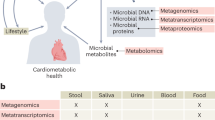
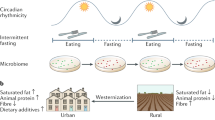
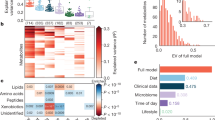
Abstract
Nutrigenomics is the application of high-throughput genomics tools in nutrition research. Applied wisely, it will promote an increased understanding of how nutrition influences metabolic pathways and homeostatic control, how this regulation is disturbed in the early phase of a diet-related disease and to what extent individual sensitizing genotypes contribute to such diseases. Ultimately, nutrigenomics will allow effective dietary-intervention strategies to recover normal homeostasis and to prevent diet-related diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
International Human Genome Sequencing Consortium. Initial sequencing and analysis of the human genome. Nature 409, 860–921 (2001).
Waterston, R. H. et al. Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002).
Venter, J. C. et al. The sequence of the human genome. Science 291, 1304–1351 (2001).
Francis, G. A., Fayard, E., Picard, F. & Auwerx, J. Nuclear receptors and the control of metabolism. Annu. Rev. Physiol. 65, 261–311 (2002).
Willett, W. C. Balancing life-style and genomics research for disease prevention. Science 296, 695–698 (2002).
Chuaqui, R. F. et al. Post-analysis follow-up and validation of microarray experiments. Nature Genet. 32 (Suppl.), 509–514 (2002).
Slonim, D. K. From patterns to pathways: gene expression data analysis comes of age. Nature Genet. 32 (Suppl.), 502–508 (2002).
Quackenbush, J. Microarray data normalization and transformation Nature Genet. 32 (Suppl.), 496–501 (2002).
Churchill, G. A. Fundamentals of experimental design for cDNA microarrays. Nature Genet. 32 (Suppl.), 490–495 (2002).
Stoeckert, C. J., Causton, H. C. & Ball, C. A. Microarray databases: standards and ontologies. Nature Genet. 32 (Suppl.), 469–473 (2002).
Roberts, M. A., Mutch, D. M. & German, J. B. Genomics: food and nutrition. Curr. Opin. Biotechnol. 12, 516–522 (2001).
Peregrin, T. The new frontier of nutrition science: nutrigenomics. J. Am. Diet Assoc. 101, 1306 (2001).
Elliott, R. & Ong, T. J. Nutritional genomics. BMJ 324, 1438–1442 (2002).
Daniel, H. Genomics and proteomics: importance for the future of nutrition research. Br. J. Nutr. 87 (Suppl.), 305–311 (2002).
van Ommen, B. & Stierum, R. Nutrigenomics: exploiting systems biology in the nutrition and health arena. Curr. Opin. Biotechnol. 13, 517–521 (2002).
Watkins, S. M., Reifsnyder, P. R., Pan, H. J., German, J. B. & Leiter, E. H. Lipid metabolome-wide effects of the PPAR-γ agonist rosiglitazone. J. Lipid Res. 43, 1809–1817 (2002).
MacBeath, G. Protein microarrays and proteomics. Nature Genet. 32 (Suppl.), 526–532 (2002).
Evans, W. E. & McLeod, H. L. Pharmacogenomics — drug disposition, drug targets, and side effects. N. Engl. J. Med. 348, 538–549 (2003).
Evans, W. E. & Johnson, J. A. Pharmacogenomics: the inherited basis for interindividual differences in drug response. Annu. Rev. Genomics Hum. Genet. 2, 9–39 (2001).
Brouwer, I. A., Zock, P. L., van Amelsvoort, L. G., Katan, M. B. & Schouten, E. G. Association between n-3 fatty acid status in blood and electrocardiographic predictors of arrhythmia risk in healthy volunteers. Am. J. Cardiol. 89, 629–631 (2002).
Sacks, F. M. & Katan, M. Randomized clinical trials on the effects of dietary fat and carbohydrate on plasma lipoproteins and cardiovascular disease. Am. J. Med. 113 (Suppl.), S13–S24 (2002).
Lu, T. T., Repa, J. J. & Mangelsdorf, D. J. Orphan nuclear receptors as eLiXiRs and FiXeRs of sterol metabolism. J. Biol. Chem. 276, 37735–37738 (2001).
Mangelsdorf, D. J. et al. The nuclear receptor superfamily: the second decade. Cell 83, 835–839 (1995).
Chawla, A., Repa, J. J., Evans, R. M. & Mangelsdorf, D. J. Nuclear receptors and lipid physiology: opening the X-files. Science 294, 1866–1870 (2001).
