Key Points
-
The variation in the numbers of genes in different organisms indicates a general process of the origin and evolution of new genes.
-
Examining young genes is a direct approach to study this process, whereas ancient genes reveal the antiquity of some origination mechanisms.
-
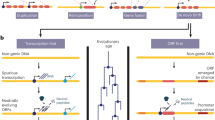
Several molecular mechanisms are involved in the creation of new gene structure, among which exon shuffling, retroposition and gene duplication have been found to be particularly important.
-
A new gene in its early stage usually undergoes rapid changes in sequence, structure and expression, which indicates a continuous evolution of function.
-
A significant role of positive Darwinian selection has been detected underlying these changes and adaptive evolution might have directed the entire origination process of new genes.
-
Direct and indirect observations of new genes in eukaryotic genomes show that genes with new functions are not as rare as was previously thought.
-
Analysis of the repeated new gene origination by retroposition in the Drosophila genome has uncovered a pattern in which new genes tend to avoid the X-chromosome linkage and most of the X-chromosome-derived autosomal new genes have evolved male-specific functions. This points to the importance of genome position in new gene origination.
Abstract
Genome data have revealed great variation in the numbers of genes in different organisms, which indicates that there is a fundamental process of genome evolution: the origin of new genes. However, there has been little opportunity to explore how genes with new functions originate and evolve. The study of ancient genes has highlighted the antiquity and general importance of some mechanisms of gene origination, and recent observations of young genes at early stages in their evolution have unveiled unexpected molecular and evolutionary processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gilbert, W. Why genes in pieces? Nature 271, 44 (1978).
van Rijk, A. & Bloemendal, H. Molecular mechanisms of exon shuffling: illegitimate recombination. Genetica 118, 245–249 (2003).
van Rijk, A. A., de Jong, W. W. & Bloemendal, H. Exon shuffling mimicked in cell culture. Proc. Natl Acad. Sci. USA 96, 8074–8079 (1999).
Moran, J. V., DeBerardinis, R. J. & Kazazian, H. H. Jr. Exon shuffling by L1 retrotransposition. Science 283, 1530–1534 (1999).
Esnault, C., Maestre, J. & Heidmann, T. Human LINE retrotransposons generate processed pseudogenes. Nature Genet. 24, 363–367 (2000).
Kaessmann, H., Zollner, S., Nekrutenko, A. & Li, W. H. Signatures of domain shuffling in the human genome. Genome Res. 12, 1642–1650 (2002). Based on analysis of human and C. elegans genomes, this paper provides new evidence that intron-phase correlation is a sign of exon shuffling that recombines protein-coding domains to form a new gene.
de Souza, S. J., Long, M., Schoenbach, L., Roy, S. W. & Gilbert, W. Intron positions correlate with module boundaries in ancient proteins. Proc. Natl Acad. Sci. USA 93, 14632–14636 (1996).
Patthy, L. Exon shuffling and other ways of module exchange. Matrix Biol. 15, 301–310, 311–312 (1996).
Ohno, S. Evolution by Gene Duplication (Springer, Berlin, 1970).
Kimura, M. The Neutral Theory of Molecular Evolution (Cambridge Univ. Press, Cambridge, 1983).
Prince, V. E. & Pickett, F. B. Splitting pairs: the diverging fates of duplicated genes. Nature Rev. Genet. 3, 827–837 (2002).
Bailey, J. A. et al. Recent segmental duplications in the human genome. Science 297, 1003–1007 (2002).
Samonte, R. V. & Eichler, E. E. Segmental duplications and the evolution of the primate genome. Nature Rev. Genet. 3, 65–72 (2002).
Makalowski, W., Mitchell, G. A. & Labuda, D. Alu sequences in the coding regions of mRNA: a source of protein variability. Trends Genet. 10, 188–193 (1994).
Makalowski, W. in The Impact of Short Interspersed Elements (SINEs) on the Host Genome (ed. Maraia, R. J.) 86–104 (Landes Company, Austin, 1995).
Nekrutenko, A. & Li, W. H. Transposable elements are found in a large number of human protein-coding genes. Trends Genet. 17, 619–621 (2001).
