Abstract
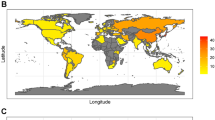
Helicobacter pylori plays an essential role in the development of various gastroduodenal diseases; however, only a small proportion of people infected with H. pylori develop these diseases. Some populations that have a high prevalence of H. pylori infection also have a high incidence of gastric cancer (for example, in East Asia), whereas others do not (for example, in Africa and South Asia). Even within East Asia, the incidence of gastric cancer varies (decreasing in the south). H. pylori is a highly heterogeneous bacterium and its virulence varies geographically. Geographic differences in the incidence of gastric cancer can be explained, at least in part, by the presence of different types of H. pylori virulence factor, especially CagA, VacA and OipA. However, it is still unclear why the pathogenicity of H. pylori increased as it migrated from Africa to East Asia during the course of evolution. H. pylori infection is also thought to be involved in the development of duodenal ulcer, which is at the opposite end of the disease spectrum to gastric cancer. This discrepancy can be explained in part by the presence of H. pylori virulence factor DupA. Despite advances in our understanding of the development of H. pylori-related diseases, further work is required to clarify the roles of H. pylori virulence factors.
Key Points
-
Experimental evidence suggests that East-Asian-type CagA is more virulent than Western-type CagA, but the concept that this explains geographic differences in the incidence of gastric cancer is still controversial
-
The number of CagA Glu-Pro-Ile-Tyr-Ala (EPIYA)-C segments (with multiple repeats increasing the virulence) may explain, to some extent, geographic differences in the incidence of gastric cancer in Western countries
-
The genotype of the vacA middle region (m1 versus m2; m1 being more virulent) may, in part, explain geographic differences in the incidence of gastric cancer in East Asian countries
-
CagA, OipA and VacA are thought to interact synergistically with each other to induce serious disease
-
The presence of functional DupA with an intact, full size dupA cluster (tfs3a) is associated with duodenal ulcers
-
Bacterial factors, environmental factors and host factors are thought to form complex interactions with each other in the actual development of disease
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
International Agency for Research on Cancer. The Globocan Project [online], (2010).
Jemal, A., Siegel, R., Xu, J. & Ward, E. Cancer Statistics, 2010. CA Cancer J. Clin. doi:10.3322/caac.20073.
Malaty, H. M. Epidemiology of Helicobacter pylori infection. Best Pract. Res. Clin. Gastroenterol. 21, 205–214 (2007).
El-Omar, E. M. Role of host genes in sporadic gastric cancer. Best Pract. Res. Clin. Gastroenterol. 20, 675–686 (2006).
Graham, D. Y. Helicobacter pylori infection in the pathogenesis of duodenal ulcer and gastric cancer: a model. Gastroenterology 113, 1983–1991 (1997).
Graham, D. Y., Lu, H. & Yamaoka, Y. African, Asian or Indian enigma, the East Asian Helicobacter pylori: facts or medical myths. J. Dig. Dis. 10, 77–84 (2009).
Falush, D. et al. Traces of human migrations in Helicobacter pylori populations. Science 299, 1582–1585 (2003).
Linz, B. et al. An African origin for the intimate association between humans and Helicobacter pylori. Nature 445, 915–918 (2007).
Moodley, Y. et al. The peopling of the Pacific from a bacterial perspective. Science 323, 527–530 (2009).
Yamaoka, Y. Helicobacter pylori typing as a tool for tracking human migration. Clin. Microbiol. Infect. 15, 829–834 (2009).
Franco, A. T. et al. Activation of beta-catenin by carcinogenic Helicobacter pylori. Proc. Natl Acad. Sci. USA 102, 10646–10651 (2005).
Franco, A. T. et al. Regulation of gastric carcinogenesis by Helicobacter pylori virulence factors. Cancer Res. 68, 379–387 (2008).
Ohnishi, N. et al. Transgenic expression of Helicobacter pylori CagA induces gastrointestinal and hematopoietic neoplasms in mouse. Proc. Natl Acad. Sci. USA 105, 1003–1008 (2008).
van Doorn, L. J. et al. Clinical relevance of the cagA, vacA, and iceA status of Helicobacter pylori. Gastroenterology 115, 58–66 (1998).
Yamaoka, Y. et al. Importance of Helicobacter pylori oipA in clinical presentation, gastric inflammation, and mucosal interleukin 8 production. Gastroenterology 123, 414–424 (2002).
