Abstract
Well-differentiated pancreatic neuroendocrine tumors (PanNETs) comprise ∼1–3% of pancreatic neoplasms. Although long considered as reasonably benign lesions, PanNETs have considerable malignant potential, with a 5-year survival of ∼65% and a 10-year survival of 45% for resected lesions. As PanNETs have a low incidence, they have been understudied, with few advances made until the completion of their exomic sequencing in the past year. In this Review, we summarize some of the latest insights into the genetics of PanNETs, and their probable implications in the context of prognosis and therapy. In particular, we discuss two genes (DAXX and ATRX) that have collectively been identified as mutated in >40% of PanNETs, and the biological and prognostic implications of these novel mutations. The identification of recurrent somatic mutations within the mTOR signaling pathway and the therapeutic implications for personalized therapy in patients with PanNETs are also discussed. Finally, this Review outlines state-of-the-art advances in the biology of PanNETs that are of emerging translational importance.
Key Points
-
Pancreatic neuroendocrine tumors (PanNETs) are fully malignant neoplasms, with the majority (∼70%) of patients presenting with advanced disease and a 5-year survival of ∼65%
-
In 2010, the WHO implemented a new classification for PanNETs that stratifies these tumors by stage and histological grade (the latter defined by mitotic counts or Ki-67 labeling index)
-
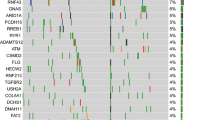
Sequencing of the PanNET exome has identified somatic mutations in MEN1, DAXX, ATRX and mTOR pathway genes in 44%, 25%, 18% and ∼16% of PanNETs, respectively
-
ATRX and DAXX are PanNET tumor suppressor genes that are involved in chromatin remodeling—mutations in these genes are correlated with the alternative lengthening of telomeres phenotype
-
Two molecularly targeted therapies—sunitinib (VEGF inhibitor) and everolimus (mTOR inhibitor)—have been independently shown to improve progression-free survival in patients with metastatic PanNETs, and are approved by the FDA
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Yao, J. C. et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J. Clin. Oncol. 26, 3063–3072 (2008).
Halfdanarson, T. R., Rabe, K. G., Rubin, J. & Petersen, G. M. Pancreatic neuroendocrine tumors (PNETs): incidence, prognosis and recent trend toward improved survival. Ann. Oncol. 19, 1727–1733 (2008).
Kouvaraki, M. A. et al. Fluorouracil, doxorubicin, and streptozocin in the treatment of patients with locally advanced and metastatic pancreatic endocrine carcinomas. J. Clin. Oncol. 22, 4762–4771 (2004).
Moertel, C. G., Hanley, J. A. & Johnson, L. A. Streptozocin alone compared with streptozocin plus fluorouracil in the treatment of advanced islet-cell carcinoma. N. Engl. J. Med. 303, 1189–1194 (1980).
Moertel, C. G., Lefkopoulo, M., Lipsitz, S., Hahn, R. G. & Klaassen, D. Streptozocin-doxorubicin, streptozocin-fluorouracil or chlorozotocin in the treatment of advanced islet-cell carcinoma. N. Engl. J. Med. 326, 519–523 (1992).
Cheng, P. N. & Saltz, L. B. Failure to confirm major objective antitumor activity for streptozocin and doxorubicin in the treatment of patients with advanced islet cell carcinoma. Cancer 86, 944–948 (1999).
McCollum, A. D. et al. Lack of efficacy of streptozocin and doxorubicin in patients with advanced pancreatic endocrine tumors. Am. J. Clin. Oncol. 27, 485–488 (2004).
Klöppel, G. Classification and pathology of gastroenteropancreatic neuroendocrine neoplasms. Endocr. Relat. Cancer 18 (Suppl. 1), S1–S16 (2011).
Rindi, G. et al. in WHO Classification of Tumours of the Digestive System (eds Bosman, F. T., Carneiro, F., Hruban, R. H. & Theise, N.) 13–14 (IARC, Lyon, 2010).
