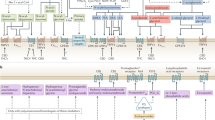
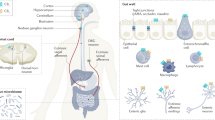
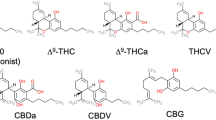
Key Points
-
Marijuana-derived cannabinoids and related compounds have been tested for the treatment of various diseases, ranging from cancer to glaucoma. Recently, these drugs have been reported to have immunomodulatory effects, so their potential for the treatment of chronic inflammatory diseases is being evaluated.
-
Marijuana-derived cannabinoids function by binding several subtypes of cannabinoid receptor in the brain and other organs. In addition, the body produces endocannabinoids that also function through binding these receptors. Compounds that are chemically related to cannabinoids have also been shown to function by binding other types of receptor, such as the NMDA (N-methyl-D-aspartate) receptor and the peroxisome-proliferative-activated receptor-γ (PPAR-γ), or by influencing other cellular components, such as lipid rafts.
-
Immune activation causes lymphocytes and macrophages to produce endocannabinoids and to alter their expression of cannabinoid receptors. These effects and endocannabinoid-mediated effects on immune-cell migration and cytokine production indicate that the endocannabinoid system is involved in the host inflammatory response.
-
Cannabinoids and related compounds have been shown to either suppress or increase the production of pro-inflammatory cytokines — such as tumour-necrosis factor, interleukin-1β (IL-1β) and IL-6 — in both patients and animal models, indicating that these drugs can modulate pro-inflammatory mediators. Depending on the model system, the effects of these drugs do not always depend on their interaction with cannabinoid receptors.
-
Cannabinoids bias the immune response away from T helper 1 (TH1)-cell responses, by mechanisms that involve cannabinoid receptors. It is possible that signalling through these receptors, expressed by T cells, B cells or antigen-presenting cells, suppresses the expression of TH1-cell-promoting cytokines and increases the expression of TH2-cell-promoting cytokines.
-
Cannabinoids and endocannabinoids regulate some of the inflammatory aspects of brain injury, through both cannabinoid-receptor-mediated and non-cannabinoid-receptor-mediated mechanisms. It is possible that these drugs reduce brain oedema and other aspects of neuroinflammation by inhibiting NMDA receptors, by functioning as antioxidants and by reducing the levels of pro-inflammatory cytokines in the brain.
-
Cannabinoids regulate the tissue response to inflammation in the colon, and it is possible that this regulation occurs on two levels: the first, involving the smooth-muscle response to pro-inflammatory mediators, thereby affecting gastrointestinal transit time; and the second, involving the direct suppression of pro-inflammatory-mediator production.
-
Plant-derived cannabinoids and synthetic derivatives are anti-inflammatory and immunosuppressive in animal models of arthritis. The mechanisms of action seem to be independent of cannabinoid receptors and cause suppression of pro-inflammatory cytokines that are produced by lymphocytes and macrophages.
-
Endocannabinoids and cannabinoid receptor 1 (CB1) might function as regulators of inflammation-induced hypotension, whereas cannabinoids that bind CB2 might attenuate vascular inflammation. Cannabinoid-based drugs that do not function by interacting with cannabinoid receptors decrease the symptoms of septic shock, which might result from the ability of these drugs to inhibit pro-inflammatory-cytokine production.
Abstract
In the nineteenth century, marijuana was prescribed by physicians for maladies ranging from eating disorders to rabies. However, as newer, more effective drugs were discovered and as the potential for abuse of marijuana was recognized, its use as a therapeutic became restricted, and only recently has its therapeutic potential been re-evaluated. Recent studies in animal models and in humans have produced promising results for the treatment of various disorders — such as obesity, cancer, and spasticity and tremor due to neuropathology — with drugs based on marijuana-derived cannabinoids. Moreover, as I discuss here, a wealth of information also indicates that these drugs have immunosuppressive and anti-inflammatory properties; therefore, on the basis of this mode of action, the therapeutic usefulness of these drugs in chronic inflammatory diseases is now being reassessed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Tomida, I., Pertwee, R. G. & Azuara-Blanco, A. Cannabinoids and glaucoma. Br. J. Ophthalmol. 88, 708–713 (2004).