Lorenc, A. & Makalowski, W. Transposable elements and vertebrate protein diversity. Genetica 118, 467–477 (2003).
Ochman, H. Lateral and oblique gene transfer. Curr. Opin. Genet. Dev. 11, 616–619 (2001).
de Koning, A. P., Brinkman, F. S., Jones, S. J. & Keeling, P. J. Lateral gene transfer and metabolic adaptation in the human parasite Trichomonas vaginalis. Mol. Biol. Evol. 17, 1769–1773 (2000).
Bergthorsson, U., Adams, K. L., Thomason, B. & Palmer, J. D. Widespread horizontal transfer of mitochondrial genes in flowering plants. Nature 424, 197–201 (2003).
Thomson, T. M. et al. Fusion of the human gene for the polyubiquitination coeffector UEV1 with Kua, a newly identified gene. Genome Res. 10, 1743–1756 (2000).
Nurminsky, D. I., Nurminskaya, M. V., De Aguiar, D. & Hartl, D. L. Selective sweep of a newly evolved sperm-specific gene in Drosophila. Nature 396, 572–575 (1998). In this study, a young D. melanogaster gene showed unusually rapid changes in its structure, from seemingly unrelated resources, which led to unexpected new functions of the gene in sperm tails.
Wang, W., Brunet, F. G., Nevo, E. & Long, M. Origin of sphinx, a young chimeric RNA gene in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 99, 4448–4453 (2002).
Wang, W., Thornton, K., Berry, A. & Long, M. Nucleotide variation along the Drosophila melanogaster fourth chromosome. Science 295, 134–137 (2002).
Haldane, J. B. S. The part played by recurrent mutation in evolution. Am. Nat. 67, 5–19 (1933).
Fisher, R. A. The sheltering of lethals. Am. Nat. 69, 446–455 (1935).
Hughes, A. L. The evolution of functionally novel proteins after gene duplication. Proc. R. Soc. Lond. B 256, 119–124 (1994).
Force, A. et al. Preservation of duplicate genes by complementary, degenerative mutations. Genetics 151, 1531–1545 (1999).
Walsh, J. B. How often do duplicated genes evolve new functions? Genetics 139, 421–428 (1995).
Walsh, B. Population-genetic model of the fates of duplicate genes. Genetica 118, 279–294 (2003).
Gu, X. Maximum-likelihood approach for gene family evolution under functional divergence. Mol. Biol. Evol. 18, 453–464 (2001).
Long, M. & Langley, C. H. Natural selection and the origin of jingwei, a chimeric processed functional gene in Drosophila. Science 260, 91–95 (1993).
Messier, W. & Stewart, C. B. Episodic adaptive evolution of primate lysozymes. Nature 385, 151–154 (1997).
Zhang, J., Rosenberg, H. F. & Nei, M. Positive Darwinian selection after gene duplication in primate ribonuclease genes. Proc. Natl Acad. Sci. USA 95, 3708–3713 (1998).
Zhang, J., Zhang, Y. P. & Rosenberg, H. F. Adaptive evolution of a duplicated pancreatic ribonuclease gene in a leaf-eating monkey. Nature Genet. 30, 411–415 (2002). In this study, the authors combine sequence analysis and recombination protein technology to detect positive selection in the adaptive evolution of a new gene duplicate to the changed digestive system in the leaf-eating colobine monkey.
Ohta, T. Further examples of evolution by gene duplication revealed through DNA sequence comparisons. Genetics 138, 1331–1337 (1994).
Johnson, M. E. et al. Positive selection of a gene family during the emergence of humans and African apes. Nature 413, 514–519 (2001).
Begun, D. J. Origin and evolution of a new gene descended from alcohol dehydrogenase in Drosophila. Genetics 145, 375–382 (1997).
Maston, G. A. & Ruvolo, M. Chorionic gonadotropin has a recent origin within primates and an evolutionary history of selection. Mol. Biol. Evol. 19, 320–335 (2002). This paper discusses how an important function (involving pregnancy establishment in human and other primates) arose by gene duplication, supported by ample biological data.
