Key Points
-
Common gastrointestinal diseases, such as IBS, functional dyspepsia and IBD, are closely linked to psychological morbidity
-
This link is driven in part through bidirectional signalling between the brain and gut, which reciprocally regulate each other
-
Growing evidence implicates the importance of immune activation, which might be overt (IBD) or more subtle (IBS, functional dyspepsia) in pathological gut–brain interactions
-
The composition of the intestinal microbiota affects behaviour and mood, which could in part rely on selective activation of distinct host cytokine responses
-
Therapeutic targeting of gut microorganisms, host immunity or psychological symptoms could hold the key to uncoupling pathological interactions between the gut and brain
Abstract
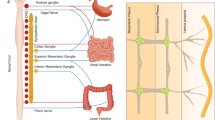
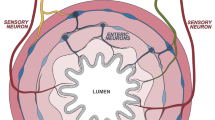
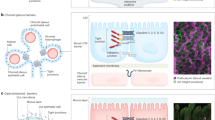
Communication between the brain and gut is not one-way, but a bidirectional highway whereby reciprocal signals between the two organ systems are exchanged to coordinate function. The messengers of this complex dialogue include neural, metabolic, endocrine and immune mediators responsive to diverse environmental cues, including nutrients and components of the intestinal microbiota (microbiota–gut–brain axis). We are now starting to understand how perturbation of these systems affects transition between health and disease. The pathological repercussions of disordered gut–brain dialogue are probably especially pertinent in functional gastrointestinal diseases, including IBS and functional dyspepsia. New insights into these pathways might lead to novel treatment strategies in these common gastrointestinal diseases. In this Review, we consider the role of the immune system as the gatekeeper and master regulator of brain–gut and gut–brain communications. Although adaptive immunity (T cells in particular) participates in this process, there is an emerging role for cells of the innate immune compartment (including innate lymphoid cells and cells of the mononuclear phagocyte system). We will also consider how these key immune cells interact with the specific components of the enteric and central nervous systems, and rapidly respond to environmental variables, including the microbiota, to alter gut homeostasis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Molodecky, N. A. et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 142, 46–54.e42 (2012).
Talley, N. J. & Ford, A. C. Functional dyspepsia. N. Engl. J. Med. 373, 1853–1863 (2015).
Talley, N. J. Functional gastrointestinal disorders as a public health problem. Neurogastroenterol. Motil. 20 (Suppl. 1), 121–129 (2008).
Talley, N. J. & Spiller, R. Irritable bowel syndrome: a little understood organic bowel disease? Lancet 360, 555–564 (2002).
Jostins, L. et al. Host–microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 491, 119–124 (2012).
Trynka, G. et al. Dense genotyping identifies and localizes multiple common and rare variant association signals in celiac disease. Nat. Genet. 43, 1193–1201 (2011).
Ek, W. E. et al. Exploring the genetics of irritable bowel syndrome: a GWA study in the general population and replication in multinational case–control cohorts. Gut 64, 1774–1782 (2015).
Tansey, E. M. Pavlov at home and abroad: his role in international physiology. Auton. Neurosci. 125, 1–11 (2006).
Furness, J. B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 9, 286–294 (2012).
Goyal, R. K. & Hirano, I. The enteric nervous system. N. Engl. J. Med. 334, 1106–1115 (1996).
Savidge, T. C. et al. Enteric glia regulate intestinal barrier function and inflammation via release of S-nitrosoglutathione. Gastroenterology 132, 1344–1358 (2007).
Bohorquez, D. V. & Liddle, R. A. The gut connectome: making sense of what you eat. J. Clin. Invest. 125, 888–890 (2015).
Mace, O. J., Tehan, B. & Marshall, F. Pharmacology and physiology of gastrointestinal enteroendocrine cells. Pharmacol. Res. Perspect. 3, e00155 (2015).
Mace, O. J. & Marshall, F. Digestive physiology of the pig symposium: gut chemosensing and the regulation of nutrient absorption and energy supply. J. Anim. Sci. 91, 1932–1945 (2013).
Hart, B. L. Biological basis of the behavior of sick animals. Neurosci. Biobehav. Rev. 12, 123–137 (1988).
Dantzer, R., O'Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci. 9, 46–56 (2008).
Okada, H., Kuhn, C., Feillet, H. & Bach, J. F. The 'hygiene hypothesis' for autoimmune and allergic diseases: an update. Clin. Exp. Immunol. 160, 1–9 (2010).
Glaser, R., Robles, T. F., Sheridan, J., Malarkey, W. B. & Kiecolt-Glaser, J. K. Mild depressive symptoms are associated with amplified and prolonged inflammatory responses after influenza virus vaccination in older adults. Arch. Gen. Psychiatry 60, 1009–1014 (2003).
McEwen, B. S. Stress, adaptation, and disease: allostasis and allostatic load. Ann. NY Acad. Sci. 840, 33–44 (1998).
Goodhand, J. R. et al. Mood disorders in inflammatory bowel disease: relation to diagnosis, disease activity, perceived stress, and other factors. Inflamm. Bowel Dis. 18, 2301–2309 (2012).
Addolorato, G., Capristo, E., Stefanini, G. F. & Gasbarrini, G. Inflammatory bowel disease: a study of the association between anxiety and depression, physical morbidity, and nutritional status. Scand. J. Gastroenterol. 32, 1013–1021 (1997).