Jansen, P. L., Müller, M. & Sturm, E. Genes and cholestasis. Hepatology 34, 1067–1074 (2001).
Chiang, J. Y. Bile acid regulation of gene expression: roles of nuclear hormone receptors. Endocr. Rev. 23, 443–463 (2002).
Plass, J. R. et al. Farnesoid X receptor and bile salts are involved in transcriptional regulation of the gene encoding the human bile salt export pump. Hepatology 35, 589–596 (2002).
Pineda Torra, I. et al. Bile acids induce the expression of the human peroxisome proliferator-activated receptor-α gene via activation of the farnesoid X receptor. Mol. Endocrinol. 17, 259–272 (2003).
Ananthanarayanan, M., Balasubramanian, N., Makishima, M., Mangelsdorf, D. J. & Suchy, F. J. Human bile salt export pump promoter is transactivated by the farnesoid X receptor/bile acid receptor. J. Biol. Chem. 276, 28857–28865 (2001).
Hwang, S. T., Urizar, N. L., Moore, D. D. & Henning, S. J. Bile acids regulate the ontogenic expression of ileal bile acid binding protein in the rat via the farnesoid X receptor. Gastroenterology 122, 1483–1492 (2002).
Lu, T. T. et al. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol. Cell 6, 507–515 (2000).
He, K. et al. Fish consumption and risk of stroke in men. JAMA 288, 3130–3136 (2002).
Albert, C. M. et al. Blood levels of long-chain n-3 fatty acids and the risk of sudden death. N. Engl. J. Med. 346, 1113–1118 (2002).
Jump, D. B. & Clarke, S. D. Regulation of gene expression by dietary fat. Annu. Rev. Nutr. 19, 63–90 (1999).
Jump, D. B. Dietary polyunsaturated fatty acids and regulation of gene transcription. Curr. Opin. Lipidol. 13, 155–164 (2002).
Kersten, S., Desvergne, B. & Wahli, W. Roles of PPARs in health and disease. Nature 405, 421–424 (2000).
Barbier, O. et al. Pleiotropic actions of peroxisome proliferator-activated receptors in lipid metabolism and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 22, 717–726 (2002).
Walczak, R. & Tontonoz, P. PPARadigms and PPARadoxes: expanding roles for PPAR-γ in the control of lipid metabolism. J. Lipid. Res. 43, 177–186 (2002).
Xu, J. et al. Peroxisome proliferator-activated receptor-α (PPAR-α) influences substrate utilization for hepatic glucose production. J. Biol. Chem. 277, 50237–50244 (2002).
Kersten, S. et al. Peroxisome proliferator-activated receptor-α mediates the adaptive response to fasting. J. Clin. Invest. 103, 1489–1498 (1999).
Jump, D. B., Thelen, A. & Mater, M. Dietary polyunsaturated fatty acids and hepatic gene expression. Lipids 34 (Suppl.), S209–S212 (1999).
Desvergne, B. & Wahli, W. Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr. Rev. 20, 649–688 (1999).
Hooper, L. V. & Gordon, J. I. Commensal host-bacterial relationships in the gut. Science 292, 1115–1118 (2001).
Stappenbeck, T. S., Hooper, L. V., Manchester, J. K., Wong, M. H. & Gordon, J. I. Laser capture microdissection of mouse intestine: characterizing mRNA and protein expression, and profiling intermediary metabolism in specified cell populations. Methods Enzymol. 356, 167–196 (2002).
Shimomura, I., Shimano, H., Horton, J. D., Goldstein, J. L. & Brown, M. S. Differential expression of exons 1a and 1c in mRNAs for sterol regulatory element binding protein-1 in human and mouse organs and cultured cells. J. Clin. Invest. 99, 838–845 (1997).
Whitney, A. R. et al. Individuality and variation in gene expression patterns in human blood. Proc. Natl Acad. Sci. USA 100, 1896–1901 (2003).
Staudt, L. M. Gene expression profiling of lymphoid malignancies. Annu. Rev. Med. 53, 303–318 (2002).
Rosenwald, A. et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N. Engl. J. Med. 346, 1937–1947 (2002).
Davis, R. E. & Staudt, L. M. Molecular diagnosis of lymphoid malignancies by gene expression profiling. Curr. Opin. Hematol. 9, 333–338 (2002).
Tang, Y., Lu, A., Aronow, B. J. & Sharp, F. R. Blood genomic responses differ after stroke, seizures, hypoglycemia, and hypoxia: blood genomic fingerprints of disease. Ann. Neurol. 50, 699–707 (2001).