Paulding, C. A., Ruvolo, M. & Haber, D. A. The Tre2 (USP6) oncogene is a hominoid-specific gene. Proc. Natl Acad. Sci. USA 100, 2507–2511 (2003).
Long, M., de Souza, S. J., Rosenberg, C. & Gilbert, W. Exon shuffling and the origin of the mitochondrial targeting function in plant cytochrome c1 precursor. Proc. Natl Acad. Sci. USA 93, 7727–7731 (1996).
Ranz, J. M., Ponce, A. R., Hartl, D. L. & Nurminsky, D. Origin and evolution of a new gene expressed in the Drosophila sperm axoneme. Genetica 118, 233–244 (2003).
Betrán, E., Wang, W., Jin, L. & Long, M. Evolution of the phosphoglycerate mutase processed gene in human and chimpanzee revealing the origin of a new primate gene. Mol. Biol. Evol. 19, 654–663 (2002).
Brown, C. J., Todd, K. M. & Rosenzweig, R. F. Multiple duplications of yeast hexose transport genes in response to selection in a glucose-limited environment. Mol. Biol. Evol. 15, 931–942 (1998). This study of experimental evolution shows that a new gene function can arise rapidly as a response to a changed environment.
Hall, B. G. The EBG system of E. coli: origin and evolution of a novel β-galactosidase for the metabolism of lactose. Genetica 118, 143–156 (2003).
McDonald, J. H. & Kreitman, M. Adaptative protein evolution at the Adh locus in. Drosophila. Nature 351, 652–654 (1991).
Henikoff, S., Ahmad, K. & Malik, H. S. The centromere paradox: stable inheritance with rapidly evolving DNA. Science 293, 1098–1102 (2001).
Malik, H. S. & Henikoff, S. Adaptive evolution of Cid, a centromere-specific histone in Drosophila. Genetics 157, 1293–1298 (2001).
Betrán, E. & Long, M. Dntf-2r: a young Drosophila retroposed gene with specific male expression under positive Darwinian selection. Genetics 164, 977–988 (2003).
Llopart, A., Comeron, J. M., Brunet, F. G., Lachaise, D. & Long, M. Intron presence–absence polymorphism in Drosophila driven by positive Darwinian selection. Proc. Natl Acad. Sci. USA 99, 8121–8126 (2002).
Cheng, C. H., Chen, L., Near, T. J. & Jin, Y. Functional antifreeze glycoprotein genes in temperate-water New Zealand nototheniid fish infer an Antarctic evolutionary origin. Mol. Biol. Evol. 28 July 2003 (doi:10.1093/molbev/msg004).
Chen, L., DeVries, A. L. & Cheng, C. H. Convergent evolution of antifreeze glycoproteins in Antarctic notothenioid fish and Arctic cod. Proc. Natl Acad. Sci. USA 94, 3817–3822 (1997). This study clearly showed how a similar environmental challenge in two different geographic locations created similar genes with the same function from different DNA materials.
Chen, L., DeVries, A. L. & Cheng, C. H. Evolution of antifreeze glycoprotein gene from a trypsinogen gene in Antarctic notothenioid fish. Proc. Natl Acad. Sci. USA 94, 3811–3816 (1997).
Cheng, C. H. & Chen, L. Evolution of an antifreeze glycoprotein. Nature 401, 443–444 (1999).
Thornton, K. & Long, M. Rapid divergence of gene duplicates on the Drosophila melanogaster X chromosome. Mol. Biol. Evol. 19, 918–925 (2002).
Charlesworth, B., Coyne, J. A. & Barton N. H. The relatives rates of evolution of sex chromosomes and autosomes. Am. Nat. 130, 113–146 (1987).
Orr, H. A. & Betancourt, A. J. Haldane's sieve and adaptation from the standing genetic variation. Genetics 157, 875–884 (2001).
Betancourt, A. J., Presgraves, D. C. & Swanson, W. J. A test for faster X evolution in Drosophila. Mol. Biol. Evol. 19, 1816–1819 (2002).