Kovacs, Z. & Kovacs, F. Depressive and anxiety symptoms, dysfunctional attitudes and social aspects in irritable bowel syndrome and inflammatory bowel disease. Int. J. Psychiatry Med. 37, 245–255 (2007).
Kurina, L. M., Goldacre, M. J., Yeates, D. & Gill, L. E. Depression and anxiety in people with inflammatory bowel disease. J. Epidemiol. Community Health 55, 716–720 (2001).
Hauser, W., Janke, K. H., Klump, B. & Hinz, A. Anxiety and depression in patients with inflammatory bowel disease: comparisons with chronic liver disease patients and the general population. Inflamm. Bowel Dis. 17, 621–632 (2011).
Mardini, H. E., Kip, K. E. & Wilson, J. W. Crohn's disease: a two-year prospective study of the association between psychological distress and disease activity. Dig. Dis. Sci. 49, 492–497 (2004).
Persoons, P. et al. The impact of major depressive disorder on the short- and long-term outcome of Crohn's disease treatment with infliximab. Aliment. Pharmacol. Ther. 22, 101–110 (2005).
Mittermaier, C. et al. Impact of depressive mood on relapse in patients with inflammatory bowel disease: a prospective 18-month follow-up study. Psychosom. Med. 66, 79–84 (2004).
Bernstein, C. N. et al. A prospective population-based study of triggers of symptomatic flares in IBD. Am. J. Gastroenterol. 105, 1994–2002 (2010).
Goodhand, J. R. et al. Factors associated with thiopurine non-adherence in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 38, 1097–1108 (2013).
Larauche, M., Mulak, A. & Tache, Y. Stress and visceral pain: from animal models to clinical therapies. Exp. Neurol. 233, 49–67 (2012).
Larauche, M., Gourcerol, G., Million, M., Adelson, D. W. & Tache, Y. Repeated psychological stress-induced alterations of visceral sensitivity and colonic motor functions in mice: influence of surgery and postoperative single housing on visceromotor responses. Stress 13, 343–354 (2010).
Bradesi, S., Eutamene, H., Garcia-Villar, R., Fioramonti, J. & Bueno, L. Acute and chronic stress differently affect visceral sensitivity to rectal distension in female rats. Neurogastroenterol. Motil. 14, 75–82 (2002).
Ren, T. H. et al. Effects of neonatal maternal separation on neurochemical and sensory response to colonic distension in a rat model of irritable bowel syndrome. Am. J. Physiol. Gastrointest. Liver Physiol. 292, G849–G856 (2007).
O'Malley, D., Cryan, J. F. & Dinan, T. G. Crosstalk between interleukin-6 and corticotropin-releasing factor modulate submucosal plexus activity and colonic secretion. Brain Behav. Immun. 30, 115–124 (2013).
Reber, S. O., Obermeier, F., Straub, R. H., Falk, W. & Neumann, I. D. Chronic intermittent psychosocial stress (social defeat/overcrowding) in mice increases the severity of an acute DSS-induced colitis and impairs regeneration. Endocrinology 147, 4968–4976 (2006).
Lennon, E. M. et al. Early life stress triggers persistent colonic barrier dysfunction and exacerbates colitis in adult IL-10−/− mice. Inflamm. Bowel Dis. 19, 712–719 (2013).
Varghese, A. K. et al. Antidepressants attenuate increased susceptibility to colitis in a murine model of depression. Gastroenterology 130, 1743–1753 (2006).
Qiu, B. S., Vallance, B. A., Blennerhassett, P. A. & Collins, S. M. The role of CD4+ lymphocytes in the susceptibility of mice to stress-induced reactivation of experimental colitis. Nat. Med. 5, 1178–1182 (1999).
Adam, B. et al. Severity of mucosal inflammation as a predictor for alterations of visceral sensory function in a rat model. Pain 123, 179–186 (2006).
Mykletun, A. et al. Prevalence of mood and anxiety disorder in self reported irritable bowel syndrome (IBS). An epidemiological population based study of women. BMC Gastroenterol. 10, 88 (2010).
Koloski, N. A. et al. The brain–gut pathway in functional gastrointestinal disorders is bidirectional: a 12-year prospective population-based study. Gut 61, 1284–1290 (2012).
Savas, L. S. et al. Irritable bowel syndrome and dyspepsia among women veterans: prevalence and association with psychological distress. Aliment. Pharmacol. Ther. 29, 115–125 (2009).
Jones, R., Latinovic, R., Charlton, J. & Gulliford, M. Physical and psychological co-morbidity in irritable bowel syndrome: a matched cohort study using the General Practice Research Database. Aliment. Pharmacol. Ther. 24, 879–886 (2006).
Tayama, J. et al. Maladjustment to academic life and employment anxiety in university students with irritable bowel syndrome. PLoS ONE 10, e0129345 (2015).
Vu, J., Kushnir, V., Cassell, B., Gyawali, C. P. & Sayuk, G. S. The impact of psychiatric and extraintestinal comorbidity on quality of life and bowel symptom burden in functional GI disorders. Neurogastroenterol. Motil. 26, 1323–1332 (2014).
Kennedy, P. J. et al. Cognitive performance in irritable bowel syndrome: evidence of a stress-related impairment in visuospatial memory. Psychol. Med. 44, 1553–1566 (2014).
