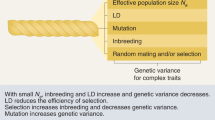
Key Points
-
High levels of inherited variation are observed for most traits and in most populations. Variation is maintained partly by mutation and partly by a balance of selective forces; however, we do not know the relative importance of these alternatives.
-
This variation allows a rapid response to natural and artificial selection. Newly arising mutations make an important contribution to long-term selection response. Selection response in large experimental populations often continues steadily for many generations, indicating that many genetic loci are involved.
-
Understanding the maintenance of variation, and the response to selection, requires that the sequence changes that cause trait differences be identified. This is challenging, because variation might depend on multiple alleles that include several interacting sites.
-
Population genetics makes predictions about the nature of quantitative trait loci (QTL). For example, balancing selection is expected to maintain alleles at high frequency, whereas mutation is likely to maintain rare alleles.
-
Predictions for the way that alleles interact, and for the size of their effects, depend on assumptions about the relationship between genotype and phenotype. One simple model indicates that QTL effects should be exponentially distributed.
Abstract
Until recently, it was impracticable to identify the genes that are responsible for variation in continuous traits, or to directly observe the effects of their different alleles. Now, the abundance of genetic markers has made it possible to identify quantitative trait loci (QTL) — the regions of a chromosome or, ideally, individual sequence variants that are responsible for trait variation. What kind of QTL do we expect to find and what can our observations of QTL tell us about how organisms evolve? The key to understanding the evolutionary significance of QTL is to understand the nature of inherited variation, not in the immediate mechanistic sense of how genes influence phenotype, but, rather, to know what evolutionary forces maintain genetic variability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Provine, W. The Origins of Theoretical Population Genetics (Chicago Univ. Press, Chicago, Illinois, 1971).
Barton, N. H. & Turelli, M. Evolutionary quantitative genetics: how little do we know? Annu. Rev. Genet. 23, 337–370 (1989).
Falconer, D. S. & Mackay, T. F. C. Introduction to Quantitative Genetics (Longman, London, 1995).
Roff, D. A. Evolutionary Quantitative Genetics (Chapman & Hall, New York, 1997).
Houle, D. Comparing evolvability and variability of quantitative traits. Genetics 130, 195–204 (1992).
Lewontin, R. C. The Genetic Basis of Evolutionary Change (Columbia Univ. Press, New York, 1974).
Bodmer, W. F. & Cavalli-Sforza, L. L. Genetics, Evolution and Man (W. H. Freeman, San Francisco, 1976).
Kondrashov, A. S. & Turelli, M. Deleterious mutations, apparent stabilising selection and the maintenance of quantitative variation. Genetics 132, 603–618 (1992).
Kingsolver, J. G. et al. The strength of phenotypic selection in natural populations. Am. Nat. 157, 245–261 (2001).A comprehensive survey of the strength of selection on quantitative traits in natural populations, which implies that stabilizing selection might be less prevalent and is harder to measure accurately than has been previously thought.
Fisher, R. A. The correlation between relatives on the supposition of Mendelian inheritance. Proc. R. Soc. Edinb. 52, 399–433 (1918).
Hill, W. G. Rates of change in quantitative traits from fixation of new mutations. Proc. Natl Acad. Sci. USA 79, 142–145 (1982).This work quantified the contribution of new mutations to artificial selection response, and predicted that this could be substantial.
Lynch, M. & Walsh, J. B. Genetics and Analysis of Quantitative Traits (Sinauer Associates, Sunderland, Massachusetts, 1998).
Yoo, B. H. Long-term selection for a quantitative character in large replicate populations of Drosophila melanogaster. II. Lethals and visible mutants with large effects. Genet. Res. 35, 19–31 (1980).
Weber, K. E. Large genetic change at small fitness cost in large populations of Drosophila melanogaster selected for wind tunnel flight: rethinking fitness surfaces. Genetics 144, 205–213 (1996).
Hill, W. G. & Keightley, P. D. in Second International Conference on Quantitative Genetics (eds Eisen, E. J., Goodman, M. M., Namkoong, G. & Weir, B. S.) 57–70 (Sinauer Associates, Sunderland, Massachusetts, 1988).
Orr, H. A. The genetics of species differences. Trends Ecol. Evol. 16, 343–358 (2001).
