Key Points
-
In addition to the well-known role of T cells in the rejection of allografts, antibodies that are produced by the host and are specific for the donor graft (that is, alloantibodies) can mediate graft rejection, which can occur at almost any time after transplantation and cause rapid or slow graft dysfunction.
-
The target antigens are the MHC class I and class II molecules and the ABO blood-group antigens, as well as potentially other minor alloantigens at the endothelial-cell surface.
-
Complement component 4d (C4d), a breakdown product of complement activation, can be detected in tissues that are undergoing antibody-mediated rejection, even though no immunoglobulin can be detected. The presence of C4d has proved to be a more useful diagnostic marker of antibody-mediated rejection than has histology.
-
Antibody and complement have diverse effects on endothelial cells, ranging from activation (which leads to the production of cytokines, chemokines, adhesion molecules and growth factors) to cell death.
-
A subset of cases of chronic rejection of renal allografts is mediated by alloantibodies, and these cases develop over many years and probably involve distinct stages of pathogenesis.
-
In some settings, antibody and complement do not seem to have adverse effects on endothelial cells, a phenomenon that is known as accommodation. Understanding the molecular basis for this might provide a strategy to inhibit the consequences of antibody-mediated rejection.
-
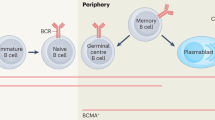
Innovative therapies that have the potential to more specifically target B cells and plasma cells are under evaluation for the prevention or treatment of antibody-mediated rejection.
Abstract
Recent studies show that alloantibodies mediate a substantial proportion of graft-rejection episodes, contributing to both early and late graft loss. Rejection that is caused by antibody is mediated by different mechanisms from rejection that is caused by T cells, thereby requiring other approaches to treatment and prevention. Antibody induces rejection acutely through the fixation of complement, resulting in tissue injury and coagulation. In addition, complement activation recruits macrophages and neutrophils, causing additional endothelial injury. Antibody and complement also induce gene expression by endothelial cells, which is thought to remodel arteries and basement membranes, leading to fixed and irreversible anatomical lesions that permanently compromise graft function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Manning, D. D., Reed, N. D. & Shaffer, C. F. Maintenance of skin xenografts of widely divergent phylogenetic origin of congenitally athymic (nude) mice. J. Exp. Med. 138, 488–494 (1973).
Pascual, M. et al. in Clinical Transplants 2001 123–130 (UCLA Immunogenetics Center, Los Angeles, 2002).
Gorer, P. The detection of antigenic differences in mouse erythrocytes by the employment of immune sera. Br. J. Exp. Pathol. 17, 42–50 (1936).
Gerlag, P. G., Koene, R. A., Hagemann, J. F. & Wijdeveld, P. G. Hyperacute rejection of skin allografts in the mouse. Sensitivity of ingrowing skin grafts to the action of alloantibody and rabbit complement. Transplantation 20, 308–313 (1975).
Jooste, S. V., Colvin, R. B., Soper, W. D. & Winn, H. J. The vascular bed as the primary target in the destruction of skin grafts by antiserum. I. Resistance of freshly placed xenografts of skin to antiserum. J. Exp. Med. 154, 1319–1331 (1981).
Jeannet, M., Pinn, V. W., Flax, M. H., Winn, H. J. & Russell, P. S. Humoral antibodies in renal allotransplantation in man. N. Engl. J. Med. 282, 111–117 (1970).
Russell, P. S., Chase, C. M. & Colvin, R. B. Alloantibody- and T cell-mediated immunity in the pathogenesis of transplant arteriosclerosis: lack of progression to sclerotic lesions in B cell-deficient mice. Transplantation 64, 1531–1536 (1997).
Halloran, P. F., Schlaut, J., Solez, K. & Srinivasa, N. S. The significance of the anti-class I antibody response. II. Clinical and pathologic features of renal transplants with anti-class I-like antibody. Transplantation 53, 550–555 (1992).