Yamaoka, Y. et al. Relationship between Helicobacter pylori iceA, cagA, and vacA status and clinical outcome: studies in four different countries. J. Clin. Microbiol. 37, 2274–2279 (1999).
Yamaoka, Y., Kodama, T., Kashima, K., Graham, D. Y. & Sepulveda, A. R. Variants of the 3′ region of the cagA gene in Helicobacter pylori isolates from patients with different H. pylori-associated diseases. J. Clin. Microbiol. 36, 2258–2263 (1998).
Yamaoka, Y. et al. Relationship between the cagA 3′ repeat region of Helicobacter pylori, gastric histology, and susceptibility to low pH. Gastroenterology 117, 342–349 (1999).
Yamaoka, Y. et al. Molecular epidemiology of Helicobacter pylori: separation of H. pylori from East Asian and non-Asian countries. Epidemiol. Infect. 124, 91–96 (2000).
Yamaoka, Y. et al. Helicobacter pylori in North and South America before Columbus. FEBS Lett. 517, 180–184 (2002).
Hatakeyama, M. Oncogenic mechanisms of the Helicobacter pylori CagA protein. Nat. Rev. Cancer 4, 688–694 (2004).
Saribasak, H., Salih, B. A., Yamaoka, Y. & Sander, E. Analysis of Helicobacter pylori genotypes and correlation with clinical outcome in Turkey. J. Clin. Microbiol. 42, 1648–1651 (2004).
Shokrzadeh, L. et al. Analysis of 3′-end variable region of the cagA gene in Helicobacter pylori isolated from Iranian population. J. Gastroenterol. Hepatol. 25, 172–177 (2010).
Uchida, T. et al. Analysis of virulence factors of Helicobacter pylori isolated from a Vietnamese population. BMC Microbiol. 9, 175 (2009).
Vilaichone, R. K. et al. Molecular epidemiology and outcome of Helicobacter pylori infection in Thailand: a cultural cross roads. Helicobacter 9, 453–459 (2004).
Xia, Y., Yamaoka, Y., Zhu, Q., Matha, I. & Gao, X. A comprehensive sequence and disease correlation analyses for the C-terminal region of CagA protein of Helicobacter pylori. PLoS ONE 4, e7736 (2009).
Truong, B. X. et al. Diverse characteristics of the CagA gene of Helicobacter pylori strains collected from patients from southern Vietnam with gastric cancer and peptic ulcer. J. Clin. Microbiol. 47, 4021–4028 (2009).
Argent, R. H. et al. Determinants and consequences of different levels of CagA phosphorylation for clinical isolates of Helicobacter pylori. Gastroenterology 127, 514–523 (2004).
Azuma, T. et al. Correlation between variation of the 3′ region of the cagA gene in Helicobacter pylori and disease outcome in Japan. J. Infect. Dis. 186, 1621–1630 (2002).
Poppe, M., Feller, S. M., Romer, G. & Wessler, S. Phosphorylation of Helicobacter pylori CagA by c-Abl leads to cell motility. Oncogene 26, 3462–3472 (2007).
Selbach, M., Moese, S., Hauck, C. R., Meyer, T. F. & Backert, S. Src is the kinase of the Helicobacter pylori CagA protein in vitro and in vivo. J. Biol. Chem. 277, 6775–6778 (2002).
Stein, M. et al. c-Src/Lyn kinases activate Helicobacter pylori CagA through tyrosine phosphorylation of the EPIYA motifs. Mol. Microbiol. 43, 971–980 (2002).
Tammer, I., Brandt, S., Hartig, R., Konig, W. & Backert, S. Activation of Abl by Helicobacter pylori: a novel kinase for CagA and crucial mediator of host cell scattering. Gastroenterology 132, 1309–1319 (2007).
Tsutsumi, R., Higashi, H., Higuchi, M., Okada, M. & Hatakeyama, M. Attenuation of Helicobacter pylori CagA x SHP-2 signaling by interaction between CagA and C-terminal Src kinase. J. Biol. Chem. 278, 3664–3670 (2003).
Backert, S., Tegtmeyer, N. & Selbach, M. The versatility of Helicobacter pylori CagA effector protein functions: The master key hypothesis. Helicobacter 15, 163–176 (2010).
Selbach, M. et al. Host cell interactome of tyrosine-phosphorylated bacterial proteins. Cell Host Microbe 5, 397–403 (2009).
Hirata, Y. et al. Functional variability of cagA gene in Japanese isolates of Helicobacter pylori. Gene 343, 165–172 (2004).