Kearsey, M. J. & Farquhar, A. G. L. QTL analysis in plants; where are we now? Heredity 80, 137–142 (1998).
Mackay, T. F. C. Quantitative trait loci in Drosophila. Nature Rev. Genet. 2, 11–20 (2001).
Mackay, T. F. C. & Langley, C. H. Molecular and phenotypic variation in the achaete-scute region of Drosophila melanogaster. Nature 348, 64–66 (1990).This paper shows that naturally occurring large insertions in a candidate gene for bristle number in Drosophila melanogaster are associated with bristle number variation.
Long, A. D., Lyman, R. F., Morgan, A. H., Langley, C. H. & Mackay, T. F. C. Both naturally occurring insertions of transposable elements and intermediate frequency polymorphisms at the achaete scute complex are associated with variation in bristle number in Drosophila melanogaster. Genetics 154, 1255–1269 (2000).
Charlesworth, B. & Langley, C. H. The population genetics of Drosophila transposable elements. Annu. Rev. Genet. 23, 251–287 (1989).
Lyman, R. F., Lai, C. Q. & Mackay, T. F. C. Linkage disequilibrium mapping of molecular polymorphisms at the scabrous locus associated with naturally occurring variation in bristle number in Drosophila melanogaster. Genet. Res. 74, 303–311 (1999).
Long, A. D., Lyman, R. F., Langley, C. H. & Mackay, T. F. C. Two sites in the Delta gene region contribute to naturally occurring variation in bristle number in Drosophila melanogaster. Genetics 149, 999–1017 (1998).
Zapata, C. & Alvarez, G. The detection of gametic disequilibrium between allozyme loci in natural populations of Drosophila. Evolution 46, 1900–1917 (1992).
Begun, D. J. & Aquadro, C. F. African and North American populations of Drosophila melanogaster are very different at the DNA level. Nature 353, 548–549 (1993).
Przeworski, M., Wall, J. D. & Andolfatto, P. Recombination and the frequency spectrum in Drosophila melanogaster and Drosophila simulans. Mol. Biol. Evol. 18, 291–298 (2001).
Teeter, K. et al. Haplotype dimorphism in a SNP collection from Drosophila melanogaster. J. Exp. Zool. 88, 63–75 (2000).
Stam, L. F. & Laurie, C. C. Molecular dissection of a major gene effect on a quantitative trait: the level of alcohol dehydrogenase expression in Drosophila melanogaster. Genetics 144, 1559–1564 (1996).A beautiful paper that studied replicated transgenic constructs of the Adh gene in Drosophila to dissect the contribution of molecular variation in different parts of the gene to variation in enzyme activity.
Laurie-Ahlberg, C. C. Genetic variation affecting the expression of enzyme-coding genes in Drosophila: an evolutionary perspective. Curr. Top. Biol. Med. Res. 12, 33–88 (1985).
Risch, N. J. Searching for genetic determinants in the new millennium. Nature 405, 847–856 (2000).
Cardon, L. R. & Bell, J. I. Association study designs for complex diseases. Nature Rev. Genet. 2, 91–99 (2001).
Weiss, K. M. & Terwilliger, J. D. How many diseases does it take to map a gene with SNPs? Nature Genet. 26, 151–157 (2001).
Graham, G. I., Wolff, D. W. & Stuber, C. W. Characterization of a yield quantitative trait locus on chromosome five of maize by fine mapping. Crop Sci. 37, 1601–1610 (1997).
Iraqi, F. et al. Fine mapping of trypanosomiasis resistance loci in murine advanced intercross lines. Mamm. Genome 11, 645–648 (2000).
Legare, M. E., Bartlett, F. S. & Frankel, W. N. A major effect QTL determined by multiple genes in epileptic EL mice. Genome Res. 10, 42–48 (2000).
Monforte, A. J. & Tanksley, S. D. Fine mapping of a quantitative trait locus (QTL) from Lycopersicon hirsutum chromosome 1 affecting fruit characteristics and agronomic traits: breaking linkage among QTL affecting different traits and dissection of heterosis for yield. Theor. Appl. Genet. 100, 471–497 (2000).
Podolin, P. L. et al. Localization of two insulin-dependent diabetes (Idd) genes to the Idd10 region on mouse chromosome 3. Mamm. Genome 9, 283–286 (1998).