Feucht, H. E. et al. Vascular deposition of complement-split products in kidney allografts with cell-mediated rejection. Clin. Exp. Immunol. 86, 464–470 (1991).
Collins, A. B. et al. Complement activation in acute humoral renal allograft rejection: diagnostic significance of C4d deposits in peritubular capillaries. J. Am. Soc. Nephrol. 10, 2208–2214 (1999). This was the first paper to show that the presence of C4d in the peritubular capillaries of renal allografts is associated with circulating donor-HLA-specific antibodies and distinctive histological features.
Pei, R., Lee, J. H., Shih, N. J., Chen, M. & Terasaki, P. I. Single human leukocyte antigen flow cytometry beads for accurate identification of human leukocyte antigen antibody specificities. Transplantation 75, 43–49 (2003).
Jaramillo, A. et al. Anti-HLA class I antibody binding to airway epithelial cells induces production of fibrogenic growth factors and apoptotic cell death: a possible mechanism for bronchiolitis obliterans syndrome. Hum. Immunol. 64, 521–529 (2003).
Takemoto, S. K. et al. National conference to assess antibody-mediated rejection in solid organ transplantation. Am. J. Transplant. 4, 1033–1041 (2004).
Racusen, L. C. et al. Antibody-mediated rejection criteria — an addition to the Banff 97 classification of renal allograft rejection. Am. J. Transplant. 3, 708–714 (2003).
Mauiyyedi, S. et al. Acute humoral rejection in kidney transplantation: II. Morphology, immunopathology, and pathologic classification. J. Am. Soc. Nephrol. 13, 779–787 (2002).
Nickeleit, V., Zeiler, M., Gudat, F., Thiel, G. & Mihatsch, M. J. Detection of the complement degradation product C4d in renal allografts: diagnostic and therapeutic implications. J. Am. Soc. Nephrol. 13, 242–251 (2002).
Herzenberg, A. M., Gill, J. S., Djurdjev, O. & Magil, A. B. C4d deposition in acute rejection: an independent long-term prognostic factor. J. Am. Soc. Nephrol. 13, 234–241 (2002).
Lorenz, M. et al. Risk factors for capillary C4d deposition in kidney allografts: evaluation of a large study cohort. Transplantation 78, 447–452 (2004).
Trpkov, K. et al. Pathologic features of acute renal allograft rejection associated with donor-specific antibody: analysis using the Banff grading schema. Transplantation 61, 1586–1592 (1996).
Bohmig, G. A. et al. Capillary C4d deposition in kidney allografts: a specific marker of alloantibody-dependent graft injury. J. Am. Soc. Nephrol. 13, 1091–1099 (2002).
Magil, A. B. & Tinckam, K. Monocytes and peritubular capillary C4d deposition in acute renal allograft rejection. Kidney Int. 63, 1888–1893 (2003).
Lederer, S. R. et al. Impact of humoral alloreactivity early after transplantation on the long-term survival of renal allografts. Kidney Int. 59, 334–341 (2001). This is a recent paper by the research group of Helmut Feucht, who was the first to stain renal allografts for C4d and show that it predicted a poor prognosis (reported in reference 9).
Haas, M., Ratner, L. E. & Montgomery, R. A. C4d staining of perioperative renal transplant biopsies. Transplantation 74, 711–717 (2002).
Rotman, S., Collins, A. B. & Colvin, R. B. C4d deposition in allografts: current concepts and interpretation. Transplant. Rev. (in the press).
Hammond, E. H. et al. Vascular (humoral) rejection in heart transplantation: pathologic observations and clinical implications. J. Heart Transplant. 8, 430–443 (1989).
John, R. et al. Immunologic sensitization in recipients of left ventricular assist devices. J. Thorac. Cardiovasc. Surg. 125, 578–591 (2003).
Smith, R. N. et al. C4d deposition in cardiac allografts correlates with alloantibody. J. Heart Lung Transplant. 24, 1202–1210 (2005). This paper shows that C4d deposition in myocardial capillaries is associated with circulating HLA-specific antibodies rather than ischaemia.