Guzman, M. Cannabinoids: potential anticancer agents. Nature Rev. Cancer 3, 745–755 (2003).
Kunos, G. & Pacher, P. Cannabinoids cool the intestine. Nature Med. 10, 678–679 (2004).
Mendizabal, V. E. & Adler-Graschinsky, E. Cannabinoid system as a potential target for drug development in the treatment of cardiovascular disease. Curr. Vasc. Pharmacol. 1, 301–313 (2003).
Mechoulam, R., Panikashvili, D. & Shohami, E. Cannabinoids and brain injury: therapeutic implications. Trends Mol. Med. 8, 58–61 (2002).
Baker, D. & Pryce, G. The therapeutic potential of cannabis in multiple sclerosis. Expert Opin. Investig. Drugs 12, 561–567 (2003).
Baker, D., Pryce, G., Giovannoni, G. & Thompson, A. J. The therapeutic potential of cannabis. Lancet Neurol. 2, 291–298 (2003).
Di Marzo, V., Bifulco, M. & De Petrocellis, L. The endocannabinoid system and its therapeutic exploitation. Nature Rev. Drug Discov. 3, 771–784 (2004).
Black, S. C. Cannabinoid receptor antagonists and obesity. Curr. Opin. Investig. Drugs 5, 389–394 (2004).
Gaoni, Y. & Mechoulam, R. Isolation, structure, and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 86, 1646–1647 (1964). This is a seminal paper that describes the isolation and synthesis of THC.
Howlett, A. C. et al. International Union of Pharmacology. XXVII. Classification of cannabinoid receptors. Pharmacol. Rev. 54, 161–202 (2002). For those wanting more on the chemistry and biology of the cannabinoid system, this is a recent and authoritative review.
Rhee, M. H. et al. Cannabinol derivatives: binding to cannabinoid receptors and inhibition of adenylylcyclase. J. Med. Chem. 40, 3228–3233 (1997).
Zurier, R. B. et al. Dimethylheptyl-THC-11 oic acid. A nonpsychoactive antiinflammatory agent with a cannabinoid template structure. Arthritis Rheum. 41, 163–170 (1998).
Burstein, S. H. The cannabinoid acids: nonpsychoactive derivatives with therapeutic potential. Pharmacol. Ther. 82, 87–96 (1999).
Liu, J., Li, H., Burstein, S. H., Zurier, R. B. & Chen, J. D. Activation and binding of peroxisome proliferator-activated receptor γ by synthetic cannabinoid ajulemic acid. Mol. Pharmacol. 63, 983–992 (2003). Cannabinoid-based drugs might function through mechanisms other than binding cannabinoid receptors, such as through binding PPAR-γ, as described in this paper.
Piomelli, D. The molecular logic of endocannabinoid signalling. Nature Rev. Neurosci. 4, 873–884 (2003). Cannabinoids are best known for their psychoactive effects on the brain. This Review, together with references 28 and 29, discusses these mechanisms in detail.
Sugiura, T., Kobayashi, Y., Oka, S. & Waku, K. Biosynthesis and degradation of anandamide and 2-arachidonoylglycerol and their possible physiological significance. Prostaglandins Leukot. Essent. Fatty Acids 66, 173–192 (2002).
Devane, W. A. et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258, 1946–1949 (1992). An important breakthrough in the field was the discovery of an endocannabinoid. This paper describes the isolation of the first endocannabinoid to be found, AEA (previously known as anandamide).
Zygmunt, P. M. et al. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature 400, 452–457 (1999).
Mechoulam, R. et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharm. 50, 83–90 (1995).
Hanus, L. et al. 2-Arachidonyl glyceryl ether, an endogenous agonist of the cannabinoid CB1 receptor. Proc. Natl Acad. Sci. USA 98, 3662–3665 (2001).
Matsuda, L. A., Lolait, S. J., Brownstein, M. J., Young, A. C. & Bonner, T. I. Structure of cannabinoid receptor and functional expression of the cloned cDNA. Nature 346, 561–564 (1990).
Munro, S., Thomas, K. L. & Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 365, 61–65 (1993). References 20–23 describe the isolation of endocannabinoids that were discovered subsequently to AEA and the isolation of the cannabinoid receptors, CB 1 and CB 2.
