Abstract
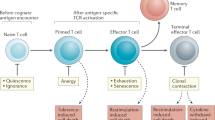
T cells constitute a heterogeneous, hierarchically organized population, comprising several maturation/differentiation states that have different capacities for clonal expansion and self-renewal. Here, we argue that the relative probabilities of proliferation, differentiation and death — the cellular events that determine the population's structure, as well as its size — are not entirely pre-programmed or fixed; instead, these events are regulated dynamically through the recurrent interaction of lymphocytes with exogenous and endogenous antigens, antigen-presenting cells and each other.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Marrack, P. et al. Homeostasis of αβ-TCR+ T cells. Nature Immunol. 1, 107–111 (2000).
Tanchot, C. & Rocha, B. The organization of mature T-cell pools. Immunol. Today 19, 575–579 (1998).
Freitas, A. A. & Rocha, B. Peripheral T cell survival. Curr. Opin. Immunol. 11, 152–156 (1999).
Freitas, A. A. & Rocha, B. Population biology of lymphocytes: the flight for survival. Annu. Rev. Immunol. 18, 83–111 (2000).
Selin, L. K. et al. Attrition of T cell memory: selective loss of LCMV epitope-specific memory CD8 T cells following infections with heterologous viruses. Immunity 11, 733–742 (1999).
Flynn, K. J. et al. Virus-specific CD8+ T cells in primary and secondary influenza pneumonia. Immunity 8, 683–691 (1998).
Blattman, J. N. et al. Estimating the precursor frequency of naive antigen-specific CD8 T cells. J. Exp. Med. 195, 657–664 (2002).
Jenkins, M. K. et al. In vivo activation of antigen-specific CD4 T cells. Annu. Rev. Immunol. 19, 23–45 (2001).
Grayson, J. M., Harrington, L. E., Lanier, J. G., Wherry, E. J. & Ahmed, R. Differential sensitivity of naive and memory CD8+ T cells to apoptosis in vivo. J. Immunol. 169, 3760–3770 (2002).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Champagne, P. et al. Skewed maturation of memory HIV-specific CD8 T lymphocytes. Nature 410, 106–111 (2001).
Brenchley, J. M. et al. Expression of CD57 defines replicative senescence and antigen-induced apoptotic death of CD8+ T cells. Blood 101, 2711–2720 (2003).
Opferman, J. T., Ober, B. T. & Ashton-Rickardt, P. G. Linear differentiation of cytotoxic effectors into memory T lymphocytes. Science 283, 1745–1748 (1999).
Roman, E. et al. CD4 effector T cell subsets in the response to influenza: heterogeneity, migration, and function. J. Exp. Med. 196, 957–968 (2002).
Wherry, E. J. et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nature Immunol. 4, 225–234 (2003).
van Lier, R. A., ten Berge, I. J. & Gamadia, L. E. Human CD8+ T-cell differentiation in response to viruses. Nature Rev. Immunol. 3, 931–939 (2003).
Hayashi, N., Liu, D., Min, B., Ben-Sasson, S. Z. & Paul, W. E. Antigen challenge leads to in vivo activation and elimination of highly polarized TH1 memory T cells. Proc. Natl Acad. Sci. USA 99, 6187–6191 (2002).
Ben-Sasson, S. Z. et al. Generation and characterization of memory CD4 T cells. Adv. Exp. Med. Biol. 512, 129–134 (2002).
Wu, C. Y. et al. Distinct lineages of TH1 cells have differential capacities for memory cell generation in vivo. Nature Immunol. 3, 852–858 (2002).
Geginat, J., Lanzavecchia, A. & Sallusto, F. Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines. Blood 101, 4260–4266 (2003).
Richter, A., Lohning, M. & Radbruch, A. Instruction for cytokine expression in T helper lymphocytes in relation to proliferation and cell cycle progression. J. Exp. Med. 190, 1439–1450 (1999).
Laouar, Y. & Crispe, I. N. Functional flexibility in T cells: independent regulation of CD4+ T cell proliferation and effector function in vivo. Immunity 13, 291–301 (2000).