Vladutu, C., McLaughlin, J. & Phillips, R. L. Fine mapping and characterization of linked quantitative trait loci involved in the transition of the maize apical meristem from vegetative to generative structures. Genetics 153, 993–1007 (1999).
Perez, D. E. & Wu, C. I. Further characterization of the Odysseus locus of hybrid sterility in Drosophila: one gene is not enough. Genetics 140, 201–206 (1995).
Doebley, J., Stec, A. & Hubbard, L. The evolution of apical dominance in maize. Nature 386, 485–488 (1997).Reports what is generally accepted to be the first map-based cloning of a QTL, teosinte branched1.
Frary, A., Nesbitt, T. C. & Frary, A. fw2.2: a quantitative trait locus key to the evolution of tomato fruit size. Science 289, 85–88 (2000).
Fridman, E., Pleban, T. & Zamir, D. A recombination hotspot delimits a wild-species quantitative trait locus for tomato sugar content to 484 bp within an invertase gene. Proc. Natl Acad. Sci. USA 97, 4718–4723 (2000).References 41 and 42 report successful positional cloning experiments for agronomic traits in tomato.
Wang, R. L., Stec, A., Hey, J., Ukens, L. & Doebley, J. The limits of selection during maize domestication. Nature 398, 236–239 (1999).
Barton, N. H. Pleiotropic models of quantitative variation. Genetics 124, 773–782 (1990).
Turelli, M. Heritable genetic variation via mutation–selection balance: Lerch's ζ meets the abdominal bristle. Theor. Popul. Biol. 25, 138–193 (1984).
Lande, R. The maintenance of genetic variability by mutation in a polygenic character with linked loci. Genet. Res. 26, 221–236 (1975).References 45 and 46 provide two mathematical analyses of the stabilizing selection model and come to different conclusions concerning the variation that can be maintained for quantitative traits, depending on the mutation rates at the individual loci involved.
Wagner, G. P. Apparent stabilizing selection and the maintenance of neutral genetic variation. Genetics 143, 617–619 (1996).
Keightley, P. D. & Eyre-Walker, A. Deleterious mutations and the evolution of sex. Science 290, 331–333 (2000).
Wagner, G. P. Multivariate mutation–selection balance with constrained pleiotropic effects. Genetics 122, 223–234 (1989).
Turelli, M. Effects of pleiotropy on predictions concerning mutation–selection balance for polygenic traits. Genetics 111, 165–195 (1985).
Keightley, P. D. & Hill, W. G. Quantitative genetic variability maintained by mutation/stabilising selection balance in finite populations. Genet. Res. 52, 33–43 (1988).
Lyman, R. F., Lawrence, F., Nuzhdin, S. & Mackay, T. F. C. Effects of single P-element insertions on bristle number and viability in Drosophila melanogaster. Genetics 143, 277–292 (1996).
Hastings, A. & Hom, C. L. Pleiotropic stabilising selection limits the number of polymorphic loci to at most the number of characters. Genetics 122, 459–463 (1989).
Wright, S. Evolution in populations in approximate equilibrium. J. Genet. 30, 257–266 (1935).
Lerner, I. M. Genetic Homeostasis (Oliver & Boyd, Edinburgh, 1954).
Gillespie, J. H. & Turelli, M. Genotype–environment interactions and the maintenance of polygenic variation. Genetics 137, 129–138 (1989).
Zhivotovsky, L. A. & Feldman, M. W. On models of quantitative genetic variation: a stabilizing selection-balance model. Genetics 130, 947–955 (1992).
Podolsky, R. H. Genetic variation for morphological and allozyme variation in relation to population size in Clarkia dudleyana, an endemic annual. Conserv. Biol. 15, 412–423 (2001).
Charlesworth, D. & Mayer, S. Genetic variability of plant characters in the partial inbreeder Collinsia heterophylla (Scrophulariaceae). Am. J. Bot. 82, 112–120 (1995).
Smith, T. B. Disruptive selection and the genetic basis of bill size polymorphism in the African finch Pyrenestes. Nature 363, 618–620 (1993).
Sasaki, A. & Ellner, S. Quantitative genetic variance maintained by fluctuating selection with overlapping generations: variance components and covariances. Evolution 51, 682–696 (1997).
