Key Points
-
Hepatitis E virus (HEV) causes varying disease severity among patient subpopulations: it is self-limiting in most young adults, but causes ∼30% mortality in pregnant women and leads to chronicity in immunocompromised patients
-
HEV has a broad but poorly characterized host range, and in industrialized countries it is primarily transmitted zoonotically through the consumption of undercooked meat
-
A prophylactic vaccine against HEV exists but is currently only licensed in China
-
There are currently no direct-acting therapy available against HEV and no non-teratogenic treatment options for pregnant women, creating a need for the development of new therapeutics
-
The molecular biology of HEV remains incompletely understood
-
New model systems are emerging to study HEV, but more refined models are needed to gain insights in the interactions of HEV with its host, including mechanisms of HEV pathogenesis
Abstract
At least 20 million hepatitis E virus (HEV) infections occur annually, with >3 million symptomatic cases and ∼60,000 fatalities. Hepatitis E is generally self-limiting, with a case fatality rate of 0.5–3% in young adults. However, it can cause up to 30% mortality in pregnant women in the third trimester and can become chronic in immunocompromised individuals, such as those receiving organ transplants or chemotherapy and individuals with HIV infection. HEV is transmitted primarily via the faecal–oral route and was previously thought to be a public health concern only in developing countries. It is now also being frequently reported in industrialized countries, where it is transmitted zoonotically or through organ transplantation or blood transfusions. Although a vaccine for HEV has been developed, it is only licensed in China. Additionally, no effective, non-teratogenic and specific treatments against HEV infections are currently available. Although progress has been made in characterizing HEV biology, the scarcity of adequate experimental platforms has hampered further research. In this Review, we focus on providing an update on the HEV life cycle. We will further discuss existing cell culture and animal models and highlight platforms that have proven to be useful and/or are emerging for studying other hepatotropic (viral) pathogens.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Khuroo, M. S., Khuroo, M. S. & Khuroo, N. S. Hepatitis E: discovery, global impact, control and cure. World J. Gastroenterol. 22, 7030–7045 (2016).
Khuroo, M. S. Study of an epidemic of non-A, non-B hepatitis. Possibility of another human hepatitis virus distinct from post-transfusion non-A, non-B type. Am. J. Med. 68, 818–824 (1980).
Wong, D. C., Purcell, R. H., Sreenivasan, M. A., Prasad, S. R. & Pavri, K. M. Epidemic and endemic hepatitis in India: evidence for a non-A, non-B hepatitis virus aetiology. Lancet 2, 876–879 (1980).
Balayan, M. S. et al. Evidence for a virus in non-A, non-B hepatitis transmitted via the fecal-oral route. Intervirology 20, 23–31 (1983). In this study, a volunteer ingested pooled stool extracts from patients infected with HEV to show for the first time that the virus is transmitted via the faecal–oral route.
Reyes, G. R. et al. Isolation of a cDNA from the virus responsible for enterically transmitted non-A, non-B hepatitis. Science 247, 1335–1339 (1990). This study is the first to clone the partial cDNA of ENANBH, and the authors renamed the virus 'hepatitis E virus'.
Arankalle, V. A., Chadha, M. S., Mehendale, S. M. & Banerjee, K. Outbreak of enterically transmitted non-A, non-B hepatitis among schoolchildren. Lancet 2, 1199–1200 (1988).
Naik, S. R., Aggarwal, R., Salunke, P. N. & Mehrotra, N. N. A large waterborne viral hepatitis E epidemic in Kanpur, India. Bull. World Health Organ. 70, 597–604 (1992).
Skidmore, S. J., Yarbough, P. O., Gabor, K. A. & Reyes, G. R. Hepatitis E virus: the cause of a waterbourne hepatitis outbreak. J. Med. Virol. 37, 58–60 (1992).
Arankalle, V. A. et al. Seroepidemiology of water-borne hepatitis in India and evidence for a third enterically-transmitted hepatitis agent. Proc. Natl Acad. Sci. USA 91, 3428–3432 (1994).
Centers for Disease Control. Enterically transmitted non-A, non-B hepatitis — East Africa. MMWR Morb. Mortal. Wkly Rep. 36, 241–244 (1987).
[No authors listed.] Leads from the MMWR. Enterically transmitted non-A, non-B hepatitis — East Africa. JAMA 257, 2704–2705 (1987).
Centers for Disease Control. Enterically transmitted non-A, non-B hepatitis — Mexico. MMWR Morb. Mortal. Wkly Rep. 36, 597–602 (1987).
[No authors listed.] Leads from the MMWR. Enterically transmitted non-A, non-B hepatitis — Mexico. JAMA 258, 2041 (1987).
Iqbal, M. et al. An outbreak of enterically transmitted non-A, non-B hepatitis in Pakistan. Am. J. Trop. Med. Hyg. 40, 438–443 (1989).
Fortier, D., Treadwell, T. L. & Koff, R. S. Enterically transmitted non-A, non-B hepatitis: importation from Mexico to Massachusetts. N. Engl. J. Med. 320, 1281–1282 (1989).
Hlady, W. G. et al. Enterically transmitted non-A, non-B hepatitis associated with an outbreak in Dhaka: epidemiology and public health implications. Trop. Doct. 20, 15–17 (1990).
Zhuang, H., Cao, X. Y., Liu, C. B. & Wang, G. M. Epidemiology of hepatitis E in China. Gastroenterol. Jpn. 26 (Suppl. 3), 135–138 (1991).
Tsega, E. et al. Outbreak of acute hepatitis E virus infection among military personnel in northern Ethiopia. J. Med. Virol. 34, 232–236 (1991).
McCarthy, M. C. et al. Acute hepatitis E infection during the 1988 floods in Khartoum, Sudan. Trans. R. Soc. Trop. Med. Hyg. 88, 177 (1994).
Guthmann, J. P. et al. A large outbreak of hepatitis E among a displaced population in Darfur, Sudan, 2004: the role of water treatment methods. Clin. Infect. Dis. 42, 1685–1691 (2006).
Corwin, A. L. et al. A waterborne outbreak of hepatitis E virus transmission in southwestern Vietnam. Am. J. Trop. Med. Hyg. 54, 559–562 (1996).
Peron, J. M. et al. Hepatitis E is an autochthonous disease in industrialized countries. Analysis of 23 patients in South-West France over a 13-month period and comparison with hepatitis A. Gastroenterol. Clin. Biol. 30, 757–762 (2006).
Preiss, J. C. et al. Autochthonous hepatitis E virus infection in Germany with sequence similarities to other European isolates. Infection 34, 173–175 (2006).
Rein, D. B., Stevens, G. A., Theaker, J., Wittenborn, J. S. & Wiersma, S. T. The global burden of hepatitis E virus genotypes 1 and 2 in 2005. Hepatology 55, 988–997 (2012).
World Health Organization. Hepatitis E. WHO http://www.who.int/mediacentre/factsheets/fs280/en/ (2015).
Ahmad, I., Holla, R. P. & Jameel, S. Molecular virology of hepatitis E virus. Virus Res. 161, 47–58 (2011).
van Cuyck-Gandre, H. et al. Short report: polymerase chain reaction detection of hepatitis E virus in north African fecal samples. Am. J. Trop. Med. Hyg. 54, 134–135 (1996).
Escriba, J. M. et al. Hepatitis E, Central African Republic. Emerg. Infect. Dis. 14, 681–683 (2008).
Goumba, A. I., Konamna, X. & Komas, N. P. Clinical and epidemiological aspects of a hepatitis E outbreak in Bangui, Central African Republic. BMC Infect. Dis. 11, 93 (2011).