The Human Genome Sequencing Consortium. Initial sequencing and analysis of the human genome. Nature 409, 860–921 (2001).
Waterston, R. H. et al. Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002).
Betrán, E. & Long, M. Expansion of genome coding regions by acquisition of new genes. Genetica 115, 65–80 (2002).
Patthy, L. Protein Evolution by Exon-shuffling (Springer, New York, 1995).
Todd, A. E., Orengo, C. A. & Thornton, J. M. Evolution of function in protein superfamilies, from a structural perspective. J. Mol. Biol. 307, 1113–1143 (2001). A detailed structural and sequence analysis of 31 enzyme superfamilies for which structural data are available. Interestingly, almost all of the families were subject to domain shuffling, which highlights the generality of the mechanism in new gene origination.
Long, M., de Souza, S. J. & Gilbert, W. Evolution of the intron–exon structure of eukaryotic genes. Curr. Opin. Genet. Dev. 5, 774–778 (1995).
Long, M., Rosenberg, C. & Gilbert, W. Intron phase correlations and the evolution of the intron/exon structure of genes. Proc. Natl Acad. Sci. USA 92, 12495–12499 (1995).
Gilbert, W., de Souza, S. J. & Long, M. Origin of genes. Proc. Natl Acad. Sci. USA 94, 7698–7703 (1997).
Long, M. Evolution of novel genes. Curr. Opin. Genet. Dev. 11, 673–680 (2001).
Long, M., de Souza, S. J., Rosenberg, C. & Gilbert, W. Relationship between “proto-splice sites” and intron phases: evidence from dicodon analysis. Proc. Natl Acad. Sci. USA 95, 219–223 (1998).
Patthy, L. Intron-dependent evolution: preferred types of exons and introns. FEBS Lett. 214, 1–7 (1987).
Roy, S. W., Lewis, B. P., Fedorov, A. & Gilbert, W. Footprints of primordial introns on the eukaryotic genome. Trends Genet. 17, 496–501 (2001). The authors described an unexpectedly significant correlation between the distribution of intron phases and the age of the host genes, which supported a model of ancient exon–intron structure with gradual intron addition.
Fedorov, A., Roy, S., Cao, X. & Gilbert, W. Phylogenetically older introns strongly correlate with module boundaries in ancient proteins. Genome Res. 13, 1155–1157 (2003).
Venter, J. C. et al. The sequence of the human genome. Science 291, 1304–1351 (2001).
Deutsch, M. & Long, M. Intron–exon structures of eukaryotic model organisms. Nucleic Acids Res. 27, 3219–3228 (1999).
Long, M. & de Souza, S. J. Intron–exon structures: from molecular to population biology. Adv. Genome Biol. 5A, 143–178 (1998).
Kazazian, H. H. J. L1 retrotransposons shape the mammalian genome. Science 289, 1152–1153 (2000).
Pickeral, O. K., Makalowski, W., Boguski, M. S. & Boeke, J. D. Frequent human genomic DNA transduction driven by LINE-1 retrotransposition. Genome Res. 10, 411–415 (2000).
Harrison, P. M. et al. Molecular fossils in the human genome: identification and analysis of the pseudogenes in chromosomes 21 and 22. Genome Res. 12, 272–280 (2002).
Sorek, R., Ast, G. & Graur, D. Alu-containing exons are alternatively spliced. Genome Res. 12, 1060–1067 (2002).
Lifschytz, E. & Lindsley, D. L. The role of X-chromosome inactivation during spermatogenesis (Drosophila-allocycly-chromosome evolution-male sterility-dosage compensation). Proc. Natl Acad. Sci. USA 69, 182–186 (1972).
McCarrey, J. R. Nucleotide sequence of the promoter region of a tissue-specific human retroposon: comparison with its housekeeping progenitor. Gene 61, 291–298 (1987).
McCarrey, J. R. Molecular evolution of the human Pgk-2 retroposon. Nucleic Acids Res. 18, 949–955 (1990).