Slatkin, M. Frequency- and density-dependent selection on a quantitative character. Genetics 93, 755–771 (1979).
Bulmer, M. G. The Mathematical Theory of Quantitative Genetics (Oxford Univ. Press, Oxford, 1985).
Burger, R. Evolution of genetic variability and the advantage of sex and recombination in changing environments. Genetics 153, 1055–1069 (1999).
Waxman, D. & Peck, J. R. Sex and adaptation in a changing environment. Genetics 153, 1041–1053 (1999).
Kondrashov, A. S. & Yampolsky, L. Y. High genetic variability under the balance between symmetric mutation and fluctuating stabilizing selection. Genet. Res. 68, 157–164 (1996).
Robertson, A. Effect of selection against extreme deviants based on deviation or on homozygosis. J. Genet. 54, 236–248 (1956).
Gillespie, J. H. Pleiotropic overdominance and the maintenance of genetic variation in polygenic characters. Genetics 107, 321–330 (1984).
Watt, W. B., Cassin, R. C. & Swan, M. S. Adaptation at specific loci. III. Field behaviour and survivorship differences among Colias PGI genotypes are predictable from in vitro biochemistry. Genetics 103, 725–729 (1983).
Kreitman, M. & Aguade, M. Excess polymorphism in the Adh region of Drosophila melanogaster. Genetics 114, 93–110 (1986).
Hughes, A. L. Adaptive Evolution of Genes and Genomes (Oxford Univ. Press, Oxford, 1999).
Burger, R. The Mathematical Theory of Selection, Recombination and Mutation (Wiley, Chichester, UK, 2000).
Fisher, R. A. The Genetical Theory of Natural Selection (Oxford Univ. Press, Oxford, 1930).
Kimura, M. The Neutral Theory of Molecular Evolution (Cambridge Univ. Press, Cambridge, 1983).
Orr, H. A. The population genetics of adaptation: the distribution of factors fixed during adaptive evolution. Evolution 52, 935–949 (1998).An analysis of Fisher's model of adaptation, which shows that the distribution of factors fixed during adaptation is expected to be approximately exponential.
Hayes, B. & Goddard, M. E. The distribution of the effects of genes affecting quantitative traits in livestock. Genet. Select. Evol. 33, 209–230 (2001).
Shrimpton, A. E. & Robertson, A. The isolation of polygenic factors controlling bristle score in Drosophila melanogaster. II. Distribution of third chromosome bristle effects within chromosome sections. Genetics 118, 445–459 (1988).
Orr, H. A. Adaptation and the cost of complexity. Evolution 54, 13–20 (2000).
True, H. L. & Lindquist, S. L. A yeast prion provides a mechanism for genetic variation and genetic diversity. Nature 407, 477–483 (2000).
Hirsh, A. E. & Fraser, H. B. Protein dispensability and rate of evolution. Nature 411, 1046–1049 (2001).
Smith, V., Chou, K. N., Lashkari, D., Botstein, D. & Borwn, P. O. Functional analysis of the genes of yeast chromosome V by genetic footprinting. Science 274, 2069–2074 (1996).
Kauffman, S. Origins of Order (Cambridge Univ. Press, Cambridge, 1992).
Kacser, H. in Evolution and Animal Breeding (eds Hill, W. G. & Mackay, T. F. C.) 219–226 (CAB International, Wallingford, UK, 1989).
Hasty, J., McMillen, D., Isaacs, F. & Collins, J. J. Computational studies of gene regulatory networks: in numero molecular biology. Nature Rev. Genet. 2, 268–279 (2001).
Weber, K. E. et al. An analysis of polygenes affecting wing shape on chromosome 3 in Drosophila melanogaster. Genetics 153, 773–786 (1999).An extremely well-replicated QTL-mapping experiment for wing shape in Drosophila that points to a highly polygenic basis of inheritance.
Keightley, P. D. Models of quantitative genetic variation of flux in metabolic pathways. Genetics 121, 869–876 (1989).
Kacser, H. & Burns, J. A. The molecular basis of dominance. Genetics 97, 639–666 (1981).A classic paper that was one of the first to explicitly model a biochemical system and relate its properties to the properties of quantitative traits, such as dominance and epistasis.
Keightley, P. D. Metabolic models of selection response. J. Theor. Biol. 182, 311–316 (1996).