Higashi, H. et al. SHP-2 tyrosine phosphatase as an intracellular target of Helicobacter pylori CagA protein. Science 295, 683–686 (2002).
Miura, M., Ohnishi, N., Tanaka, S., Yanagiya, K. & Hatakeyama, M. Differential oncogenic potential of geographically distinct Helicobacter pylori CagA isoforms in mice. Int. J. Cancer 125, 2497–2504 (2009).
Churin, Y. et al. Helicobacter pylori CagA protein targets the c-Met receptor and enhances the motogenic response. J. Cell Biol. 161, 249–255 (2003).
Kwok, T. et al. Helicobacter exploits integrin for type IV secretion and kinase activation. Nature 449, 862–866 (2007).
Ren, S., Higashi, H., Lu, H., Azuma, T. & Hatakeyama, M. Structural basis and functional consequence of Helicobacter pylori CagA multimerization in cells. J. Biol. Chem. 281, 32344–32352 (2006).
Saadat, I. et al. Helicobacter pylori CagA targets PAR1/MARK kinase to disrupt epithelial cell polarity. Nature 447, 330–333 (2007).
Umeda, M. et al. Helicobacter pylori CagA causes mitotic impairment and induces chromosomal instability. J. Biol. Chem. 284, 22166–22172 (2009).
Nesic, D. et al. Helicobacter pylori CagA inhibits PAR1-MARK family kinases by mimicking host substrates. Nat. Struct. Mol. Biol. 17, 130–132 (2010).
Suzuki, M. et al. Helicobacter pylori CagA phosphorylation-independent function in epithelial proliferation and inflammation. Cell Host Microbe 5, 23–34 (2009).
Yamaoka, Y., Kita, M., Kodama, T., Sawai, N. & Imanishi, J. Helicobacter pylori cagA gene and expression of cytokine messenger RNA in gastric mucosa. Gastroenterology 110, 1744–1752 (1996).
Yamaoka, Y. et al. Chemokines in the gastric mucosa in Helicobacter pylori infection. Gut 42, 609–617 (1998).
Yamaoka, Y. et al. Relation between clinical presentation, Helicobacter pylori density, interleukin 1β and 8 production, and cagA status. Gut 45, 804–811 (1999).
Yamaoka, Y. et al. Relation between cytokines and Helicobacter pylori in gastric cancer. Helicobacter 6, 116–124 (2001).
Censini, S. et al. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc. Natl Acad. Sci. USA 93, 14648–14653 (1996).
Fischer, W. et al. Systematic mutagenesis of the Helicobacter pylori cag pathogenicity island: essential genes for CagA translocation in host cells and induction of interleukin-8. Mol. Microbiol. 42, 1337–1348 (2001).
Al-Ghoul, L. et al. Analysis of the type IV secretion system-dependent cell motility of Helicobacter pylori-infected epithelial cells. Biochem. Biophys. Res. Commun. 322, 860–866 (2004).
Brandt, S., Kwok, T., Hartig, R., Konig, W. & Backert, S. NF-kappaB activation and potentiation of proinflammatory responses by the Helicobacter pylori CagA protein. Proc. Natl Acad. Sci. USA 102, 9300–9305 (2005).
Kim, S. Y., Lee, Y. C., Kim, H. K. & Blaser, M. J. Helicobacter pylori CagA transfection of gastric epithelial cells induces interleukin-8. Cell Microbiol. 8, 97–106 (2006).
Kersulyte, D. et al. Differences in genotypes of Helicobacter pylori from different human populations. J. Bacteriol. 182, 3210–3218 (2000).
Holcombe, C. Helicobacter pylori: the African enigma. Gut 33, 429–431 (1992).
Atherton, J. C. The pathogenesis of Helicobacter pylori-induced gastro-duodenal diseases. Annu. Rev. Pathol. Mech. Dis. 1, 63–96 (2006).
Cover, T. L. & Blanke, S. R. Helicobacter pylori VacA, a paradigm for toxin multifunctionality. Nat. Rev. Microbiol. 3, 320–332 (2005).
Kusters, J. G., van Vliet, A. H. & Kuipers, E. J. Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev. 19, 449–490 (2006).
Boncristiano, M. et al. The Helicobacter pylori vacuolating toxin inhibits T cell activation by two independent mechanisms. J. Exp. Med. 198, 1887–1897 (2003).