Klimstra, D. S. et al. in WHO Classification of Tumours of the Digestive System (eds Bosman, F. T., Carneiro, F., Hruban, R. H. & Theise, N.) 322–326 (IARC, Lyon, 2010).
Yachida, S. et al. Small cell and large cell neuroendocrine carcinomas of the pancreas are genetically similar and distinct from well-differentiated pancreatic neuroendocrine tumors. Am. J. Surg. Pathol. 36, 173–184 (2012).
Halfdanarson, T. R., Rubin J., Farnell M. B., Grant C. S. & Petersen G. M. Pancreatic endocrine neoplasms: epidemiology and prognosis of pancreatic endocrine tumors. Endocr. Relat. Cancer 15, 409–427 (2008).
Whipple, A. O. The surgical therapy of hyperinsulinism. J. Int. Chir. 3, 237–276 (1938).
Zollinger, R. M. & Ellison, E. H. Primary peptic ulcerations of the jejunum associated with islet cell tumors of the pancreas. Ann. Surg. 142, 709–723 (1955).
Hermann, G., Konukiewitz, B., Schmitt, A., Perren, A. & Klöppel, G. Hormonally defined pancreatic and duodenal neuroendocrine tumors differ in their transcription factor signatures: expression of ISL1, PDX1, NGN3, and CDX2. Virchows Arch. 459, 147–154 (2011).
Verner, J. V. & Morrison, A. B. Islet cell tumor and a syndrome of refractory watery diarrhea and hypokalemia. Am. J. Med. 25, 374–380 (1958).
Kindmark, H. et al. Endocrine pancreatic tumors with glucagon hypersecretion: a retrospective study of 23 cases during 20 years. Med. Oncol. 24, 330–337 (2007).
Larsson, L. I. et al. Pancreatic somatostatinoma. Clinical features and physiological implications. Lancet 1, 666–668 (1977).
Garbrecht, N. et al. Somatostatin-producing neuroendocrine tumors of the duodenum and pancreas: incidence, types, biological behavior, association with inherited syndromes, and functional activity. Endocr. Relat. Cancer 15, 229–241 (2008).
Panzuto, F. et al. Metastatic and locally advanced pancreatic endocrine carcinomas: analysis of factors associated with disease progression. J. Clin. Oncol. 29, 2372–2377 (2011).
Alexakis, N. et al. Hereditary pancreatic endocrine tumours. Pancreatology 4, 417–433 (2004).
Jensen, R. T., Berna, M. J., Bingham, D. B. & Norton, J. A. Inherited pancreatic endocrine tumor syndromes: advances in molecular pathogenesis, diagnosis, management, and controversies. Cancer 113 (7 Suppl.), 1807–1843 (2008).
Anlauf, M. et al. Microadenomatosis of the endocrine pancreas in patients with and without the multiple endocrine neoplasia type 1 syndrome. Am. J. Surg. Pathol. 30, 560–574 (2006).
Périgny, M. et al. Pancreatic endocrine microadenomatosis in patients with von Hippel-Lindau disease: characterization by VHL/HIF pathway proteins expression. Am. J. Surg. Pathol. 33, 739–748 (2009).
Perren, A. et al. Multiple endocrine neoplasia type 1 (MEN1): loss of one MEN1 allele in tumors and monohormonal endocrine cell clusters but not in islet hyperplasia of the pancreas. J. Clin. Endocrinol. Metab. 92, 1118–1128 (2007).
Pipeleers-Marichal, M. et al. Gastrinomas in the duodenums of patients with multiple endocrine neoplasia type 1 and the Zollinger-Ellison syndrome. N. Engl. J. Med. 322, 723–727 (1990).
Triponez, F. et al. Epidemiology data on 108 MEN 1 patients from the GTE with isolated nonfunctioning tumors of the pancreas. Ann. Surg. 243, 265–672 (2006).
Carty, S. E. et al. The variable penetrance and spectrum of manifestations of multiple endocrine neoplasia type 1. Surgery 124, 1106–1113 (1998).
Dean, P. G. et al. Are patients with multiple endocrine neoplasia type I prone to premature death? World J. Surg. 24, 1437–1441 (2000).
