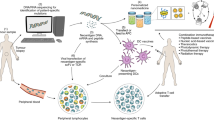
Abstract
Cancer immunotherapy can successfully promote long-term anticancer immune responses, although there is still only a limited number of patients who benefit from such treatment, and it can sometimes have severe treatment-associated adverse events. Compared with systemic immunomodulation, local immunomodulation may enable more effective treatment at lower doses and, at the same time, prevent systemic toxicity. Local delivery of engineered three-dimensional scaffolds may fulfil this role by acting as synthetic immune niches that boost anticancer immunity. In this Opinion article, we highlight the potential of scaffold-based adoptive cell transfer and scaffold-based cancer vaccines that, although applied locally, can promote systemic antitumour immunity. Furthermore, we discuss how scaffold-based cancer immunotherapy may contribute to the development of the next generation of cancer treatments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2015).
Larkin, J. et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373, 23–34 (2015).
Michot, J. M. et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur. J. Cancer 54, 139–148 (2016).
Dudley, M. E. et al. Randomized selection design trial evaluating CD8+-enriched versus unselected tumor-infiltrating lymphocytes for adoptive cell therapy for patients with melanoma. J. Clin. Oncol. 31, 2152–2159 (2013).
Stevanovic, S. et al. Complete regression of metastatic cervical cancer after treatment with human papillomavirus-targeted tumor-infiltrating T cells. J. Clin. Oncol. 33, 1543–1550 (2015).
Junker, N. et al. Bimodal ex vivo expansion of T cells from patients with head and neck squamous cell carcinoma: a prerequisite for adoptive cell transfer. Cytotherapy 13, 822–834 (2011).
Ahmed, N. et al. Human epidermal growth factor receptor 2 (HER2) -specific chimeric antigen receptor-modified T cells for the immunotherapy of HER2-Positive sarcoma. J. Clin. Oncol. 33, 1688–1696 (2015).
Rosenberg, S. A. et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 17, 4550–4557 (2011).
Rosenberg, S. A. & Restifo, N. P. Adoptive cell transfer as personalized immunotherapy for human cancer. Science 348, 62–68 (2015).
Tel, J. et al. Natural human plasmacytoid dendritic cells induce antigen-specific T-cell responses in melanoma patients. Cancer Res. 73, 1063–1075 (2013).
Schreibelt, G. et al. Effective clinical responses in metastatic melanoma patients after vaccination with primary myeloid dendritic cells. Clin. Cancer Res. 22, 2155–2166 (2016).
van der Burg, S. H., Arens, R., Ossendorp, F., van Hall, T. & Melief, C. J. Vaccines for established cancer: overcoming the challenges posed by immune evasion. Nat. Rev. Cancer 16, 219–233 (2016).
Wesley, J. D., Whitmore, J., Trager, J. & Sheikh, N. An overview of sipuleucel-T: autologous cellular immunotherapy for prostate cancer. Hum. Vaccin Immunother. 8, 520–527 (2012).
van Hooren, L. et al. Local checkpoint inhibition of CTLA-4 as a monotherapy or in combination with anti-PD1 prevents the growth of murine bladder cancer. Eur. J. Immunol. 47, 385–393 (2017).
Fransen, M. F., van der Sluis, T. C., Ossendorp, F., Arens, R. & Melief, C. J. Controlled local delivery of CTLA-4 blocking antibody induces CD8+ T-cell-dependent tumor eradication and decreases risk of toxic side effects. Clin. Cancer Res. 19, 5381–5389 (2013).
Sandin, L. C. et al. Local CTLA4 blockade effectively restrains experimental pancreatic adenocarcinoma growth in vivo. Oncoimmunology 3, e27614 (2014).
Fransen, M. F., Arens, R. & Melief, C. J. Local targets for immune therapy to cancer: tumor draining lymph nodes and tumor microenvironment. Int. J. Cancer 132, 1971–1976 (2013).