Gurganus, M. C., Nuzhdin, S. V., Leips, J. W. & Mackay, T. F. C. High-resolution mapping of quantitative trait loci for sternopleural bristle number in Drosophila melanogaster. Genetics 152, 1585–1604 (1999).
Long, A. D. et al. High resolution genetic mapping of genetic factors affecting abdominal bristle number in Drosophila melanogaster. Genetics 139, 1273–1291 (1995).
Routman, E. J. & Cheverud, J. M. Gene effects on a quantitative trait: two-locus epistatic effects measured at microsatellite markers and at estimated QTL. Evolution 51, 1654–1662 (1997).
Eshed, Y. & Zamir, D. Less-than-additive epistatic interactions of quantitative trait loci in tomato. Genetics 143, 1807–1817 (1996).
Lukens, L. N. & Doebley, J. Epistatic and environmental interactions for quantitative trait loci involved in maize evolution. Genet. Res. 74, 291–302 (1999).
Kondrashov, A. S. Deleterious mutations and the evolution of sexual reproduction. Nature 336, 435–441 (1988).
Lehman, N. & Joyce, G. F. Evolution in vitro: analysis of a lineage of ribozymes. Curr. Biol. 3, 723–734 (1993).
McKenzie, J. A. & O'Farrell, K. Modification of developmental instability and fitness — malathion resistance in the Australian sheep blowfly, Lucilia cuprina. Genetica 89, 67–76 (1993).
Schrag, S. J., Perrot, V. & Levin, B. R. Adaptation to the fitness costs of antibiotic resistance in Escherichia coli. Proc. R. Soc. Lond. B 264, 1287–1291 (1997).
Stern, D. L. Evolutionary developmental biology and the problem of variation. Evolution 54, 1079–1091 (2000).
Weatherall, D. J. Phenotype–genotype relationships in monogenic disease: lessons from the thalassaemias. Nature Rev. Genet. 2, 245–255 (2001).
Dekkers, J. C. M. & Dentine, M. R. Quantitative genetic variance associated with chromosomal markers in segregating populations. Theor. Appl. Genet. 81, 212–220 (1991).
Visscher, P. M. & Haley, C. S. Detection of putative quantitative trait loci in line crosses under infinitesimal genetic models. Theor. Appl. Genet. 93, 691–702 (1996).
Noor, M. A., Cunningham, A. L. & Larkin, J. C. Consequences of recombination rate variation on quantitative trait locus mapping studies. Simulations based on the Drosophila melanogaster genome. Genetics 159, 581–588 (2001).
Hyne, V. & Kearsey, M. J. QTL analysis — further uses of marker regression. Theor. Appl. Genet. 91, 471–476 (1995).
Beavis, W. D. in Proceedings of the Corn and Sorghum Industry Research Conference 250–266 (American Seed Trade Association, Washington DC, 1994).
White, S. & Doebley, J. Of genes and genomes and the origin of maize. Trends Genet. 14, 327–332 (1998).
Doebley, J. & Stec, A. Genetic analysis of the morphological differences between maize and teosinte. Genetics 129, 285–295 (1991).
Dorweiler, J. et al. Teosinte glume architecture 1: a genetic locus controlling a key step in maize evolution. Science 262, 233–235 (1993).
Doebley, J., Stec, A. & Gustus, C. Teosinte branched 1 and the origin of maize: evidence for epistasis and the evolution of dominance. Genetics 141, 333–346 (1995).
Shrimpton, A. E. & Robertson, A. The isolation of polygenic factors controlling bristle score in Drosophila melanogaster. I. Allocation of third chromosome sternopleural bristle effect to chromosome sections. Genetics 118, 437–443 (1988).
Paterson, A. H. et al. Resolution of quantitative traits into Mendelian factors by using a complete linkage map of restriction fragment length polymorphisms. Nature 335, 721–726 (1988).
Darvasi, A. Experimental strategies for the genetic dissection of complex traits in animal models. Nature Genet. 18, 19–24 (1998).
Breese, E. L. & Mather, K. The organization of polygenic activity within a chromosome in Drosophila. 1. Hair characters. Heredity 11, 373–395 (1957).
Adams, M. D., Celniker, S. E. & Holt, R. A. The genome sequence of Drosophila melanogaster. Science 287, 2185–2195 (2000).