[No authors listed.] Hepatitis E, Chad. Wkly Epidemiol. Rec. 79, 313 (2004).
Coursaget, P. et al. Outbreak of enterically-transmitted hepatitis due to hepatitis A and hepatitis E viruses. J. Hepatol. 28, 745–750 (1998).
Ahmed, J. A. et al. Hepatitis E outbreak, Dadaab refugee camp, Kenya, 2012. Emerg. Infect. Dis. 19, 1010–1012 (2013).
Benjelloun, S. et al. Seroepidemiological study of an acute hepatitis E outbreak in Morocco. Res. Virol. 148, 279–287 (1997).
Isaacson, M., Frean, J., He, J., Seriwatana, J. & Innis, B. L. An outbreak of hepatitis E in Northern Namibia, 1983. Am. J. Trop. Med. Hyg. 62, 619–625 (2000).
Bile, K. et al. Contrasting roles of rivers and wells as sources of drinking water on attack and fatality rates in a hepatitis E epidemic in Somalia. Am. J. Trop. Med. Hyg. 51, 466–474 (1994).
Mushahwar, I. K., Dawson, G. J., Bile, K. M. & Magnius, L. O. Serological studies of an enterically transmitted non-A, non-B hepatitis in Somalia. J. Med. Virol. 40, 218–221 (1993).
Centers for Disease Control and Prevention. Investigation of hepatitis E outbreak among refugees — Upper Nile, South Sudan, 2012–2013. MMWR Morb. Mortal. Wkly Rep. 62, 581–586 (2013).
Rayis, D. A., Jumaa, A. M., Gasim, G. I., Karsany, M. S. & Adam, I. An outbreak of hepatitis E and high maternal mortality at Port Sudan, Eastern Sudan. Pathog. Glob. Health 107, 66–68 (2013).
Teshale, E. H. et al. Hepatitis E epidemic, Uganda. Emerg. Infect. Dis. 16, 126–129 (2010).
Clemente-Casares, P., Ramos-Romero, C., Ramirez-Gonzalez, E. & Mas, A. Hepatitis E virus in industrialized countries: the silent threat. Biomed. Res. Int. 2016, 9838041 (2016).
Colson, P. et al. Pig liver sausage as a source of hepatitis E virus transmission to humans. J. Infect. Dis. 202, 825–834 (2010).
Lewis, H. C., Wichmann, O. & Duizer, E. Transmission routes and risk factors for autochthonous hepatitis E virus infection in Europe: a systematic review. Epidemiol. Infect. 138, 145–166 (2010).
Li, T. C. et al. Hepatitis E virus transmission from wild boar meat. Emerg. Infect. Dis. 11, 1958–1960 (2005).
Miyamura, T. Hepatitis E virus infection in developed countries. Virus Res. 161, 40–46 (2011).
Ditah, I. et al. Current epidemiology of hepatitis E virus infection in the United States: low seroprevalence in the National Health and Nutrition Evaluation Survey. Hepatology 60, 815–822 (2014).
Murrison, L. B. & Sherman, K. E. The enigma of hepatitis E virus. Gastroenterol. Hepatol. (N.Y.) 13, 484–491 (2017).
Avellon, A., Morago, L., Garcia- Galera del Carmen, M., Munoz, M. & Echevarria, J. M. Comparative sensitivity of commercial tests for hepatitis E genotype 3 virus antibody detection. J. Med. Virol. 87, 1934–1939 (2015).
Vollmer, T., Diekmann, J., Eberhardt, M., Knabbe, C. & Dreier, J. Monitoring of anti-hepatitis E virus antibody seroconversion in asymptomatically infected blood donors: systematic comparison of nine commercial anti-HEV IgM and IgG assays. Viruses 8, E232 (2016).
Harritshoj, L. H. et al. Low transfusion transmission of hepatitis E among 25,637 single-donation, nucleic acid-tested blood donors. Transfusion 56, 2225–2232 (2016).
Gallian, P. et al. Hepatitis E virus infections in blood donors, France. Emerg. Infect. Dis. 20, 1914–1917 (2014).
Vollmer, T. et al. Novel approach for detection of hepatitis E virus infection in German blood donors. J. Clin. Microbiol. 50, 2708–2713 (2012).
Hogema, B. M. et al. Incidence and duration of hepatitis E virus infection in Dutch blood donors. Transfusion 56, 722–728 (2016).
Sauleda, S. et al. Seroprevalence of hepatitis E virus (HEV) and detection of HEV RNA with a transcription-mediated amplification assay in blood donors from Catalonia (Spain). Transfusion 55, 972–979 (2015).
Gallian, P. et al. Comparison of hepatitis E virus nucleic acid test screening platforms and RNA prevalence in French blood donors. Transfusion 57, 223–224 (2017).
Domanovic, D. et al. Hepatitis E and blood donation safety in selected European countries: a shift to screening? Euro Surveill. 22, 30514 (2017).
Roth, N. J. et al. Low hepatitis E virus RNA prevalence in a large-scale survey of United States source plasma donors. Transfusion http://dx.doi.org/10.1111/trf.14285 (2017).
Meng, X. J. Expanding host range and cross-species infection of hepatitis E virus. PLoS Pathog. 12, e1005695 (2016).
Hoofnagle, J. H., Nelson, K. E. & Purcell, R. H. Hepatitis E. N. Engl. J. Med. 367, 1237–1244 (2012).
Meng, X. J. Zoonotic and foodborne transmission of hepatitis E virus. Semin. Liver Dis. 33, 41–49 (2013).
Kamar, N. et al. Hepatitis E virus and chronic hepatitis in organ-transplant recipients. N. Engl. J. Med. 358, 811–817 (2008). This paper shows that HEV infection could become chronic in organ transplant recipients; previously the virus was thought to cause only acute disease.
Van der Poel, W. H. Food and environmental routes of Hepatitis E virus transmission. Curr. Opin. Virol. 4, 91–96 (2014).
Widen, F. et al. Molecular epidemiology of hepatitis E virus in humans, pigs and wild boars in Sweden. Epidemiol. Infect. 139, 361–371 (2011).
Jimenez de Oya, N. et al. Widespread distribution of hepatitis E virus in Spanish pig herds. BMC Res. Notes 4, 412 (2011).
Rose, N. et al. High prevalence of Hepatitis E virus in French domestic pigs. Comp. Immunol. Microbiol. Infect. Dis. 34, 419–427 (2011).
Halbur, P. G. et al. Comparative pathogenesis of infection of pigs with hepatitis E viruses recovered from a pig and a human. J. Clin. Microbiol. 39, 918–923 (2001).
Mykytczuk, O., Harlow, J., Bidawid, S., Corneau, N. & Nasheri, N. Prevalence and molecular characterization of the hepatitis E virus in retail pork products marketed in Canada. Food Environ. Virol. 9, 208–218 (2017).
Szabo, K. et al. Detection of hepatitis E virus RNA in raw sausages and liver sausages from retail in Germany using an optimized method. Int. J. Food Microbiol. 215, 149–156 (2015).
Pavio, N., Merbah, T. & Thebault, A. Frequent hepatitis E virus contamination in food containing raw pork liver, France. Emerg. Infect. Dis. 20, 1925–1927 (2014).
Barnaud, E., Rogee, S., Garry, P., Rose, N. & Pavio, N. Thermal inactivation of infectious hepatitis E virus in experimentally contaminated food. Appl. Environ. Microbiol. 78, 5153–5159 (2012).