Michaels, P. J. et al. Humoral rejection in cardiac transplantation: risk factors, hemodynamic consequences and relationship to transplant coronary artery disease. J. Heart Lung Transplant. 22, 58–69 (2003).
Reed, E. F. et al. Monitoring of soluble HLA alloantigens and anti-HLA antibodies identifies heart allograft recipients at risk of transplant-associated coronary artery disease. Transplantation 61, 566–572 (1996).
Behr, T. M. et al. Detection of humoral rejection in human cardiac allografts by assessing the capillary deposition of complement fragment C4d in endomyocardial biopsies. J. Heart Lung Transplant. 18, 904–912 (1999).
Magro, C. M. et al. Use of C4d as a diagnostic adjunct in lung allograft biopsies. Am. J. Transplant. 3, 1143–1154 (2003).
Krukemeyer, M. G. et al. Description of B lymphocytes and plasma cells, complement, and chemokines/receptors in acute liver allograft rejection. Transplantation 78, 65–70 (2004).
Terasaki, P. I. & Ozawa, M. Predicting kidney graft failure by HLA antibodies: a prospective trial. Am. J. Transplant. 4, 438–443 (2004).
Piazza, A. et al. Impact of donor-specific antibodies on chronic rejection occurrence and graft loss in renal transplantation: posttransplant analysis using flow cytometric techniques. Transplantation 71, 1106–1112 (2001).
Pelletier, R. P. et al. Clinical significance of MHC-reactive alloantibodies that develop after kidney or kidney–pancreas transplantation. Am. J. Transplant. 2, 134–141 (2002).
Worthington, J. E., Martin, S., Al-Husseini, D. M., Dyer, P. A. & Johnson, R. W. Posttransplantation production of donor HLA-specific antibodies as a predictor of renal transplant outcome. Transplantation 75, 1034–1040 (2003). This report shows that circulating antibodies that are specific for donor HLA class I or class II antigens predict the subsequent development of chronic rejection of a renal allograft.
Mauiyyedi, S. et al. Chronic humoral rejection: identification of antibody-mediated chronic renal allograft rejection by C4d deposits in peritubular capillaries. J. Am. Soc. Nephrol. 12, 574–582 (2001). The paper identifies and establishes the immunopathological criteria for a new form of chronic renal-allograft rejection that is mediated by antibodies.
Regele, H. et al. Capillary deposition of complement split product C4d in renal allografts is associated with basement membrane injury in peritubular and glomerular capillaries: a contribution of humoral immunity to chronic allograft rejection. J. Am. Soc. Nephrol. 13, 2371–2380 (2002). This was the first paper to show that the presence of C4d in the peritubular capillaries of renal allografts precedes the development of chronic allograft glomerulopathy. It also shows that C4d deposition correlates with lamination of the peritubular capillary basement membrane.
Vongwiwatana, A., Gourishankar, S., Campbell, P. M., Solez, K. & Halloran, P. F. Peritubular capillary changes and C4d deposits are associated with transplant glomerulopathy but not IgA nephropathy. Am. J. Transplant. 4, 124–129 (2004).
Sijpkens, Y. W. et al. Immunologic risk factors and glomerular C4d deposits in chronic transplant glomerulopathy. Kidney Int. 65, 2409–2418 (2004).
Erlich, H. A., Opelz, G. & Hansen, J. HLA DNA typing and transplantation. Immunity 14, 347–356 (2001).
Race, R. R. & Sanger, R. Blood Groups in Man (Blackwell Scientific, Oxford, 1958).
Chao, N. J. Minors come of age: minor histocompatibility antigens and graft-versus-host disease. Biol. Blood Marrow Transplant. 10, 215–223 (2004).
Derhaag, J. G., Duijvestijn, A. M., Damoiseaux, J. G. & van Breda Vriesman, P. J. Effects of antibody reactivity to major histocompatibility complex (MHC) and non-MHC alloantigens on graft endothelial cells in heart allograft rejection. Transplantation 69, 1899–1906 (2000).