Schmitt, A. M. et al. VHL inactivation is an important pathway for the development of malignant sporadic pancreatic endocrine tumors. Endocr. Relat. Cancer 16, 1219–1227 (2009).
Hammel, P. R. et al. Pancreatic involvement in von Hippel-Lindau disease. The Groupe Francophone d'Etude de la Maladie de von Hippel-Lindau. Gastroenterology 119, 1087–1095 (2000).
Lonser, R. R. et al. von Hippel-Lindau disease. Lancet 361, 2059–2067 (2003).
Mao, C., Shah, A., Hanson, D. J. & Howard, J. M. Von Recklinghausen's disease associated with duodenal somatostatinoma: contrast of duodenal versus pancreatic somatostatinomas. J. Surg. Oncol. 59, 67–73 (1995).
Fujisawa, T. et al. Malignant endocrine tumor of the pancreas associated with von Recklinghausen's disease. J. Gastroenterol. 37, 59–67 (2002).
Ilgren, E. B. & Westmoreland, D. Tuberous sclerosis: unusual associations in four cases. J. Clin. Pathol. 37, 272–278 (1984).
Chandrasekharappa, S. C. et al. Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276, 404–407 (1997).
Marx, S. et al. Multiple endocrine neoplasia type 1: clinical and genetic topics. Ann. Intern. Med. 129, 484–494 (1998).
Agarwal, S. K. et al. Menin interacts with the AP1 transcription factor JunD and represses JunD-activated transcription. Cell 96, 143–152 (1999).
Hughes, C. M. et al. Menin associates with a trithorax family histone methyltransferase complex and with the hoxc8 locus. Mol. Cell 13, 587–597 (2004).
Yokoyama, A. & Cleary, M. L. Menin critically links MLL proteins with LEDGF on cancer-associated target genes. Cancer Cell 14, 36–46 (2008).
Karnik, S. K. et al. Menin regulates pancreatic islet growth by promoting histone methylation and expression of genes encoding p27Kip1 and p18INK4c. Proc. Natl Acad. Sci. USA 102, 14659–14664 (2005).
Schnepp, R. W. et al. Mutation of tumor suppressor gene Men1 acutely enhances proliferation of pancreatic islet cells. Cancer Res. 66, 5707–5715 (2006).
Bertolino, P., Tong, W. M., Galendo, D., Wang, Z. Q. & Zhang, C. X. Heterozygous Men1 mutant mice develop a range of endocrine tumors mimicking multiple endocrine neoplasia type 1. Mol. Endocrinol. 17, 1880–1892 (2003).
Crabtree, J. S. et al. A mouse model of multiple endocrine neoplasia, type 1, develops multiple endocrine tumors. Proc. Natl Acad. Sci. USA 98, 1118–1123 (2001).
Crabtree, J. S. et al. Of mice and MEN1: Insulinomas in a conditional mouse knockout. Mol. Cell. Biol. 23, 6075–6085 (2003).
Toliat, M. R., Berger, W., Ropers, H. H., Neuhaus, P. & Wiedenmann, B. Mutations in the MEN I gene in sporadic neuroendocrine tumours of gastroenteropancreatic system. Lancet 350, 1223 (1997).
Moore, P. S. et al. Role of disease-causing genes in sporadic pancreatic endocrine tumors: MEN1 and VHL. Genes Chromosomes Cancer 32, 177–181 (2001).
Corbo, V. et al. MEN1 in pancreatic endocrine tumors: analysis of gene and protein status in 169 sporadic neoplasms reveals alterations in the vast majority of cases. Endocr. Relat. Cancer 17, 771–783 (2010).
Hay, N. The Akt-mTOR tango and its relevance to cancer. Cancer Cell 8, 179–183 (2005).
Speel, E. J. et al. Genetic differences in endocrine pancreatic tumor subtypes detected by comparative genomic hybridization. Am. J. Pathol. 155, 1787–1794 (1999).
Perren, A. et al. Mutation and expression analyses reveal differential subcellular compartmentalization of PTEN in endocrine pancreatic tumors compared to normal islet cells. Am. J. Pathol. 157, 1097–1103 (2000).
