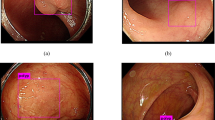
Abstract
Several studies have raised warnings about the limited effectiveness of colonoscopy for the prevention of colorectal cancer (CRC), especially of the proximal colon. Two major categories of factors might be responsible for the development of interval cancers, namely technical, endoscopist-dependent factors and biological characteristics of the cancer that lead to more rapid tumour progression. Recognition of endoscopist-dependent factors is critical, as these factors are probably amenable to correction through improved awareness and education of endoscopists, using quality metrics (such as adenoma detection rates and cecal intubation rates) for objective evaluation and feedback. In this article, the current literature regarding the incidence of, and potential explanations for, interval CRCs is outlined. Although there is probably an interaction between technical and biology-related factors—and an attempt to dissect the biology from the technology might be fraught with difficulties—a structured analysis of individual cases of interval cancer might help in the continuous monitoring of the quality of colonoscopy, and ultimately might reduce the number of interval CRCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lakoff, J., Paszat, L. F., Saskin, R. & Rabeneck, L. Risk of developing proximal versus distal colorectal cancer after a negative colonoscopy: a population-based study. Clin. Gastroenterol. Hepatol. 6, 1117–1121 (2008).
Brenner, H., Chang-Claude, J., Seiler, C. M., Rickert, A. & Hoffmeister, M. Protection from colorectal cancer after colonoscopy: a population-based, case-control study. Ann. Intern. Med. 154, 22–30 (2011).
Singh, H., Nugent, Z., Demers, A. A. & Bernstein, C. N. Rate and predictors of early/missed colorectal cancers after colonoscopy in manitoba: a population-based study. Am. J. Gastroenterol. 105, 2588–2596 (2010).
Baxter, N. N. et al. Association of colonoscopy and death from colorectal cancer. Ann. Intern. Med. 150, 1–8 (2009).
Rex, D. K. Preventing colorectal cancer and cancer mortality with colonoscopy: what we know and what we don't know. Endoscopy 42, 320–323 (2010).
Rex, D. K., Bond, J. H. & Feld, A. D. Medical-legal risks of incident cancers after clearing colonoscopy. Am. J. Gastroenterol. 96, 952–957 (2001).
Kaminski, M. F. et al. Quality indicators for colonoscopy and the risk of interval cancer. N. Engl. J. Med. 362, 1795–1803 (2010).
le Clercq, C. et al. Interval colorectal cancers frequently have subtle macroscopic appearance: a 10 year-experience in an academic center. Gastroenterology 140, S112–S113 (2011).
Baxter, N. N. et al. Analysis of administrative data finds endoscopist quality measures associated with postcolonoscopy colorectal cancer. Gastroenterology 140, 65–72 (2011).
Imperiale, T. F. et al. Five-year risk of colorectal neoplasia after negative screening colonoscopy. N. Engl. J. Med. 359, 1218–1224 (2008).
Bressler, B. et al. Rates of new or missed colorectal cancers after colonoscopy and their risk factors: a population-based analysis. Gastroenterology 132, 96–102 (2007).
Rabeneck, L., Paszat, L. F. & Saskin, R. Endoscopist specialty is associated with incident colorectal cancer after a negative colonoscopy. Clin. Gastroenterol. Hepatol. 8, 275–279 (2010).
Ferlay, J., Parkin, D. M. & Steliarova-Foucher, E. Estimates of cancer incidence and mortality in Europe in 2008. Eur. J. Cancer 46, 765–781 (2010).
Gorski, T. F., Rosen, L., Riether, R., Stasik, J. & Khubchandani, I. Colorectal cancer after surveillance colonoscopy: false-negative examination or fast growth? Dis. Colon Rectum 42, 877–880 (1999).
Haseman, J. H., Lemmel, G. T., Rahmani, E. Y. & Rex, D. K. Failure of colonoscopy to detect colorectal cancer: evaluation of 47 cases in 20 hospitals. Gastrointest. Endosc. 45, 451–455 (1997).
Chen, S. C. & Rex, D. K. Endoscopist can be more powerful than age and male gender in predicting adenoma detection at colonoscopy. Am. J. Gastroenterol. 102, 856–861 (2007).
Robertson, D. J. et al. Colorectal cancer in patients under close colonoscopic surveillance. Gastroenterology 129, 34–41 (2005).