Wu, G. D. et al. Vascular endothelial cell apoptosis induced by anti-donor non-MHC antibodies: a possible injury pathway contributing to chronic allograft rejection. J. Heart Lung Transplant. 21, 1174–1187 (2002).
Kooijmans-Coutinho, M. F. et al. Interstitial rejection, vascular rejection, and diffuse thrombosis of renal allografts. Predisposing factors, histology, immunohistochemistry, and relation to outcome. Transplantation 61, 1338–1344 (1996).
Sumitran-Holgersson, S., Wilczek, H. E., Holgersson, J. & Soderstrom, K. Identification of the nonclassical HLA molecules, MICA, as targets for humoral immunity associated with irreversible rejection of kidney allografts. Transplantation 74, 268–277 (2002).
Mizutani, K. et al. Serial ten-year follow-up of HLA and MICA antibody production prior to kidney graft failure. Am. J. Transplant. 5, 1–8 (2005).
Dragun, D. et al. Angiotensin II type 1-receptor activating antibodies in renal-allograft rejection. N. Engl. J. Med. 352, 558–569 (2005).
Jurcevic, S. et al. Antivimentin antibodies are an independent predictor of transplant-associated coronary artery disease after cardiac transplantation. Transplantation 71, 886–892 (2001).
Morgun, A. et al. Pre- and post-transplant anti-myosin and anti-heat shock protein antibodies and cardiac transplant outcome. J. Heart Lung Transplant. 23, 204–209 (2004).
Austen, W. G. Jr et al. Murine hindlimb reperfusion injury can be initiated by a self-reactive monoclonal IgM. Surgery 136, 401–406 (2004).
Mitchison, N. A. T-cell–B-cell cooperation. Nature Rev. Immunol. 4, 308–312 (2004).
Baratin, M., Bonin, K. & Daniel, C. Peripheral priming of alloreactive T cells by the direct pathway of allorecognition. Eur. J. Immunol. 34, 3305–3314 (2004).
Steele, D. J. et al. Two levels of help for B cell alloantibody production. J. Exp. Med. 183, 699–703 (1996).
Edwards, J. C. et al. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N. Engl. J. Med. 350, 2572–2581 (2004).
Asiedu, C. K., Dong, S. S., Lobashevsky, A., Jenkins, S. M. & Thomas, J. M. Tolerance induced by anti-CD3 immunotoxin plus 15-deoxyspergualin associates with donor-specific indirect pathway unresponsiveness. Cell. Immunol. 223, 103–112 (2003).
Epstein, M. M., Di Rosa, F., Jankovic, D., Sher, A. & Matzinger, P. Successful T cell priming in B cell-deficient mice. J. Exp. Med. 182, 915–922 (1995).
Williams, G. S., Oxenius, A., Hengartner, H., Benoist, C. & Mathis, D. CD4+ T cell responses in mice lacking MHC class II molecules specifically on B cells. Eur. J. Immunol. 28, 3763–3772 (1998).
Shapiro-Shelef, M. & Calame, K. Regulation of plasma-cell development. Nature Rev. Immunol. 5, 230–242 (2005).
Kerjaschki, D. et al. Lymphatic neoangiogenesis in human kidney transplants is associated with immunologically active lymphocytic infiltrates. J. Am. Soc. Nephrol. 15, 603–612 (2004).
Sarwal, M. et al. Molecular heterogeneity in acute renal allograft rejection identified by DNA microarray profiling. N. Engl. J. Med. 349, 125–138 (2003).
Cassese, G. et al. Inflamed kidneys of NZB/W mice are a major site for the homeostasis of plasma cells. Eur. J. Immunol. 31, 2726–2732 (2001).
Desvaux, D. et al. γ-Interferon expression and poor clinical outcome. Nephrol. Dial. Transplant. 19, 933–939 (2004).