Missiaglia, E. et al. Pancreatic endocrine tumors: expression profiling evidences a role for AKT-mTOR pathway. J. Clin. Oncol. 28, 245–255 (2010).
Gumbs, A. A. et al. Review of the clinical, histological, and molecular aspects of pancreatic endocrine neoplasms. J. Surg. Oncol. 81, 45–53 (2002).
Oberg, K. Genetics and molecular pathology of neuroendocrine gastrointestinal and pancreatic tumors (gastroenteropancreatic neuroendocrine tumors). Curr. Opin. Endocrinol. Diabetes Obes. 16, 72–78 (2009).
Chung, D. C. et al. Overexpression of cyclin D1 occurs frequently in human pancreatic endocrine tumors. J. Clin. Endocrinol. Metab. 85, 4373–4378 (2000).
Maitra, A. et al. Global expression analysis of well-differentiated pancreatic endocrine neoplasms using oligonucleotide microarrays. Clin. Cancer Res. 9, 5988–5995 (2003).
Jiao, Y. et al. DAXX/ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science 331, 1199–1203 (2011).
Jones, S. et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 321, 1801–1806 (2008).
Samuels, Y. et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 304, 554 (2004).
Elsaesser, S. J. & Allis, C. D. HIRA and Daxx constitute two independent histone H3.3-containing predeposition complexes. Cold Spring Harb. Symp. Quant. Biol. 75, 27–34 (2010).
Goldberg, A. D. et al. Distinct factors control histone variant H3.3 localization at specific genomic regions. Cell 140, 678–691 (2010).
Lewis, P. W., Elsaesser, S. J., Noh, K. M., Stadler, S. C. & Allis, C. D. Daxx is an H3.3-specific histone chaperone and cooperates with ATRX in replication-independent chromatin assembly at telomeres. Proc. Natl Acad. Sci. USA 107, 14075–14080 (2010).
Meeker, A. K. et al. Telomere shortening is an early somatic DNA alteration in human prostate tumorigenesis. Cancer Res. 62, 6405–6409 (2002).
van Heek, N. T. et al. Telomere shortening is nearly universal in pancreatic intraepithelial neoplasia. Am. J. Pathol. 161, 1541–1547 (2002).
Heaphy, C. M. et al. Altered telomeres in tumors with ATRX and DAXX mutations. Science 333, 425 (2011).
Heaphy, C. M. et al. Prevalence of the alternative lengthening of telomeres telomere maintenance mechanism in human cancer subtypes. Am. J. Pathol. 179, 1608–1615 (2011).
Hakin-Smith, V. et al. Alternative lengthening of telomeres and survival in patients with glioblastoma multiforme. Lancet 361, 836–838 (2003).
Ulaner, G. A. et al. Absence of a telomere maintenance mechanism as a favorable prognostic factor in patients with osteosarcoma. Cancer Res. 63, 1759–1763 (2003).
Thompson, S. L., Bakhoum, S. F. & Compton, D. A. Mechanisms of chromosomal instability. Curr. Biol. 20, R285–R295 (2010).
Bardeesy, N. et al. Both p16(Ink4a) and the p19(Arf)-p53 pathway constrain progression of pancreatic adenocarcinoma in the mouse. Proc. Natl Acad. Sci. USA 103, 5947–5952 (2006).
Hingorani, S. R. et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 4, 437–450 (2003).
Hingorani, S. R. et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 7, 469–483 (2005).
Fendrich, V. & Bartsch, D. K. Surgical treatment of gastrointestinal neuroendocrine tumors. Langenbecks Arch. Surg. 396, 299–311 (2011).
Eriksson, B. New drugs in neuroendocrine tumors: rising of new therapeutic philosophies? Curr. Opin. Oncol. 22, 381–386 (2010).
Rinke, A. et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J. Clin. Oncol. 27, 4656–4663 (2009).
Faivre, S., Sablin, M. P., Dreyer, C. & Raymond, E. Novel anticancer agents in clinical trials for well-differentiated neuroendocrine tumors. Endocrinol. Metab. Clin. North Am. 39, 811–826 (2010).