Marabelle, A., Kohrt, H., Caux, C. & Levy, R. Intratumoral immunization: a new paradigm for cancer therapy. Clin. Cancer Res. 20, 1747–1756 (2014).
Van der Jeught, K. et al. Targeting the tumor microenvironment to enhance antitumor immune responses. Oncotarget 6, 1359–1381 (2015).
Munn, D. H. & Mellor, A. L. The tumor-draining lymph node as an immune-privileged site. Immunol. Rev. 213, 146–158 (2006).
Schreiber, R. D., Old, L. J. & Smyth, M. J. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science 331, 1565–1570 (2011).
Gajewski, T. F. et al. Immune resistance orchestrated by the tumor microenvironment. Immunol. Rev. 213, 131–145 (2006).
Bhatia, A. & Kumar, Y. Cellular and molecular mechanisms in cancer immune escape: a comprehensive review. Expert Rev. Clin. Immunol. 10, 41–62 (2014).
Fridman, W. H., Pages, F., Sautes-Fridman, C. & Galon, J. The immune contexture in human tumours: impact on clinical outcome. Nat. Rev. Cancer 12, 298–306 (2012).
Lee, J. H. et al. Quantitative analysis of melanoma-induced cytokine-mediated immunosuppression in melanoma sentinel nodes. Clin. Cancer Res. 11, 107–112 (2005).
Torisu-Itakura, H. et al. Molecular characterization of inflammatory genes in sentinel and nonsentinel nodes in melanoma. Clin. Cancer Res. 13, 3125–3132 (2007).
Kohrt, H. E. et al. Profile of immune cells in axillary lymph nodes predicts disease-free survival in breast cancer. PLoS Med. 2, e284 (2005).
Gai, X. D., Li, C., Song, Y., Lei, Y. M. & Yang, B. X. In situ analysis of FOXP3 regulatory T cells and myeloid dendritic cells in human colorectal cancer tissue and tumor-draining lymph node. Biomed. Rep. 1, 207–212 (2013).
Vence, L. et al. Circulating tumor antigen-specific regulatory T cells in patients with metastatic melanoma. Proc. Natl Acad. Sci. USA 104, 20884–20889 (2007).
Diaz-Montero, C. M. et al. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol. Immunother. 58, 49–59 (2009).
Thomas, S. N., Vokali, E., Lund, A. W., Hubbell, J. A. & Swartz, M. A. Targeting the tumor-draining lymph node with adjuvanted nanoparticles reshapes the anti-tumor immune response. Biomaterials 35, 814–824 (2014).
Jeanbart, L. et al. Enhancing efficacy of anticancer vaccines by targeted delivery to tumor-draining lymph nodes. Cancer Immunol. Res. 2, 436–447 (2014).
Peggs, K. S., Quezada, S. A. & Allison, J. P. Cancer immunotherapy: co-stimulatory agonists and co-inhibitory antagonists. Clin. Exp. Immunol. 157, 9–19 (2009).
Sluijter, B. et al. Arming the melanoma SLN through local administration of CpG-B and GM-CSF: recruitment and activation of BDCA3/CD141+ DC and enhanced cross-presentation. Cancer Immunol. Res. (2015).
van den Hout, M. F. et al. Local delivery of CpG-B and GM-CSF induces concerted activation of effector and regulatory T cells in the human melanoma sentinel lymph node. Cancer Immunol. Immunother. 65, 405–415 (2016).
Andtbacka, R. H. et al. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J. Clin. Oncol. 33, 2780–2788 (2015).
Kaufman, H. L. et al. Systemic versus local responses in melanoma patients treated with talimogene laherparepvec from a multi-institutional phase II study. J. Immunother. Cancer 4, 12 (2016).
Brody, J. D. et al. In situ vaccination with a TLR9 agonist induces systemic lymphoma regression: a phase I/II study. J. Clin. Oncol. 28, 4324–4332 (2010).
