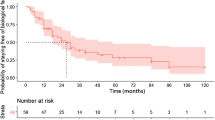
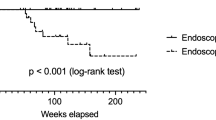
Abstract
The natural history of Crohn's disease is characterized by progression to complicated and disabling disease, often necessitating surgical interventions. There is either circumstantial or direct evidence to support the disease-modifying potential of several therapeutic agents. Healing of endoscopic lesions is an emerging surrogate marker of disease modification, as mucosal lesions are considered to reflect ongoing inflammation and tissue damage that lead to the formation of fistulas and fibrotic strictures, which are the main indications for surgery. In contrast to systemic steroids, both azathioprine and anti-tumor necrosis factor (TNF) agents have demonstrated the potential of mucosal healing. Prevention of hospitalization and surgery in the short and medium term has been demonstrated for the anti-TNF agents infliximab and adalimumab. The evidence supporting a role for medical therapy in the prevention of fibrotic wall thickening and in the obliteration of fistula tracks is limited and should be the focus of further prospective studies. These studies should validate predictors of complicated disease and randomized studies should be performed in high-risk groups to investigate whether early introduction of immunosuppressive agents or biologic therapies slows down disease progression and alters the natural history of the disease.
Key Points
-
Disease modification in Crohn's disease should be aimed at avoiding the development of complicated disease, which is associated with hospitalization and surgery
-
Endoscopic mucosal healing is currently the best surrogate marker of disease modification and has been observed after treatment with traditional immunosuppressive agents and with anti-tumor necrosis factor (TNF) agents
-
Early, aggressive immunosuppressive therapy with thiopurines and/or anti-TNF agents results in higher rates of clinical remission; the influence of this strategy on the prevention of complicated disease needs to be established
-
Predictors of early complicated disease are still ill-defined and elucidating these predictors would enable the selection of patients for early immunosuppressive therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Munkholm, P., Langholz, E., Davidsen, M. & Binder, V. Disease activity courses in a regional cohort of Crohn's disease patients. Scand. J. Gastroenterol. 30, 699–706 (1995).
Silverstein, M. D. et al. Clinical course and costs of care for Crohn's disease: Markov model analysis of a population-based cohort. Gastroenterology 117, 49–57 (1999).
Veloso, F. T., Ferreira, J. T., Barros, L. & Almeida, S. Clinical outcome of Crohn's disease: analysis according to the Vienna classification and clinical activity. Inflamm. Bowel Dis. 7, 306–313 (2001).
Farmer, R. G., Hawk, W. A. & Turnbull R. B. Jr. Clinical patterns in Crohn's disease: a statistical study of 615 cases. Gastroenterology 68, 627–635 (1975).
Louis, E. et al. Behavior of Crohn's disease according to the Vienna classification: changing pattern over the course of the disease. Gut 49, 777–782 (2001).
Cosnes, J. et al. Long-term evolution of disease behavior of Crohn's disease. Inflamm. Bowel Dis. 8, 244–250 (2002).
Michelassi, F., Balestracci, T., Chappell, R. & Block, G. E. Primary and recurrent Crohn's disease. Experience with 1379 patients. Ann. Surg. 214, 230–238 (1991).
Greenstein, A. J. et al. Perforating and non-perforating indications for repeated operations in Crohn's disease: evidence for two clinical forms. Gut 29, 588–592 (1988).
Rutgeerts, P. et al. Predictability of the postoperative course of Crohn's disease. Gastroenterology 99, 956–963 (1990).
Mekhjian, H. S. et al. National Cooperative Crohn's Disease Study: factors determining recurrence of Crohn's disease after surgery. Gastroenterology 77, 907–913 (1979).
Whelan, G., Farmer, R. G., Fazio, V. W. & Goormastic, M. Recurrence after surgery in Crohn's disease. Relationship to location of disease (clinical pattern) and surgical indication. Gastroenterology 88, 1826–1833 (1985).
Beaugerie, L. et al. Predictors of Crohn's disease. Gastroenterology 130, 650–656 (2006).
Wolters, F. L. et al. Disease outcome of inflammatory bowel disease patients: general outline of a Europe-wide population-based 10-year clinical follow-up study. Scand. J. Gastroenterol. Suppl. 243, 46–54 (2006).
Lichtenstein, G. R. et al. Factors associated with the development of intestinal strictures or obstructions in patients with Crohn's disease. Am. J. Gastroenterol. 101, 1030–1038 (2006).
Buning, C. et al. Mutations in the NOD2/CARD15 gene in Crohn's disease are associated with ileocecal resection and are a risk factor for reoperation. Aliment. Pharmacol. Ther. 19, 1073–1078 (2004).
Franchimont, D. P., Louis, E., Croes, F. & Belaiche, J. Clinical pattern of corticosteroid dependent Crohn's disease. Eur. J. Gastroenterol. Hepatol. 10, 821–825 (1998).
Munkholm, P., Langholz, E., Davidsen, M. & Binder, V. Frequency of glucocorticoid resistance and dependency in Crohn's disease. Gut 35, 360–362 (1994).