Canavan, J. B., Bennett, K., Feely, J., O'Morain, C. A. & O'Connor, H. J. Significant psychological morbidity occurs in irritable bowel syndrome: a case–control study using a pharmacy reimbursement database. Aliment. Pharmacol. Ther. 29, 440–449 (2009).
Qi, R. et al. Intrinsic brain abnormalities in irritable bowel syndrome and effect of anxiety and depression. Brain Imaging Behav. http://dx.doi.org/10.1007/s11682-015-9478-1 (2015).
Icenhour, A. et al. Neural circuitry of abdominal pain-related fear learning and reinstatement in irritable bowel syndrome. Neurogastroenterol. Motil. 27, 114–127 (2015).
Song, G. H. et al. Cortical effects of anticipation and endogenous modulation of visceral pain assessed by functional brain MRI in irritable bowel syndrome patients and healthy controls. Pain 126, 79–90 (2006).
Hong, J. Y. et al. Altered brain responses in subjects with irritable bowel syndrome during cued and uncued pain expectation. Neurogastroenterol. Motil. 28, 127–138 (2016).
Grzesiak, M. et al. The lifetime prevalence of anxiety disorders among patients with irritable bowel syndrome. Adv. Clin. Exp. Med. 23, 987–992 (2014).
Hillila, M. T., Siivola, M. T. & Farkkila, M. A. Comorbidity and use of health-care services among irritable bowel syndrome sufferers. Scand. J. Gastroenterol. 42, 799–806 (2007).
Talley, N. J., Fett, S. L., Zinsmeister, A. R. & Melton, L. J. 3rd. Gastrointestinal tract symptoms and self-reported abuse: a population-based study. Gastroenterology 107, 1040–1049 (1994).
Talley, N. J., Fett, S. L. & Zinsmeister, A. R. Self-reported abuse and gastrointestinal disease in outpatients: association with irritable bowel-type symptoms. Am. J. Gastroenterol. 90, 366–371 (1995).
Drossman, D. A. et al. Sexual and physical abuse in women with functional or organic gastrointestinal disorders. Ann. Intern. Med. 113, 828–833 (1990).
Koloski, N. A., Jones, M. & Talley, N. J. Evidence that independent gut-to-brain and brain-to-gut pathways operate in the irritable bowel syndrome and functional dyspepsia: a 1-year population-based prospective study. Aliment. Pharmacol. Ther. 44, 592–600 (2016).
Thabane, M., Kottachchi, D. T. & Marshall, J. K. Systematic review and meta-analysis: the incidence and prognosis of post-infectious irritable bowel syndrome. Aliment. Pharmacol. Ther. 26, 535–544 (2007).
Marshall, J. K. et al. Incidence and epidemiology of irritable bowel syndrome after a large waterborne outbreak of bacterial dysentery. Gastroenterology 131, 445–450 (2006).
Ford, A. C. et al. Prevalence of uninvestigated dyspepsia 8 years after a large waterborne outbreak of bacterial dysentery: a cohort study. Gastroenterology 138, 1727–1736 (2010).
Wouters, M. M. et al. Psychological comorbidity increases the risk for postinfectious IBS partly by enhanced susceptibility to develop infectious gastroenteritis. Gut 65, 1279–1288 (2016).
Dunlop, S. P., Jenkins, D., Neal, K. R. & Spiller, R. C. Relative importance of enterochromaffin cell hyperplasia, anxiety, and depression in postinfectious IBS. Gastroenterology 125, 1651–1659 (2003).
Neurath, M. F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 14, 329–342 (2014).
Isidro, R. A. & Appleyard, C. B. Colonic macrophage polarization in homeostasis, inflammation, and cancer. Am. J. Physiol. Gastrointest. Liver Physiol. 311, G59–G73 (2016).
La Flamme, A. C. et al. Type II-activated murine macrophages produce IL-4. PLoS ONE 7, e46989 (2012).
Goldberg, R., Prescott, N., Lord, G. M., MacDonald, T. T. & Powell, N. The unusual suspects — innate lymphoid cells as novel therapeutic targets in IBD. Nat. Rev. Gastroenterol. Hepatol. 12, 271–283 (2015).
Goto, Y. et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science 345, 1254009 (2014).
Zimmerman, N. P., Vongsa, R. A., Wendt, M. K. & Dwinell, M. B. Chemokines and chemokine receptors in mucosal homeostasis at the intestinal epithelial barrier in inflammatory bowel disease. Inflamm. Bowel Dis. 14, 1000–1011 (2008).
Charo, I. F. & Ransohoff, R. M. The many roles of chemokines and chemokine receptors in inflammation. N. Engl. J. Med. 354, 610–621 (2006).
Habtezion, A., Nguyen, L. P., Hadeiba, H. & Butcher, E. C. Leukocyte trafficking to the small intestine and colon. Gastroenterology 150, 340–354 (2016).
Feagan, B. G. et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 369, 699–710 (2013).
Sandborn, W. J. et al. Vedolizumab as induction and maintenance therapy for Crohn's disease. N. Engl. J. Med. 369, 711–721 (2013).
Ulrich-Lai, Y. M. & Herman, J. P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 10, 397–409 (2009).
Elenkov, I. J., Wilder, R. L., Chrousos, G. P. & Vizi, E. S. The sympathetic nerve — an integrative interface between two supersystems: the brain and the immune system. Pharmacol. Rev. 52, 595–638 (2000).