Bushnell, D. L. Jr et al. 90Y-edotreotide for metastatic carcinoid refractory to octreotide. J. Clin. Oncol. 28, 1652–1659 (2010).
Kwekkeboom, D. J. et al. Treatment with the radiolabeled somatostatin analog [177 Lu-DOTA 0, Tyr3]octreotate: toxicity, efficacy, and survival. J. Clin. Oncol. 26, 2124–2130 (2008).
Rindi, G. & Wiedenmann, B. Neuroendocrine neoplasms of the gut and pancreas: new insights. Nat. Rev. Endocrinol. 8, 54–64 (2012).
Broder, L. E. & Carter, S. K. Pancreatic islet cell carcinoma. II. Results of therapy with streptozotocin in 52 patients. Ann. Intern. Med. 79, 108–118 (1973).
Chernicoff, D., Bukowski, R. M., Groppe, C. W. Jr & Hewlett, J. S. Combination chemotherapy for islet cell carcinoma and metastatic carcinoid tumors with 5-fluorouracil and streptozotocin. Cancer Treat. Rep. 63, 795–796 (1979).
Kulke, M. H. et al. Phase II study of temozolomide and thalidomide in patients with metastatic neuroendocrine tumors. J. Clin. Oncol. 24, 401–406 (2006).
Ekeblad, S. et al. Temozolomide as monotherapy is effective in treatment of advanced malignant neuroendocrine tumors. Clin. Cancer Res. 13, 2986–2991 (2007).
Strosberg, J. R. et al. First-line chemotherapy with capecitabine and temozolomide in patients with metastatic pancreatic endocrine carcinomas. Cancer 117, 268–275 (2011).
Eriksson, B. et al. Medical treatment and long-term survival in a prospective study of 84 patients with endocrine pancreatic tumors. Cancer 65, 1883–1890 (1990).
Alexandraki, K. I. & Kaltsas, G. Gastroenteropancreatic neuroendocrine tumors: new insights in the diagnosis and therapy. Endocrine 41, 40–52 (2012).
Liu, L. & Gerson, S. L. Targeted modulation of MGMT: clinical implications. Clin. Cancer Res. 12, 328–331 (2006).
Kulke, M. H. et al. O6-methylguanine DNA methyltransferase deficiency and response to temozolomide-based therapy in patients with neuroendocrine tumors. Clin. Cancer Res. 15, 338–345 (2009).
Wang, X. & Sun, S. Y. Enhancing mTOR-targeted cancer therapy. Expert Opin. Ther. Targets 13, 1193–1203 (2009).
Yao, J. C. et al. Daily oral everolimus activity in patients with metastatic pancreatic neuroendocrine tumors after failure of cytotoxic chemotherapy: a phase II trial. J. Clin. Oncol. 28, 69–76 (2010).
Yao, J. C. et al. Efficacy of RAD001 (everolimus) and octreotide LAR in advanced low- to intermediate-grade neuroendocrine tumors: results of a phase II study. J. Clin. Oncol. 26, 4311–4318 (2008).
Yao, J. C. et al. Everolimus for advanced pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 514–523 (2011).
Pommier, R. F. et al. Impact of prior chemotherapy on progression-free survival in patients (pts) with advanced pancreatic neuroendocrine tumors (pNET): Results from the RADIANT-3 trial [abstract]. J. Clin. Oncol. 29 (Suppl.), 4103 (2011).
Shah, M. H. et al. Everolimus in patients with advanced pancreatic neuroendocrine tumors (pNET): Impact of somatostatin analog use on progression-free survival in the RADIANT-3 trial [abstract]. J. Clin. Oncol. 29 (Suppl.), 4010 (2011).
Strosberg, J. R., Lincy, J., Winkler, R. E. & Wolin, E. M. Everolimus in patients with advanced pancreatic neuroendocrine tumors (pNET): Updated results of a randomized, double-blind, placebo-controlled, multicenter, phase III trial (RADIANT-3) [abstract]. J. Clin. Oncol. 29 (Suppl.), 4009 (2011).
ClinicalTrials.gov [online], (2011).