Ben-Sasson, S. Z., Gerstel, R., Hu-Li, J. & Paul, W. E. Cell division is not a 'clock' measuring acquisition of competence to produce IFN-γ or IL-4. J. Immunol. 166, 112–120 (2001).
Auphan-Anezin, N., Verdeil, G. & Schmitt-Verhulst, A. M. Distinct thresholds for CD8 T cell activation lead to functional heterogeneity: CD8 T cell priming can occur independently of cell division. J. Immunol. 170, 2442–2448 (2003).
Grossman, Z. Recognition of self, balance of growth and competition: horizontal networks regulate immune responsiveness. Eur. J. Immunol. 12, 747–756 (1982).
Grossman, Z. The stem cell concept revisited: self-renewal capacity is a dynamic property of hemopoietic cells. Leuk. Res. 10, 937–950 (1986).
Grossman, Z. Cellular tolerance as a dynamic state of the adaptable lymphocyte. Immunol. Rev. 133, 45–73 (1993).
Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
Lanzavecchia, A., Lezzi, G. & Viola, A. From TCR engagement to T cell activation: a kinetic view of T cell behavior. Cell 96, 1–4 (1999).
Langenkamp, A., Messi, M., Lanzavecchia, A. & Sallusto, F. Kinetics of dendritic cell activation: impact on priming of TH1, TH2 and nonpolarized T cells. Nature Immunol. 1, 311–316 (2000).
Wang, X. & Mosmann, T. In vivo priming of CD4 T cells that produce interleukin (IL)-2 but not IL-4 or interferon (IFN)-γ, and can subsequently differentiate into IL-4- or IFN-γ-secreting cells. J. Exp. Med. 194, 1069–1080 (2001).
Veiga-Fernandes, H., Walter, U., Bourgeois, C., McLean, A. & Rocha, B. Response of naive and memory CD8+ T cells to antigen stimulation in vivo. Nature Immunol. 1, 47–53 (2000).
Morrison, S. J., Uchida, N. & Weissman, I. L. The biology of hematopoietic stem cells. Annu. Rev. Cell Dev. Biol. 11, 35–71 (1995).
Blau, H. M., Brazelton, T. R. & Weimann, J. M. The evolving concept of a stem cell: entity or function? Cell 105, 829–841 (2001).
Badovinac, V. P., Porter, B. B. & Harty, J. T. Programmed contraction of CD8+ T cells after infection. Nature Immunol. 3, 619–626 (2002).
Blattman, J. N., Cheng, L. E. & Greenberg, P. D. CD8+ T cell responses: it's all downhill after their prime. Nature Immunol. 3, 601–602 (2002).
Badovinac, V. P., Messingham, K. A., Hamilton, S. E. & Harty, J. T. Regulation of CD8+ T cells undergoing primary and secondary responses to infection in the same host. J. Immunol. 170, 4933–4942 (2003).
Grossman, Z. Recognition of self and regulation of specificity at the level of cell populations. Immunol. Rev. 79, 119–138 (1984).
Grossman, Z. & Paul, W. E. Autoreactivity, dynamic tuning and selectivity. Curr. Opin. Immunol. 13, 687–698 (2001).
Butz, E. A. & Bevan, M. J. Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8, 167–175 (1998).
Kedl, R. M. et al. T cells compete for access to antigen-bearing antigen-presenting cells. J. Exp. Med. 192, 1105–1113 (2000).
Kedl, R. M., Schaefer, B. C., Kappler, J. W. & Marrack, P. T cells down-modulate peptide–MHC complexes on APCs in vivo. Nature Immunol. 3, 27–32 (2002).
Kedl, R. M., Kappler, J. W. & Marrack, P. Epitope dominance, competition and T cell affinity maturation. Curr. Opin. Immunol. 15, 120–127 (2003).
Wong, P. & Pamer, E. G. Feedback regulation of pathogen-specific T cell priming. Immunity 18, 499–511 (2003).
Shevach, E. M. Regulatory T cells in autoimmmunity. Annu. Rev. Immunol. 18, 423–449 (2000).