McAllister, S. D. & Glass, M. CB1 and CB2 receptor-mediated signalling: a focus on endocannabinoids. Prostaglandins Leukot. Essent. Fatty Acids 66, 161–171 (2002).
Klein, T. W. et al. The cannabinoid system and immune modulation. J. Leukoc. Biol. 74, 486–496 (2003).
Klein, T., Newton, C. & Friedman, H. Cannabinoid receptors and immunity. Immunol. Today 19, 373–381 (1998).
Berdyshev, E. V. Cannabinoid receptors and the regulation of immune response. Chem. Phys. Lipids 108, 169–190 (2000).
Schlicker, E. & Kathmann, M. Modulation of transmitter release via presynaptic cannabinoid receptors. Trends Pharmacol. Sci. 22, 565–572 (2001).
Christie, M. J. & Vaughan, C. W. Cannabinoids act backwards. Nature 410, 527–530 (2001).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nature Rev. Immunol. 4, 499–511 (2004).
Noverr, M. C., Erb-Downward, J. R. & Huffnagle, G. B. Production of eicosanoids and other oxylipins by pathogenic eukaryotic microbes. Clin. Microbiol. Rev. 16, 517–533 (2003).
DiMarzo, V., DePetrocellis, L., Sepe, N. & Buono, A. Biosynthesis of anandamide and related acylethanolamides in mouse J774 macrophages and N18 neuroblastoma cells. Biochem. J. 316, 977–984 (1996).
DiMarzo, V. et al. Biosynthesis and inactivation of the endocannabinoid 2-arachidonoylglycerol in circulating and tumoral macrophages. Eur. J. Biochem. 264, 258–267 (1999).
Maccarrone, M., Bari, M., Battista, N. & Finazzi-Agro, A. Endocannabinoid degradation, endotoxic shock and inflammation. Curr. Drug Targets Inflamm. Allergy 1, 53–63 (2002).
Matias, I. et al. Presence and regulation of the endocannabinoid system in human dendritic cells. Eur. J. Biochem. 269, 3771–3778 (2002).
Maccarrone, M. et al. Lipopolysaccharide downregulates fatty acid amide hydrolase expression and increases anandamide levels in human peripheral lymphocytes. Arch. Biochem. Biophys. 393, 321–328 (2001). References 32–36 discuss the metabolism of endocannabinoids in immune cells.
Wyss-Coray, T. & Mucke, L. Inflammation in neurodegenerative disease — a double-edged sword. Neuron 35, 419–432 (2002).
Moser, B., Wolf, M., Walz, A. & Loetscher, P. Chemokines: multiple levels of leukocyte migration control. Trends Immunol. 25, 75–84 (2004).
Oka, S. et al. 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, induces the migration of EoL-1 human eosinophilic leukemia cells and human peripheral blood eosinophils. J. Leukoc. Biol. 76, 1002–1009 (2004).
Rayman, N. et al. Distinct expression profiles of the peripheral cannabinoid receptor in lymphoid tissues depending on receptor activation status. J. Immunol. 172, 2111–2117 (2004).
Maestroni, G. J. The endogenous cannabinoid 2-arachidonoyl glycerol as in vivo chemoattractant for dendritic cells and adjuvant for TH1 response to a soluble protein. FASEB J. 18, 1914–1916 (2004).
Alberich Jorda, M. et al. The peripheral cannabinoid receptor Cb2, frequently expressed on AML blasts, either induces a neutrophilic differentiation block or confers abnormal migration properties in a ligand-dependent manner. Blood 104, 526–534 (2004). The many putative roles of cannabinoid receptors in immune-cell biology remain undefined. This paper indicates that these receptors might have a role in leukaemia.
Szabo, I. et al. Heterologous desensitization of opioid receptors by chemokines inhibits chemotaxis and enhances the perception of pain. Proc. Natl Acad. Sci. USA 99, 10276–10281 (2002).
Lee, S. F., Newton, C., Widen, R., Friedman, H. & Klein, T. W. Differential expression of cannabinoid CB2 receptor mRNA in mouse immune cell subpopulations and following B cell stimulation. Eur. J. Pharmacol. 423, 235–241 (2001).
Daaka, Y., Friedman, H. & Klein, T. W. Cannabinoid receptor proteins are increased in Jurkat, human T-cell line after mitogen activation. J. Pharmacol. Exp. Ther. 276, 776–783 (1996). This report was among the first to show that cannabinoid-receptor expression changes with variations in immune-cell activation.