Felten, D. L., Felten, S. Y., Carlson, S. L., Olschowka, J. A. & Livnat, S. Noradrenergic and peptidergic innervation of lymphoid tissue. J. Immunol. 135, 755s–765s (1985).
Mawdsley, J. E., Macey, M. G., Feakins, R. M., Langmead, L. & Rampton, D. S. The effect of acute psychologic stress on systemic and rectal mucosal measures of inflammation in ulcerative colitis. Gastroenterology 131, 410–419 (2006).
Vanuytsel, T. et al. Psychological stress and corticotropin-releasing hormone increase intestinal permeability in humans by a mast cell-dependent mechanism. Gut 63, 1293–1299 (2014).
Nakano, K. et al. Dopamine induces IL-6-dependent IL-17 production via D1-like receptor on CD4 naive T cells and D1-like receptor antagonist SCH-23390 inhibits cartilage destruction in a human rheumatoid arthritis/SCID mouse chimera model. J. Immunol. 186, 3745–3752 (2011).
Khan, N. A. & Poisson, J. P. 5-HT3 receptor-channels coupled with Na+ influx in human T cells: role in T cell activation. J. Neuroimmunol. 99, 53–60 (1999).
Aune, T. M., McGrath, K. M., Sarr, T., Bombara, M. P. & Kelley, K. A. Expression of 5HT1a receptors on activated human T cells. Regulation of cyclic AMP levels and T cell proliferation by 5-hydroxytryptamine. J. Immunol. 151, 1175–1183 (1993).
Buttari, B. et al. Neuropeptide Y induces potent migration of human immature dendritic cells and promotes a Th2 polarization. FASEB J. 28, 3038–3049 (2014).
Payan, D. G., Brewster, D. R., Missirian-Bastian, A. & Goetzl, E. J. Substance P recognition by a subset of human T lymphocytes. J. Clin. Invest. 74, 1532–1539 (1984).
Johnson, M. C., McCormack, R. J., Delgado, M., Martinez, C. & Ganea, D. Murine T-lymphocytes express vasoactive intestinal peptide receptor 1 (VIP-R1) mRNA. J. Neuroimmunol. 68, 109–119 (1996).
Sanders, V. M. et al. Differential expression of the β2-adrenergic receptor by Th1 and Th2 clones: implications for cytokine production and B cell help. J. Immunol. 158, 4200–4210 (1997).
Sanders, V. M. The role of adrenoceptor-mediated signals in the modulation of lymphocyte function. Adv. Neuroimmunol. 5, 283–298 (1995).
Sanders, V. M. & Straub, R. H. Norepinephrine, the β-adrenergic receptor, and immunity. Brain Behav. Immun. 16, 290–332 (2002).
Panina-Bordignon, P. et al. β2-agonists prevent Th1 development by selective inhibition of interleukin 12. J. Clin. Invest. 100, 1513–1519 (1997).
Ramer-Quinn, D. S., Baker, R. A. & Sanders, V. M. Activated T helper 1 and T helper 2 cells differentially express the beta-2-adrenergic receptor: a mechanism for selective modulation of T helper 1 cell cytokine production. J. Immunol. 159, 4857–4867 (1997).
Takenaka, M. C. et al. Norepinephrine controls effector T cell differentiation through β2-adrenergic receptor-mediated inhibition of NF-κB and AP-1 in dendritic cells. J. Immunol. 196, 637–644 (2016).
Orand, A. et al. Catecholaminergic gene polymorphisms are associated with GI symptoms and morphological brain changes in irritable bowel syndrome. PLoS ONE 10, e0135910 (2015).
Gabanyi, I. et al. Neuro-immune interactions drive tissue programming in intestinal macrophages. Cell 164, 378–391 (2016).
Kurowska-Stolarska, M. et al. IL-33 amplifies the polarization of alternatively activated macrophages that contribute to airway inflammation. J. Immunol. 183, 6469–6477 (2009).
Li, D. et al. IL-33 promotes ST2-dependent lung fibrosis by the induction of alternatively activated macrophages and innate lymphoid cells in mice. J. Allergy Clin. Immunol. 134, 1422–1432.e11 (2014).
Cirillo, C. et al. Evidence for neuronal and structural changes in submucous ganglia of patients with functional dyspepsia. Am. J. Gastroenterol. 110, 1205–1215 (2015).
Girodet, P. O. et al. Alternative macrophage activation is increased in asthma. Am. J. Respir. Cell Mol. Biol. 55, 467–475 (2016).
Airaksinen, M. S. & Saarma, M. The GDNF family: signalling, biological functions and therapeutic value. Nat. Rev. Neurosci. 3, 383–394 (2002).
Ibiza, S. et al. Glial-cell-derived neuroregulators control type 3 innate lymphoid cells and gut defence. Nature 535, 440–443 (2016).
Limsui, D. et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm. Bowel Dis. 13, 175–181 (2007).
Midhagen, G. & Hallert, C. High rate of gastrointestinal symptoms in celiac patients living on a gluten-free diet: controlled study. Am. J. Gastroenterol. 98, 2023–2026 (2003).
Isgar, B., Harman, M., Kaye, M. D. & Whorwell, P. J. Symptoms of irritable bowel syndrome in ulcerative colitis in remission. Gut 24, 190–192 (1983).
Farrokhyar, F., Marshall, J. K., Easterbrook, B. & Irvine, E. J. Functional gastrointestinal disorders and mood disorders in patients with inactive inflammatory bowel disease: prevalence and impact on health. Inflamm. Bowel Dis. 12, 38–46 (2006).