McCarrey, J. R. Evolution of tissue-specific gene expression in mammals: how a new phosphoglycerate kinase was formed and refined. BioScience 44, 20–27 (1994).
Pan, Y., Decker, W. K., Huq, A. H. & Craigen, W. J. Retrotransposition of glycerol kinase-related genes from the X chromosome to autosomes: functional and evolutionary aspects. Genomics 59, 282–290 (1999).
Elliott, D. J. et al. An evolutionarily conserved germ cell-specific hnRNP is encoded by a retrotransposed gene. Hum. Mol. Genet. 9, 2117–2124 (2000).
Betrán, E., Thornton, K. & Long, M. Retroposed new genes out of the X in Drosophila. Genome Res. 12, 1854–1859 (2002).
Kaminker, J. S. et al. The transposable elements of the Drosophila melanogaster euchromatin: a genomics perspective. Genome Biol. 3, 0084 (2002).
Swanson, W. J., Clark, A. G., Waldrip-Dail, H. M., Wolfner, M. F. & Aquadro, C. F. Evolutionary EST analysis identifies rapidly evolving male reproductive proteins in Drosophila. Proc. Natl Acad. Sci. USA 98, 7375–7379 (2001).
Avner, P. & Heard, E. X-chromosome inactivation: counting, choice and initiation. Nature Rev. Genet. 2, 59–67 (2001).
Franke, A. & Baker, B. S. Dosage compensation rox! Curr. Opin. Cell Biol. 12, 351–354 (2000).
Richler, C. et al. Splicing components are excluded from the transcriptionally inactive XY body in male meiotic nuclei. Mol. Biol. Cell 5, 1341–1352 (1994).
Wu, C -I. & Xu, E. Y. Sexual antagonism and X inactivation — the SAXI hypothesis. Trends Genet. 19, 243–247 (2003).
Wang, P. J., McCarrey, J. R., Yang, F. & Page, D. C. An abundance of X-linked genes expressed in spermatogonia. Nature Genet. 27, 422–426 (2001).
Rice, W. R. Sex chromosomes and the evolution of sexual dimorphism. Evolution 38, 735–742 (1984).
Parisi, M. et al. Paucity of genes on the Drosophila X chromosome showing male-biased expression. Science 299, 697–700 (2003).
Ranz, J. M., Castillo-Davis, C. I., Meiklejohn, C. D. & Hartl, D. L. Sex-dependent gene expression and evolution of the Drosophila transcriptome. Science 300, 1742–1745 (2003).
Reinke, V. et al. A global profile of germline gene expression in C. elegans. Mol. Cell 6, 605–616 (2000).
Kelly, W. G. et al. X-chromosome silencing in the germline of C. elegans. Development 129, 479–492 (2002).
Langley, C. H., Montgomery, E. & Quattlebaum, W. F. Restriction map variation in the Adh region of Drosophila. Proc. Natl Acad. Sci. USA 79, 5631–5635 (1982).
Jeffs, P. & Ashburner, M. Processed pseudogenes in Drosophila. Proc. R. Soc. Lond. B 244, 151–159 (1991).
Wang, W., Zhang, J., Alvarez, C., Llopart, A. & Long, M. The origin of the Jingwei gene and the complex modular structure of its parental gene, yellow emperor, in Drosophila melanogaster. Mol. Biol. Evol. 17, 1294–1301 (2000).
Long, M., Wang, W. & Zhang, J. Origin of new genes and source for N-terminal domain of the chimerical gene, jingwei, in Drosophila. Gene 238, 135–141 (1999).
Petrov, D. A., Lozovskaya, E. R. & Hartl, D. L. High intrinsic rate of DNA loss in Drosophila. Nature 384, 346–349 (1996).
Weiner, A. M., Deininger, P. L. & Efstratiadis, A. Nonviral retroposons: genes, pseudogenes, and transposable elements generated by the reverse flow of genetic information. Annu. Rev. Biochem. 55, 631–661 (1986).
Brosius, J. Retroposons — seeds of evolution. Science 251, 753 (1991).