Woo, P. C. et al. New hepatitis E virus genotype in bactrian camels, Xinjiang, China, 2013. Emerg. Infect. Dis. 22, 2219–2221 (2016).
Woo, P. C. et al. New hepatitis E virus genotype in camels, the Middle East. Emerg. Infect. Dis. 20, 1044–1048 (2014).
Rasche, A. et al. Hepatitis E virus infection in dromedaries, North and East Africa, United Arab Emirates, and Pakistan, 1983–2015. Emerg. Infect. Dis. 22, 1249–1252 (2016).
Kumar, S., Subhadra, S., Singh, B. & Panda, B. K. Hepatitis E virus: the current scenario. Int. J. Infect. Dis. 17, e228–e233 (2013).
Navaneethan, U., Al Mohajer, M. & Shata, M. T. Hepatitis E and pregnancy: understanding the pathogenesis. Liver Int. 28, 1190–1199 (2008).
Perez-Gracia, M. T., Suay-Garcia, B. & Mateos-Lindemann, M. L. Hepatitis E and pregnancy: current state. Rev. Med. Virol. http://dx.doi.org/10.1002/rmv.1929 (2017).
Sharapov, M. B. et al. Acute viral hepatitis morbidity and mortality associated with hepatitis E virus infection: Uzbekistan surveillance data. BMC Infect. Dis. 9, 35 (2009).
Pischke, S. & Wedemeyer, H. Hepatitis E virus infection: multiple faces of an underestimated problem. J. Hepatol. 58, 1045–1046 (2013).
Colson, P., Dhiver, C., Poizot-Martin, I., Tamalet, C. & Gerolami, R. Acute and chronic hepatitis E in patients infected with human immunodeficiency virus. J. Viral Hepat. 18, 227–228 (2011).
Dalton, H. R., Bendall, R. P., Keane, F. E., Tedder, R. S. & Ijaz, S. Persistent carriage of hepatitis E virus in patients with HIV infection. N. Engl. J. Med. 361, 1025–1027 (2009).
Khuroo, M. S. & Khuroo, M. S. Hepatitis E: an emerging global disease — from discovery towards control and cure. J. Viral Hepat. 23, 68–79 (2016).
Fujiwara, S. et al. Chronic hepatitis E: a review of the literature. J. Viral Hepat. 21, 78–89 (2014).
Kamar, N. et al. Factors associated with chronic hepatitis in patients with hepatitis E virus infection who have received solid organ transplants. Gastroenterology 140, 1481–1489 (2011).
Abravanel, F. et al. Hepatitis E virus reinfections in solid-organ-transplant recipients can evolve into chronic infections. J. Infect. Dis. 209, 1900–1906 (2014).
Borentain, P. et al. Hepatocellular carcinoma complicating hepatitis E virus-related cirrhosis. Hepatology http://dx.doi.org/10.1002/hep.29508 (2017).
Kamar, N. et al. Hepatitis E virus-induced neurological symptoms in a kidney-transplant patient with chronic hepatitis. Am. J. Transplant. 10, 1321–1324 (2010).
van den Berg, B. et al. Guillain-Barre syndrome associated with preceding hepatitis E virus infection. Neurology 82, 491–497 (2014).
Bazerbachi, F., Haffar, S., Garg, S. K. & Lake, J. R. Extra-hepatic manifestations associated with hepatitis E virus infection: a comprehensive review of the literature. Gastroenterol. Rep. (Oxf.) 4, 1–15 (2016).
Pischke, S. et al. Hepatitis E virus: Infection beyond the liver? J. Hepatol. 66, 1082–1095 (2017).
McLean, B. N., Gulliver, J. & Dalton, H. R. Hepatitis E virus and neurological disorders. Pract. Neurol. 17, 282–288 (2017).
Mallet, V. et al. Hepatitis E virus-induced primary cutaneous CD30(+) T cell lymphoproliferative disorder. J. Hepatol. http://dx.doi.org/10.1016/j.jhep.2017.08.011 (2017).
Comont, T. et al. Acute hepatitis E infection associated with Guillain-Barre syndrome in an immunocompetent patient. Rev. Med. Interne 35, 333–336 (2014).
Zhou, X. et al. Hepatitis E virus infects neurons and brains. J. Infect. Dis. 215, 1197–1206 (2017).
Choi, C. & Chae, C. Localization of swine hepatitis E virus in liver and extrahepatic tissues from naturally infected pigs by in situ hybridization. J. Hepatol. 38, 827–832 (2003).
Kamar, N. et al. Influence of immunosuppressive therapy on the natural history of genotype 3 hepatitis-E virus infection after organ transplantation. Transplantation 89, 353–360 (2010).
Kamar, N. et al. Ribavirin for chronic hepatitis E virus infection in transplant recipients. N. Engl. J. Med. 370, 1111–1120 (2014). In this study, ribavirin monotherapy leads to HEV clearance in 95% of patients in a cohort of SOT recipients with chronic infection.
Khuroo, M. S. & Kamili, S. Aetiology, clinical course and outcome of sporadic acute viral hepatitis in pregnancy. J. Viral Hepat. 10, 61–69 (2003).
Debing, Y. et al. A mutation in the hepatitis E virus RNA polymerase promotes its replication and associates with ribavirin treatment failure in organ transplant recipients. Gastroenterology 147, 1008–1011.e7; quiz e15–e16 (2014). This paper reports the discovery of a mutation in the HEV polymerase in two patients who were nonresponsive to ribavirin therapy; in vitro , this mutation increased the replication efficiency of the virus.
Debing, Y. et al. Hepatitis E virus mutations associated with ribavirin treatment failure result in altered viral fitness and ribavirin sensitivity. J. Hepatol. 65, 499–508 (2016).
Mazzola, A. et al. Chronic hepatitis E viral infection after liver transplantation: a regression of fibrosis following antiviral therapy. Transplantation 101, 2083–2087 (2017).
Alric, L., Bonnet, D., Laurent, G., Kamar, N. & Izopet, J. Chronic hepatitis E virus infection: successful virologic response to pegylated interferon-α therapy. Ann. Intern. Med. 153, 135–136 (2010).
Kamar, N. et al. Three-month pegylated interferon-alpha-2a therapy for chronic hepatitis E virus infection in a haemodialysis patient. Nephrol. Dial. Transplant. 25, 2792–2795 (2010).
Dao Thi, V. L. et al. Sofosbuvir inhibits hepatitis E virus replication in vitro and results in an additive effect when combined with ribavirin. Gastroenterology 150, 82–85.e84 (2016).
van der Valk, M., Zaaijer, H. L., Kater, A. P. & Schinkel, J. Sofosbuvir shows antiviral activity in a patient with chronic hepatitis E virus infection. J. Hepatol. 66, 242–243 (2017).
Todt, D. et al. In vivo evidence for ribavirin-induced mutagenesis of the hepatitis E virus genome. Gut 65, 1733–1743 (2016).
Lhomme, S. et al. Mutation in the hepatitis E virus polymerase and outcome of ribavirin therapy. Antimicrob. Agents Chemother. 60, 1608–1614 (2015).
Zhao, Q. et al. Antigenic determinants of hepatitis E virus and vaccine-induced immunogenicity and efficacy. J. Gastroenterol. 48, 159–168 (2013).
Lee, G. Y. et al. Hepatitis E virus infection: epidemiology and treatment implications. World J. Virol. 4, 343–355 (2015).