Ford, A. C., Talley, N. J., Walker, M. M. & Jones, M. P. Increased prevalence of autoimmune diseases in functional gastrointestinal disorders: case–control study of 23471 primary care patients. Aliment. Pharmacol. Ther. 40, 827–834 (2014).
Bashashati, M. et al. Cytokine imbalance in irritable bowel syndrome: a systematic review and meta-analysis. Neurogastroenterol. Motil. 26, 1036–1048 (2014).
Dinan, T. G. et al. Hypothalamic–pituitary–gut axis dysregulation in irritable bowel syndrome: plasma cytokines as a potential biomarker? Gastroenterology 130, 304–311 (2006).
Dinan, T. G. et al. Enhanced cholinergic-mediated increase in the pro-inflammatory cytokine IL-6 in irritable bowel syndrome: role of muscarinic receptors. Am. J. Gastroenterol. 103, 2570–2576 (2008).
Liebregts, T. et al. Immune activation in patients with irritable bowel syndrome. Gastroenterology 132, 913–920 (2007).
Liebregts, T. et al. Small bowel homing T cells are associated with symptoms and delayed gastric emptying in functional dyspepsia. Am. J. Gastroenterol. 106, 1089–1098 (2011).
Gao, J. Correlation between anxiety-depression status and cytokines in diarrhea-predominant irritable bowel syndrome. Exp. Ther. Med. 6, 93–96 (2013).
Aerssens, J. et al. Alterations in mucosal immunity identified in the colon of patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 6, 194–205 (2008).
Macsharry, J. et al. Mucosal cytokine imbalance in irritable bowel syndrome. Scand. J. Gastroenterol. 43, 1467–1476 (2008).
Barbara, G. et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology 126, 693–702 (2004).
Guilarte, M. et al. Diarrhoea-predominant IBS patients show mast cell activation and hyperplasia in the jejunum. Gut 56, 203–209 (2007).
Martinez, C. et al. Diarrhoea-predominant irritable bowel syndrome: an organic disorder with structural abnormalities in the jejunal epithelial barrier. Gut 62, 1160–1168 (2013).
Weston, A. P., Biddle, W. L., Bhatia, P. S. & Miner, P. B. Jr. Terminal ileal mucosal mast cells in irritable bowel syndrome. Dig. Dis. Sci. 38, 1590–1595 (1993).
Akbar, A. et al. Increased capsaicin receptor TRPV1-expressing sensory fibres in irritable bowel syndrome and their correlation with abdominal pain. Gut 57, 923–929 (2008).
Cremon, C. et al. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am. J. Gastroenterol. 104, 392–400 (2009).
Barbara, G. et al. Mast cell-dependent excitation of visceral-nociceptive sensory neurons in irritable bowel syndrome. Gastroenterology 132, 26–37 (2007).
Buhner, S. et al. Submucous rather than myenteric neurons are activated by mucosal biopsy supernatants from irritable bowel syndrome patients. Neurogastroenterol. Motil. 24, 1134–e572 (2012).
Wouters, M. M. et al. Histamine receptor H1-mediated sensitization of TRPV1 mediates visceral hypersensitivity and symptoms in patients with irritable bowel syndrome. Gastroenterology 150, 875–887.e89 (2016).
Santos, J., Yang, P. C., Soderholm, J. D., Benjamin, M. & Perdue, M. H. Role of mast cells in chronic stress induced colonic epithelial barrier dysfunction in the rat. Gut 48, 630–636 (2001).
Ronkainen, J. et al. Prevalence of oesophageal eosinophils and eosinophilic oesophagitis in adults: the population-based Kalixanda study. Gut 56, 615–620 (2007).
Vanheel, H. et al. Impaired duodenal mucosal integrity and low-grade inflammation in functional dyspepsia. Gut 63, 262–271 (2014).
Walker, M. M. et al. Duodenal eosinophilia and early satiety in functional dyspepsia: confirmation of a positive association in an Australian cohort. J. Gastroenterol. Hepatol. 29, 474–479 (2014).
Walker, M. M. et al. Implications of eosinophilia in the normal duodenal biopsy — an association with allergy and functional dyspepsia. Aliment. Pharmacol. Ther. 31, 1229–1236 (2010).
Futagami, S. et al. Migration of eosinophils and CCR2-/CD68-double positive cells into the duodenal mucosa of patients with postinfectious functional dyspepsia. Am. J. Gastroenterol. 105, 1835–1842 (2010).
Powell, N., Walker, M. M. & Talley, N. J. Gastrointestinal eosinophils in health, disease and functional disorders. Nat. Rev. Gastroenterol. Hepatol. 7, 146–156 (2010).
Kindt, S. et al. Immune dysfunction in patients with functional gastrointestinal disorders. Neurogastroenterol. Motil. 21, 389–398 (2009).
Ohman, L., Isaksson, S., Lundgren, A., Simren, M. & Sjovall, H. A controlled study of colonic immune activity and β7+ blood T lymphocytes in patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 3, 980–986 (2005).
Spiller, R. C. et al. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut 47, 804–811 (2000).
Elsenbruch, S. et al. Are there alterations of neuroendocrine and cellular immune responses to nutrients in women with irritable bowel syndrome? Am. J. Gastroenterol. 99, 703–710 (2004).