Gebert, B., Fischer, W., Weiss, E., Hoffmann, R. & Haas, R. Helicobacter pylori vacuolating cytotoxin inhibits T lymphocyte activation. Science 301, 1099–1102 (2003).
Sundrud, M. S., Torres, V. J., Unutmaz, D. & Cover, T. L. Inhibition of primary human T cell proliferation by Helicobacter pylori vacuolating toxin (VacA) is independent of VacA effects on IL-2 secretion. Proc. Natl Acad. Sci. USA 101, 7727–7732 (2004).
Mimuro, H. et al. Helicobacter pylori dampens gut epithelial self-renewal by inhibiting apoptosis, a bacterial strategy to enhance colonization of the stomach. Cell Host Microbe 2, 250–263 (2007).
Oldani, A. et al. Helicobacter pylori counteracts the apoptotic action of its VacA toxin by injecting the CagA protein into gastric epithelial cells. PLoS Pathog. 5, e1000603 (2009).
Tegtmeyer, N. et al. Importance of EGF receptor, HER2/Neu and Erk1/2 kinase signaling for host cell elongation and scattering induced by the Helicobacter pylori CagA protein: antagonistic effects of the vacuolating cytotoxin VacA. Cell. Microbiol. 11, 488–505 (2009).
Yokoyama, K. et al. Functional antagonism between Helicobacter pylori CagA and vacuolating toxin VacA in control of the NFAT signaling pathway in gastric epithelial cells. Proc. Natl Acad. Sci. USA 102, 9661–9666 (2005).
Cover, T. L. & Blaser, M. J. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J. Biol. Chem. 267, 10570–10575 (1992).
Leunk, R. D. Production of a cytotoxin by Helicobacter pylori. Rev. Infect. Dis. 13 (Suppl. 8), S686–S689 (1991).
Atherton, J. C. et al. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J. Biol. Chem. 270, 17771–17777 (1995).
Letley, D. P., Lastovica, A., Louw, J. A., Hawkey, C. J. & Atherton, J. C. Allelic diversity of the Helicobacter pylori vacuolating cytotoxin gene in South Africa: rarity of the vacA s1a genotype and natural occurrence of an s2/m1 allele. J. Clin. Microbiol. 37, 1203–1205 (1999).
Sugimoto, M., Zali, M. R. & Yamaoka, Y. The association of vacA genotypes and Helicobacter pylori-related gastroduodenal diseases in the Middle East. Eur. J. Clin. Microbiol. Infect. Dis. 28, 1227–1236 (2009).
Sugimoto, M. & Yamaoka, Y. The association of vacA genotype and Helicobacter pylori-related disease in Latin American and African populations. Clin. Microbiol. Infect. 15, 835–842 (2009).
Rhead, J. L. et al. A new Helicobacter pylori vacuolating cytotoxin determinant, the intermediate region, is associated with gastric cancer. Gastroenterology 133, 926–936 (2007).
Hussein, N. R. et al. Differences in virulence markers between Helicobacter pylori strains from Iraq and those from Iran: potential importance of regional differences in H. pylori-associated disease. J. Clin. Microbiol. 46, 1774–1779 (2008).
Basso, D. et al. Clinical relevance of Helicobacter pylori cagA and vacA gene polymorphisms. Gastroenterology 135, 91–99 (2008).
Ogiwara, H., Graham, D. Y. & Yamaoka, Y. vacA i-region subtyping. Gastroenterology 134, 1267 (2008).
Ogiwara, H. et al. Role of deletion located between the intermediate and middle regions of the Helicobacter pylori vacA gene in cases of gastroduodenal diseases. J. Clin. Microbiol. 47, 3493–3500 (2009).
Figueiredo, C., Machado, J. C. & Yamaoka, Y. Pathogenesis of Helicobacter pylori Infection. Helicobacter 10 (Suppl. 1), 14–20 (2005).
Fujimoto, S. et al. Helicobacter pylori BabA expression, gastric mucosal injury, and clinical outcome. Clin. Gastroenterol. Hepatol. 5, 49–58 (2007).
Lu, H., Yamaoka, Y. & Graham, D. Y. Helicobacter pylori virulence factors: facts and fantasies. Curr. Opin. Gastroenterol. 21, 653–659 (2005).
Yamaoka, Y. et al. Helicobacter pylori outer membrane proteins and gastroduodenal disease. Gut 55, 775–781 (2006).