Farrar, W. D., Sawhney, M. S., Nelson, D. B., Lederle, F. A. & Bond, J. H. Colorectal cancers found after a complete colonoscopy. Clin. Gastroenterol. Hepatol. 4, 1259–1264 (2006).
Kahi, C. J., Hewett, D. G. & Rex, D. K. Relationship of non-polypoid colorectal neoplasms to quality of colonoscopy. Gastrointest. Endosc. Clin. N. Am. 20, 407–415 (2010).
Martinez, M. E. et al. A pooled analysis of advanced colorectal neoplasia diagnoses after colonoscopic polypectomy. Gastroenterology 136, 832–841 (2009).
Morini, S. et al. Diverticular disease as a risk factor for sigmoid colon adenomas. Dig. Liver Dis. 34, 635–639 (2002).
Leung, K. et al. Ongoing colorectal cancer risk despite surveillance colonoscopy: the Polyp Prevention Trial Continued Follow-up Study. Gastrointest. Endosc. 71, 111–117 (2010).
Loeve, F., van Ballegooijen, M., Snel, P. & Habbema, J. D. Colorectal cancer risk after colonoscopic polypectomy: a population-based study and literature search. Eur. J. Cancer 41, 416–422 (2005).
Imperiale, T. F., Glowinski, E. A., Juliar, B. E., Azzouz, F. & Ransohoff, D. F. Variation in polyp detection rates at screening colonoscopy. Gastrointest. Endosc. 69, 1288–1295 (2009).
Arain, M. A. et al. CIMP status of interval colon cancers: another piece to the puzzle. Am. J. Gastroenterol. 105, 1189–1195 (2009).
Hewett, D. G., Kahi, C. J. & Rex, D. K. Efficacy and effectiveness of colonoscopy: how do we bridge the gap? Gastrointest. Endosc. Clin. N. Am. 20, 673–684 (2010).
Barclay, R. L., Vicari, J. J., Doughty, A. S., Johanson, J. F. & Greenlaw, R. L. Colonoscopic withdrawal times and adenoma detection during screening colonoscopy. N. Engl. J. Med. 355, 2533–2541 (2006).
Lee, R. H. et al. Quality of colonoscopy withdrawal technique and variability in adenoma detection rates (with videos). Gastrointest. Endosc. 74, 128–134 (2011).
Rex, D. K. Looking over your shoulder during colonoscopy: potential roles for videorecording colonoscopy withdrawals. Gastrointest. Endosc. 75, 134–137 (2012).
Pabby, A. et al. Analysis of colorectal cancer occurrence during surveillance colonoscopy in the dietary Polyp Prevention Trial. Gastrointest. Endosc. 61, 385–391 (2005).
Atkin, W. S., Morson, B. C. & Cuzick, J. Long-term risk of colorectal cancer after excision of rectosigmoid adenomas. N. Engl. J. Med. 326, 658–662 (1992).
Sawhney, M. S. et al. Microsatellite instability in interval colon cancers. Gastroenterology 131, 1700–1705 (2006).
Shaukat, A., Arain, M., Thaygarajan, B., Bond, J. H. & Sawhney, M. Is BRAF mutation associated with interval colorectal cancers? Dig. Dis. Sci. 55, 2352–2356 (2010).
Gervaz, P., Bucher, P. & Morel, P. Two colons-two cancers: paradigm shift and clinical implications. J. Surg. Oncol. 88, 261–266 (2004).
Iacopetta, B. Are there two sides to colorectal cancer? Int. J. Cancer 101, 403–408 (2002).
Li, F. Y. & Lai, M. D. Colorectal cancer, one entity or three. J. Zhejiang Univ. Sci. B 10, 219–229 (2009).
Soetikno, R. M. et al. Prevalence of nonpolypoid (flat and depressed) colorectal neoplasms in asymptomatic and symptomatic adults. JAMA 299, 1027–1035 (2008).
Soetikno, R. M., Gotoda, T., Nakanishi, Y. & Soehendra, N. Endoscopic submucosal resection. Gastrointest. Endosc. 57, 5675–5679 (2003).
Konishi, K. et al. BRAF mutations and phosphorylation status of mitogen-activated protein kinases in the development of flat and depressed-type colorectal neoplasias. Br. J. Cancer 94, 311–317 (2006).