Bal, S. M. et al. IL-1β, IL-4 and IL-12 control the fate of group 2 innate lymphoid cells in human airway inflammation in the lungs. Nat. Immunol. 17, 636–645 (2016).
Suto, G., Kiraly, A. & Tache, Y. Interleukin 1β inhibits gastric emptying in rats: mediation through prostaglandin and corticotropin-releasing factor. Gastroenterology 106, 1568–1575 (1994).
Hermann, G. & Rogers, R. C. Tumor necrosis factor-alpha in the dorsal vagal complex suppresses gastric motility. Neuroimmunomodulation 2, 74–81 (1995).
Benakis, C. et al. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nat. Med. 22, 516–523 (2016).
Felger, J. C. & Lotrich, F. E. Inflammatory cytokines in depression: neurobiological mechanisms and therapeutic implications. Neuroscience 246, 199–229 (2013).
Emch, G. S., Hermann, G. E. & Rogers, R. C. TNF-α activates solitary nucleus neurons responsive to gastric distension. Am. J. Physiol. Gastrointest. Liver Physiol. 279, G582–G586 (2000).
Soulet, D. & Rivest, S. Bone-marrow-derived microglia: myth or reality? Curr. Opin. Pharmacol. 8, 508–518 (2008).
Rivest, S. Molecular insights on the cerebral innate immune system. Brain Behav. Immun. 17, 13–19 (2003).
Jack, C. S. et al. TLR signaling tailors innate immune responses in human microglia and astrocytes. J. Immunol. 175, 4320–4330 (2005).
Arnett, H. A. et al. TNFα promotes proliferation of oligodendrocyte progenitors and remyelination. Nat. Neurosci. 4, 1116–1122 (2001).
Klein, M. et al. Innate immunity to pneumococcal infection of the central nervous system depends on Toll-like receptor (TLR) 2 and TLR4. J. Infect. Dis. 198, 1028–1036 (2008).
Zhang, S. Y. et al. TLR3 deficiency in patients with herpes simplex encephalitis. Science 317, 1522–1527 (2007).
Bogie, J. F., Stinissen, P. & Hendriks, J. J. Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol. 128, 191–213 (2014).
Liu, Y. et al. LPS receptor (CD14): a receptor for phagocytosis of Alzheimer's amyloid peptide. Brain 128, 1778–1789 (2005).
Casellas, F., Aguade, S. & Molero, J. Intestinal permeability in inflammatory bowel disease. Am. J. Gastroenterol. 81, 502 (1986).
Mujagic, Z. et al. Small intestinal permeability is increased in diarrhoea predominant IBS, while alterations in gastroduodenal permeability in all IBS subtypes are largely attributable to confounders. Aliment. Pharmacol. Ther. 40, 288–297 (2014).
Dlugosz, A. et al. Increased serum levels of lipopolysaccharide and antiflagellin antibodies in patients with diarrhea-predominant irritable bowel syndrome. Neurogastroenterol. Motil. 27, 1747–1754 (2015).
McDonnell, M. et al. Systemic Toll-like receptor ligands modify B-cell responses in human inflammatory bowel disease. Inflamm. Bowel Dis. 17, 298–307 (2011).
Dunn, A. J., Swiergiel, A. H. & de Beaurepaire, R. Cytokines as mediators of depression: what can we learn from animal studies? Neurosci. Biobehav. Rev. 29, 891–909 (2005).
Kronfol, Z. & House, J. D. Lymphocyte mitogenesis, immunoglobulin and complement levels in depressed patients and normal controls. Acta Psychiatr. Scand. 80, 142–147 (1989).
Maes, M. et al. Evidence for a systemic immune activation during depression: results of leukocyte enumeration by flow cytometry in conjunction with monoclonal antibody staining. Psychol. Med. 22, 45–53 (1992).
Miyaoka, H. et al. Depression from interferon therapy in patients with hepatitis C. Am. J. Psychiatry 156, 1120 (1999).
Loftis, J. M. et al. Vulnerability to somatic symptoms of depression during interferon-alpha therapy for hepatitis C: a 16-week prospective study. J. Psychosom. Res. 74, 57–63 (2013).
Dantzer, R. & Kelley, K. W. Twenty years of research on cytokine-induced sickness behavior. Brain Behav. Immun. 21, 153–160 (2007).
Kapas, L. et al. Somnogenic, pyrogenic, and anorectic activities of tumor necrosis factor-alpha and TNF-alpha fragments. Am. J. Physiol. 263, R708–R715 (1992).
Kent, S., Rodriguez, F., Kelley, K. W. & Dantzer, R. Reduction in food and water intake induced by microinjection of interleukin-1 beta in the ventromedial hypothalamus of the rat. Physiol. Behav. 56, 1031–1036 (1994).
Besedovsky, H., del Rey, A., Sorkin, E. & Dinarello, C. A. Immunoregulatory feedback between interleukin-1 and glucocorticoid hormones. Science 233, 652–654 (1986).
Silverman, M. N., Pearce, B. D. & Miller, A. H. in Cytokines and Mental Health ( ed Kronfol, Z. ) 85–122 (Springer Science, 2003).
Mark, K. S. & Miller, D. W. Increased permeability of primary cultured brain microvessel endothelial cell monolayers following TNF-α exposure. Life Sci. 64, 1941–1953 (1999).