Wu, T. et al. Difference of T cell and B cell activation in two homologous proteins with similar antigenicity but great distinct immunogenicity. Mol. Immunol. 44, 3261–3266 (2007).
Tsarev, S. A. et al. Recombinant vaccine against hepatitis E: dose response and protection against heterologous challenge. Vaccine 15, 1834–1838 (1997).
Shrestha, M. P. et al. Safety and efficacy of a recombinant hepatitis E vaccine. N. Engl. J. Med. 356, 895–903 (2007).
Zhu, F. C. et al. Efficacy and safety of a recombinant hepatitis E vaccine in healthy adults: a large-scale, randomised, double-blind placebo-controlled, phase 3 trial. Lancet 376, 895–902 (2010). This study reports that the HEV239 vaccine was found to be 100% effective (95% CI 72.1–100) after three doses in a phase III clinical trial in China.
Joshi, S. S. & Arankalle, V. A. Enhanced humoral response in pregnant mice immunized with liposome encapsulated recombinant neutralizing epitope protein of Hepatitis-E virus. Virol. J. 12, 70 (2015).
World Health Organization. Vaccines prequalification priority list 2015–2016. WHO http://www.who.int/immunization_standards/vaccine_quality/priority_pq_vaccines_2015_16/en/ (2016).
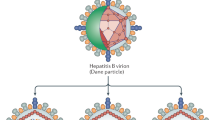
Guu, T. S. et al. Structure of the hepatitis E virus-like particle suggests mechanisms for virus assembly and receptor binding. Proc. Natl Acad. Sci. USA 106, 12992–12997 (2009). This paper reports the crystal structure of the HEV virus-like particle, composed of self-assembling capsid proteins, which was determined to a 3.5 Å resolution.
Feng, Z. et al. A pathogenic picornavirus acquires an envelope by hijacking cellular membranes. Nature 496, 367–371 (2013).
Takahashi, M. et al. Monoclonal antibodies raised against the ORF3 protein of hepatitis E virus (HEV) can capture HEV particles in culture supernatant and serum but not those in feces. Arch. Virol. 153, 1703–1713 (2008). This study is the first to show that HEV particles in culture supernatant and faeces have distinct properties.
Yin, X., Ambardekar, C., Lu, Y. & Feng, Z. Distinct entry mechanisms for nonenveloped and quasi-enveloped hepatitis E viruses. J. Virol. 90, 4232–4242 (2016). In this study, HEV is formally stated to be a quasi-enveloped virus, with the enveloped and non-enveloped forms having different entry mechanisms.
Chapuy-Regaud, S. et al. Characterization of the lipid envelope of exosome encapsulated HEV particles protected from the immune response. Biochimie 141, 70–79 (2017).
Nagashima, S. et al. The characterization of the quasi-enveloped hepatitis E virus particles released by the cellular exosomal pathway. J. Virol http://dx.doi.org/10.1128/JVI.00822-17 (2017).
Zhang, M., Purcell, R. H. & Emerson, S. U. Identification of the 5′ terminal sequence of the SAR-55 and MEX-14 strains of hepatitis E virus and confirmation that the genome is capped. J. Med. Virol. 65, 293–295 (2001).
Tam, A. W. et al. Hepatitis E virus (HEV): molecular cloning and sequencing of the full-length viral genome. Virology 185, 120–131 (1991).
Nair, V. P. et al. Endoplasmic reticulum stress induced synthesis of a novel viral factor mediates efficient replication of genotype-1 hepatitis E virus. PLoS Pathog. 12, e1005521 (2016).
Fry, K. E. et al. Hepatitis E virus (HEV): strain variation in the nonstructural gene region encoding consensus motifs for an RNA-dependent RNA polymerase and an ATP/GTP binding site. Virus Genes 6, 173–185 (1992).
Rozanov, M. N., Koonin, E. V. & Gorbalenya, A. E. Conservation of the putative methyltransferase domain: a hallmark of the 'Sindbis-like' supergroup of positive-strand RNA viruses. J. Gen. Virol. 73, 2129–2134 (1992).
Magden, J. et al. Virus-specific mRNA capping enzyme encoded by hepatitis E virus. J. Virol. 75, 6249–6255 (2001).
Koonin, E. V. et al. Computer-assisted assignment of functional domains in the nonstructural polyprotein of hepatitis E virus: delineation of an additional group of positive-strand RNA plant and animal viruses. Proc. Natl Acad. Sci. USA 89, 8259–8263 (1992). In this paper, it is predicted, based on sequence homology, that HEV contains a domain similar to the PCP of rubella virus; however, to date, protease function has not been conclusively proven for HEV.
Chen, J. P., Strauss, J. H., Strauss, E. G. & Frey, T. K. Characterization of the rubella virus nonstructural protease domain and its cleavage site. J. Virol. 70, 4707–4713 (1996).
Golubtsov, A., Kaariainen, L. & Caldentey, J. Characterization of the cysteine protease domain of Semliki Forest virus replicase protein nsP2 by in vitro mutagenesis. FEBS Lett. 580, 1502–1508 (2006).
Jore, J., De Geus, B., Jackson, R. J., Pouwels, P. H. & Enger-Valk, B. E. Poliovirus protein 3CD is the active protease for processing of the precursor protein P1 in vitro. J. Gen. Virol. 69, 1627–1636 (1988).
Jameel, S., Zafrullah, M., Ozdener, M. H. & Panda, S. K. Expression in animal cells and characterization of the hepatitis E virus structural proteins. J. Virol. 70, 207–216 (1996).
Ding, Q. et al. Hepatitis E virus ORF3 is a functional ion channel required for release of infectious particles. Proc. Natl Acad. Sci. USA 114, 1147–1152 (2017). In this paper, HEV ORF3, a viral factor critical for release, is shown to exhibit ion channel activity; therefore, HEV is now known to contain a viroporin.
Nagashima, S. et al. A PSAP motif in the ORF3 protein of hepatitis E virus is necessary for virion release from infected cells. J. Gen. Virol. 92, 269–278 (2011). This study confirms that a specific PSAP motif within the HEV ORF3 protein is involved in viral release.
Martin-Serrano, J., Zang, T. & Bieniasz, P. D. Role of ESCRT-I in retroviral budding. J. Virol. 77, 4794–4804 (2003).
Kalia, M., Chandra, V., Rahman, S. A., Sehgal, D. & Jameel, S. Heparan sulfate proteoglycans are required for cellular binding of the hepatitis E virus ORF2 capsid protein and for viral infection. J. Virol. 83, 12714–12724 (2009).
Kapur, N., Thakral, D., Durgapal, H. & Panda, S. K. Hepatitis E virus enters liver cells through receptor-dependent clathrin-mediated endocytosis. J. Viral Hepat. 19, 436–448 (2012).
Holla, P., Ahmad, I., Ahmed, Z. & Jameel, S. Hepatitis E virus enters liver cells through a dynamin-2, clathrin and membrane cholesterol-dependent pathway. Traffic 16, 398–416 (2015). Little was known about host factors involved in HEV entry until this study, which used specific small molecule inhibitors to identify cellular proteins mediating entry of fluorescent HEV-like particles.
Graff, J., Torian, U., Nguyen, H. & Emerson, S. U. A bicistronic subgenomic mRNA encodes both the ORF2 and ORF3 proteins of hepatitis E virus. J. Virol. 80, 5919–5926 (2006). This study provided the first evidence that HEV ORF2 and ORF3 proteins are translated from a subgenomic RNA.