Chiu, C. W., Nozawa, H. & Hanahan, D. Survival benefit with proapoptotic molecular and pathologic responses from dual targeting of mammalian target of rapamycin and epidermal growth factor receptor in a preclinical model of pancreatic neuroendocrine carcinogenesis. J. Clin. Oncol. 28, 4425–4433 (2010).
O'Reilly, K. E. et al. mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res. 66, 1500–1508 (2006).
Joyce, J. A. et al. Stage-specific vascular markers revealed by phage display in a mouse model of pancreatic islet tumorigenesis. Cancer Cell 4, 393–403 (2003).
Lindahl, P., Johansson, B. R., Levéen, P. & Betsholtz, C. Pericyte loss and microaneurysm formation in PDGF-B-deficient mice. Science 277, 242–245 (1997).
Fjällskog, M. L., Hessman, O., Eriksson, B. & Janson, E. T. Upregulated expression of PDGF receptor beta in endocrine pancreatic tumors and metastases compared to normal endocrine pancreas. Acta Oncol. 46, 741–746 (2007).
Hansel, D. E. et al. Liver metastases arising from well-differentiated pancreatic endocrine neoplasms demonstrate increased VEGF-C expression. Mod. Pathol. 16, 652–659 (2003).
Hanahan, D. Heritable formation of pancreatic beta-cell tumours in transgenic mice expressing recombinant insulin/simian virus 40 oncogenes. Nature 315, 115–122 (1985).
Bergers, G., Song, S., Meyer-Morse, N., Bergsland, E. & Hanahan, D. Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J. Clin. Invest. 111, 1287–1295 (2003).
Pàez-Ribes, M. et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15, 220–231 (2009).
Pietras, K. & Hanahan, D. A multitargeted, metronomic, and maximum-tolerated dose “chemo-switch” regimen is antiangiogenic, producing objective responses and survival benefit in a mouse model of cancer. J. Clin. Oncol. 23, 939–952 (2005).
Faivre, S. et al. Safety, pharmacokinetic, and antitumor activity of SU11248, a novel oral multitarget tyrosine kinase inhibitor, in patients with cancer. J. Clin. Oncol. 24, 25–35 (2006).
Kulke, M. H. et al. Activity of sunitinib in patients with advanced neuroendocrine tumors. J. Clin. Oncol. 26, 3403–3410 (2008).
Raymond, E. et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 501–513 (2011).
Raymond, E. et al. Updated overall survival (OS) and progression-free survival (PFS) by blinded independent central review (BICR) of sunitinib (SU) versus placebo (PBO) for patients (Pts) with advanced unresectable pancreatic neuroendocrine tumors (NET). J. Clin. Oncol. 29 (Suppl.), 4008 (2011).
Acknowledgements
The authors would like to acknowledge the support of the Sol Goldman Pancreatic Cancer Research Center and the Caring for Carcinoid Foundation. The authors are grateful to Drs Alan Meeker and Christopher Heaphy (Johns Hopkins University) for assistance with Figure 3 of this Review and helpful suggestions.
Author information
Authors and Affiliations
Contributions
R. F. de Wilde and A. Maitra researched data for the article. All authors contributed equally to discussion of the content, writing the manuscript and reviewing/editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
de Wilde, R., Edil, B., Hruban, R. et al. Well-differentiated pancreatic neuroendocrine tumors: from genetics to therapy. Nat Rev Gastroenterol Hepatol 9, 199–208 (2012). https://doi.org/10.1038/nrgastro.2012.9
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2012.9
This article is cited by
-
Metabolic intervention by low carbohydrate diet suppresses the onset and progression of neuroendocrine tumors
Cell Death & Disease (2023)
-
Cdk5 drives formation of heterogeneous pancreatic neuroendocrine tumors
Oncogenesis (2021)
-
Inherited Endocrine Neoplasia— A Comprehensive Review from Gland to Gene
Current Genetic Medicine Reports (2019)
-
Robotic enucleations of pancreatic benign or low-grade malignant tumors: preliminary results and comparison with robotic demolitive resections
Surgical Endoscopy (2019)