Lopez-Ramirez, M. A. et al. Role of caspases in cytokine-induced barrier breakdown in human brain endothelial cells. J. Immunol. 189, 3130–3139 (2012).
Cohen, S. S. et al. Effects of interleukin-6 on the expression of tight junction proteins in isolated cerebral microvessels from yearling and adult sheep. Neuroimmunomodulation 20, 264–273 (2013).
Maruo, N., Morita, I., Shirao, M. & Murota, S. IL-6 increases endothelial permeability in vitro. Endocrinology 131, 710–714 (1992).
Bsibsi, M. et al. Toll-like receptor 3 on adult human astrocytes triggers production of neuroprotective mediators. Glia 53, 688–695 (2006).
Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Kostic, A. D., Xavier, R. J. & Gevers, D. The microbiome in inflammatory bowel disease: current status and the future ahead. Gastroenterology 146, 1489–1499 (2014).
Chassaing, B. et al. Crohn disease — associated adherent-invasive E. coli bacteria target mouse and human Peyer's patches via long polar fimbriae. J. Clin. Invest. 121, 966–975 (2011).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl Acad. Sci. USA 105, 16731–16736 (2008).
Machiels, K. et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 63, 1275–1283 (2014).
Carroll, I. M., Ringel-Kulka, T., Siddle, J. P. & Ringel, Y. Alterations in composition and diversity of the intestinal microbiota in patients with diarrhea-predominant irritable bowel syndrome. Neurogastroenterol. Motil. 24, 521–e248 (2012).
Sundin, J. et al. Altered faecal and mucosal microbial composition in post-infectious irritable bowel syndrome patients correlates with mucosal lymphocyte phenotypes and psychological distress. Aliment. Pharmacol. Ther. 41, 342–351 (2015).
Giamarellos-Bourboulis, E. et al. Molecular assessment of differences in the duodenal microbiome in subjects with irritable bowel syndrome. Scand. J. Gastroenterol. 50, 1076–1087 (2015).
Jeffery, I. B. et al. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 61, 997–1006 (2012).
Ikeda, M., Hamada, K., Sumitomo, N., Okamoto, H. & Sakakibara, B. Serum amyloid A, cytokines, and corticosterone responses in germfree and conventional mice after lipopolysaccharide injection. Biosci. Biotechnol. Biochem. 63, 1006–1010 (1999).
Souza, D. G. et al. The essential role of the intestinal microbiota in facilitating acute inflammatory responses. J. Immunol. 173, 4137–4146 (2004).
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
Atarashi, K. et al. Th17 cell induction by adhesion of microbes to intestinal epithelial cells. Cell 163, 367–380 (2015).
Seo, S. U. et al. Distinct commensals induce interleukin-1β via NLRP3 inflammasome in inflammatory monocytes to promote intestinal inflammation in response to injury. Immunity 42, 744–755 (2015).
Lakhdari, O. et al. Functional metagenomics: a high throughput screening method to decipher microbiota-driven NF-κB modulation in the human gut. PLoS ONE 5, e13092 (2010).
Powell, N. et al. The transcription factor T-bet regulates intestinal inflammation mediated by interleukin-7 receptor+ innate lymphoid cells. Immunity 37, 674–684 (2012).
Furusawa, Y. et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504, 446–450 (2013).
Smith, P. M. et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341, 569–573 (2013).
Round, J. L. & Mazmanian, S. K. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl Acad. Sci. USA 107, 12204–12209 (2010).
Bailey, M. T. et al. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav. Immun. 25, 397–407 (2011).
Sudo, N. et al. Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice. J. Physiol. 558, 263–275 (2004).
Clarke, G. et al. The microbiome–gut–brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatry 18, 666–673 (2013).
Neufeld, K. M., Kang, N., Bienenstock, J. & Foster, J. A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 23, 255–e119 (2011).
Diaz Heijtz, R. et al. Normal gut microbiota modulates brain development and behavior. Proc. Natl Acad. Sci. USA 108, 3047–3052 (2011).
Galley, J. D. et al. Exposure to a social stressor disrupts the community structure of the colonic mucosa-associated microbiota. BMC Microbiol. 14, 189 (2014).
Galley, J. D. & Bailey, M. T. Impact of stressor exposure on the interplay between commensal microbiota and host inflammation. Gut Microbes 5, 390–396 (2014).
Galley, J. D. et al. The structures of the colonic mucosa-associated and luminal microbial communities are distinct and differentially affected by a prolonged murine stressor. Gut Microbes 5, 748–760 (2014).
Bailey, M. T. et al. Stressor exposure disrupts commensal microbial populations in the intestines and leads to increased colonization by Citrobacter rodentium. Infect. Immun. 78, 1509–1519 (2010).
Gareau, M. G. et al. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60, 307–317 (2011).
Desbonnet, L., Clarke, G., Shanahan, F., Dinan, T. G. & Cryan, J. F. Microbiota is essential for social development in the mouse. Mol. Psychiatry 19, 146–148 (2014).
Houlden, A. et al. Brain injury induces specific changes in the caecal microbiota of mice via altered autonomic activity and mucoprotein production. Brain Behav. Immun. 57, 10–20 (2016).
Kabouridis, P. S. & Pachnis, V. Emerging roles of gut microbiota and the immune system in the development of the enteric nervous system. J. Clin. Invest. 125, 956–964 (2015).