Maloy, K. J. & Powrie, F. Regulatory T cells in the control of immune pathology. Nature Immunol. 2, 816–822 (2001).
Kaech, S. M. & Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nature Immunol. 2, 415–422 (2001).
van Stipdonk, M. J., Lemmens, E. E. & Schoenberger, S. P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nature Immunol. 2, 423–429 (2001).
Bird, J. J. et al. Helper T cell differentiation is controlled by the cell cycle. Immunity 9, 229–237 (1998).
Kaech, S. M., Hemby, S., Kersh, E. & Ahmed, R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell 111, 837–851 (2002).
Grossman, Z. & Paul, W. E. Self-tolerance: context dependent tuning of T cell antigen recognition. Semin. Immunol. 12, 197–203; discussion 257–344 (2000).
Grossman, Z. & Herberman, R. B. T-cell homeostasis in HIV infection is neither failing nor blind: modified cell counts reflect an adaptive response of the host. Nature Med. 3, 486–490 (1997).
Grossman, Z. & Paul, W. E. Adaptive cellular interactions in the immune system: the tunable activation threshold and the significance of subthreshold responses. Proc. Natl Acad. Sci. USA 89, 10365–10369 (1992).
Tanchot, C., Barber, D. L., Chiodetti, L. & Schwartz, R. H. Adaptive tolerance of CD4+ T cells in vivo: multiple thresholds in response to a constant level of antigen presentation. J. Immunol. 167, 2030–2039 (2001).
Bhandoola, A. et al. Peripheral expression of self-MHC-II influences the reactivity and self-tolerance of mature CD4+ T cells: evidence from a lymphopenic T cell model. Immunity 17, 425–436 (2002).
Almeida, A. R., Legrand, N., Papiernik, M. & Freitas, A. A. Homeostasis of peripheral CD4+ T cells: IL-2Rα and IL-2 shape a population of regulatory cells that controls CD4+ T cell numbers. J. Immunol. 169, 4850–4860 (2002).
Apostolou, I., Sarukhan, A., Klein, L. & von Boehmer, H. Origin of regulatory T cells with known specificity for antigen. Nature Immunol. 3, 756–763 (2002).
Stockinger, B., Barthlott, T. & Kassiotis, G. T cell regulation: a special job or everyone's responsibility? Nature Immunol. 2, 757–758 (2001).
Barthlott, T., Kassiotis, G. & Stockinger, B. T cell regulation as a side effect of homeostasis and competition. J. Exp. Med. 197, 451–460 (2003).
Zhang, X., Sun, S., Hwang, I., Tough, D. F. & Sprent, J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 8, 591–599 (1998).
Ku, C. C., Murakami, M., Sakamoto, A., Kappler, J. & Marrack, P. Control of homeostasis of CD8+ memory T cells by opposing cytokines. Science 288, 675–678 (2000).
Jonuleit, H., Schmitt, E., Schuler, G., Knop, J. & Enk, A. H. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 192, 1213–1222 (2000).
Adorini, L., Penna, G., Giarratana, N. & Uskokovic, M. Tolerogenic dendritic cells induced by vitamin D receptor ligands enhance regulatory T cells inhibiting allograft rejection and autoimmune diseases. J. Cell. Biochem. 88, 227–233 (2003).
Qin, S. et al. 'Infectious' transplantation tolerance. Science 259, 974–977 (1993).
Harrington, L. E., Galvan, M., Baum, L. G., Altman, J. D. & Ahmed, R. Differentiating between memory and effector CD8 T cells by altered expression of cell surface O-glycans. J. Exp. Med. 191, 1241–1246 (2000).
Langenkamp, A. et al. Kinetics and expression patterns of chemokine receptors in human CD4+ T lymphocytes primed by myeloid or plasmacytoid dendritic cells. Eur. J. Immunol. 33, 474–482 (2003).
Turner, S. J., Cross, R., Xie, W. & Doherty, P. C. Concurrent naive and memory CD8+ T cell responses to an influenza A virus. J. Immunol. 167, 2753–2758 (2001).