Faubion, W. A. Jr et al. The natural history of corticosteroid therapy for inflammatory bowel disease: a population-based study. Gastroenterology 121, 255–260 (2001).
Rutgeerts, P. et al. A comparison of budesonide with prednisolone for active Crohn's disease. N. Engl. J. Med. 331, 842–845 (1994).
Sandborn, W. J., Lofberg, R., Feagan, B. G., Hanauer, S. B., Campieri, M. & Greenberg, G. R. Budesonide for maintenance of remission in patients with Crohn's disease in medically induced remission: a predetermined pooled analysis of four randomized, double-blind, placebo-controlled trials. Am. J. Gastroenterol. 100, 1780–1787 (2005).
Ewe, K., Bottger, T., Buhr, H. J., Ecker, K. W. & Otto, H. F. Low-dose budesonide treatment for prevention of postoperative recurrence of Crohn's disease: a multicentre randomized placebo-controlled trial. German Budesonide Study Group. Eur. J. Gastroenterol. Hepatol. 11, 277–282 (1999).
Hellers, G. et al. Oral budesonide for prevention of postsurgical recurrence in Crohn's disease. The IOIBD Budesonide Study Group. Gastroenterology 116, 294–300 (1999).
Travis, S. P. L. et al. for the European Crohn's and Colitis Organisation (ECCO). European evidence based consensus on the diagnosis and management of Crohn's disease: current management. Gut 55 (Suppl. 1), 16–35 (2006).
Candy, S. et al. A controlled double blind study of azathioprine in the management of Crohn's disease. Gut 37, 674–678 (1995).
Markowitz, J. et al. A multicenter trial of 6-mercaptopurine and prednisone in children with newly diagnosed Crohn's disease. Gastroenterology 119, 895–902 (2000).
Feagan, B. G. et al. Methotrexate for the treatment of Crohn's disease. The North American Crohn's Study Group Investigators. N. Engl. J. Med. 332, 292–297 (1995).
Feagan, B. G. et al. A comparison of methotrexate with placebo for the maintenance of remission in Crohn's disease. North American Crohn's Study Group Investigators. N. Engl. J. Med. 342, 1627–1632 (2000).
Weisman, M. H. Natural history and treatment decisions in rheumatoid arthritis revisited. Arthritis Care Res. 2, S75–S83 (1989).
Kozarek, R. A. et al. Methotrexate induces clinical and histological remission in patients with refractory inflammatory bowel disease. Ann. Intern. Med. 110, 353–356 (1989).
Panaccione, R. The use of methotrexate is associated with mucosal healing in Crohn's disease. Gastroenterology 128 (Suppl.), A49 (2005).
Fraser, A. G. et al. The efficacy of methotrexate for maintaining remission in inflammatory bowel disease. Aliment. Pharmacol. Ther. 16, 693–697 (2002).
Lemann, M. et al. Methotrexate in Crohn's disease: long-term efficacy and toxicity. Am. J. Gastroenterol. 95, 1730–1734 (2000).
Vandeputte, L. et al. Methotrexate in refractory Crohn's disease. Inflamm. Bowel Dis. 5, 11–15 (1999).
Cosnes, J. et al. Impact of the increasing use of immunosuppressants in Crohn's disease on the need for intestinal surgery. Gut 54, 237–241 (2005).
Ardizzone, S. et al. Azathioprine and mesalazine for prevention of relapse after conservative surgery for Crohn's disease. Gastroenterology 127, 730–740 (2004).
Hanauer, S. B. et al. Postoperative maintenance of Crohn's disease remission with 6-mercaptopurine, mesalazine, or placebo: a 2-year trial. Gastroenterology 127, 723–729 (2004).
D'Haens, G. R. et al. Therapy of metronidazole with azathioprine to prevent postoperative recurrence of Crohn's disease: a controlled randomized trial. Gastroenterology 135, 1123–1129 (2008).
Domènech, E. et al. Impact of azathioprine on the prevention of postoperative Crohn's disease recurrence: results of a prospective, observational, long-term follow-up study. Inflamm. Bowel Dis. 14, 508–513 (2008).
Rutgeerts, P. Strategies in the prevention of post-operative recurrence in Crohn's disease. Best Pract. Res. Clin. Gastroenterol. 17, 63–73 (2003).
Hanauer, S. B. et al. ACCENT I Study Group. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet 359, 1541–1549 (2002).
Sands, B. E. et al. A randomized controlled trial of infliximab maintenance therapy for fistulizing Crohn's disease (ACCENT II). N. Engl. J. Med. 350, 876–885 (2004).
Targan, S. R. et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. Crohn's Disease cA2 Study Group. N. Engl. J. Med. 337, 1029–1035 (1997).
Mease, P. TNFalpha therapy in psoriatic arthritis and psoriasis. Ann. Rheum. Dis. 63, 755–758 (2004).
