontaneous uterine tumors in low molecular mass polypeptide-2 knockout mice. Cancer Res. 62, 24–27 (2002).
Nishikawa, H. et al. Accelerated chemically induced tumor development mediated by CD4+CD25+ regulatory T cells in wild-type hosts. Proc. Natl Acad. Sci. USA 102, 9253–9257 (2005).
Mitra-Kaushik, S., Harding, J., Hess, J., Schreiber, R. & Ratner, L. Enhanced tumorigenesis in HTLV-1 tax-transgenic mice deficient in interferon-γ. Blood 104, 3305–3311 (2004).
Street, S. E. et al. Innate immune surveillance of spontaneous B cell lymphomas by natural killer cells and γδ T cells. J. Exp. Med. 199, 879–884 (2004).
van den Broek, M. F. et al. Decreased tumor surveillance in perforin-deficient mice. J. Exp. Med. 184, 1781–1790 (1996).
Cretney, E. et al. Increased susceptibility to tumor initiation and metastasis in TNF-related apoptosis-inducing ligand-deficient mice. J. Immunol. 168, 1356–1361 (2002).
Zerafa, N. et al. TRAIL deficiency accelerates hematological malignancies. J. Immunol. 175, 5586–5590 (2005).
Hayakawa, Y., Rovero, S., Forni, G. & Smyth, M. J. α-Galactosylceramide (KRN7000) suppression of chemical- and oncogene-dependent carcinogenesis. Proc. Natl Acad. Sci. USA 100, 9464–9469 (2003).
Barchet, W., Blasius, A., Cella, M. & Colonna, M. Plasmacytoid dendritic cells: in search of their niche in immune responses. Immunol. Res. 32, 75–84 (2005).
Ikeda, H., Old, L. J. & Schreiber, R. D. The roles of IFNγ in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev. 13, 95–109 (2002).
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964 (2006).
Acknowledgements
The authors are grateful to the following individuals: L. J. Old and M. J. Smyth for past and continuing collaborations and for critical comment during the preparation of this Review; G. P. Linette for helpful comment on the clinical use of IFNs in cancer immunotherapy; and all past and present members of the Schreiber laboratory for their essential contributions to the development of the cancer-immunoediting hypothesis. This work was supported by grants from the National Cancer Institute (United States), the Ludwig Institute for Cancer Research (United States) and the Cancer Research Institute (United States).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Glossary
- Transformation
-
The process of conversion of a normal cell into a cancer cell.
- Cancer immunosurveillance
-
Immunological protection of the host against the development of cancer, resulting from immune effector functions stimulated by immune recognition of either stress ligands or antigens expressed on transformed cells.
- Hallmarks of cancer
-
A specific set of features that must be acquired by a malignant cell. These features are self-sufficiency in growth signals, insensitivity to growth-inhibitory signals, evasion of apoptotic cell death, limitless replicative potential, sustained angiogenesis, tissue invasion and evasion of the antitumour immune response.
- Primary tumorigenesis
-
Transformation and tumour formation that occurs entirely in the original host, in contrast to tumour formation in a host owing to transplantation of cells that were transformed in another host or in vitro.
- γδ T cell
-
T cells express either a T-cell receptor (TCR) composed of an α-subunit and a β-subunit (αβ-TCR) or a TCR composed of a γ-subunit and a δ-subunit (γδ-TCR). T cells that express αβ-TCRs mainly recognize antigenic peptides bound to conventional MHC class I or class II molecules. T cells that express γδ-TCRs are less abundant, and the ligands for these receptors are less well characterized.
- Natural killer T cell
-
(NKT cell). A T cell that expresses both natural killer (NK)-cell receptors and an αβ T-cell receptor. NKT cells are characterized by cytolytic activity and by rapid production of cytokines (including interferon-γ and interleukin-4), and they might regulate the function of other T cells.
- Epigenetic
-
Any heritable influence on the function of a chromosome or gene that is not caused by a change in DNA sequence.
- Regressor tumour
-
A cell line derived from a primary tumour that is rejected when transplanted into naive syngeneic hosts that are immunocompetent but grows progressively in syngeneic hosts that are immunocompromised.
- Angiostatic molecule
-
A molecule that blocks or opposes the growth of blood vessels and the formation of new blood vessels.
Rights and permissions
About this article
Cite this article
Dunn, G., Koebel, C. & Schreiber, R. Interferons, immunity and cancer immunoediting. Nat Rev Immunol 6, 836–848 (2006). https://doi.org/10.1038/nri1961
Issue Date:
DOI: https://doi.org/10.1038/nri1961
This article is cited by
-
Immunotherapy for Brain Tumors: Where We Have Been, and Where Do We Go From Here?
Current Treatment Options in Oncology (2024)
-
BRAF D594A mutation defines a unique biological and immuno-modulatory subgroup associated with functional CD8+ T cell infiltration in colorectal cancer
Journal of Translational Medicine (2023)
-
The crucial regulatory role of type I interferon in inflammatory diseases
Cell & Bioscience (2023)
-
The diagnostic/prognostic roles and biological function of the IFIT family members in acute myeloid leukemia
BMC Medical Genomics (2023)
-
Novel role of immune-related non-coding RNAs as potential biomarkers regulating tumour immunoresponse via MICA/NKG2D pathway
Biomarker Research (2023)