Boldrick, J. C. et al. Stereotyped and specific gene expression programs in human innate immune responses to bacteria. Proc. Natl Acad. Sci. USA 99, 972–977 (2002).
Staudt, L. M. Gene expression physiology and pathophysiology of the immune system. Trends Immunol. 22, 35–40 (2001).
Shaffer, A. L. et al. Signatures of the immune response. Immunity 15, 375–385 (2001).
Grody, W. W. Molecular genetic risk screening. Annu. Rev. Med. 54, 473–490 (2003).
Bailey, L. B. & Gregory, J. F. Polymorphisms of methylenetetrahydrofolate reductase and other enzymes: metabolic significance, risks and impact on folate requirement. J. Nutr. 129, 919–922 (1999).
Omer, R. E. et al. Peanut butter intake, GSTM1 genotype and hepatocellular carcinoma: a case-control study in Sudan. Cancer Causes Control 12, 23–32 (2001).
Sachidanandam, R. et al. A map of human genome sequence variation containing 1.42 million single nucleotide polymorphisms. Nature 409, 928–933 (2001).
Potter, J. D. At the interfaces of epidemiology, genetics and genomics. Nature Rev. Genet. 2, 142–147 (2001).
Kong, A. et al. A high-resolution recombination map of the human genome. Nature Genet. 31, 241–247 (2002).
Boomsma, D., Busjahn, A. & Peltonen, L. Classical twin studies and beyond. Nature Rev. Genet. 3, 872–882 (2002).
Cascante, M. et al. Metabolic control analysis in drug discovery and disease. Nature Biotechnol. 20, 243–249 (2002).
Ideker, T. et al. Integrated genomic and proteomic analyses of a systematically perturbed metabolic network. Science 292, 929–934 (2001).
Ideker, T., Galitski, T. & Hood, L. A new approach to decoding life: systems biology. Annu. Rev. Genomics Hum. Genet. 2, 343–372 (2001).
Jansen, R. C. Studying complex biological systems using multifactorial perturbation. Nature Rev. Genet. 4, 145–151 (2003).
Kitano, H. Systems biology: a brief overview. Science 295, 1662–1664 (2002).
Watkins, S. M. & German, J. B. Toward the implementation of metabolomic assessments of human health and nutrition. Curr. Opin. Biotechnol. 13, 512–516 (2002).
Ideker, T., Ozier, O., Schwikowski, B. & Siegel, A. F. Discovering regulatory and signalling circuits in molecular interaction networks. Bioinformatics 18 (Suppl.) 233–240 (2002).
Bradham, C. A., Plumpe, J., Manns, M. P., Brenner, D. A. & Trautwein, C. Mechanisms of hepatic toxicity. I. TNF-induced liver injury. Am. J. Physiol. 275, 387–392 (1998).
Streetz, K. L., Wustefeld, T., Klein, C., Manns, M. P. & Trautwein, C. Mediators of inflammation and acute phase response in the liver. Cell. Mol. Biol. 47, 661–673 (2001).
Diehl, A. M. Cytokine regulation of liver injury and repair. Immunol. Rev. 174, 160–171 (2000).
Ruminy, P. et al. Gene transcription in hepatocytes during the acute phase of a systemic inflammation: from transcription factors to target genes. Inflamm. Res. 50, 383–390 (2001).
Pineda Torra, I., Gervois, P. & Staels, B. Peroxisome proliferator-activated receptor-α in metabolic disease, inflammation, atherosclerosis and aging. Curr. Opin. Lipidol. 10, 151–159 (1999).
Streetz, K. et al. Tumor necrosis factor-α in the pathogenesis of human and murine fulminant hepatic failure. Gastroenterology 119, 446–460 (2000).
Clark, J. M., Brancati, F. L. & Diehl, A. M. Nonalcoholic fatty liver disease. Gastroenterology 122, 1649–1657 (2002).
Das, U. N. Is metabolic syndrome X an inflammatory condition? Exp. Biol. Med. 227, 989–997 (2002).
Evans, J. L., Goldfine, I. D., Maddux, B. A. & Grodsky, G. M. Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr. Rev. 23, 599–622 (2002).
Libby, P. Inflammation in atherosclerosis. Nature 420, 868–874 (2002).
Tilg, H. & Diehl, A. M. Cytokines in alcoholic and nonalcoholic steatohepatitis. N. Engl. J. Med. 343, 1467–1476 (2000).