Noe, S. N., Newton, C., Widen, R., Friedman, H. & Klein, T. W. Anti-CD40, anti-CD3, and IL-2 stimulation induce contrasting changes in CB1 mRNA expression in mouse splenocytes. J. Neuroimmunol. 110, 161–167 (2000).
Gardner, B. et al. Autocrine and paracrine regulation of lymphocyte CB2 receptor expression by TGF-β. Biochem. Biophys. Res. Commun. 290, 91–96 (2002).
Klein, T. W., Newton, C., Zhu, W., Daaka, Y. & Friedman, H. Δ9-Tetrahydrocannabinol, cytokines and immunity to Legionella pneumophila. Proc. Soc. Exp. Biol. Med. 209, 205–212 (1995).
Carlisle, S., Marciano-Cabral, F., Staab, A., Ludwick, C. & Cabral, G. Differential expression of the CB2 cannabinoid receptor by rodent macrophages and macrophage-like cells in relation to cell activation. Int. Immunopharmacol. 2, 69–82 (2002).
Walter, L. et al. Nonpsychotropic cannabinoid receptors regulate microglial cell migration. J. Neurosci. 23, 1398–1405 (2003). Although CB 2 has been reported to be expressed in the brain, this was the first report to indicate that it might be expressed by brain microglial cells.
Bromley, S. K. et al. The immunological synapse. Annu. Rev. Immunol. 19, 375–396 (2001).
Blanchard, D. K., Newton, C., Klein, T. W., Stewart, W. E. & Friedman, H. In vitro and in vivo suppressive effects of Δ9-tetrahydrocannabinol on interferon production by murine spleen cells. Int. J. Immunopharmacol. 8, 819–824 (1986).
Cabral, G. A., Lockmuller, J. C. & Mishkin, E. M. Δ9-Tetrahydrocannabinol decreases α/β interferon response to herpes simplex virus type 2 in the B6C3F1 mouse. Proc. Soc. Exp. Biol. Med. 181, 305–311 (1986).
Klein, T. W., Lane, B., Newton, C. A. & Friedman, H. The cannabinoid system and cytokine network. Proc. Soc. Exp. Biol. Med. 225, 1–8 (2000).
Smith, S. R., Terminelli, C. & Denhardt, G. Effects of cannabinoid receptor agonist and antagonist ligands on production of inflammatory cytokines and anti-inflammatory interleukin-10 in endotoxemic mice. J. Pharmacol. Exp. Ther. 293, 136–150 (2000).
Shohami, E., Gallily, R., Mechoulam, R., Bass, R. & Ben-Hur, T. Cytokine production in the brain following closed head injury: dexanabinol (HU-211) is a novel TNF-α inhibitor and an effective neuroprotectant. J. Neuroimmunol. 72, 169–177 (1997). Using an animal model of closed head injury, this paper shows that non-psychoactive cannabinoids can suppress the production of pro-inflammatory cytokines.
Doble, A. The role of excitotoxicity in neurodegenerative disease: implications for therapy. Pharmacol. Ther. 81, 163–221 (1999).
Di Filippo, C., Rossi, F., Rossi, S. & D'Amico, M. Cannabinoid CB2 receptor activation reduces mouse myocardial ischemia–reperfusion injury: involvement of cytokine/chemokines and PMN. J. Leukoc. Biol. 75, 453–459 (2004).
Baldwin, G. C. et al. Marijuana and cocaine impair alveolar macrophage function and cytokine production. Am. J. Respir. Crit. Care Med. 156, 1606–1613 (1997). This is one of the few reports that shows a direct suppressive effect of marijuana smoking on immune-cell function.
Klein, T. W., Newton, C., Widen, R. & Friedman, H. Δ9-Tetrahydrocannabinol injection induces cytokine-mediated mortality of mice infected with Legionella pneumophila. J. Pharmacol. Exp. Ther. 267, 635–640 (1993).
Zhu, W., Newton, C., Daaka, Y., Friedman, H. & Klein, T. W. Δ9-Tetrahydrocannabinol enhances the secretion of interleukin 1 from endotoxin-stimulated macrophages. J. Pharmacol. Exp. Ther. 270, 1334–1339 (1994).