Collins, J., Borojevic, R., Verdu, E. F., Huizinga, J. D. & Ratcliffe, E. M. Intestinal microbiota influence the early postnatal development of the enteric nervous system. Neurogastroenterol. Motil. 26, 98–107 (2014).
Luczynski, P. et al. Adult microbiota-deficient mice have distinct dendritic morphological changes: differential effects in the amygdala and hippocampus. Eur. J. Neurosci. 44, 2654–2666 (2016).
Ford, A. C. et al. Effect of antidepressants and psychological therapies, including hypnotherapy, in irritable bowel syndrome: systematic review and meta-analysis. Am. J. Gastroenterol. 109, 1350–1365 (2014).
Lackner, J. M. et al. Rapid response to cognitive behavior therapy predicts treatment outcome in patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 8, 426–432 (2010).
Everitt, H. et al. Assessing Cognitive behavioural Therapy in Irritable Bowel (ACTIB): protocol for a randomised controlled trial of clinical-effectiveness and cost-effectiveness of therapist delivered cognitive behavioural therapy and web-based self-management in irritable bowel syndrome in adults. BMJ Open 5, e008622 (2015).
Lackner, J. M. et al. The Irritable Bowel Syndrome Outcome Study (IBSOS): rationale and design of a randomized, placebo-controlled trial with 12 month follow up of self- versus clinician-administered CBT for moderate to severe irritable bowel syndrome. Contemp. Clin. Trials 33, 1293–1310 (2012).
Dunlop, S. P. et al. Randomized, double-blind, placebo-controlled trial of prednisolone in post-infectious irritable bowel syndrome. Aliment. Pharmacol. Ther. 18, 77–84 (2003).
Feldmann, M. Many cytokines are very useful therapeutic targets in disease. J. Clin. Invest. 118, 3533–3536 (2008).
Stefanini, G. F. et al. Oral cromolyn sodium in comparison with elimination diet in the irritable bowel syndrome, diarrheic type. Multicenter study of 428 patients. Scand. J. Gastroenterol. 30, 535–541 (1995).
Lunardi, C. et al. Double-blind cross-over trial of oral sodium cromoglycate in patients with irritable bowel syndrome due to food intolerance. Clin. Exp. Allergy 21, 569–572 (1991).
Bahi, A., Schwed, J. S., Walter, M., Stark, H. & Sadek, B. Anxiolytic and antidepressant-like activities of the novel and potent non-imidazole histamine H3 receptor antagonist ST-1283. Drug Des. Devel. Ther. 8, 627–637 (2014).
Spiller, R. Review article: probiotics and prebiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 28, 385–396 (2008).
Marchesi, J. R. et al. The gut microbiota and host health: a new clinical frontier. Gut 65, 330–339 (2016).
Moran, C. & Shanahan, F. Editorial: probiotics and IBS — where are we now? Aliment. Pharmacol. Ther. 40, 318 (2014).
Pinn, D. M., Aroniadis, O. C. & Brandt, L. J. Is fecal microbiota transplantation (FMT) an effective treatment for patients with functional gastrointestinal disorders (FGID)? Neurogastroenterol. Motil. 27, 19–29 (2015).
O'Mahony, L. et al. Lactobacillus and bifidobacterium in irritable bowel syndrome: symptom responses and relationship to cytokine profiles. Gastroenterology 128, 541–551 (2005).
Pimentel, M. et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N. Engl. J. Med. 364, 22–32 (2011).
Chumpitazi, B. P. et al. Randomised clinical trial: gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 42, 418–427 (2015).
Halmos, E. P., Power, V. A., Shepherd, S. J., Gibson, P. R. & Muir, J. G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 146, 67–75.e5 (2014).
McIntosh, K. et al. FODMAPs alter symptoms and the metabolome of patients with IBS: a randomised controlled trial. Gut http://dx.doi.org/10.1136/gutjnl-2015-311339 (2016).
Zucchelli, M. et al. Association of TNFSF15 polymorphism with irritable bowel syndrome. Gut 60, 1671–1677 (2011).
Swan, C. et al. Identifying and testing candidate genetic polymorphisms in the irritable bowel syndrome (IBS): association with TNFSF15 and TNFα. Gut 62, 985–994 (2013).
Author information
Authors and Affiliations
Contributions
N.P. and M.M.W. researched data for the article, N.P. wrote the article and N.P., M.M.W. and N.J.T. made substantial contributions to discussion of content and reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Powell, N., Walker, M. & Talley, N. The mucosal immune system: master regulator of bidirectional gut–brain communications. Nat Rev Gastroenterol Hepatol 14, 143–159 (2017). https://doi.org/10.1038/nrgastro.2016.191
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2016.191
This article is cited by
-
Oral administration of DNA alginate nanovaccine induced immune-protection against Helicobacter pylori in Balb/C mice
BMC Immunology (2024)
-
Gastrointestinal and brain barriers: unlocking gates of communication across the microbiota–gut–brain axis
Nature Reviews Gastroenterology & Hepatology (2024)
-
A comprehensive review on phytochemicals as potential therapeutic agents for stress-induced gastric ulcer
Journal of Umm Al-Qura University for Applied Sciences (2024)
-
Modelling host–microbiome interactions in organ-on-a-chip platforms
Nature Reviews Bioengineering (2023)
-
Proenkephalin deletion in hematopoietic cells induces intestinal barrier failure resulting in clinical feature similarities with irritable bowel syndrome in mice
Communications Biology (2023)