Hidalgo, L. G. & Halloran, P. F. Role of IFN-γ in allograft rejection. Crit. Rev. Immunol. 22, 317–349 (2002).
Auchincloss, H. Jr & Sachs, D. H. Xenogeneic transplantation. Annu. Rev. Immunol. 16, 433–470 (1998).
Wasowska, B. A. et al. Passive transfer of alloantibodies restores acute cardiac rejection in IgKO mice. Transplantation 71, 727–736 (2001).
Rahimi, S. et al. Non-complement- and complement-activating antibodies synergize to cause rejection of cardiac allografts. Am. J. Transplant. 4, 326–334 (2004).
Menoret, S. et al. Characterization of human CD55 and CD59 transgenic pigs and kidney xenotransplantation in the pig-to-baboon combination. Transplantation 77, 1468–1471 (2004).
Azimzadeh, A. et al. Hyperacute lung rejection in the pig-to-human model. 2. Synergy between soluble and membrane complement inhibition. Xenotransplantation 10, 120–131 (2003).
Prodinger, W. M., Wurzner, R., Stoiber, H. & Dierich, M. P. in Fundamentals of Immunology (ed. Paul, W. E.) 1077–1103 (Lippincott Williams & Wilkins, Philadelphia, 2003).
van den Elsen, J. M. et al. X-ray crystal structure of the C4d fragment of human complement component C4. J. Mol. Biol. 322, 1103–1115 (2002).
Baldwin, W. M. 3rd, Kasper, E. K., Zachary, A. A., Wasowska, B. A. & Rodriguez, E. R. Beyond C4d: other complement-related diagnostic approaches to antibody-mediated rejection. Am. J. Transplant. 4, 311–318 (2004).
Sund, S. et al. Complement activation in early protocol kidney graft biopsies after living-donor transplantation. Transplantation 75, 1204–1213 (2003).
Nakashima, S., Qian, Z., Rahimi, S., Wasowska, B. A. & Baldwin, W. M. 3rd. Membrane attack complex contributes to destruction of vascular integrity in acute lung allograft rejection. J. Immunol. 169, 4620–4627 (2002).
Saadi, S. & Platt, J. L. Humoral rejection and endothelial cell activation, 2001. Xenotransplantation 9, 239–241 (2002).
Albrecht, E. A. et al. C5a-induced gene expression in human umbilical vein endothelial cells. Am. J. Pathol. 164, 849–859 (2004).
Monsinjon, T. et al. Regulation by complement C3a and C5a anaphylatoxins of cytokine production in human umbilical vein endothelial cells. FASEB J. 17, 1003–1014 (2003).
Saadi, S., Holzknecht, R. A., Patte, C. P. & Platt, J. L. Endothelial cell activation by pore-forming structures: pivotal role for interleukin-1α. Circulation 101, 1867–1873 (2000).
Benzaquen, L. R., Nicholson-Weller, A. & Halperin, J. A. Terminal complement proteins C5b–9 release basic fibroblast growth factor and platelet-derived growth factor from endothelial cells. J. Exp. Med. 179, 985–992 (1994).
Selvan, R. S., Kapadia, H. B. & Platt, J. L. Complement-induced expression of chemokine genes in endothelium: regulation by IL-1-dependent and -independent mechanisms. J. Immunol. 161, 4388–4395 (1998).
Kilgore, K. S. et al. Sublytic concentrations of the membrane attack complex of complement induce endothelial interleukin-8 and monocyte chemoattractant protein-1 through nuclear factor-κB activation. Am. J. Pathol. 150, 2019–2031 (1997).
Ikeda, K. et al. C5a induces tissue factor activity on endothelial cells. Thromb. Haemost. 77, 394–398 (1997).
Saadi, S., Holzknecht, R. A., Patte, C. P., Stern, D. M. & Platt, J. L. Complement-mediated regulation of tissue factor activity in endothelium. J. Exp. Med. 182, 1807–1814 (1995).