Smith, S. R., Terminelli, C. & Denhardt, G. Modulation of cytokine responses in Corynebacterium parvum-primed endotoxemic mice by centrally administered cannabinoid ligands. Eur. J. Pharmacol. 425, 73–83 (2001).
Derocq, J. M. et al. Genomic and functional changes induced by the activation of the peripheral cannabinoid receptor CB2 in the promyelocytic cells HL-60. Possible involvement of the CB2 receptor in cell differentiation. J. Biol. Chem. 275, 15621–15628 (2000).
Kishimoto, S. et al. 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, induces accelerated production of chemokines in HL-60 cells. J. Biochem. (Tokyo) 135, 517–524 (2004).
Pryce, G. et al. Cannabinoids inhibit neurodegeneration in models of multiple sclerosis. Brain 126, 2191–2202 (2003).
Ni, X. et al. WIN 55212-2, a cannabinoid receptor agonist, attenuates leukocyte/endothelial interactions in an experimental autoimmune encephalomyelitis model. Mult. Scler. 10, 158–164 (2004).
Kawai, T. et al. Selective diapedesis of TH1 cells induced by endothelial cell RANTES. J. Immunol. 163, 3269–3278 (1999).
Savinov, A. Y., Wong, F. S. & Chervonsky, A. V. IFN-γ affects homing of diabetogenic T cells. J. Immunol. 167, 6637–6643 (2001).
Cabral, G. & Dove Pettit, D. Drugs and immunity: cannabinoids and their role in decreased resistance to infectious diseases. J. Neuroimmunol. 83, 116–123 (1998).
Newton, C. A., Klein, T. W. & Friedman, H. Secondary immunity to Legionella pneumophila and TH1 activity are suppressed by Δ9-tetrahydrocannabinol injection. Infect. Immun. 62, 4015–4020 (1994). This was the first report to show that cannabinoids bias the immune response away from T H 1-cell responses.
Klein, T. W., Newton, C. A., Nakachi, N. & Friedman, H. Δ9-Tetrahydrocannabinol treatment suppresses immunity and early IFNγ, IL-12, and IL-12 receptor β2 responses to Legionella pneumophila infection. J. Immunol. 164, 6461–6466 (2000).
Braun, M. C. & Kelsall, B. L. Regulation of interleukin-12 production by G-protein-coupled receptors. Microbes Infect. 3, 99–107 (2001).
Zhu, L. X. et al. Δ9-Tetrahydrocannabinol inhibits antitumor immunity by a CB2 receptor-mediated, cytokine-dependent pathway. J. Immunol. 165, 373–380 (2000).
Piccirillo, C. A. & Thornton, A. M. Cornerstone of peripheral tolerance: naturally occurring CD4+CD25+ regulatory T cells. Trends Immunol. 25, 374–380 (2004).
Weiner, H. L. Induction and mechanism of action of transforming growth factor-β-secreting TH3 regulatory cells. Immunol. Rev. 182, 207–214 (2001).
Yuan, M. et al. Δ9-Tetrahydrocannabinol regulates TH1/TH2 cytokine balance in activated human T cells. J. Neuroimmunol. 133, 124–131 (2002).
Pacifici, R. et al. Modulation of the immune system in cannabis users. JAMA 289, 1929–1931 (2003). This report provided the first evidence from a human study to support the findings in animals that indicate that marijuana-derived cannabinoids bias T H -cell responses.
Croxford, J. L. & Miller, S. D. Immunoregulation of a viral model of multiple sclerosis using the synthetic cannabinoid R(+)WIN55,212. J. Clin. Invest. 111, 1231–1240 (2003).
Arevalo-Martin, A., Vela, J. M., Molina-Holgado, E., Borrell, J. & Guaza, C. Therapeutic action of cannabinoids in a murine model of multiple sclerosis. J. Neurosci. 23, 2511–2516 (2003).
Das, J. et al. A critical role for NF-κB in Gata3 expression and TH2 differentiation in allergic airway inflammation. Nature Immunol. 2, 45–50 (2001).
Klein, T. W. et al. Cannabinoid receptors and T helper biasing. J. Neuroimmunol. 147, 91–94 (2004).
Herring, A. C. & Kaminski, N. E. Cannabinol-mediated inhibition of nuclear factor-κB, cAMP response element-binding protein, and interleukin-2 secretion by activated thymocytes. J. Pharmacol. Exp. Ther. 291, 1156–1163 (1999).