Agrawal, S., Gupta, D. & Panda, S. K. The 3′ end of hepatitis E virus (HEV) genome binds specifically to the viral RNA-dependent RNA polymerase (RdRp). Virology 282, 87–101 (2001).
Nanda, S. K., Panda, S. K., Durgapal, H. & Jameel, S. Detection of the negative strand of hepatitis E virus RNA in the livers of experimentally infected rhesus monkeys: evidence for viral replication. J. Med. Virol. 42, 237–240 (1994). This study demonstrates that HEV can replicate in the livers of rhesus monkeys, which was proven through the detection of negative-strand RNA, a technique that was later used to validate other animal models for HEV and to discover extrahepatic sites of HEV replication.
Varma, S. P. et al. Hepatitis E virus replication involves alternating negative- and positive-sense RNA synthesis. J. Gen. Virol. 92, 572–581 (2011).
Xia, X., Huang, R. & Li, D. Studies on the subgenomic RNAs of hepatitis E virus [Chinese]. Wei Sheng Wu Xue Bao 40, 622–627 (2000).
Yarbough, P. O. et al. Hepatitis E virus: identification of type-common epitopes. J. Virol. 65, 5790–5797 (1991).
Mahilkar, S., Paingankar, M. S. & Lole, K. S. Hepatitis E virus RNA-dependent RNA polymerase: RNA template specificities, recruitment and synthesis. J. Gen. Virol. 97, 2231–2242 (2016).
Montpellier, C. et al. Hepatitis E virus lifecycle and identification of 3 forms of the ORF2 capsid protein. Gastroenterology http://dx.doi.org/10.1053/j.gastro.2017.09.020 (2017). In this study, pORF2 is first discovered to be processed into multiple isoforms, which are secreted from the cell through distinct pathways as either infectious or non-infectious particles.
Zafrullah, M., Ozdener, M. H., Kumar, R., Panda, S. K. & Jameel, S. Mutational analysis of glycosylation, membrane translocation, and cell surface expression of the hepatitis E virus ORF2 protein. J. Virol. 73, 4074–4082 (1999).
Graff, J. et al. Mutations within potential glycosylation sites in the capsid protein of hepatitis E virus prevent the formation of infectious virus particles. J. Virol. 82, 1185–1194 (2008).
Surjit, M., Jameel, S. & Lal, S. K. The ORF2 protein of hepatitis E virus binds the 5′ region of viral RNA. J. Virol. 78, 320–328 (2004).
Surjit, M., Jameel, S. & Lal, S. K. Cytoplasmic localization of the ORF2 protein of hepatitis E virus is dependent on its ability to undergo retrotranslocation from the endoplasmic reticulum. J. Virol. 81, 3339–3345 (2007).
Lenggenhager, D. et al. Visualization of hepatitis E virus RNA and proteins in the human liver. J. Hepatol. 67, 471–479 (2017).
Suzuki, T. et al. The human polyoma JC virus agnoprotein acts as a viroporin. PLoS Pathog. 6, e1000801 (2010).
Nieva, J. L., Madan, V. & Carrasco, L. Viroporins: structure and biological functions. Nat. Rev. Microbiol. 10, 563–574 (2012).
Zafrullah, M., Ozdener, M. H., Panda, S. K. & Jameel, S. The ORF3 protein of hepatitis E virus is a phosphoprotein that associates with the cytoskeleton. J. Virol. 71, 9045–9053 (1997).
Korkaya, H. et al. The ORF3 protein of hepatitis E virus binds to Src homology 3 domains and activates MAPK. J. Biol. Chem. 276, 42389–42400 (2001).
Parvez, M. K. & Al-Dosari, M. S. Evidence of MAPK-JNK1/2 activation by hepatitis E virus ORF3 protein in cultured hepatoma cells. Cytotechnology 67, 545–550 (2015).
Kar-Roy, A., Korkaya, H., Oberoi, R., Lal, S. K. & Jameel, S. The hepatitis E virus open reading frame 3 protein activates ERK through binding and inhibition of the MAPK phosphatase. J. Biol. Chem. 279, 28345–28357 (2004).
Tyagi, S., Surjit, M. & Lal, S. K. The 41-amino-acid C-terminal region of the hepatitis E virus ORF3 protein interacts with bikunin, a kunitz-type serine protease inhibitor. J. Virol. 79, 12081–12087 (2005).
Ratra, R., Kar-Roy, A. & Lal, S. K. The ORF3 protein of hepatitis E virus interacts with hemopexin by means of its 26 amino acid N-terminal hydrophobic domain II. Biochemistry 47, 1957–1969 (2008).
Ratra, R., Kar-Roy, A. & Lal, S. K. ORF3 protein of hepatitis E virus interacts with the Bβ chain of fibrinogen resulting in decreased fibrinogen secretion from HuH-7 cells. J. Gen. Virol. 90, 1359–1370 (2009).
Kannan, H., Fan, S., Patel, D., Bossis, I. & Zhang, Y. J. The hepatitis E virus open reading frame 3 product interacts with microtubules and interferes with their dynamics. J. Virol. 83, 6375–6382 (2009).
Moin, S. M., Chandra, V., Arya, R. & Jameel, S. The hepatitis E virus ORF3 protein stabilizes HIF-1α and enhances HIF-1-mediated transcriptional activity through p300/CBP. Cell. Microbiol. 11, 1409–1421 (2009).
Chandra, V., Kalia, M., Hajela, K. & Jameel, S. The ORF3 protein of hepatitis E virus delays degradation of activated growth factor receptors by interacting with CIN85 and blocking formation of the Cbl-CIN85 complex. J. Virol. 84, 3857–3867 (2010).
Chandra, V. et al. The hepatitis E virus ORF3 protein regulates the expression of liver-specific genes by modulating localization of hepatocyte nuclear factor 4. PLoS ONE 6, e22412 (2011).
Wang, C. et al. HEV-ORF3 encoding phosphoprotein interacts with hepsin. Hepat. Mon. 14, e13902 (2014).
Chandra, V., Kar-Roy, A., Kumari, S., Mayor, S. & Jameel, S. The hepatitis E virus ORF3 protein modulates epidermal growth factor receptor trafficking, STAT3 translocation, and the acute-phase response. J. Virol. 82, 7100–7110 (2008).
Nagashima, S. et al. The membrane on the surface of hepatitis E virus particles is derived from the intracellular membrane and contains trans-Golgi network protein 2. Arch. Virol. 159, 979–991 (2014).
Surjit, M., Oberoi, R., Kumar, R. & Lal, S. K. Enhanced α1 microglobulin secretion from Hepatitis E virus ORF3-expressing human hepatoma cells is mediated by the tumor susceptibility gene 101. J. Biol. Chem. 281, 8135–8142 (2006).
Yamada, K. et al. ORF3 protein of hepatitis E virus is essential for virion release from infected cells. J. Gen. Virol. 90, 1880–1891 (2009).
Emerson, S. U. et al. Release of genotype 1 hepatitis E virus from cultured hepatoma and polarized intestinal cells depends on open reading frame 3 protein and requires an intact PXXP motif. J. Virol. 84, 9059–9069 (2010).
Kenney, S. P., Wentworth, J. L., Heffron, C. L. & Meng, X. J. Replacement of the hepatitis E virus ORF3 protein PxxP motif with heterologous late domain motifs affects virus release via interaction with TSG101. Virology 486, 198–208 (2015).
Nagashima, S. et al. Hepatitis E virus egress depends on the exosomal pathway, with secretory exosomes derived from multivesicular bodies. J. Gen. Virol. 95, 2166–2175 (2014).