Ren, B., Thelen, A. P., Peters, J. M., Gonzalez, F. J. & Jump, D. B. Polyunsaturated fatty acid suppression of hepatic fatty acid synthase and S14 gene expression does not require peroxisome proliferator-activated receptor-α. J. Biol. Chem. 272, 26827–26832 (1997).
Dallongeville, J. et al. Peroxisome proliferator-activated receptor-α is not rate-limiting for the lipoprotein-lowering action of fish oil. J. Biol. Chem. 276, 4634–4639 (2001).
Clagett-Dame, M. & DeLuca, H. F. The role of vitamin A in mammalian reproduction and embryonic development. Annu. Rev. Nutr. 22, 347–381 (2002).
Stanford, W. L., Cohn, J. B. & Cordes, S. P. Gene-trap mutagenesis: past, present and beyond. Nature Rev. Genet. 2, 756–768 (2001).
Copeland, N. G., Jenkins, N. A. & Court, D. L. Recombineering: a powerful new tool for mouse functional genomics. Nature Rev. Genet. 2, 769–779 (2001).
Lewandoski, M. Conditional control of gene expression in the mouse. Nature Rev. Genet. 2, 743–755 (2001).
Weindruch, R., Kayo, T., Lee, C. K. & Prolla, T. A. Gene expression profiling of aging using DNA microarrays. Mech. Ageing Dev. 123, 177–193 (2002).
Cao, S. X., Dhahbi, J. M., Mote, P. L. & Spindler, S. R. Genomic profiling of short- and long-term caloric restriction effects in the liver of aging mice. Proc. Natl Acad. Sci. USA 98, 10630–10635 (2001).
Lee, C. K., Allison, D. B., Brand, J., Weindruch, R. & Prolla, T. A. Transcriptional profiles associated with aging and middle age-onset caloric restriction in mouse hearts. Proc. Natl Acad. Sci. USA 99, 14988–14993 (2002).
Prolla, T. A. DNA microarray analysis of the aging brain. Chem. Senses 27, 299–306 (2002).
Sreekumar, R., Halvatsiotis, P., Schimke, J. C. & Nair, K. S. Gene expression profile in skeletal muscle of type 2 diabetes and the effect of insulin treatment. Diabetes 51, 1913–1920 (2002).
Maier, S. & Olek, A. Diabetes: a candidate disease for efficient DNA methylation profiling. J. Nutr. 132, 2440–2443 (2002).
Shalev, A. et al. Oligonucleotide microarray analysis of intact human pancreatic islets: identification of glucose-responsive genes and a highly regulated TGFβ signaling pathway. Endocrinology 143, 3695–3698 (2002).
Shih, D. Q. et al. Hepatocyte nuclear factor-1α is an essential regulator of bile acid and plasma cholesterol metabolism. Nature Genet. 27, 375–382 (2001).
Naiki, T. et al. Analysis of gene expression profile induced by hepatocyte nuclear factor 4α in hepatoma cells using an oligonucleotide microarray. J. Biol. Chem. 277, 14011–14019 (2002).
Ross, S. E. et al. Microarray analyses during adipogenesis: understanding the effects of Wnt signaling on adipogenesis and the roles of liver X receptor-α in adipocyte metabolism. Mol. Cell. Biol. 22, 5989–5999 (2002).
Kersten, S. et al. The peroxisome proliferator-activated receptor-α regulates amino acid metabolism. FASEB J. 15, 1971–1978 (2001).
Lichtlen, P. et al. Target gene search for the metal-responsive transcription factor MTF-1. Nucleic Acids Res. 29, 1514–1523 (2001).
Blanchard, R. K., Moore, J. B., Green, C. L. & Cousins, R. J. Modulation of intestinal gene expression by dietary zinc status: effectiveness of cDNA arrays for expression profiling of a single nutrient deficiency. Proc. Natl Acad. Sci. USA 98, 13507–13513 (2001).
Xiao, J. et al. The effect of chronic exposure to fatty acids on gene expression in clonal insulin-producing cells: studies using high density oligonucleotide microarray. Endocrinology 142, 4777–4784 (2001).
Endo, Y., Fu, Z., Abe, K., Arai, S. & Kato, H. Dietary protein quantity and quality affect rat hepatic gene expression. J. Nutr. 132, 3632–3637 (2002).
Mariadason, J. M., Corner, G. A. & Augenlicht, L. H. Genetic reprogramming in pathways of colonic cell maturation induced by short chain fatty acids: comparison with trichostatin A, sulindac, and curcumin and implications for chemoprevention of colon cancer. Cancer Res. 60, 4561–4572 (2000).