Foulds, J., Burke, M., Steinberg, M., Williams, J. M. & Ziedonis, D. M. Advances in pharmacotherapy for tobacco dependence. Expert Opin. Emerg. Drugs 9, 39–53 (2004).
Howlett, A. C. et al. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology 47, 345–358 (2004).
Baker, D. et al. Cannabinoids control spasticity and tremor in a multiple sclerosis model. Nature 404, 84–87 (2000). The central effects of cannabinoids that bind CB 1 might have a role in controlling spasticity and tremor in individuals with multiple sclerosis, as shown in animals with chronic, relapsing EAE.
De Petrocellis, L. et al. The endogenous cannabinoid anandamide inhibits human breast cancer cell proliferation. Proc. Natl Acad. Sci. USA 95, 8375–8380 (1998).
Galve-Roperh, I. et al. Anti-tumoral action of cannabinoids: involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation. Nature Med. 6, 313–319 (2000).
Bifulco, M. et al. A new strategy to block tumor growth by inhibiting endocannabinoid inactivation. FASEB J. 18, 1606–1608 (2004). In this paper, a novel mechanism is reported for endocannabinoids in the inhibition of tumour growth.
Lyman, W. D., Sonett, J. R., Brosnan, C. F., Elkin, R. & Bornstein, M. B. Δ9-Tetrahydrocannabinol: a novel treatment for experimental autoimmune encephalomyelitis. J. Neuroimmunol. 23, 73–81 (1989).
Wirguin, I. et al. Suppression of experimental autoimmune encephalomyelitis by cannabinoids. Immunopharmacology 28, 209–214 (1994).
Newton, C. A. et al. The THC-induced suppression of TH1 polarization in response to Legionella pneumophila infection is not mediated by increases in corticosterone and PGE2 . J. Leukoc. Biol. 76, 854–861 (2004).
Kingery, W. S. A critical review of controlled clinical trials for peripheral neuropathic pain and complex regional pain syndromes. Pain 73, 123–139 (1997).
Calignano, A., La Rana, G., Giuffrida, A. & Piomelli, D. Control of pain initiation by endogenous cannabinoids. Nature 394, 277–281 (1998).
Ibrahim, M. M. et al. Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc. Natl Acad. Sci. USA 100, 10529–10533 (2003). This study shows that cannabinoids might inhibit pain by binding CB 2 , as well as CB 1.
Svendsen, K. B., Jensen, T. S. & Bach, F. W. Does the cannabinoid dronabinol reduce central pain in multiple sclerosis? Randomised double blind placebo controlled crossover trial. BMJ 329, 253 (2004).
Baker, D. et al. Endocannabinoids control spasticity in a multiple sclerosis model. Faseb J. 15, 300–302 (2001).
Zajicek, J. et al. Cannabinoids for treatment of spasticity and other symptoms related to multiple sclerosis (CAMS study): multicentre randomised placebo-controlled trial. Lancet 362, 1517–1526 (2003).
Vaney, C. et al. Efficacy, safety and tolerability of an orally administered cannabis extract in the treatment of spasticity in patients with multiple sclerosis: a randomized, double-blind, placebo-controlled, crossover study. Mult. Scler. 10, 417–424 (2004).
Wade, D. T., Makela, P., Robson, P., House, H. & Bateman, C. Do cannabis-based medicinal extracts have general or specific effects on symptoms in multiple sclerosis? A double-blind, randomized, placebo-controlled study on 160 patients. Mult. Scler. 10, 434–441 (2004).
Panikashvili, D. et al. An endogenous cannabinoid (2-AG) is neuroprotective after brain injury. Nature 413, 527–531 (2001).
Knoller, N. et al. Dexanabinol (HU-211) in the treatment of severe closed head injury: a randomized, placebo-controlled, Phase II clinical trial. Crit. Care Med. 30, 548–554 (2002).
Pharmos Corporation. Dexanabinol did not demonstrate efficacy [online], http://www.pharmoscorp.com/news/pr/pr122004.html (2004).
Molina-Holgado, F. et al. Endogenous interleukin-1 receptor antagonist mediates anti-inflammatory and neuroprotective actions of cannabinoids in neurons and glia. J. Neurosci. 23, 6470–6474 (2003).
