Key Points
-
For many years, it has been understood that commensal bacteria contribute to the development of the gut-associated lymphoid tissue. Recently, it has been shown that certain systemic immune functions are also impaired in the absence of commensal bacteria.
-
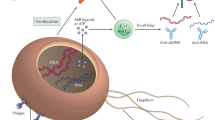
Bacteroides fragilis, a commensal bacterium that is found in all mammals, synthesizes zwitterionic polysaccharides (ZPSs), which are unique in structure and function compared with all other known bacterial polysaccharides. Unlike other polysaccharides, ZPSs activate CD4+ T cells specifically and are therefore T-cell-dependent antigens.
-
ZPSs contain both a positively and a negatively charged motif in each repeating unit. This property is required for the interactions of ZPSs with the immune system.
-
The unique chemical structure of ZPSs mediates their uptake by professional antigen-presenting cells (APCs), directs their processing in the endosomes of APCs and contributes to their presentation to CD4+ T cells by MHC class II molecules. Therefore, ZPSs are non-peptide antigens that bind MHC class II molecules and induce immune responses.
-
ZPSs protect laboratory animals from T-cell-mediated pathologies such as abscess formation and fibrosis, through inducing production of the anti-inflammatory molecule interleukin-10.
-
Colonization of germ-free animals with B. fragilis corrects several immune defects that are found in the absence of bacteria, including a reduction in the proportion of the total splenic lymphocyte population that consists of CD4+ T cells, incomplete development of the spleen and imbalanced production of T helper 1 (TH1) and TH2 cytokines. ZPSs are necessary and sufficient for correcting these defects.
-
The 'hygiene hypothesis' states that recent societal changes have altered our natural exposure to bacteria, with consequent increases in the rates of certain immune and atopic disorders. ZPSs might be the missing link in our understanding of the molecular basis of the increase in incidence of allergic disorders, such as inflammatory bowel disease and asthma.
Abstract
This article explores the fascinating relationship between the mammalian immune system and the bacteria that are present in the mammalian gut. Every human is an ecosystem that hosts 1013–1014 bacteria. We review the evidence that immunomodulatory molecules produced by commensal bacteria in the gut have a beneficial influence on the development of certain immune responses, through eliciting the clonal expansion of CD4+ T-cell populations. This process seems to contribute to the overall health of the host by offering protection against various diseases and might provide supporting evidence at a molecular level for the 'hygiene hypothesis' of allergic immune disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Merrell, D. S. & Falkow, S. Frontal and stealth attack strategies in microbial pathogenesis. Nature 430, 250–256 (2004).
Monack, D. M., Mueller, A. & Falkow, S. Persistent bacterial infections: the interface of the pathogen and the host immune system. Nature Rev. Microbiol. 2, 747–765 (2004).
Hooper, L. V., Bry, L., Falk, P. G. & Gordon, J. I. Host–microbial symbiosis in the mammalian intestine: exploring an internal ecosystem. Bioessays 20, 336–343 (1998).
Hooper, L. V., Falk, P. G. & Gordon, J. I. Analyzing the molecular foundations of commensalism in the mouse intestine. Curr. Opin. Microbiol. 3, 79–85 (2000).
Hooper, L. V. et al. Molecular analysis of commensal host–microbial relationships in the intestine. Science 291, 881–884 (2001).
Ley, R. E., Peterson, D. A. & Gordon, J. I. Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell 124, 837–848 (2006). A fascinating and informative review of the evolution of host–microorganism relationships and their influence on many aspects of the physiology of both species.
Hughes, K. L. Recent knowledge of the strict anaerobes of the gut. Aust. Vet. J. 48, 508–514 (1972).
Mueller, C. & Macpherson, A. J. Layers of mutualism with commensal bacteria protect us from intestinal inflammation. Gut 55, 276–284 (2006).