Dempsey, P. W., Allison, M. E., Akkaraju, S., Goodnow, C. C. & Fearon, D. T. C3d of complement as a molecular adjuvant: bridging innate and acquired immunity. Science 271, 348–350 (1996).
Qian, Z. et al. C6 produced by macrophages contributes to cardiac allograft rejection. Am. J. Pathol. 155, 1293–1302 (1999).
Ota, H. et al. Terminal complement components mediate release of von Willebrand factor and adhesion of platelets in arteries of allografts. Transplantation 79, 276–281 (2005).
Pratt, J. R., Basheer, S. A. & Sacks, S. H. Local synthesis of complement component C3 regulates acute renal transplant rejection. Nature Med. 8, 582–587 (2002).
Smith, J. D., Lawson, C., Yacoub, M. H. & Rose, M. L. Activation of NF-κB in human endothelial cells induced by monoclonal and allospecific HLA antibodies. Int. Immunol. 12, 563–571 (2000).
Jin, Y. P. et al. Ligation of HLA class I molecules on endothelial cells induces phosphorylation of Src, paxillin, and focal adhesion kinase in an actin-dependent manner. J. Immunol. 168, 5415–5423 (2002). This is the most recent in a series of papers from this research group. It identifies the activation pathways in endothelial cells that are triggered by HLA-specific antibodies.
Yuan, F. F. et al. Association of Fcγ receptor IIA polymorphisms with acute renal-allograft rejection. Transplantation 78, 766–769 (2004).
Koch, C. A., Khalpey, Z. I. & Platt, J. L. Accommodation: preventing injury in transplantation and disease. J. Immunol. 172, 5143–5148 (2004).
Alexandre, G. P. J. et al. Present experience in a series of 26 ABO-incompatible living donor renal allografts. Transplant. Proc. 19, 4538–4544 (1987).
Shishido, S. et al. ABO-incompatible living-donor kidney transplantation in children. Transplantation 72, 1037–1042 (2001).
Ogawa, H. et al. Mouse-heart grafts expressing an incompatible carbohydrate antigen. II. Transition from accommodation to tolerance. Transplantation 77, 366–373 (2004).
Soares, M. P., Brouard, S., Smith, R. N. & Bach, F. H. Heme oxygenase-1, a protective gene that prevents the rejection of transplanted organs. Immunol. Rev. 184, 275–285 (2001).
Tabata, T. et al. Accommodation after lung xenografting from hamster to rat. Transplantation 75, 607–612 (2003).
Salama, A. D. et al. Transplant accommodation in highly sensitized patients: a potential role for Bcl-xL and alloantibody. Am. J. Transplant. 1, 260–269 (2001). This paper shows antibody-induced increases in endothelial-cell expression of BCL-X L , a potential molecular mechanism of accommodation in human renal transplants.
Narayanan, K., Jaramillo, A., Phelan, D. L. & Mohanakumar, T. Pre-exposure to sub-saturating concentrations of HLA class I antibodies confers resistance to endothelial cells against antibody complement-mediated lysis by regulating Bad through the phosphatidylinositol 3-kinase/Akt pathway. Eur. J. Immunol. 34, 2303–2312 (2004).
Park, W. D. et al. Accommodation in ABO-incompatible kidney allografts, a novel mechanism of self-protection against antibody-mediated injury. Am. J. Transplant. 3, 952–960 (2003).
Lee, P. C. et al. All chronic rejection failures of kidney transplants were preceded by the development of HLA antibodies. Transplantation 74, 1192–1194 (2002).
Mason, J. C. et al. Decay-accelerating factor induction on vascular endothelium by vascular endothelial growth factor (VEGF) is mediated via a VEGF receptor-2 (VEGF-R2)- and protein kinase C-α/ε (PKCα/ε)-dependent cytoprotective signaling pathway and is inhibited by cyclosporin A. J. Biol. Chem. 279, 41611–41618 (2004).
Shimizu, I. et al. DAF prevents acute humoral rejection induced by low titres of anti-αGal antibodies in GAL KO mice. Transplantation (in the press).