Kim, Y. H. et al. In situ vaccination against mycosis fungoides by intratumoral injection of a TLR9 agonist combined with radiation: a phase 1/2 study. Blood 119, 355–363 (2012).
Sandin, L. C. et al. Locally delivered CD40 agonist antibody accumulates in secondary lymphoid organs and eradicates experimental disseminated bladder cancer. Cancer Immunol. Res. 2, 80–90 (2014).
Fransen, M. F., Sluijter, M., Morreau, H., Arens, R. & Melief, C. J. Local activation of CD8 T cells and systemic tumor eradication without toxicity via slow release and local delivery of agonistic CD40 antibody. Clin. Cancer Res. 17, 2270–2280 (2011).
Ellmark, P., Mangsbo, S. M., Furebring, C., Norlen, P. & Totterman, T. H. Tumor-directed immunotherapy can generate tumor-specific T cell responses through localized co-stimulation. Cancer Immunol. Immunother. 66, 1–7 (2017).
Fontana, F., Liu, D., Hirvonen, J. & Santos, H. A. Delivery of therapeutics with nanoparticles: what's new in cancer immunotherapy? Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. http://dx.doi.org/10.1002/wnan.1421 (2017).
Kranz, L. M. et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature 534, 396–401 (2016).
Qian, X. et al. In vivo tumor targeting and spectroscopic detection with surface-enhanced Raman nanoparticle tags. Nat. Biotechnol. 26, 83–90 (2008).
Rosalia, R. A. et al. CD40-targeted dendritic cell delivery of PLGA-nanoparticle vaccines induce potent anti-tumor responses. Biomaterials 40, 88–97 (2015).
Cruz, L. J. et al. Targeted PLGA nano- but not microparticles specifically deliver antigen to human dendritic cells via DC-SIGN in vitro. J. Control Release 144, 118–126 (2010).
Eggermont, L. J., Paulis, L. E., Tel, J. & Figdor, C. G. Towards efficient cancer immunotherapy: advances in developing artificial antigen-presenting cells. Trends Biotechnol. 32, 456–465 (2014).
Goc, J. et al. Dendritic cells in tumor-associated tertiary lymphoid structures signal a Th1 cytotoxic immune contexture and license the positive prognostic value of infiltrating CD8+ T cells. Cancer Res. 74, 705–715 (2014).
Sautes-Fridman, C. et al. Tertiary lymphoid structures in cancers: prognostic value, regulation, and manipulation for therapeutic intervention. Front. Immunol. 7, 407 (2016).
Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 3, 589–601 (2006).
Edele, F. et al. Efficiency of dendritic cell vaccination against B16 melanoma depends on the immunization route. PLoS ONE 9, e105266 (2014).
Chen, L., Fabian, K. L., Taylor, J. L. & Storkus, W. J. Therapeutic use of dendritic cells to promote the extranodal priming of anti-tumor immunity. Front. Immunol. 4, 388 (2013).
Hori, Y., Winans, A. M., Huang, C. C., Horrigan, E. M. & Irvine, D. J. Injectable dendritic cell-carrying alginate gels for immunization and immunotherapy. Biomaterials 29, 3671–3682 (2008).
Hori, Y., Stern, P. J., Hynes, R. O. & Irvine, D. J. Engulfing tumors with synthetic extracellular matrices for cancer immunotherapy. Biomaterials 30, 6757–6767 (2009).
Hori, Y., Winans, A. M. & Irvine, D. J. Modular injectable matrices based on alginate solution/microsphere mixtures that gel in situ and co-deliver immunomodulatory factors. Acta Biomater. 5, 969–982 (2009).
Verma, V. et al. Activated dendritic cells delivered in tissue compatible biomatrices induce in-situ anti-tumor CTL responses leading to tumor regression. Oncotarget 7, 39894–39906 (2016).