Matsuda, T., Saito, Y., Hotta, K., Sano, Y. & Fujii, T. Prevalence and clinicopathological features of nonpolypoid colorectal neoplasms: should we pay more attention to identifying flat and depressed lesions? Dig. Endosc. 22 (Suppl. 1), S57–S62 (2010).
Rondagh, E. J. et al. Endoscopic appearance of proximal colorectal neoplasms and potential implications for colonoscopic cancer prevention. Gastrointest. Endosc. 75, 1218–1225 (2012).
Snover, D. C. Update on the serrated pathway to colorectal carcinoma. Hum. Pathol. 42, 1–10 (2011).
Rondagh, E. J. et al. Endoscopic red flags for the detection of high-risk serrated polyps: an observational study. Endoscopy 43, 1052–1058 (2011).
Sweetser, S., Smyrk, T. C. & Sugumar, A. Serrated polyps: critical precursors to colorectal cancer. Expert Rev. Gastroenterol. Hepatol. 5, 627–635 (2011).
Kahi, C. J., Hewett, D. G., Norton, D. L., Eckert, G. J. & Rex, D. K. Prevalence and variable detection of proximal colon serrated polyps during screening colonoscopy. Clin. Gastroenterol. Hepatol. 9, 42–46 (2011).
Hetzel, J. T. et al. Variation in the detection of serrated polyps in an average risk colorectal cancer screening cohort. Am. J. Gastroenterol. 105, 2656–2664 (2010).
Participants in the Paris Workshop. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest. Endosc. 58, S3–S43 (2003).
Lasisi, F. & Rex, D. K. Improving protection against proximal colon cancer by colonoscopy. Expert Rev. Gastroenterol. Hepatol. 5, 745–754 (2011).
Jass, J. R. et al. Neoplastic progression occurs through mutator pathways in hyperplastic polyposis of the colorectum. Gut 47, 43–49 (2000).
East, J. E., Saunders, B. P. & Jass, J. R. Sporadic and syndromic hyperplastic polyps and serrated adenomas of the colon: classification, molecular genetics, natural history, and clinical management. Gastroenterol. Clin. North Am. 37, 25–46 (2008).
Sillars-Hardebol, A. H., Carvalho, B., van Engeland, M., Fijneman, R. J. & Meijer, G. A. The adenoma hunt in colorectal cancer screening: defining the target. J. Pathol. 226, 1–6 (2012).
Mulder, S. A. et al. Exposure to colorectal examinations before a colorectal cancer diagnosis: a case-control study. Eur. J. Gastroenterol. Hepatol. 22, 437–443 (2010).
Kahi, C. J., Imperiale, T. F., Juliar, B. E. & Rex, D. K. Effect of screening colonoscopy on colorectal cancer incidence and mortality. Clin. Gastroenterol. Hepatol 7, 770–775 (2009).
Acknowledgements
G. A. Meijer receives research support from the Center for Translational Molecular Medicine (www.ctmm.nl).
Author information
Authors and Affiliations
Contributions
S. Sanduleanu contributed to all aspects of this manuscript. A. Masclee and G. A. Meijer contributed equally to the discussion of content and reviewing/editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
S. Sanduleanu declares that she is a consultant for, on the speakers bureau and receives grant/research support from Pentax Nederland B.V. A. Masclee receives grant/research support from Pentax Ned-erland B.V. G. Meijer receive grant/research support and is a con-sultant for the Center for Translational Molecular Medicine.
Rights and permissions
About this article
Cite this article
Sanduleanu, S., Masclee, A. & Meijer, G. Interval cancers after colonoscopy—insights and recommendations. Nat Rev Gastroenterol Hepatol 9, 550–554 (2012). https://doi.org/10.1038/nrgastro.2012.136
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2012.136
This article is cited by
-
Periodontitis is associated with an increased risk for proximal colorectal neoplasms
Scientific Reports (2019)
-
Clinicopathologic and genetic characteristics of interval colorectal carcinomas favor origin from missed or incompletely excised precursors
Modern Pathology (2019)
-
Detection of colonic neoplasia in vivo using near-infrared-labeled peptide targeting cMet
Scientific Reports (2019)
-
Lead Time Bias May Contribute to the Shorter Life Expectancy in Post-colonoscopy Colorectal Cancer
Digestive Diseases and Sciences (2019)
-
Comparison of Small Versus Large Volume Split Dose Preparation for Colonoscopy: A Study of Colonoscopy Outcomes
Digestive Diseases and Sciences (2018)