Qi, Y. et al. Hepatitis E virus produced from cell culture has a lipid envelope. PLoS ONE 10, e0132503 (2015).
Paingankar, M. S. & Arankalle, V. A. Identification and characterization of cellular proteins interacting with Hepatitis E virus untranslated regions. Virus Res. 208, 98–109 (2015).
Hurley, J. H. The ESCRT complexes. Crit. Rev. Biochem. Mol. Biol. 45, 463–487 (2010).
Tam, A. W. et al. In vitro infection and replication of hepatitis E virus in primary cynomolgus macaque hepatocytes. Virology 238, 94–102 (1997). This study reports the first use of primary hepatocytes to study HEV in vitro.
Panda, S. K., Ansari, I. H., Durgapal, H., Agrawal, S. & Jameel, S. The in vitro-synthesized RNA from a cDNA clone of hepatitis E virus is infectious. J. Virol. 74, 2430–2437 (2000).
Huang, R. et al. Cell culture of sporadic hepatitis E virus in China. Clin. Diagn. Lab. Immunol. 6, 729–733 (1999).
Kazachkov, Y. et al. Hepatitis E virus in cultivated cells. Arch. Virol. 127, 399–402 (1992).
Tanaka, T. et al. Development and characterization of a genotype 4 hepatitis E virus cell culture system using a HE-JF5/15F strain recovered from a fulminant hepatitis patient. J. Clin. Microbiol. 47, 1906–1910 (2009).
Yamada, K. et al. Construction of an infectious cDNA clone of hepatitis E virus strain JE03-1760F that can propagate efficiently in cultured cells. J. Gen. Virol. 90, 457–462 (2009).
Shukla, P. et al. Cross-species infections of cultured cells by hepatitis E virus and discovery of an infectious virus-host recombinant. Proc. Natl Acad. Sci. USA 108, 2438–2443 (2011). In this study, a cell-culture-adapted HEV strain, Kernow-C1/p6, is discovered and found to contain a human ribosomal insert that expands its host range and leads to more efficient replication; this HEV strain has been widely used for subsequent in vitro studies.
Shukla, P. et al. Adaptation of a genotype 3 hepatitis E virus to efficient growth in cell culture depends on an inserted human gene segment acquired by recombination. J. Virol. 86, 5697–5707 (2012).
Nguyen, H. T. et al. A naturally occurring human/hepatitis E recombinant virus predominates in serum but not in faeces of a chronic hepatitis E patient and has a growth advantage in cell culture. J. Gen. Virol. 93, 526–530 (2012).
Yin, X. et al. Hepatitis E virus persists in the presence of a type III interferon response. PLoS Pathog. 13, e1006417 (2017).
Xu, L. et al. RIG-I is a key antiviral interferon-stimulated gene against hepatitis E virus dispensable of interferon production. Hepatology 65, 1823–1839 (2017).
Rogee, S. et al. New models of hepatitis E virus replication in human and porcine hepatocyte cell lines. J. Gen. Virol. 94, 549–558 (2013).
Elkayam, T., Amitay-Shaprut, S., Dvir-Ginzberg, M., Harel, T. & Cohen, S. Enhancing the drug metabolism activities of C3A — a human hepatocyte cell line — by tissue engineering within alginate scaffolds. Tissue Eng. 12, 1357–1368 (2006).
Dembele, L. et al. Persistence and activation of malaria hypnozoites in long-term primary hepatocyte cultures. Nat. Med. 20, 307–312 (2014).
Mitaka, T. The current status of primary hepatocyte culture. Int. J. Exp. Pathol. 79, 393–409 (1998).
Thomas, E. & Liang, T. J. Experimental models of hepatitis B and C — new insights and progress. Nat. Rev. Gastroenterol. Hepatol. 13, 362–374 (2016).
Verrier, E. R., Colpitts, C. C., Schuster, C., Zeisel, M. B. & Baumert, T. F. Cell culture models for the investigation of hepatitis B and D virus infection. Viruses 8, E261 (2016).
Dunn, J. C., Tompkins, R. G. & Yarmush, M. L. Long-term in vitro function of adult hepatocytes in a collagen sandwich configuration. Biotechnol. Prog. 7, 237–245 (1991).
Dunn, J. C., Yarmush, M. L., Koebe, H. G. & Tompkins, R. G. Hepatocyte function and extracellular matrix geometry: long-term culture in a sandwich configuration. FASEB J. 3, 174–177 (1989).
Guillouzo, A., Morel, F., Ratanasavanh, D., Chesne, C. & Guguen-Guillouzo, C. Long-term culture of functional hepatocytes. Toxicol. In Vitro 4, 415–427 (1990).
Chong, T. W. et al. Primary human hepatocytes in spheroid formation to study hepatitis C infection. J. Surg. Res. 130, 52–57 (2006).
Kidambi, S. et al. Oxygen-mediated enhancement of primary hepatocyte metabolism, functional polarization, gene expression, and drug clearance. Proc. Natl Acad. Sci. USA 106, 15714–15719 (2009).
Khetani, S. R. & Bhatia, S. N. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 26, 120–126 (2008).
Bhatia, S. N., Balis, U. J., Yarmush, M. L. & Toner, M. Effect of cell-cell interactions in preservation of cellular phenotype: cocultivation of hepatocytes and nonparenchymal cells. FASEB J. 13, 1883–1900 (1999).
Shlomai, A. et al. Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc. Natl Acad. Sci. USA 111, 12193–12198 (2014).
March, S. et al. A microscale human liver platform that supports the hepatic stages of Plasmodium falciparum and vivax. Cell Host Microbe 14, 104–115 (2013).
Ploss, A. et al. Persistent hepatitis C virus infection in microscale primary human hepatocyte cultures. Proc. Natl Acad. Sci. USA 107, 3141–3145 (2010).
Winer, B. Y. et al. Long-term hepatitis B infection in a scalable hepatic co-culture system. Nat. Commun. 8, 125 (2017).
Helsen, N. et al. Stem cell-derived hepatocytes: a novel model for hepatitis E virus replication. J. Hepatol. 64, 565–573 (2016). This paper establishes hepatocyte-like cells derived from human pluripotent stem cells as an in vitro model to study HEV.
Yamanaka, S. Induced pluripotent stem cells: past, present, and future. Cell Stem Cell 10, 678–684 (2012).
Ramanan, V., Scull, M. A., Sheahan, T. P., Rice, C. M. & Bhatia, S. N. New methods in tissue engineering: improved models for viral infection. Annu. Rev. Virol. 1, 475–499 (2014).
Irion, S., Nostro, M. C., Kattman, S. J. & Keller, G. M. Directed differentiation of pluripotent stem cells: from developmental biology to therapeutic applications. Cold Spring Harb. Symp. Quant. Biol. 73, 101–110 (2008).
Davidson, M. D., Kukla, D. A. & Khetani, S. R. Microengineered cultures containing human hepatic stellate cells and hepatocytes for drug development. Integr. Biol. (Camb.) 9, 662–677 (2017).
Ma, X. et al. Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting. Proc. Natl Acad. Sci. USA 113, 2206–2211 (2016).
Chen, A. A. et al. Humanized mice with ectopic artificial liver tissues. Proc. Natl Acad. Sci. USA 108, 11842–11847 (2011).
March, S. et al. Micropatterned coculture of primary human hepatocytes and supportive cells for the study of hepatotropic pathogens. Nat. Protoc. 10, 2027–2053 (2015).