Acknowledgements
The authors thank their colleagues from the Division of Human Nutrition, the Centre of Human Nutrigenomics and the Innovative Cluster Nutrigenomics group for critical discussions. The work of the authors is supported by the Dutch Scientific Organization (NOW), the Dutch Diabetes Foundation, the Wageningen Centre of Food Sciences, the Dutch Dairy Foundation for Nutrition and Health, the Innovative Research Programme (IOP) Genomics and the Food Technology, Agrobiotechnology, Nutrition and Health Sciences (VLAG) research school.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- ACUTE-PHASE RESPONSE
-
The early and immediate set of homeostatic control reactions that are induced during inflammation.
- BILE CANALICULUS
-
A half tubule that is formed by the apical membranes of two hepatocytes, and is limited laterally by their smooth surfaces.
- CANALICULAR MEMBRANE
-
The apical membrane of liver epithelial cells (hepatocytes) that lines the bile canaliculus. Members of the ABC-transporter superfamily that are localized in this membrane are responsible for bile secretion.
- HEPATOCYTES
-
Epithelial cells that are the main functional units of the liver, and comprise 80% of the organ's cytoplasmic mass.
- INDUCIBLE EXPRESSION SYSTEMS
-
Expression systems that regulate mammalian gene expression with, for example, tetracycline or its derivatives (Tet-On/Tet-Off gene expression systems).
- INFLAMMATION
-
The complex series of reactions that occur in the host as a response to injury, trauma or infection of a tissue, which prevent ongoing tissue damage, isolate and destroy the infective organism and activate the repair processes that are necessary to return the organism to normal function.
- KETOGENESIS
-
The production of ketone bodies — such as acetoacetate and β-hydroxybutyrate — which are the intermediate products of fatty-acid catabolism and can be used to provide energy.
- LASER CAPTURE MICRODISSECTION
-
A method in which cells are cut out from a tissue sample using a laser beam, allowing single cell expression analysis.
- LYMPHOCYTE
-
A type of white blood cell that is responsible for the adaptive immune response; for example, B lymphocytes and T lymphocytes.
- MACRONUTRIENTS
-
Organic compounds, including proteins, amino acids, carbohydrates and lipids, that are required in large amounts in the diet.
- METABOLOMICS
-
The study of the metabolome, which is the entire metabolic content of a cell or organism, at a given time.
- MICRONUTRIENTS
-
Dietary compounds, including vitamins and minerals that are required in small amounts in the diet.
- NUTRIGENETICS
-
The relationship between genotype and the risk of developing diet-related diseases, such as cancer, diabetes type II and cardio-vascular diseases.
- NUTRIGENOMICS
-
The study of the genome-wide influences of nutrition or dietary components on the transcriptome, proteome and metabolome, of cells, tissues or organisms, at a given time.
- PHARMACOGENOMICS
-
A term often used to mean the influence of DNA-sequence variation — in drug targets, Phase I or Phase II drug-metabolizing enzymes, and transporters — on the effect of a drug, which ultimately allows physicians to design individualized therapy.
- PROTEOMICS
-
The study of proteomes (the complete collection of proteins in a cell or tissue at a given time), which attempts to determine their role inside cells and the molecules with which they interact.
- RNA INTERFERENCE
-
(RNAi). The process by which double-stranded RNA silences homologous genes.
- SATURATION
-
The binding state of a C–C bond in a fatty acid molecule.
- SYSTEMS BIOLOGY
-
The study of whole biological systems (cells, tissues and organisms) using holistic methods.
- TRANSCRIPTOME
-
The complete collection of gene transcripts in a cell or a tissue at a given time.
- TRANSDOMINANT NEGATIVE ADENOVIRAL CONSTRUCT
-
A recombinant adenovirus that infects cells, resulting in the high-level expression of a mutant protein that, for example, specifically blocks a given signalling pathway (superrepressor) by competing with the endogenous protein.
Rights and permissions
About this article
Cite this article
Müller, M., Kersten, S. Nutrigenomics: goals and strategies. Nat Rev Genet 4, 315–322 (2003). https://doi.org/10.1038/nrg1047
Issue Date:
DOI: https://doi.org/10.1038/nrg1047
This article is cited by
-
Emergence of nutrigenomics and dietary components as a complementary therapy in cancer prevention
Environmental Science and Pollution Research (2022)
-
Personalized nutrition approach in pediatrics: a narrative review
Pediatric Research (2021)
-
Nutritional programming in fishes: insights from mammalian studies
Reviews in Fish Biology and Fisheries (2020)
-
Effect of the consumption of natural products on some biochemical parameters of a type 2 diabetic population
Journal of Diabetes & Metabolic Disorders (2020)