Manara, L. et al. Functional assessment of neuronal cannabinoid receptors in the muscular layers of human ileum and colon. Dig. Liver Dis. 34, 262–269 (2002).
Izzo, A. A. et al. Cannabinoid CB1-receptor mediated regulation of gastrointestinal motility in mice in a model of intestinal inflammation. Br. J. Pharmacol. 134, 563–570 (2001).
Pinto, L. et al. Endocannabinoids as physiological regulators of colonic propulsion in mice. Gastroenterology 123, 227–234 (2002).
Mathison, R., Ho, W., Pittman, Q. J., Davison, J. S. & Sharkey, K. A. Effects of cannabinoid receptor-2 activation on accelerated gastrointestinal transit in lipopolysaccharide-treated rats. Br. J. Pharmacol. 142, 1247–1254 (2004).
Massa, F. et al. The endogenous cannabinoid system protects against colonic inflammation. J. Clin. Invest. 113, 1202–1209 (2004). The endocannabinoid system might control and regulate intestinal inflammatory responses, as reported in this paper.
Dajani, E. Z. et al. 1′,1′-Dimethylheptyl-Δ8-tetrahydrocannabinol-11-oic acid: a novel, orally effective cannabinoid with analgesic and anti-inflammatory properties. J. Pharmacol. Exp. Ther. 291, 31–38 (1999).
Zurier, R. B., Rossetti, R. G., Burstein, S. H. & Bidinger, B. Suppression of human monocyte interleukin-1β production by ajulemic acid, a nonpsychoactive cannabinoid. Biochem. Pharmacol. 65, 649–655 (2003).
Burstein, S. & Zurier, R. B. Pain reduction and lack of psychotropic effects with ajulemic acid. Arthritis Rheum. 50, 4078–4079 (2004).
Malfait, A. M. et al. The nonpsychoactive cannabis constituent cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis. Proc. Natl Acad. Sci. USA 97, 9561–9566 (2000).
Gallily, R. et al. γ-Irradiation enhances apoptosis induced by cannabidiol, a non-psychotropic cannabinoid, in cultured HL-60 myeloblastic leukemia cells. Leuk. Lymphoma 44, 1767–1773 (2003).
Sumariwalla, P. F. et al. A novel synthetic, nonpsychoactive cannabinoid acid (HU-320) with antiinflammatory properties in murine collagen-induced arthritis. Arthritis Rheum. 50, 985–998 (2004). References 112–114 use animal models to show the anti-inflammatory potential of non-psychoactive cannabinoids.
Varga, K., Wagner, J. A., Bridgen, D. T. & Kunos, G. Platelet- and macrophage-derived endogenous cannabinoids are involved in endotoxin-induced hypotension. Faseb J. 12, 1035–1044 (1998).
Wang, Y. et al. Simultaneous measurement of anandamide and 2-arachidonoylglycerol by polymyxin B-selective adsorption and subsequent high-performance liquid chromatography analysis: increase in endogenous cannabinoids in the sera of patients with endotoxic shock. Anal. Biochem. 294, 73–82 (2001).
Gallily, R. et al. Protection against septic shock and suppression of tumor necrosis factor α and nitric oxide production by dexanabinol (HU-211), a nonpsychotropic cannabinoid. J. Pharmacol. Exp. Ther. 283, 918–924 (1997).
Veldhuis, W. B. et al. Neuroprotection by the endogenous cannabinoid anandamide and arvanil against in vivo excitotoxicity in the rat: role of vanilloid receptors and lipoxygenases. J. Neurosci. 23, 4127–4133 (2003).
Breivogel, C. S., Griffin, G., Di Marzo, V. & Martin, B. R. Evidence for a new G protein-coupled cannabinoid receptor in mouse brain. Mol. Pharmacol. 60, 155–163 (2001). This paper indicates that there might be more than two cannabinoid receptors.
Biswas, K. K. et al. Membrane cholesterol but not putative receptors mediates anandamide-induced hepatocyte apoptosis. Hepatology 38, 1167–1177 (2003). This paper shows that lipid rafts might be affected by cannabinoid-based drugs.
Vogt, A. B., Spindeldreher, S. & Kropshofer, H. Clustering of MHC–peptide complexes prior to their engagement in the immunological synapse: lipid raft and tetraspan microdomains. Immunol. Rev. 189, 136–151 (2002).