Tsao, C. T. et al. Thermoreversible poly(ethylene glycol)-g-chitosan hydrogel as a therapeutic T lymphocyte depot for localized glioblastoma immunotherapy. Biomacromolecules 15, 2656–2662 (2014).
Monette, A., Ceccaldi, C., Assaad, E., Lerouge, S. & Lapointe, R. Chitosan thermogels for local expansion and delivery of tumor-specific T lymphocytes towards enhanced cancer immunotherapies. Biomaterials 75, 237–249 (2016).
Stephan, S. B. et al. Biopolymer implants enhance the efficacy of adoptive T-cell therapy. Nat. Biotechnol. 33, 97–101 (2015).
Liu, Y. et al. In situ modulation of dendritic cells by injectable thermosensitive hydrogels for cancer vaccines in mice. Biomacromolecules 15, 3836–3845 (2014).
Ali, O. A., Huebsch, N., Cao, L., Dranoff, G. & Mooney, D. J. Infection-mimicking materials to program dendritic cells in situ. Nat. Mater. 8, 151–158 (2009).
Ali, O. A. et al. Biomaterial-based vaccine induces regression of established intracranial glioma in rats. Pharm. Res. 28, 1074–1080 (2011).
Ali, O. A., Lewin, S. A., Dranoff, G. & Mooney, D. J. Vaccines combined with immune checkpoint antibodies promote cytotoxic T-cell activity and tumor eradication. Cancer Immunol. Res. 4, 95–100 (2016).
Ali, O. A. et al. Identification of immune factors regulating antitumor immunity using polymeric vaccines with multiple adjuvants. Cancer Res. 74, 1670–1681 (2014).
Ali, O. A., Tayalia, P., Shvartsman, D., Lewin, S. & Mooney, D. J. Inflammatory cytokines presented from polymer matrices differentially generate and activate DCs in situ. Adv. Funct. Mater. 23, 4621–4628 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT01753089 (2017).
Kim, J. et al. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 33, 64–72 (2015).
Bencherif, S. A. et al. Injectable preformed scaffolds with shape-memory properties. Proc. Natl Acad. Sci. USA 109, 19590–19595 (2012).
Guvendiren, M., Lu, H. D. & Burdick, J. A. Shear-thinning hydrogels for biomedical applications. Soft Matter 8, 260–272 (2012).
Bencherif, S. A. et al. Injectable cryogel-based whole-cell cancer vaccines. Nat. Commun. 6, 7556 (2015).
Singh, A., Suri, S. & Roy, K. In-situ crosslinking hydrogels for combinatorial delivery of chemokines and siRNA-DNA carrying microparticles to dendritic cells. Biomaterials 30, 5187–5200 (2009).
Singh, A. et al. An injectable synthetic immune-priming center mediates efficient T-cell class switching and T-helper 1 response against B cell lymphoma. J. Control Release 155, 184–192 (2011).
Lei, C. et al. Local release of highly loaded antibodies from functionalized nanoporous support for cancer immunotherapy. J. Am. Chem. Soc. 132, 6906–6907 (2010).
Kwong, B., Gai, S. A., Elkhader, J., Wittrup, K. D. & Irvine, D. J. Localized immunotherapy via liposome-anchored Anti-CD137 + IL-2 prevents lethal toxicity and elicits local and systemic antitumor immunity. Cancer Res. 73, 1547–1558 (2013).
Kwong, B., Liu, H. & Irvine, D. J. Induction of potent anti-tumor responses while eliminating systemic side effects via liposome-anchored combinatorial immunotherapy. Biomaterials 32, 5134–5147 (2011).
Chen, M., Ouyang, H., Zhou, S., Li, J. & Ye, Y. PLGA-nanoparticle mediated delivery of anti-OX40 monoclonal antibody enhances anti-tumor cytotoxic T cell responses. Cell. Immunol. 287, 91–99 (2014).
Park, J. et al. Combination delivery of TGF-β inhibitor and IL-2 by nanoscale liposomal polymeric gels enhances tumour immunotherapy. Nat. Mater. 11, 895–905 (2012).