Hoogkamp-Korstanje, J. A., Lindner, J. G., Marcelis, J. H., den Daas-Slagt, H. & de Vos, N. M. Composition and ecology of the human intestinal flora. Antonie Van Leeuwenhoek 45, 35–40 (1979).
Sartor, R. B. Targeting enteric bacteria in treatment of inflammatory bowel diseases: why, how, and when. Curr. Opin. Gastroenterol. 19, 358–365 (2003).
Guarner, F. & Malagelada, J. R. Gut flora in health and disease. Lancet 361, 512–519 (2003).
Rastall, R. A. Bacteria in the gut: friends and foes and how to alter the balance. J. Nutr. 134, 2022S–2026S (2004).
Saier, M. H. Jr. & Mansour, N. M. Probiotics and prebiotics in human health. J. Mol. Microbiol. Biotechnol. 10, 22–25 (2005).
Sartor, R. B. Probiotic therapy of intestinal inflammation and infections. Curr. Opin. Gastroenterol. 21, 44–50 (2005).
O'Hara, A. M. & Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 7, 688–693 (2006).
Mazmanian, S. K., Liu, C. H., Tzianabos, A. O. & Kasper, D. L. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118 (2005). A report on the outcome of colonization of mammals by B. fragilis and the role of PSA in immune-system maturation. This was the first study to identify a molecular component of gut bacteria involved in mediating communication with a host during beneficial mutualism.
Macpherson, A. J. & Harris, N. L. Interactions between commensal intestinal bacteria and the immune system. Nature Rev. Immunol. 4, 478–485 (2004).
Sonnenburg, E. D. et al. A hybrid two-component system protein of a prominent human gut symbiont couples glycan sensing in vivo to carbohydrate metabolism. Proc. Natl Acad. Sci. USA 103, 8834–8839 (2006).
Gordon, J. I. A genomic view of our symbiosis with members of the gut microbiota. J. Pediatr. Gastroenterol. Nutr. 40 (Suppl. 1), 28 (2005).
Hooper, L. V., Stappenbeck, T. S., Hong, C. V. & Gordon, J. I. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nature Immunol. 4, 269–273 (2003). The first study to show that, during colonization, commensal bacteria induce the host to express antimicrobial proteins, identifying a mechanism by which commensal bacteria in the gut influence the microbial ecology of the gut and shape innate immunity.
Strachan, D. P. Hay fever, hygiene, and household size. BMJ 299, 1259–1260 (1989). The seminal report that proposed a link between increased incidence of specific human immune disorders and environmental bacteria. This article introduced the hygiene hypothesis.
Noverr, M. C. & Huffnagle, G. B. The 'microflora hypothesis' of allergic diseases. Clin. Exp. Allergy 35, 1511–1520 (2005).
Nikaido, H. Structure and functions of the cell envelope of Gram-negative bacteria. Rev. Infect. Dis. 10, S279–S281 (1988).
Upreti, R. K., Kumar, M. & Shankar, V. Bacterial glycoproteins: functions, biosynthesis and applications. Proteomics 3, 363–379 (2003).
Coutinho, A., Moller, G., Anderson, J. & Bullock, W. W. In vitro activation of mouse lymphocytes in serum-free medium: effect of T and B cell mitogens on proliferation and antibody synthesis. Eur. J. Immunol. 3, 299–306 (1973).
Coutinho, A. & Moller, G. B cell mitogenic properties of thymus-independent antigens. Nature New Biol. 245, 12–14 (1973).
McGhee, J. R. et al. Mucosal immunoregulation: environmental lipopolysaccharide and GALT T lymphocytes regulate the IgA response. Microbiol. Immunol. 28, 261–280 (1984).
Barrett, D. J. Human immune responses to polysaccharide antigens: an analysis of bacterial polysaccharide vaccines in infants. Adv. Pediatr. 32, 139–158 (1985).
Sood, R. K. & Fattom, A. Capsular polysaccharide–protein conjugate vaccines and intravenous immunoglobulins. Expert Opin. Investig. Drugs 7, 333–347 (1998).