Javaud, C., Dupuy, F., Maftah, A., Julien, R. & Petit, J -M. The fucosyltransferase gene family: an amazing summary of the underlying mechanisms of gene evolution. Genetica 118, 157–170 (2003).
Hughes, A. Adaptive Evolution of Genes and Genomes, (Oxford Univ. Press, Oxford, 2000).
Courseaux, A. & Nahon, J. L. Birth of two chimeric genes in the Hominidae lineage. Science 291, 1293–1297 (2001). This paper reported two chimeric genes — PMCHL1 and very young PMCHL2 — in Hominidae, which showed that an intron-containing gene structure can be retroposed to another genomic location.
Goodier, J. L., Ostertag, E. M. & Kazazian, H. H. Jr. Transduction of 3′-flanking sequences is common in L1 retrotransposition. Hum. Mol. Genet. 9, 653–657 (2000).
Brosius, J. RNAs from all categories generate retrosequences that may be exapted as novel genes or regulatory elements. Gene 238, 115–134 (1999).
Brosius, J. The contribution of RNAs and retroposition to evolutionary novelties. Genetica 118, 99–116 (2003).
Makalowski, W. Genomic scrap yard: how genomes utilize all that junk. Gene 259, 61–67 (2000).
Lorenc, A. & Makalowski, W. Transposable elements and vertebrate protein diversity. Genetica 118, 183–191 (2003).
Ragan, M. A. On surrogate methods for detecting lateral gene transfer. FEMS Microbiol. Lett. 201, 187–191 (2001).
McCarthy, A. D. & Hardie, D. G. Fatty acid synthase — an example of protein evolution by gene fusion. Trends Biochem. Sci. 4, 60–63 (1984).
Snel, B., Bork, P. & Huynen, M. Gene fusion versus gene fission. Trends Genet. 16, 9–11 (2000).
Martignetti, J. A. & Brosius, J. Neural BC1 RNA as an evolutionary marker: guinea pig remains a rodent. Proc. Natl Acad. Sci. USA 90, 9698–9702 (1993).
Martignetti, J. A. & Brosius, J. BC200 RNA: a neural RNA polymerase III product encoded by a monomeric Alu element. Proc. Natl Acad. Sci. USA 90, 11563–11567 (1993).
Zhang, J., Webb, D. M. & Podlaha, O. Accelerated protein evolution and origins of human-specific features: Foxp2 as an example. Genetics 162, 1825–1835 (2002).
Enard, W. et al. Molecular evolution of FOXP2, a gene involved in speech and language. Nature 418, 869–872 (2002).
Brosius, J. & Gould, S. J. On “genomenclature”: a comprehensive (and respectful) taxonomy for pseudogenes and other “junk DNA”. Proc. Natl Acad. Sci. USA 89, 10706–10710 (1992).
Gogolevskaya, I. K. & Kramerov, D. A. Evolutionary history of 4.5SI RNA and indication that it is functional. J. Mol. Evol. 54, 354–364 (2002).
Long, M. Protein-coding segments: evolution of exon–intron gene structure. Nature Encyclopedia of Life Sciences [online], <http://www.els.net> (doi:10.1038/npg.els.0000887) (2000).
Acknowledgements
We thank J. Sporfford, J. Zhang and the referees for their helpful suggestions. We also thank all members of the M.L. laboratory, past and present, for their devoted contributions to the studies on the origin of new genes and their evolution. M.L. is funded by a David and Lucile Packard Foundation Fellowship, a National Science Foundation CAREER Award and a National Institutes of Health grant.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
FlyBase
LocusLink
Saccharomyces Genome Database
FURTHER INFORMATION
Glossary
- NEW GENE
-
A gene that has originated recently in the relevant evolutionary timescale.
- ECTOPIC RECOMBINATION
-
Recombination between nonhomologous sequences.
- ILLEGITIMATE RECOMBINATION
-
Nonhomologous sequence recombination at the genomic DNA level.
- L1 RETRO-ELEMENT
-
A member of the long interspersed transposable element (LINE) family, which is a type of large repetitive DNA sequence that inserts itself throughout the genome through retroposition. L1 retro-elements are ∼6,400 base pairs long and are abundant in the human genome.