Grubbs, B. C., Benson, B. A. & Dalmasso, A. P. Characteristics of CD59 up-regulation induced in porcine endothelial cells by αGal ligation and its association with protection from complement. Xenotransplantation 10, 387–397 (2003).
Williams, J. M. et al. Acute vascular rejection and accommodation: divergent outcomes of the humoral response to organ transplantation. Transplantation 78, 1471–1478 (2004).
Hillebrands, J. L. et al. Origin of neointimal endothelium and α-actin-positive smooth muscle cells in transplant arteriosclerosis. J. Clin. Invest. 107, 1411–1422 (2001).
Koestner, S. C. et al. Histo-blood group type change of the graft from B to O after ABO mismatched heart transplantation. Lancet 363, 1523–1525 (2004).
Lagaaij, E. L. et al. Endothelial cell chimerism after renal transplantation and vascular rejection. Lancet 357, 33–37 (2001).
Mohiuddin, M. M., Ogawa, H., Yin, D. P., Shen, J. & Galili, U. Antibody-mediated accommodation of heart grafts expressing an incompatible carbohydrate antigen. Transplantation 75, 258–262 (2003). This report describes a pilot study of gene expression in ABO-incompatible human cardiac allografts with accommodation.
Toyoda, M., Petrosyan, A., Pao, A. & Jordan, S. C. Immunomodulatory effects of combination of pooled human gammaglobulin and rapamycin on cell proliferation and apoptosis in the mixed lymphocyte reaction. Transplantation 78, 1134–1138 (2004).
Jordan, S. C. et al. Intravenous immune globulin treatment inhibits crossmatch positivity and allows for successful transplantation of incompatible organs in living-donor and cadaver recipients. Transplantation 76, 631–636 (2003).
Frank, M. M., Miletic, V. D. & Jiang, H. Immunoglobulin in the control of complement action. Immunol. Res. 22, 137–146 (2000).
Vieira, C. A. et al. Rituximab for reduction of anti-HLA antibodies in patients awaiting renal transplantation: 1. Safety, pharmacodynamics, and pharmacokinetics. Transplantation 77, 542–548 (2004). This paper describes encouraging results from a Phase I clinical trial of rituximab in pre-sensitized patients who are awaiting a renal transplant. It shows some efficacy at the inhibition of presumed pre-existing memory B cells and long-lived plasma cells.
Gloor, J. M. et al. Overcoming a positive crossmatch in living-donor kidney transplantation. Am. J. Transplant. 3, 1017–1023 (2003).
Tyden, G., Kumlien, G. & Fehrman, I. Successful ABO-incompatible kidney transplantations without splenectomy using antigen-specific immunoadsorption and rituximab. Transplantation 76, 730–731 (2003).
McHeyzer-Williams, L. J. & McHeyzer-Williams, M. G. Antigen-specific memory B cell development. Annu. Rev. Immunol. 23, 487–513 (2005).
Craxton, A., Magaletti, D., Ryan, E. J. & Clark, E. A. Macrophage- and dendritic cell-dependent regulation of human B-cell proliferation requires the TNF family ligand BAFF. Blood 101, 4464–4471 (2003).
Gross, J. A. et al. TACI–Ig neutralizes molecules critical for B cell development and autoimmune disease: impaired B cell maturation in mice lacking BLyS. Immunity 15, 289–302 (2001).
O'Connor, B. P. et al. BCMA is essential for the survival of long-lived bone marrow plasma cells. J. Exp. Med. 199, 91–98 (2004).
West, L. J. et al. ABO-incompatible heart transplantation in infants. N. Engl. J. Med. 344, 793–800 (2001).
Fan, X. et al. Donor-specific B-cell tolerance after ABO-incompatible infant heart transplantation. Nature Med. 10, 1227–1233 (2004).