Yamaoka, Y. & Alm, R. in Helicobacter pylori: Molecular Genetics and Cellular Biology (ed. Yamaoka, Y.) 37–60 (Caister Academic Press, 2008).
Yamaoka, Y. Increasing evidence of the roles of Helicobacter pylori SabA in the pathogenesis of gastroduodenal disease. J. Infect. Dev. Ctries 2, 174–181 (2008).
Yamaoka, Y. Roles of H. pylori BabA in gastroduodenal pathogenesis. World J. Gastroenterol. 14, 4265–4272 (2008).
Yamaoka, Y., Kwon, D. H. & Graham, D. Y. A M(r) 34,000 proinflammatory outer membrane protein (oipA) of Helicobacter pylori. Proc. Natl Acad. Sci. USA 97, 7533–7538 (2000).
Odenbreit, S., Kavermann, H., Puls, J. & Haas, R. CagA tyrosine phosphorylation and interleukin-8 induction by Helicobacter pylori are independent from alpAB, HopZ and bab group outer membrane proteins. Int. J. Med. Microbiol. 292, 257–266 (2002).
Ando, T. et al. Polymorphisms of Helicobacter pylori HP0638 reflect geographic origin and correlate with cagA status. J. Clin. Microbiol. 40, 239–246 (2002).
Dossumbekova, A. et al. Helicobacter pylori HopH (OipA) and bacterial pathogenicity: genetic and functional genomic analysis of hopH gene polymorphisms. J. Infect. Dis. 194, 1346–1355 (2006).
Akanuma, M. et al. The evaluation of putative virulence factors of Helicobacter pylori for gastroduodenal disease by use of a short-term Mongolian gerbil infection model. J. Infect. Dis. 185, 341–347 (2002).
Yamaoka, Y. Helicobacter pylori outer membrane proteins and gastric inflammation: author's reply. Gut 55, 1361 (2006).
Yamaoka, Y. et al. Role of interferon-stimulated responsive element-like element in interleukin-8 promoter in Helicobacter pylori infection. Gastroenterology 126, 1030–1043 (2004).
Choi, I. J., Fujimoto, S., Yamauchi, K., Graham, D. Y. & Yamaoka, Y. Helicobacter pylori environmental interactions: effect of acidic conditions on H. pylori-induced gastric mucosal interleukin-8 production. Cell. Microbiol. 9, 2457–2469 (2007).
Lu, H. et al. Functional and intracellular signaling differences associated with the Helicobacter pylori AlpAB adhesin from Western and East Asian strains. J. Biol. Chem. 282, 6242–6254 (2007).
Kudo, T. et al. Pattern of transcription factor activation in Helicobacter pylori-infected Mongolian gerbils. Gastroenterology 132, 1024–1038 (2007).
Wu, J. Y. et al. Balance between polyoma enhancing activator 3 and activator protein 1 regulates Helicobacter pylori-stimulated matrix metalloproteinase 1 expression. Cancer Res. 66, 5111–5120 (2006).
Lu, H. et al. Regulation of interleukin-6 promoter activation in gastric epithelial cells infected with Helicobacter pylori. Mol. Biol. Cell 16, 4954–4966 (2005).
Tabassam, F. H., Graham, D. Y. & Yamaoka, Y. OipA plays a role in Helicobacter pylori-induced focal adhesion kinase activation and cytoskeletal re-organization. Cell. Microbiol. 10, 1008–1020 (2008).
Tabassam, F. H., Graham, D. Y. & Yamaoka, Y. Helicobacter pylori activate epidermal growth factor receptor- and phosphatidylinositol 3-OH kinase-dependent Akt and glycogen synthase kinase 3β phosphorylation. Cell. Microbiol. 11, 70–82 (2009).
Yamaoka, Y. et al. Helicobacter pylori infection in mice: Role of outer membrane proteins in colonization and inflammation. Gastroenterology 123, 1992–2004 (2002).
Lu, H., Hsu, P. I., Graham, D. Y. & Yamaoka, Y. Duodenal ulcer promoting gene of Helicobacter pylori. Gastroenterology 128, 833–848 (2005).
Hussein, N. R. et al. Helicobacter pylori dupA is polymorphic, and its active form induces proinflammatory cytokine secretion by mononuclear cells. J. Infect. Dis. 202, 261–269 (2010).
Schmidt, H. M. et al. The prevalence of the duodenal ulcer promoting gene (dupA) in Helicobacter pylori isolates varies by ethnic group and is not universally associated with disease development: a case-control study. Gut Pathog. 1, 5 (2009).