Lander, E. S. & Botstein, D. Mapping Mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics 121, 185–199 (1989).
Thoday, J. M. Location of polygenes. Nature 191, 368–370 (1961).
Yoo, B. H. Long-term selection for a quantitative character in large replicate populations of Drosophila melanogaster. I. Response to selection. Genet. Res. 35, 1–17 (1980).
Yoo, B. H. Long-term selection for a quantitative character in large replicate populations of Drosophila melanogaster. III. The nature of residual genetic variability. Theor. Appl. Genet. 57, 25–32 (1980).
Barton, N. H. The geometry of adaptation. Nature 395, 751–752 (1998).
Acknowledgements
We are grateful to the Biotechnology and Biological Sciences Research Council and the Royal Society for their support, and to W. Hill, T. Mackay, M. Slatkin, M. Turelli, B. Walsh and an anonymous referee for their helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Glossary
- QUANTITATIVE TRAIT LOCI
-
(QTL). Genetic loci identified through the statistical analysis of complex traits (such as plant height or body weight). These traits are typically affected by more than one gene and also by the environment.
- HERITABILITY
-
The fraction of the phenotypic variance due to additive genetic variance (VA/VP).
- ENVIRONMENTAL VARIANCE
-
The variance in the trait among genetically identical individuals. This variation might be due to the different environmental conditions experienced by different individuals, or to essentially random factors.
- GENETIC VARIANCE
-
The variance of trait values that can be ascribed to genetic differences between individuals.
- STABILIZING SELECTION
-
Intermediate phenotypes have greater fitness than extreme phenotypes.
- DIRECTIONAL SELECTION
-
Natural selection that acts to promote the fixation of a particular allele.
- DISRUPTIVE SELECTION
-
Intermediate phenotypes have lower fitness than extreme phenotypes; the opposite of stabilizing selection.
- INFINITESIMAL MODEL
-
A simple model of the inheritance of quantitative traits, which assumes an infinite number of unlinked loci, each with an infinitesimal effect.
- EFFECTIVE POPULATION SIZE
-
The size of the ideal population in which the effects of random drift would be the same as observed in the actual population.
- LINKAGE DISEQUILIBRIUM
-
The condition in which the frequency of a particular haplotype is significantly greater than that expected from the product of the observed allelic frequencies at each locus.
- SELECTIVE SWEEP
-
After the fixation of a new favourable mutation, the surrounding region of the genome is also fixed; neutral diversity is therefore 'swept' out of the population.
- BALANCER CHROMOSOME
-
Chromosome with recessive lethal mutations and inverted segments that suppress recombination.
- BALANCING SELECTION
-
Selection that acts to maintain two or more alleles in a population.
- OVERDOMINANCE
-
The phenotype of the heterozygote is greater than that of either homozygote. Overdominance for fitness can lead to the maintenance of both alleles in the population.
- DOMINANCE
-
A genetic interaction between the two alleles at a locus, such that the phenotype of heterozygotes deviates from the average of the two homozygotes.
- EPISTASIS
-
In the context of quantitative genetics, epistasis refers to any genetic interaction in which the combined phenotypic effect of two or more loci is less than (negative epistasis) or greater than (positive epistasis) the sum of effects at individual loci.
- DIRECTIONAL DOMINANCE
-
The phenotype of individuals that are heterozygous for the multiple loci that affect a trait deviates from the average of the phenotypes of homozygous individuals.
Rights and permissions
About this article
Cite this article
Barton, N., Keightley, P. Understanding quantitative genetic variation. Nat Rev Genet 3, 11–21 (2002). https://doi.org/10.1038/nrg700
Issue Date:
DOI: https://doi.org/10.1038/nrg700
This article is cited by
-
On the genetic architecture of rapidly adapting and convergent life history traits in guppies
Heredity (2022)
-
Parallel reduction in flowering time from de novo mutations enable evolutionary rescue in colonizing lineages
Nature Communications (2022)
-
Stress-induced changes in body surface temperature are repeatable, but do not differ between urban and rural birds
Oecologia (2022)
-
A proximal LAVA method for genome-wide association and prediction of traits with mixed inheritance patterns
BMC Bioinformatics (2021)
-
Opportunities and limits of combining microbiome and genome data for complex trait prediction
Genetics Selection Evolution (2021)