Fischer-Stenger, K., Dove Pettit, D. A. & Cabral, G. A. Δ9-Tetrahydrocannabinol inhibition of tumor necrosis factor-α: suppression of post-translational events. J. Pharmacol. Exp. Ther. 267, 1558–1565 (1993).
Chang, Y. H., Lee, S. T. & Lin, W. W. Effects of cannabinoids on LPS-stimulated inflammatory mediator release from macrophages: involvement of eicosanoids. J. Cell. Biochem. 81, 715–723 (2001).
Facchinetti, F., Del Giudice, E., Furegato, S., Passarotto, M. & Leon, A. Cannabinoids ablate release of TNFα in rat microglial cells stimulated with lipopolysaccharide. Glia 41, 161–168 (2003).
Acknowledgements
I express sincere appreciation of H. Friedman and C. Newton for years of collaboration, resulting in many novel findings. I also thank the National Institute on Drug Abuse (United States) and the National Institute of Allergy and Infectious Diseases (United States) for continued support.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Glossary
- CANNABINOID RECEPTORS
-
G-protein-coupled receptors for Δ9-tetrahydrocannabinol, its synthetic analogues and endocannabinoids. They have been identified in most vertebrate phyla. Two subtypes are known: cannabinoid receptor 1 (CB1) and CB2.
- ENDOCANNABINOIDS
-
Endogenous agonists for cannabinoid receptors that are present in animals. They are metabolites of eicosanoid fatty acids.
- CANNABIMIMETIC
-
Δ9-Tetrahydrocannabinol (THC)-like in pharmacological terms. A compound is usually accepted to be cannabimimetic if it produces four characteristic effects of THC in an in vivo assay known as the 'mouse tetrad model'. These effects are hypomotility, hypothermia, analgesia and a sustained immobility of posture (catalepsy).
- NMDA RECEPTOR
-
(N-methyl-D-aspartate receptor). NMDA is a synthetic amino acid with affinity for NMDA receptors, which mediate excitatory effects in the brain when they are stimulated by endogenous ligands such as glutamic acid. Overstimulation can lead to neuronal excitotoxicity.
- VANILLOID RECEPTORS
-
Cation channels that are expressed by nerve sensory fibres and are involved in the perception of pain. These receptors are ligand-, proton- and heat-activated and are targets for capsaicin — the hot component of chillies.
- DIAPEDESIS
-
The last step in the leukocyte–endothelial-cell adhesion cascade. This cascade includes tethering, triggering, tight adhesion and transmigration. Diapedesis is the migration of leukocytes across the endothelium, which occurs by squeezing through the junctions between adjacent endothelial cells.
- GLUTAMATERGIC SYNAPTIC TRANSMISSION
-
Glutamic acid is the main excitatory transmitter in the central nervous system, where it mediates fast synaptic transmission. It is released from the terminal of a glutamatergic nerve, crosses the synaptic cleft and acts on postsynaptic receptors.
- AIR-POUCH INFLAMMATORY RESPONSE
-
An experimental model of acute inflammation. Skin pouches are established on the backs of mice, by subcutaneous injection of air on several consecutive days. Subsequently, inflammation is induced by injection of interleukin-1β and tumour-necrosis factor into the pouch cavity.
- INDOMETHACIN
-
A cyclooxygenase inhibitor and thereby a non-steroidal anti-inflammatory drug.
Rights and permissions
About this article
Cite this article
Klein, T. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat Rev Immunol 5, 400–411 (2005). https://doi.org/10.1038/nri1602
Issue Date:
DOI: https://doi.org/10.1038/nri1602
This article is cited by
-
Anti-inflammatory effects of recreational marijuana in virally suppressed youth with HIV-1 are reversed by use of tobacco products in combination with marijuana
Retrovirology (2022)
-
New AKT-dependent mechanisms of anti-COVID-19 action of high-CBD Cannabis sativa extracts
Cell Death Discovery (2022)
-
Cannabinoid receptors distribution in mouse cortical plasma membrane compartments
Molecular Brain (2021)
-
Cannabinoids, the Endocannabinoid System, and Cognitive Functions: Enemies or Friends?
Neuroscience and Behavioral Physiology (2021)
-
Endocannabinoid system and its modulation of brain, gut, joint and skin inflammation
Molecular Biology Reports (2021)