Lee, C. J., Lee, L. H., Lu, C. S. & Wu, A. Bacterial polysaccharides as vaccines — immunity and chemical characterization. Adv. Exp. Med. Biol. 491, 453–471 (2001).
Briles, D. E., Hollingshead, S. K., Nabors, G. S., Paton, J. C. & Brooks-Walter, A. The potential for using protein vaccines to protect against otitis media caused by Streptococcus pneumoniae. Vaccine 19 (Suppl. 1), 87–95 (2000).
Kelly, D. F., Moxon, E. R. & Pollard, A. J. Haemophilus influenzae type b conjugate vaccines. Immunology 113, 163–174 (2004).
Lesinski, G. B. & Westerink, M. A. Vaccines against polysaccharide antigens. Curr. Drug Targets Infect. Disord. 1, 325–334 (2001).
Weintraub, A. Immunology of bacterial polysaccharide antigens. Carbohydr. Res. 338, 2539–2547 (2003).
Lesinski, G. B. & Westerink, M. A. Novel vaccine strategies to T-independent antigens. J. Microbiol. Methods 47, 135–149 (2001).
Onderdonk, A. B., Kasper, D. L., Cisneros, R. L. & Bartlett, J. G. The capsular polysaccharide of Bacteroides fragilis as a virulence factor: comparison of the pathogenic potential of encapsulated and unencapsulated strains. J. Infect. Dis. 136, 82–89 (1977).
Onderdonk, A. B. et al. Experimental animal models for anaerobic infections. Rev. Infect. Dis. 1, 291–301 (1979).
Polk, B. F. & Kasper, D. L. Bacteroides fragilis subspecies in clinical isolates. Ann. Intern. Med. 86, 569–571 (1977).
Kasper, D. L. et al. Isolation and identification of encapsulated strains of Bacteroides fragilis. J. Infect. Dis. 136, 75–81 (1977).
Tzianabos, A. O. & Kasper, D. L. Role of T cells in abscess formation. Curr. Opin. Microbiol. 5, 92–96 (2002).
Kasper, D. L. The polysaccharide capsule of Bacteroides fragilis subspecies fragilis: immunochemical and morphologic definition. J. Infect. Dis. 133, 79–87 (1976).
Zaleznik, D. F. & Kasper, D. L. The role of anaerobic bacteria in abscess formation. Annu. Rev. Med. 33, 217–229 (1982).
Lindberg, A. A., Weintraub, A., Kasper, D. L. & Lonngren, J. Virulence factors in infections with Bacteroides fragilis: isolation and characterization of capsular polysaccharide and lipopolysaccharide. Scand. J. Infect. Dis. Suppl. 35, 45–52 (1982).
Bartlett, J. G., Onderdonk, A. B., Louie, T., Kasper, D. L. & Gorbach, S. L. A review. Lessons from an animal model of intra-abdominal sepsis. Arch. Surg. 113, 853–857 (1978).
Kasper, D. L., Onderdonk, A. B., Crabb, J. & Bartlett, J. G. Protective efficacy of immunization with capsular antigen against experimental infection with Bacteroides fragilis. J. Infect. Dis. 140, 724–731 (1979).
Onderdonk, A. B., Markham, R. B., Zaleznik, D. F., Cisneros, R. L. & Kasper, D. L. Evidence for T cell-dependent immunity to Bacteroides fragilis in an intraabdominal abscess model. J. Clin. Invest. 69, 9–16 (1982).
Kasper, D. L. & Onderdonk, A. B. Infection with Bacteroides fragilis: pathogenesis and immunoprophylaxis in an animal model. Scand. J. Infect. Dis. Suppl. 31, 28–33 (1982).
Shapiro, M. E., Onderdonk, A. B., Kasper, D. L. & Finberg, R. W. Cellular immunity to Bacteroides fragilis capsular polysaccharide. J. Exp. Med. 155, 1188–1197 (1982).