- ALU ELEMENT
-
An interspersed DNA sequence of ∼300 base pairs (bp) that is found in the genomes of primates, which can be cleaved by the restriction enzyme AluI. They are composed of a head-to-tail dimer, with the first monomer ∼140-bp long and the second ∼170-bp long. In humans, there are 300,000–600,000 copies of Alu elements.
- MOBILE ELEMENTS
-
Also known as transposable elements. DNA sequences in the genome that replicate and insert themselves into various positions in the genome.
- SUBFUNCTIONALIZATION
-
The process of 'partitioning' the ancestral functions of a locus among its duplicates. For example, if a single-copy gene that is normally expressed in two tissues subsequently duplicates, and each duplicate is then expressed in a different tissue, subfunctionalization has occurred.
- NEOFUNCTIONALIZATION
-
The evolution of a new function by a duplicate gene.
- PURIFYING SELECTION
-
Selection against deleterious alleles.
- REPLACEMENT CHANGES
-
(Substitutions). Changes in the nucleotide sequences of coding genes that result in changes in the peptide sequence (that is, the replacement of an amino acid). These contrast with silent (or synonymous) changes in coding sequences, which do not result in changes in the peptide.
- ADAPTIVE EVOLUTION
-
An evolutionary process that is directed by natural selection, which makes a population better adapted to live in an environment.
- KA/KS
-
KA is the rate of substitution at non-synonymous sites and KS is the rate of substitution at synonymous sites. The ratio between the two (KA/KS) is often used to infer selection: a KA/KS that is <1 indicates a functional constraint; a KA/KS that is equal to 1 indicates a lack of functional constraint; and a KA/KS that is >1 indicates positive Darwinian selection.
- POPULATION GENETIC ANALYSIS
-
The process of making inferences about the evolutionary and demographic history of a gene (or organism) on the basis of data on genetic variation in a species.
- SELECTIVE CONSTRAINT
-
A limit on evolutionary change.
- MCDONALD-KREITMAN TEST
-
A statistical test that is commonly used for the comparison of between-species divergence and within-species polymorphism at replacement and synonymous sites to infer adaptive protein evolution.
- CONVERGENT EVOLUTION
-
Independent evolution from different ancestors that leads to similar characteristics.
- NOTOTHENIOID
-
The most abundant group of Antarctic fish.
- TRYPSINOGEN
-
A large diverse protein family of serine peptidases.
- INTRON PHASE
-
The relative position of an intron within or between codons. Phase zero, one and two are defined by the position of an intron between two codons or after the first or second nucleotide of a codon, respectively.
- GERMLINE INACTIVATION
-
The early inactivation of the sex chromosomes in germline cells in the heterogametic sex.
- DOSAGE COMPENSATION
-
The phenomenon whereby the expression levels of sex-linked genes are made equal in males and females of heterogametic species.
- XIST TRANSCRIPT
-
A non-coding RNA that is transcribed by an X-linked gene known as Xist (X-inactive-specific transcription), which has a role in the somatic transcriptional inactivation of one X chromosome in female mammals. This is believed to occur through the interaction of transcripts from Xist and the related gene Tsix.
Rights and permissions
About this article
Cite this article
Long, M., Betrán, E., Thornton, K. et al. The origin of new genes: glimpses from the young and old. Nat Rev Genet 4, 865–875 (2003). https://doi.org/10.1038/nrg1204
Issue Date:
DOI: https://doi.org/10.1038/nrg1204
This article is cited by
-
Evolution of chemosensory tissues and cells across ecologically diverse Drosophilids
Nature Communications (2024)
-
Super-enhancers complexes zoom in transcription in cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
Identification of deep intronic variants of PAH in phenylketonuria using full-length gene sequencing
Orphanet Journal of Rare Diseases (2023)
-
De novo genes with an lncRNA origin encode unique human brain developmental functionality
Nature Ecology & Evolution (2023)
-
Orphan gene expressed in flame cone cells uniquely found in seahorse epithelium
Cell and Tissue Research (2023)