Bose, P. D. et al. Evidence of extrahepatic replication of hepatitis E virus in human placenta. J. Gen. Virol. 95, 1266–1271 (2014).
de Deus, N. et al. Detection of hepatitis E virus in liver, mesenteric lymph node, serum, bile and faeces of naturally infected pigs affected by different pathological conditions. Vet. Microbiol. 119, 105–114 (2007).
Williams, T. P. et al. Evidence of extrahepatic sites of replication of the hepatitis E virus in a swine model. J. Clin. Microbiol. 39, 3040–3046 (2001).
Huang, R. et al. Hepatitis E virus (87A strain) propagated in A549 cells. J. Med. Virol. 47, 299–302 (1995).
Takahashi, H. et al. A549 and PLC/PRF/5 cells can support the efficient propagation of swine and wild boar hepatitis E virus (HEV) strains: demonstration of HEV infectivity of porcine liver sold as food. Arch. Virol. 157, 235–246 (2012).
Jirintai, S. et al. Molecular analysis of hepatitis E virus from farm rabbits in Inner Mongolia, China and its successful propagation in A549 and PLC/PRF/5 cells. Virus Res. 170, 126–137 (2012).
Montalvo Villalba, M. C. et al. Hepatitis E virus in bottlenose dolphins Tursiops truncatus. Dis. Aquat. Organ. 123, 13–18 (2017).
Panda, S. K., Datta, R., Kaur, J., Zuckerman, A. J. & Nayak, N. C. Enterically transmitted non-A, non-B hepatitis: recovery of virus-like particles from an epidemic in south Delhi and transmission studies in rhesus monkeys. Hepatology 10, 466–472 (1989).
Uchida, T. et al. Animal model, virology and gene cloning of hepatitis E. Gastroenterol. Jpn 26 (Suppl. 3), 148–151 (1991).
Arankalle, V. A., Chadha, M. S. & Chobe, L. P. Long-term serological follow up and cross-challenge studies in rhesus monkeys experimentally infected with hepatitis E virus. J. Hepatol. 30, 199–204 (1999).
Yu, C. et al. Pathogenesis of hepatitis E virus and hepatitis C virus in chimpanzees: similarities and differences. J. Virol. 84, 11264–11278 (2010). The authors compare host responses to HCV and HEV in chimpanzees, which are the only non-human host for both viruses.
Shata, M. T. et al. Characterization of hepatitis E-specific cell-mediated immune response using IFN-γ ELISPOT assay. J. Immunol. Methods 328, 152–161 (2007).
Li, X., Kamili, S. & Krawczynski, K. Quantitative detection of hepatitis E virus RNA and dynamics of viral replication in experimental infection. J. Viral Hepat. 13, 835–839 (2006).
Emerson, S. U. et al. Recombinant hepatitis E virus genomes infectious for primates: importance of capping and discovery of a cis-reactive element. Proc. Natl Acad. Sci. USA 98, 15270–15275 (2001). The authors infect rhesus monkeys and chimpanzees with capped versus uncapped HEV transcripts and find improved infection efficiency and a more severe clinical phenotype with the capped transcript.
Meng, X. J. et al. Genetic and experimental evidence for cross-species infection by swine hepatitis E virus. J. Virol. 72, 9714–9721 (1998). In this study, HEV from humans is found to be capable of infecting swine, and sequence analysis reveals that human and swine HEV strains in the USA are similar; these data strongly suggest the possibility of zoonotic transmission of HEV from swine to humans, which is now known to be the primary cause of HEV in developed nations.
Krawczynski, K., Meng, X. J. & Rybczynska, J. Pathogenetic elements of hepatitis E and animal models of HEV infection. Virus Res. 161, 78–83 (2011).
Tsarev, S. A. et al. Variation in course of hepatitis E in experimentally infected cynomolgus monkeys. J. Infect. Dis. 167, 1302–1306 (1993).
Tsarev, S. A. et al. Infectivity titration of a prototype strain of hepatitis E virus in cynomolgus monkeys. J. Med. Virol. 43, 135–142 (1994).
Soe, S. et al. Enterically transmitted non-A, non-B hepatitis in cynomolgus monkeys: morphology and probable mechanism of hepatocellular necrosis. Liver 9, 135–145 (1989).
Tsarev, S. A. et al. Successful passive and active immunization of cynomolgus monkeys against hepatitis E. Proc. Natl Acad. Sci. USA 91, 10198–10202 (1994).
Arankalle, V. A., Chadha, M. S., Chobe, L. P., Nair, R. & Banerjee, K. Cross-challenge studies in rhesus monkeys employing different Indian isolates of hepatitis E virus. J. Med. Virol. 46, 358–363 (1995).
Zhang, M. et al. Recombinant vaccine against hepatitis E: duration of protective immunity in rhesus macaques. Vaccine 20, 3285–3291 (2002).
Arankalle, V. A., Chadha, M. S., Banerjee, K., Srinivasan, M. A. & Chobe, L. P. Hepatitis E virus infection in pregnant rhesus monkeys. Indian J. Med. Res. 97, 4–8 (1993).
Tsarev, S. A. et al. Experimental hepatitis E in pregnant rhesus monkeys: failure to transmit hepatitis E virus (HEV) to offspring and evidence of naturally acquired antibodies to HEV. J. Infect. Dis. 172, 31–37 (1995).
Clayson, E. T. et al. Detection of hepatitis E virus infections among domestic swine in the Kathmandu Valley of Nepal. Am. J. Trop. Med. Hyg. 53, 228–232 (1995).
Meng, X. J. et al. A novel virus in swine is closely related to the human hepatitis E virus. Proc. Natl Acad. Sci. USA 94, 9860–9865 (1997). This paper is the first to discover HEV in pigs, which are now known to be the major reservoir for HEV in developed nations.
Meng, X. J. et al. Experimental infection of pigs with the newly identified swine hepatitis E virus (swine HEV), but not with human strains of HEV. Arch. Virol. 143, 1405–1415 (1998).
Purcell, R. H. et al. Pre-clinical immunogenicity and efficacy trial of a recombinant hepatitis E vaccine. Vaccine 21, 2607–2615 (2003).
Usmanov, R. K. et al. Experimental hepatitis E infection in piglets [Russian]. Vopr. Virusol. 36, 212–216 (1991).
Arankalle, V. A., Chobe, L. P., Walimbe, A. M., Yergolkar, P. N. & Jacob, G. P. Swine HEV infection in south India and phylogenetic analysis (1985–1999). J. Med. Virol. 69, 391–396 (2003).
Cooper, K. et al. Identification of genotype 3 hepatitis E virus (HEV) in serum and fecal samples from pigs in Thailand and Mexico, where genotype 1 and 2 HEV strains are prevalent in the respective human populations. J. Clin. Microbiol. 43, 1684–1688 (2005).
Kasorndorkbua, C. et al. Infectious swine hepatitis E virus is present in pig manure storage facilities on United States farms, but evidence of water contamination is lacking. Appl. Environ. Microbiol. 71, 7831–7837 (2005).
Feagins, A. R., Opriessnig, T., Huang, Y. W., Halbur, P. G. & Meng, X. J. Cross-species infection of specific-pathogen-free pigs by a genotype 4 strain of human hepatitis E virus. J. Med. Virol. 80, 1379–1386 (2008).
Cao, D. et al. Pig model mimicking chronic hepatitis E virus infection in immunocompromised patients to assess immune correlates during chronicity. Proc. Natl Acad. Sci. USA 114, 6914–6923 (2017). This study demonstrates that pigs that were iatrogenically immunosuppressed developed chronic HEV infection and, therefore, are an important potential model for HEV infection in the context of immunosuppression (e.g. in SOT recipients).