Shapiro, M. E., Onderdonk, A. B., Kasper, D. L. & Finberg, R. W. Immune T cells prevent Bacteroides fragilis abscesses. Curr. Surg. 40, 123–126 (1983).
Shapiro, M. E. et al. Cellular control of abscess formation: role of T cells in the regulation of abscesses formed in response to Bacteroides fragilis. J. Immunol. 137, 341–346 (1986).
Zaleznik, D. F., Finberg, R. W., Shapiro, M. E., Onderdonk, A. B. & Kasper, D. L. A soluble suppressor T cell factor protects against experimental intraabdominal abscesses. J. Clin. Invest. 75, 1023–1027 (1985).
Brubaker, J. O., Li, Q., Tzianabos, A. O., Kasper, D. L. & Finberg, R. W. Mitogenic activity of purified capsular polysaccharide A from Bacteroides fragilis: differential stimulatory effect on mouse and rat lymphocytes in vitro. J. Immunol. 162, 2235–2242 (1999).
Tzianabos, A. O. et al. T cells activated by zwitterionic molecules prevent abscesses induced by pathogenic bacteria. J. Biol. Chem. 275, 6733–6740 (2000).
Kalka-Moll, W. M. et al. Effect of molecular size on the ability of zwitterionic polysaccharides to stimulate cellular immunity. J. Immunol. 164, 719–724 (2000).
Ruiz-Perez, B. et al. Modulation of surgical fibrosis by microbial zwitterionic polysaccharides. Proc. Natl Acad. Sci. USA 102, 16753–16758 (2005). The first study to show that ZPSs induce production of IL-10 and that this process is required for protection against T-cell-mediated inflammation. Therefore, IL-10 was identified as the low-molecular-weight component that had been described two decades earlier as being responsible for protection against the formation of intra-abdominal abscesses.
Rouleau, M. et al. IL-10 transgenic mice present a defect in T cell development reminiscent of SCID patients. J. Immunol. 163, 1420–1427 (1999).
Groux, H. & Cottrez, F. The complex role of interleukin-10 in autoimmunity. J. Autoimmun. 20, 281–285 (2003).
Stephen, T. L. et al. Effect of B7-2 and CD40 signals from activated antigen-presenting cells on the ability of zwitterionic polysaccharides to induce T-cell stimulation. Infect. Immun. 73, 2184–2189 (2005).
Van Gool, S. W. et al. Blocking CD40–CD154 and CD80/CD86–CD28 interactions during primary allogeneic stimulation results in T cell anergy and high IL-10 production. Eur. J. Immunol. 29, 2367–2375 (1999).
Kronfeld, K., Abken, H. & Seliger, B. B7-1 and B7-2 act differentially in the induction of a T cell response: their impact for a HLA-matched and HLA-mismatched anti-tumor immunotherapy. Int. J. Cancer 117, 794–799 (2005).
Krinos, C. M. et al. Extensive surface diversity of a commensal microorganism by multiple DNA inversions. Nature 414, 555–558 (2001).
Kuwahara, T. et al. Genomic analysis of Bacteroides fragilis reveals extensive DNA inversions regulating cell surface adaptation. Proc. Natl Acad. Sci. USA 101, 14919–14924 (2004).
Cerdeno-Tarraga, A. M. et al. Extensive DNA inversions in the B. fragilis genome control variable gene expression. Science 307, 1463–1465 (2005).
Baumann, H., Tzianabos, A. O., Brisson, J. R., Kasper, D. L. & Jennings, H. J. Structural elucidation of two capsular polysaccharides from one strain of Bacteroides fragilis using high-resolution NMR spectroscopy. Biochemistry 31, 4081–4089 (1992).
Tzianabos, A. O., Onderdonk, A. B., Rosner, B., Cisneros, R. L. & Kasper, D. L. Structural features of polysaccharides that induce intra-abdominal abscesses. Science 262, 416–419 (1993). The first paper to characterize the structure–function relationship between ZPSs and intra-abdominal abscesses, showing that the zwitterionic properties of ZPSs were required for interaction with the immune system.