Xu, K. et al. Injectable hyaluronic acid-tyramine hydrogels incorporating interferon-α2a for liver cancer therapy. J. Control Release 166, 203–210 (2013).
Roeven, M. W. et al. Efficient nontoxic delivery of PD-L1 and PD-L2 siRNA into dendritic cell vaccines using the cationic lipid SAINT-18. J. Immunother. 38, 145–154 (2015).
Teo, P. Y. et al. Ovarian cancer immunotherapy using PD-L1 siRNA targeted delivery from folic acid-functionalized polyethylenimine: strategies to enhance T cell killing. Adv. Healthc Mater. 4, 1180–1189 (2015).
Pradhan, P. et al. The effect of combined IL10 siRNA and CpG ODN as pathogen-mimicking microparticles on Th1/Th2 cytokine balance in dendritic cells and protective immunity against B cell lymphoma. Biomaterials 35, 5491–5504 (2014).
Suematsu, S. & Watanabe, T. Generation of a synthetic lymphoid tissue-like organoid in mice. Nat. Biotechnol. 22, 1539–1545 (2004).
Okamoto, N., Chihara, R., Shimizu, C., Nishimoto, S. & Watanabe, T. Artificial lymph nodes induce potent secondary immune responses in naive and immunodeficient mice. J. Clin. Invest. 117, 997–1007 (2007).
Kobayashi, Y. & Watanabe, T. Gel-trapped lymphorganogenic chemokines trigger artificial tertiary lymphoid organs and mount adaptive immune responses in vivo. Front. Immunol. 7, 316 (2016).
Tang, H. et al. Facilitating T cell infiltration in tumor microenvironment overcomes resistance to PD-L1 blockade. Cancer Cell 30, 500 (2016).
Adutler-Lieber, S. et al. Engineering of synthetic cellular microenvironments: implications for immunity. J. Autoimmun 54, 100–111 (2014).
Pashuck, E. T. & Stevens, M. M. Designing regenerative biomaterial therapies for the clinic. Sci. Transl. Med. 4, 160sr4 (2012).
Purwada, A., Roy, K. & Singh, A. Engineering vaccines and niches for immune modulation. Acta Biomater. 10, 1728–1740 (2014).
Hotaling, N. A., Tang, L., Irvine, D. J. & Babensee, J. E. Biomaterial strategies for immunomodulation. Annu. Rev. Biomed. Eng. 17, 317–349 (2015).
Acknowledgements
The authors thank A.B. van Spriel for critically reviewing the manuscript. This work was supported by the Institute of Chemical Immunology (grant 024.002.009). C.G.F. is a recipient of the Netherlands Organisation for Scientific Research (NWO) Spinoza Prize, the European Research Council advanced grant PATHFINDER (269019) and KWO grant 2009–4402 of the Dutch Cancer Society.
Author information
Authors and Affiliations
Contributions
C.G.F., J.W. and J.T. contributed equally to researching the data for the article, to discussing the content and to reviewing and editing the manuscript before submission. J.W. was responsible for writing the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Weiden, J., Tel, J. & Figdor, C. Synthetic immune niches for cancer immunotherapy. Nat Rev Immunol 18, 212–219 (2018). https://doi.org/10.1038/nri.2017.89
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2017.89
This article is cited by
-
A nanoadjuvant that dynamically coordinates innate immune stimuli activation enhances cancer immunotherapy and reduces immune cell exhaustion
Nature Nanotechnology (2023)
-
Biomaterial-based platforms for tumour tissue engineering
Nature Reviews Materials (2023)
-
The immune subtypes and landscape of sarcomas
BMC Immunology (2022)
-
Short Review on Advances in Hydrogel-Based Drug Delivery Strategies for Cancer Immunotherapy
Tissue Engineering and Regenerative Medicine (2022)
-
Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity
Signal Transduction and Targeted Therapy (2021)