Cossaboom, C. M., Cordoba, L., Dryman, B. A. & Meng, X. J. Hepatitis E virus in rabbits, Virginia, USA. Emerg. Infect. Dis. 17, 2047–2049 (2011).
Cossaboom, C. M. et al. Cross-species infection of pigs with a novel rabbit, but not rat, strain of hepatitis E virus isolated in the United States. J. Gen. Virol. 93, 1687–1695 (2012).
Liu, P. et al. Transmission of hepatitis E virus from rabbits to cynomolgus macaques. Emerg. Infect. Dis. 19, 559–565 (2013).
Wang, L., Xia, J., Wang, L. & Wang, Y. Experimental infection of rabbits with genotype 3 hepatitis E virus produced both chronicity and kidney injury. Gut 66, 561–562 (2017).
Xia, J. et al. Experimental infection of pregnant rabbits with hepatitis E virus demonstrating high mortality and vertical transmission. J. Viral Hepat. 22, 850–857 (2015).
Huang, F. et al. Experimental infection of Balb/c nude mice with Hepatitis E virus. BMC Infect. Dis. 9, 93 (2009).
Mercer, D. F. et al. Hepatitis C virus replication in mice with chimeric human livers. Nat. Med. 7, 927–933 (2001).
Bissig, K. D. et al. Human liver chimeric mice provide a model for hepatitis B and C virus infection and treatment. J. Clin. Invest. 120, 924–930 (2010).
Lutgehetmann, M. et al. Humanized chimeric uPA mouse model for the study of hepatitis B and D virus interactions and preclinical drug evaluation. Hepatology 55, 685–694 (2012).
de Jong, Y. P. et al. Broadly neutralizing antibodies abrogate established hepatitis C virus infection. Sci. Transl Med. 6, 254ra129 (2014).
Winer, B. Y. et al. Recapitulation of treatment response patterns in a novel humanized mouse model for chronic hepatitis B virus infection. Virology 502, 63–72 (2017).
Carpentier, A. et al. Engrafted human stem cell-derived hepatocytes establish an infectious HCV murine model. J. Clin. Invest. 124, 4953–4964 (2014).
VanBuskirk, K. M. et al. Preerythrocytic, live-attenuated Plasmodium falciparum vaccine candidates by design. Proc. Natl Acad. Sci. USA 106, 13004–13009 (2009).
Sacci, J. B. Jr et al. Plasmodium falciparum infection and exoerythrocytic development in mice with chimeric human livers. Int. J. Parasitol. 36, 353–360 (2006).
Mikolajczak, S. A. et al. Plasmodium vivax liver stage development and hypnozoite persistence in human liver-chimeric mice. Cell Host Microbe 17, 526–535 (2015).
Vaughan, A. M. et al. Complete Plasmodium falciparum liver-stage development in liver-chimeric mice. J. Clin. Invest. 122, 3618–3628 (2012).
Soulard, V. et al. Plasmodium falciparum full life cycle and Plasmodium ovale liver stages in humanized mice. Nat. Commun. 6, 7690 (2015).
Allweiss, L. et al. Human liver chimeric mice as a new model of chronic hepatitis E virus infection and preclinical drug evaluation. J. Hepatol. 64, 1033–1040 (2016).
Sayed, I. M. et al. Transmission of hepatitis E virus infection to human-liver chimeric FRG mice using patient plasma. Antiviral Res. 141, 150–154 (2017).
Sayed, I. M. et al. Study of hepatitis E virus infection of genotype 1 and 3 in mice with humanised liver. Gut 66, 920–929 (2017).
van de Garde, M. D. et al. Hepatitis E virus (HEV) genotype 3 infection of human liver chimeric mice as a model for chronic HEV infection. J. Virol. 90, 4394–4401 (2016). This paper demonstrates that human liver chimeric mice are able to support chronic infection with HEV genotype 3, establishing a mouse model to study the virus.
Washburn, M. L. et al. A humanized mouse model to study hepatitis C virus infection, immune response, and liver disease. Gastroenterology 140, 1334–1344 (2011).
Bility, M. T. et al. Hepatitis B virus infection and immunopathogenesis in a humanized mouse model: induction of human-specific liver fibrosis and M2-like macrophages. PLoS Pathog. 10, e1004032 (2014).
Billerbeck, E. et al. Humanized mice efficiently engrafted with fetal hepatoblasts and syngeneic immune cells develop human monocytes and NK cells. J. Hepatol. 65, 334–343 (2016).
Kabrane-Lazizi, Y. et al. Evidence for widespread infection of wild rats with hepatitis E virus in the United States. Am. J. Trop. Med. Hyg. 61, 331–335 (1999).
Debing, Y. et al. A rat model for hepatitis E virus. Dis. Model. Mech. 9, 1203–1210 (2016). This study shows that rats infected with a rat–human chimeric HEV strain are responsive to antiviral therapy, suggesting they could be a potential animal model for HEV studies.
Purcell, R. H. et al. Hepatitis E virus in rats, Los Angeles, California, USA. Emerg. Infect. Dis. 17, 2216–2222 (2011).
Hong, Y. et al. Experimental infection of Z:ZCLA Mongolian gerbils with human hepatitis E virus. World J. Gastroenterol. 21, 862–867 (2015).
Li, W. et al. Experimental infection of Mongolian gerbils by a genotype 4 strain of swine hepatitis E virus. J. Med. Virol. 81, 1591–1596 (2009).
Yang, Y. et al. Effect of swine hepatitis E virus on the livers of experimentally infected Mongolian gerbils by swine hepatitis E virus. Virus Res. 208, 171–179 (2015).
Lee, G. H. et al. Chronic infection with camelid hepatitis E virus in a liver transplant recipient who regularly consumes camel meat and milk. Gastroenterology 150, 355–357.e3 (2016).
Acknowledgements
The authors thank members of the Ploss laboratory for critical discussions of the manuscript. The work was supported in part by grants from Princeton University and an Investigator in Pathogenesis Award by the Burroughs Wellcome Fund (to A.P.). Q.D. is supported by a postdoctoral fellowship from the New Jersey Commission for Cancer Research. The authors apologize to all those whose work could not be cited due to space constraints.
Author information
Authors and Affiliations
Contributions
I.N. and A.P. wrote the article. All authors contributed equally to researching data for the article, discussion of content and reviewing/editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
A.P. and Q.D. are inventors on a patent application defining a hepatitis E virus transcomplementation system for antiviral drug screening and the viroporin function of ORF3 as an antiviral drug target.
Rights and permissions
About this article
Cite this article
Nimgaonkar, I., Ding, Q., Schwartz, R. et al. Hepatitis E virus: advances and challenges. Nat Rev Gastroenterol Hepatol 15, 96–110 (2018). https://doi.org/10.1038/nrgastro.2017.150
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2017.150
This article is cited by
-
The burden of HEV-related acute liver failure in Bangladesh, China and India: a systematic review and meta-analysis
BMC Public Health (2023)
-
Prevalence, incidence, and outcomes of hepatitis E virus coinfection in patients with chronic hepatitis C
Scientific Reports (2023)
-
Complete remission of primary membranous nephropathy following hepatitis E infection
CEN Case Reports (2023)
-
A frantic confusion: beyond rabies and anti-N-methyl-D-aspartate encephalitis
Journal of NeuroVirology (2023)
-
Hepatitis E Virus in the Iberian Peninsula: A Systematic Review
Food and Environmental Virology (2023)