Tzianabos, A. O., Onderdonk, A. B., Smith, R. S. & Kasper, D. L. Structure–function relationships for polysaccharide-induced intra-abdominal abscesses. Infect. Immun. 62, 3590–3593 (1994).
Tzianabos, A. O., Onderdonk, A. B. & Kasper, D. L. Bacterial structure and functional relation to abscess formation. Infect. Agents Dis. 3, 256–265 (1994).
Tzianabos, A. O., Kasper, D. L., Cisneros, R. L., Smith, R. S. & Onderdonk, A. B. Polysaccharide-mediated protection against abscess formation in experimental intra-abdominal sepsis. J. Clin. Invest. 96, 2727–2731 (1995).
Wang, Y., Kalka-Moll, W. M., Roehrl, M. H. & Kasper, D. L. Structural basis of the abscess-modulating polysaccharide A2 from Bacteroides fragilis. Proc. Natl Acad. Sci. USA 97, 13478–13483 (2000). The first study to solve the NMR structure for PSA, showing the arrangement of surface-exposed charges and predicting possible interactions with MHC class II molecules.
Viret, C. & Janeway, C. A. Jr. MHC and T cell development. Rev. Immunogenet. 1, 91–104 (1999).
Kronenberg, M. & Gapin, L. The unconventional lifestyle of NKT cells. Nature Rev. Immunol. 2, 557–568 (2002).
Kinjo, Y. et al. Natural killer T cells recognize diacylglycerol antigens from pathogenic bacteria. Nature Immunol. 7, 978–986 (2006).
Kalka-Moll, W. M. et al. Zwitterionic polysaccharides stimulate T cells by MHC class II-dependent interactions. J. Immunol. 169, 6149–6153 (2002).
Petersson, K., Forsberg, G. & Walse, B. Interplay between superantigens and immunoreceptors. Scand. J. Immunol. 59, 345–355 (2004).
Cobb, B. A. & Kasper, D. L. Zwitterionic capsular polysaccharides: the new MHCII-dependent antigens. Cell. Microbiol. 7, 1398–1403 (2005).
Cobb, B. A., Wang, Q., Tzianabos, A. O. & Kasper, D. L. Polysaccharide processing and presentation by the MHCII pathway. Cell 117, 677–687 (2004). The report that definitively showed that PSA is internalized into, and processed in, the endosomes of APCs and subsequently presented by MHC class II molecules to CD4+ T cells.
Davidson, H. W., Reid, P. A., Lanzavecchia, A. & Watts, C. Processed antigen binds to newly synthesized MHC class II molecules in antigen-specific B lymphocytes. Cell 67, 105–116 (1991).
Chung, D. R. et al. CD4+ T cells regulate surgical and postinfectious adhesion formation. J. Exp. Med. 195, 1471–1478 (2002).
Falk, P. G., Hooper, L. V., Midtvedt, T. & Gordon, J. I. Creating and maintaining the gastrointestinal ecosystem: what we know and need to know from gnotobiology. Microbiol. Mol. Biol. Rev. 62, 1157–1170 (1998).
Hooper, L. V. & Gordon, J. I. Commensal host–bacterial relationships in the gut. Science 292, 1115–1118 (2001).
Trexler, P. C. An isolator system for the maintenance of aseptic environments. Lancet 1, 91–93 (1973).
Dobber, R., Hertogh-Huijbregts, A., Rozing, J., Bottomly, K. & Nagelkerken, L. The involvement of the intestinal microflora in the expansion of CD4+ T cells with a naive phenotype in the periphery. Dev. Immunol. 2, 141–150 (1992).
Pereira, P. et al. Autonomous activation of B and T cells in antigen-free mice. Eur. J. Immunol. 16, 685–688 (1986).
Bowman, L. M. & Holt, P. G. Selective enhancement of systemic TH1 immunity in immunologically immature rats with an orally administered bacterial extract. Infect. Immun. 69, 3719–3727 (2001).