Yamaoka, Y. Roles of the plasticity regions of Helicobacter pylori in gastroduodenal pathogenesis. J. Med. Microbiol. 57, 545–553 (2008).
Arachchi, H. S. J. et al. Prevalence of duodenal ulcer-promoting gene (dupA) of Helicobacter pylori in patients with duodenal ulcer in North Indian population. Helicobacter 12, 591–597 (2007).
Zhang, Z. et al. The Helicobacter pylori duodenal ulcer promoting gene, dupA in China. BMC Gastroenterol. 8, 49 (2008).
Douraghi, M. et al. dupA as a risk determinant in Helicobacter pylori infection. J. Med. Microbiol. 57, 554–562 (2008).
Gomes, L. I. et al. Lack of association between Helicobacter pylori infection with dupA-positive strains and gastroduodenal diseases in Brazilian patients. Int. J. Med. Microbiol. 298, 223–230 (2007).
Pacheco, A. R. et al. Involvement of the Helicobacter pylori plasticity region and cag pathogenicity island genes in the development of gastroduodenal diseases. Eur. J. Clin. Microbiol. Infect. Dis. 27, 1053–1059 (2008).
Nguyen, L. T. et al. Helicobacter pylori dupA gene is not associated with clinical outcomes in the Japanese population. Clin. Microbiol. Infect. 16, 1264–1269 (2010).
Argent, R. H., Burette, A., Miendje Deyi, V. Y. & Atherton, J. C. The presence of dupA in Helicobacter pylori is not significantly associated with duodenal ulceration in Belgium, South Africa, China, or North America. Clin. Infect. Dis. 45, 1204–1206 (2007).
Hussein, N. R. The association of dupA and Helicobacter pylori-related gastroduodenal diseases. Eur. J. Clin. Microbiol. Infect. Dis. 29, 817–821 (2010).
Queiroz, D. M. et al. dupA polymorphisms and risk of distal gastric cancer. Gastroenterology 134, A-85 (2008).
Kersulyte, D. et al. Cluster of type IV secretion genes in Helicobacter pylori's plasticity zone. J. Bacteriol. 185, 3764–3772 (2003).
Kersulyte, D. et al. Helicobacter pylori's plasticity zones are novel transposable elements. PLoS ONE 4, e6859 (2009).
Fischer, W. et al. Strain-specific genes of Helicobacter pylori: genome evolution driven by a novel type IV secretion system and genomic island transfer. Nucleic Acids Res. doi: 10.1093/nar/gkq378.
Couturier, M. R., Tasca, E., Montecucco, C. & Stein, M. Interaction with CagF is required for translocation of CagA into the host via the Helicobacter pylori type IV secretion system. Infect. Immun. 74, 273–281 (2006).
Pattis, I., Weiss, E., Laugks, R., Haas, R. & Fischer, W. The Helicobacter pylori CagF protein is a type IV secretion chaperone-like molecule that binds close to the C-terminal secretion signal of the CagA effector protein. Microbiology 153, 2896–2909 (2007).
Jimenez-Soto, L. F. et al. Helicobacter pylori type IV secretion apparatus exploits beta1 integrin in a novel RGD-independent manner. PLoS Pathog. 5, e1000684 (2009).
Acknowledgements
This report is based on work supported in part by grants from the NIH (DK62813), and grants-in-aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan (22390085 and 22659087).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Yamaoka, Y. Mechanisms of disease: Helicobacter pylori virulence factors. Nat Rev Gastroenterol Hepatol 7, 629–641 (2010). https://doi.org/10.1038/nrgastro.2010.154
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2010.154
This article is cited by
-
Polymorphism in autophagy-related genes LRP1 and CAPZA1 may promote gastric mucosal atrophy
Genes and Environment (2023)
-
Comparative genomics of two Vietnamese Helicobacter pylori strains, CHC155 from a non-cardia gastric cancer patient and VN1291 from a duodenal ulcer patient
Scientific Reports (2023)
-
Is Helicobacter pylori infection associated with osteoporosis? a systematic review and meta-analysis
Journal of Bone and Mineral Metabolism (2023)
-
Helicobacter pylori infection: is there circulating vacuolating cytotoxin A or cytotoxin-associated gene A protein?
Gut Pathogens (2022)
-
Helicobacter pylori cagA status and gastric mucosa-associated lymphoid tissue lymphoma: a systematic review and meta-analysis
Journal of Health, Population and Nutrition (2022)