Baraliakos, X., Listing, J., Rudwaleit, M., Brandt, J., Sieper, J. & Braun, J. Radiographic progression in patients with ankylosing spondylitis after 2 years of treatment with the tumour necrosis factor alpha antibody infliximab. Ann. Rheum. Dis. 64, 1462–1466 (2005).
D'Haens, G. et al. Endoscopic and histological healing with infliximab anti-tumor necrosis factor antibodies in Crohn's disease: a European multicenter trial. Gastroenterology 116, 1029–1034 (1999).
Rutgeerts, P. et al. Comparison of scheduled and episodic treatment strategies of infliximab in Crohn's disease. Gastroenterology 126, 402–413 (2004).
Rutgeerts, P. et al. Scheduled maintenance treatment with infliximab is superior to episodic treatment for the healing of mucosal ulceration associated with Crohn's disease. Gastrointest. Endosc. 63, 433–442 (2006).
Colombel, J. F. et al. Endoscopic mucosal improvement in patients with active Crohn's disease treated with certolizumab pegol: first results of the music clinical trial. Am. J. Gastroenterol. 103, A1107 (2008).
Feagan, B. G. et al. Effects of adalimumab therapy on incidence of hospitalization and surgery in Crohn's disease: results from the CHARM study. Gastroenterology 135, 1493–1499 (2008).
Regueiro, M. et al. Infliximab prevents Crohn's disease recurrence after ileal resection. Gastroenterology 136, 441–450 (2009).
Griffiths, A. M. Enteral feeding in inflammatory bowel disease. Curr. Opin. Clin. Nutr. Metab. Care 9, 314–318 (2006).
Fell, J. M. et al. Mucosal healing and a fall in mucosal pro-inflammatory cytokine mRNA induced by a specific oral polymeric diet in paediatric Crohn's disease. Aliment. Pharmacol. Ther. 14, 281–289 (2000).
Esaki, M. et al. Preventive effect of nutritional therapy against postoperative recurrence of Crohn disease, with reference to findings determined by intra-operative enteroscopy. Scand. J. Gastroenterol. 40, 1431–1437 (2005).
Lochs, H. et al. ESPEN guidelines on enteral nutrition: gastroenterology. Clin. Nutr. 25, 260–274 (2006).
Lionetti, P. et al. Response to infliximab is related to disease duration in paediatric Crohn's disease. Aliment. Pharmacol. Ther. 18, 425–431 (2003).
Kugathasan, S. et al. Prolonged duration of response to infliximab in early but not late pediatric Crohn's disease. Am. J. Gastroenterol. 95, 3189–3194 (2000).
Hyams, J. et al. Induction and maintenance infliximab therapy for the treatment of moderate-to-severe Crohn's disease in children. Gastroenterology 13, 863–873 (2007).
Rudwaleit, M. et al. Prediction of a major clinical response (BASDAI 50) to tumour necrosis factor alpha blockers in ankylosing spondylitis. Ann. Rheum. Dis. 63, 665–670 (2004).
Goekoop-Ruiterman, Y. P. et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum. 52, 3381–3390 (2005).
D'Haens, G. et al. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn's disease: an open randomised trial. Lancet 371, 660–667 (2008).
Kandiel, A. et al. Increased risk of lymphoma among inflammatory bowel disease patients treated with azathioprine and 6-mercaptopurine. Gut 54, 1121–1125 (2005).
Bongartz, T. et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 295, 2275–2285 (2006).
Lichtenstein, G. R. et al. Serious infections and mortality in association with therapies for Crohn's disease: TREAT registry. Clin. Gastroenterol. Hepatol. 4, 621–630 (2006).
Dubinsky, M. C. et al. Serum immune responses predict rapid disease progression among children with Crohn's disease: immune responses predict disease progression. Am. J. Gastroenterol. 101, 360–367 (2006).
Mow, W. S. et al. Association of antibody responses to microbial antigens and complications of small bowel Crohn's disease. Gastroenterology 126, 414–424 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
G. Van Assche, S. Vermeire and P. Rutgeerts serve as consultants for Abbott, Merck and Schering-Plough, are members of the speaker bureau for Abbott, Merck, Schering-Plough and UCB and receive research support from Abbott and Centocor.
Rights and permissions
About this article
Cite this article
Van Assche, G., Vermeire, S. & Rutgeerts, P. The potential for disease modification in Crohn's disease. Nat Rev Gastroenterol Hepatol 7, 79–85 (2010). https://doi.org/10.1038/nrgastro.2009.220
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2009.220
This article is cited by
-
Does Similarity Breed Contempt? A Review of the Use of Biosimilars in Inflammatory Bowel Disease
Digestive Diseases and Sciences (2021)
-
Mucosal microbiota and gene expression are associated with long-term remission after discontinuation of adalimumab in ulcerative colitis
Scientific Reports (2020)
-
Systematic review with meta-analysis: loss of response and requirement of anti-TNFα dose intensification in Crohn’s disease
Journal of Gastroenterology (2017)
-
Advancing host-directed therapy for tuberculosis
Nature Reviews Immunology (2015)
-
Surgery and diagnostic imaging in abdominal Crohn’s disease
Journal of Ultrasound (2015)