Moskophidis, D., Lechner, F., Pircher, H. & Zinkernagel, R. M. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature 362, 758–761 (1993).
Brander, C. et al. Persistent HIV-1-specific CTL clonal expansion despite high viral burden post in utero HIV-1 infection. J. Immunol. 162, 4796–4800 (1999).
Islam, S. A. et al. Persistence of human immunodeficiency virus type 1-specific cytotoxic T-lymphocyte clones in a subject with rapid disease progression. J. Virol. 75, 4907–4911 (2001).
Douek, D. C. et al. Changes in thymic function with age and during the treatment of HIV infection. Nature 396, 690–695 (1998).
Kong, F. K., Chen, C. L., Six, A., Hockett, R. D. & Cooper, M. D. T cell receptor gene deletion circles identify recent thymic emigrants in the peripheral T cell pool. Proc. Natl Acad. Sci. USA 96, 1536–1540 (1999).
Poulin, J. F. et al. Direct evidence for thymic function in adult humans. J. Exp. Med. 190, 479–486 (1999).
Hazenberg, M. D. et al. Increased cell division but not thymic dysfunction rapidly affects the T-cell receptor excision circle content of the naive T cell population in HIV-1 infection. Nature Med. 6, 1036–1042 (2000).
Douek, D. C. et al. Evidence for increased T cell turnover and decreased thymic output in HIV infection. J. Immunol. 167, 6663–6668 (2001).
Grossman, Z. & Singer, A. Tuning of activation thresholds explains flexibility in the selection and development of T cells in the thymus. Proc. Natl Acad. Sci. USA 93, 14747–14752 (1996).
Starr, T. K., Jameson, S. C. & Hogquist, K. A. Positive and negative selection of T cells. Annu. Rev. Immunol. 21, 139–176 (2003).
Jameson, S. C. Maintaining the norm: T-cell homeostasis. Nature Rev. Immunol. 2, 547–556 (2002).
Min, B. et al. Neonates support lymphopenia-induced proliferation. Immunity 18, 131–140 (2003).
Kieper, W. C. & Jameson, S. C. Homeostatic expansion and phenotypic conversion of naive T cells in response to self peptide–MHC ligands. Proc. Natl Acad. Sci. USA 96, 13306–13311 (1999).
Ge, Q., Rao, V. P., Cho, B. K., Eisen, H. N. & Chen, J. Dependence of lymphopenia-induced T cell proliferation on the abundance of peptide–MHC epitopes and strength of their interaction with T cell receptors. Proc. Natl Acad. Sci. USA 98, 1728–1733 (2001).
Kassiotis, G., Zamoyska, R. & Stockinger, B. Involvement of avidity for major histocompatibility complex in homeostasis of naive and memory T cells. J. Exp. Med. 197, 1007–1016 (2003).
Min, B., Foucras, G., Meier-Schellersheim, M. & Paul, W. E. Spontaneous proliferation, a response of naïve CD4 T cells determined by the diversity of the memory cell repertoire. Proc. Natl Acad. Sci. USA 101, 3874–3879 (2004).
Moses, C. T., Thorstenson, K. M., Jameson, S. C. & Khoruts, A. Competition for self ligands restrains homeostatic proliferation of naive CD4 T cells. Proc. Natl Acad. Sci. USA 100, 1185–1190 (2003).
Troy, A. E. & Shen, H. Cutting edge: homeostatic proliferation of peripheral T lymphocytes is regulated by clonal competition. J. Immunol. 170, 672–676 (2003).
Tanchot, C. et al. Conversion of naive T cells to a memory-like phenotype in lymphopenic hosts is not related to a homeostatic mechanism that fills the peripheral naive T cell pool. J. Immunol. 168, 5042–5046 (2002).
Ge, Q., Hu, H., Eisen, H. N. & Chen, J. Different contributions of thymopoiesis and homeostasis-driven proliferation to the reconstitution of naive and memory T cell compartments. Proc. Natl Acad. Sci. USA 99, 2989–2994 (2002).
Goldrath, A. W. & Bevan, M. J. Selecting and maintaining a diverse T-cell repertoire. Nature 402, 255–262 (1999).