Knechtle, S. J. et al. Campath-1H induction plus rapamycin monotherapy for renal transplantation: results of a pilot study. Am. J. Transplant. 3, 722–730 (2003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- ACUTE REJECTION
-
The rapid (within days) clinical deterioration of graft function, which can occur days to years after transplantation. It can be caused by T-cell-dependent reactivity to donor alloantigen (that is, cellular rejection) and/or alloantibody reactivity to donor antigens on the endothelium (that is, antibody-mediated rejection).
- CHRONIC REJECTION
-
The slow (within months to years) clinical deterioration of graft function. It can be caused by T-cell- and/or antibody-mediated reaction to donor alloantigens, which results in a slow, progressive decline in graft function and is typically associated with stenotic intimal hyperplasia of the arteries.
- PRE-SENSITIZATION
-
Immunological reactivity to a donor that is present before transplantation: for example, as a result of a previous transplant, pregnancy or blood transfusion. It is typically detected by the measurement of serum antibody that is specific for donor cells or surrogate targets.
- SERUM CREATININE
-
Creatinine is a component of urine and the final product in the metabolism of creatine. An increase in serum concentration is used as a marker of kidney dysfunction.
- DE NOVO ANTIBODIES
-
Antibodies that first appear after transplantation (that is, the patient is not pre-sensitized).
- NEO-LYMPHOID ORGAN
-
The organization within a tissue of high endothelial venules, lymphoid follicles and dendritic cells. This can be mediated by persistent inflammation.
- COMPLEMENT-FIXING ANTIBODIES
-
Not all antibodies fix or activate complement. In humans, IgM and the IgG subclasses IgG1 and IgG3 readily fix complement, whereas IgG2 is less effective. The IgG subclass IgG4 and other classes of immunoglobulin do not fix complement or activate the classical complement pathway.
- ENDOTHELIAL-CELL ACTIVATION
-
Marked by phenotypic changes that usually include MHC class II expression, tissue-factor activity and increased leukocyte adhesion to the endothelium, all of which are induced by interferon. This often occurs together with morphological changes, including hypertrophy (indicated by a cuboidal appearance), increased biosynthesis and increased permeability.
- ANTIBODY-DEPENDENT CELL-MEDIATED CYTOTOXICITY
-
(ADCC). A mechanism by which natural killer (NK) cells kill other cells: for example, virus-infected target cells that are coated with antibodies. The Fc portions of the coating antibodies interact with the Fc receptor that is expressed by NK cells (FcγRIII; CD16), thereby initiating a signalling cascade that results in the release of cytotoxic granules (containing perforin and granzyme B), which induce apoptosis of the antibody-coated cell.
- INTRAVENOUS IMMUNOGLOBULIN
-
Immunoglobulin that is pooled from a large number of individuals. It is used as a replacement for patients who have been depleted of immunoglobulins and for the immunomodulatory treatment of patients with some immune disorders.
- MIXED LYMPHOCYTE REACTION
-
A tissue-culture technique for testing T-cell reactivity. The proliferation of responder T cells (in this case, recipient T cells) that is induced by exposure to inactivated stimulator cells (in this case, donor T cells) is determined by measuring the incorporation of 3H-thymidine into the DNA of dividing cells.
Rights and permissions
About this article
Cite this article
Colvin, R., Smith, R. Antibody-mediated organ-allograft rejection. Nat Rev Immunol 5, 807–817 (2005). https://doi.org/10.1038/nri1702
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri1702
This article is cited by
-
HLA class II antibody activation of endothelial cells induces M2 macrophage differentiation in peripheral blood
Clinical and Experimental Nephrology (2023)
-
Prevention of vascular-allograft rejection by protecting the endothelial glycocalyx with immunosuppressive polymers
Nature Biomedical Engineering (2021)
-
Emerging monitoring technologies in kidney transplantation
Pediatric Nephrology (2021)
-
Peptide- and Protein-Graphene Oxide Conjugate Materials for Controlling Mesenchymal Stem Cell Fate
Regenerative Engineering and Translational Medicine (2021)
-
Immune reaction and regulation in transplantation based on pluripotent stem cell technology
Inflammation and Regeneration (2020)