Rook, G. A. & Brunet, L. R. Give us this day our daily germs. Biologist (London) 49, 145–149 (2002).
Macatonia, S. E. et al. Dendritic cells produce IL-12 and direct the development of TH1 cells from naive CD4+ T cells. J. Immunol. 154, 5071–5079 (1995).
Kononen, E., Jousimies-Somer, H. & Asikainen, S. Relationship between oral Gram-negative anaerobic bacteria in saliva of the mother and the colonization of her edentulous infant. Oral Microbiol. Immunol. 7, 273–276 (1992).
Cash, H. L., Whitham, C. V., Behrendt, C. L. & Hooper, L. V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 313, 1126–1130 (2006).
Hogquist, K. A., Baldwin, T. A. & Jameson, S. C. Central tolerance: learning self-control in the thymus. Nature Rev. Immunol. 5, 772–782 (2005).
Nagler-Anderson, C. Man the barrier! Strategic defences in the intestinal mucosa. Nature Rev. Immunol. 1, 59–67 (2001).
Prioult, G. & Nagler-Anderson, C. Mucosal immunity and allergic responses: lack of regulation and/or lack of microbial stimulation? Immunol. Rev. 206, 204–218 (2005).
Macpherson, A. J., Geuking, M. B. & McCoy, K. D. Immune responses that adapt the intestinal mucosa to commensal intestinal bacteria. Immunology 115, 153–162 (2005).
Kelly, D. et al. Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear–cytoplasmic shuttling of PPAR-γ and RelA. Nature Immunol. 5, 104–112 (2004). The study that defined a previously unknown cellular pathway of anti-inflammatory responses mediated by commensal bacteria, showing the importance of this pathway in protection against intestinal inflammation caused by bacterial infection.
Pathmakanthan, S., Li, C. K., Cowie, J. & Hawkey, C. J. Lactobacillus plantarum 299: beneficial in vitro immunomodulation in cells extracted from inflamed human colon. J. Gastroenterol. Hepatol. 19, 166–173 (2004).
Wills-Karp, M., Santeliz, J. & Karp, C. L. The germless theory of allergic disease: revisiting the hygiene hypothesis. Nature Rev. Immunol. 1, 69–75 (2001).
Bjorksten, B. The environmental influence on childhood asthma. Allergy 54 (Suppl. 49), 17–23 (1999).
Kalliomaki, M. & Isolauri, E. Pandemic of atopic diseases — a lack of microbial exposure in early infancy? Curr. Drug Targets Infect. Disord. 2, 193–199 (2002).
Alm, J. S. et al. An anthroposophic lifestyle and intestinal microflora in infancy. Pediatr. Allergy Immunol. 13, 402–411 (2002).
Dhanasekar, G., Izzat, A. B. & D'Souza, A. R. Immunotherapy for allergic rhinitis — a United Kingdom survey and short review. J. Laryngol. Otol. 119, 799–805 (2005).
Umetsu, D. T., McIntire, J. J., Akbari, O., Macaubas, C. & DeKruyff, R. H. Asthma: an epidemic of dysregulated immunity. Nature Immunol. 3, 715–720 (2002).
Bender, B. G. & Rand, C. Medication non-adherence and asthma treatment cost. Curr. Opin. Allergy Clin. Immunol. 4, 191–195 (2004).
Shi, H. N. & Walker, A. Bacterial colonization and the development of intestinal defences. Can. J. Gastroenterol. 18, 493–500 (2004).
Stock, P. et al. Induction of T helper type 1-like regulatory cells that express Foxp3 and protect against airway hyper-reactivity. Nature Immunol. 5, 1149–1156 (2004).
Kenzel, S. & Henneke, P. The innate immune system and its relevance to neonatal sepsis. Curr. Opin. Infect. Dis. 19, 264–270 (2006).