Kimmig, S. et al. Two subsets of naive T helper cells with distinct T cell receptor excision circle content in human adult peripheral blood. J. Exp. Med. 195, 789–794 (2002).
Berzins, S. P., Boyd, R. L. & Miller, J. F. The role of the thymus and recent thymic migrants in the maintenance of the adult peripheral lymphocyte pool. J. Exp. Med. 187, 1839–1848 (1998).
Berzins, S. P., Godfrey, D. I., Miller, J. F. & Boyd, R. L. A central role for thymic emigrants in peripheral T cell homeostasis. Proc. Natl Acad. Sci. USA 96, 9787–9791 (1999).
Di Rosa, F., Ramaswamy, S., Ridge, J. P. & Matzinger, P. On the lifespan of virgin T lymphocytes. J. Immunol. 163, 1253–1257 (1999).
Goronzy, J. J. & Weyand, C. M. Thymic function and peripheral T-cell homeostasis in rheumatoid arthritis. Trends Immunol. 22, 251–255 (2001).
Seder, R. A. & Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nature Immunol. 4, 835–842 (2003).
Sun, J. C. & Bevan, M. J. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300, 339–342 (2003).
Shedlock, D. J. & Shen, H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300, 337–339 (2003).
Bray, D. Protein molecules as computational elements in living cells. Nature 376, 307–312 (1995).
Germain, R. N. & Stefanova, I. The dynamics of T cell receptor signaling: complex orchestration and the key roles of tempo and cooperation. Annu. Rev. Immunol. 17, 467–522 (1999).
Stefanova, I. et al. TCR ligand discrimination is enforced by competing ERK positive and SHP-1 negative feedback pathways. Nature Immunol. 4, 248–254 (2003).
Mueller, D. L. Tuning the immune system: competing positive and negative feedback loops. Nature Immunol. 4, 210–211 (2003).
Bitmansour, A. D., Douek, D. C., Maino, V. C. & Picker, L. J. Direct ex vivo analysis of human CD4+ memory T cell activation requirements at the single clonotype level. J. Immunol. 169, 1207–1218 (2002).
Azzam, H. S. et al. Fine tuning of TCR signaling by CD5. J. Immunol. 166, 5464–5472 (2001).
Smith, K. et al. Sensory adaptation in naive peripheral CD4 T cells. J. Exp. Med. 194, 1253–1261 (2001).
Bitegye, C., Hannier, S., Guerif, S., Valitutti, S. & Demotz, S. Tuning of T cell clone size and activation threshold by control of CD25 expression through mitogen-activated protein kinase pathways. Int. Arch. Allergy Immunol. 127, 322–332 (2002).
Stamou, P. et al. Chronic exposure to low levels of antigen in the periphery causes reversible functional impairment correlating with changes in CD5 levels in monoclonal CD8 T cells. J. Immunol. 171, 1278–1284 (2003).
Anderton, S. M. & Wraith, D. C. Selection and fine-tuning of the autoimmune T-cell repertoire. Nature Rev. Immunol. 2, 487–498 (2002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Grossman, Z., Min, B., Meier-Schellersheim, M. et al. Concomitant regulation of T-cell activation and homeostasis. Nat Rev Immunol 4, 387–395 (2004). https://doi.org/10.1038/nri1355
Issue Date:
DOI: https://doi.org/10.1038/nri1355
This article is cited by
-
Reference Genes for Expression Studies in Human CD8+ Naïve and Effector Memory T Cells under Resting and Activating Conditions
Scientific Reports (2020)
-
Comparison of two commonly used methods for stimulating T cells
Biotechnology Letters (2019)
-
The speed of change: towards a discontinuity theory of immunity?
Nature Reviews Immunology (2013)
-
Therapeutic vaccination against autologous cancer stem cells with mRNA-transfected dendritic cells in patients with glioblastoma
Cancer Immunology, Immunotherapy (2013)
-
CD69 limits early inflammatory diseases associated with immune response to Listeria monocytogenes infection
Immunology & Cell Biology (2010)