Wessels, M. R. et al. Stimulation of protective antibodies against type Ia and Ib group B streptococci by a type Ia polysaccharide–tetanus toxoid conjugate vaccine. Infect. Immun. 61, 4760–4766 (1993).
Acknowledgements
We thank J. McCoy for editorial advice. S.K.M. acknowledges fellowships from the Helen Hay Whitney Foundation (USA) and The William Randolph Hearst Foundations (USA). Work in D.L.K.'s laboratory is supported by funding from the National Institutes of Health (USA). Work in S.K.M.'s laboratory is supported by funding from the Crohn's and Colitis Foundation of America. We apologize to the many authors whose work could not be mentioned owing to space constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Immunoglobulin class switching
-
The somatic-recombination process by which the class of immunoglobulin expressed by naive B cells is switched from IgM to IgG, IgA or IgE on exposure to antigen.
- Obligate anaerobe
-
An anaerobic organism can grow in the absence of oxygen. Obligate anaerobes die when exposed to atmospheric levels of oxygen, unlike facultative anaerobes, which can use oxygen when it is present.
- Sterile caecal contents
-
A preparation of the cell-free material that is present in the caecum of the gastrointestinal tract. The preparation contains all of the soluble materials that are released by commensal bacteria but does not contain viable organisms.
- Immunodominance
-
The result of an antigen(s) within a complex mixture (such as whole virus) or an epitope(s) within a protein being preferentially recognized during an immune response.
- HLA-DM
-
An MHC-class-II-like molecule that facilitates the formation of high-affinity peptide–MHC complexes through the release of MHC-class-II-associated invariant-chain peptide (CLIP) in endosomes. The release of CLIP allows the binding of peptides derived from phagocytosed particles for presentation to CD4+ T cells.
- Immunological synapse
-
A region that can form between two cells of the immune system that are in close contact. This region was named the immunological synapse because of similarities to the synapses that occur in the nervous system; it originally referred only to the interaction between a T cell and an antigen-presenting cell. The immunological synapse involves adhesion molecules, as well as antigen receptors and cytokine receptors.
- Gut-associated lymphoid tissue
-
(GALT). The tissues and cells that constitute the immune system associated with the gastrointestinal tract. This system includes structures such as the Peyer's patches, inducible lymphoid follicles, cryptopatches and the mesenteric lymph nodes, as well as circulating and non-circulating immune cells of the lamina propria, and intra-epithelial lymphocytes.
- Germ-free mice
-
Animals that are born and raised in sterile isolator chambers and are devoid of colonization by any foreign microorganisms, including bacteria, viruses, fungi and protozoa. The experimental colonization of animals with known microorganisms (an approach that is known as gnotobiology) allows the effect of a specific microorganism on the biological functions of an animal to be investigated.
- Lymphoid follicles
-
Anatomical zones in the primary or secondary lymphoid tissues that contain aggregates of lymphocytes, mainly B cells, which are surrounded by T cells. Follicles might also contain structures known as germinal centres, which are areas of proliferating and differentiating B cells.
Rights and permissions
About this article
Cite this article
Mazmanian, S., Kasper, D. The love–hate relationship between bacterial polysaccharides and the host immune system. Nat Rev Immunol 6, 849–858 (2006). https://doi.org/10.1038/nri1956
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri1956
This article is cited by
-
Strengths and weaknesses of pneumococcal conjugate vaccines
Glycoconjugate Journal (2023)
-
Histone modifications and DNA methylation act cooperatively in regulating symbiosis genes in the sea anemone Aiptasia
BMC Biology (2022)
-
Effects of waterborne Pb/Cu mixture on Chinese toad, Bufo gargarizans tadpoles: morphological, histological, and intestinal microbiota assessment
Environmental Science and Pollution Research (2022)
-
Cell-based high-throughput screening of polysaccharide biosynthesis hosts
Microbial Cell Factories (2021)
-
Combined effects of glycan chain length and linkage type on the immunogenicity of glycoconjugate vaccines
npj Vaccines (2021)