Key Points
-
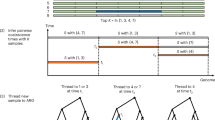
The inheritance of identical haplotypes from a common ancestor creates long tracts of homozygous genotypes known as runs of homozygosity (ROH).
-
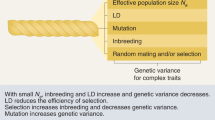
ROH are ubiquitous across human populations, and they correlate with pedigree inbreeding. Larger populations have fewer, shorter ROH, whereas isolated or bottlenecked populations have more, somewhat longer ROH. Admixed groups have the fewest ROH, whereas consanguineous communities carry very long ROH. Native American populations have the highest burdens of ROH in the world.
-
ROH can be detected in microarray or whole-genome sequencing (WGS) data, using either observational approaches, for example, that implemented in PLINK, or model-based approaches. Simulations show that PLINK outperforms many other methods.
-
ROH are non-randomly distributed across the genome, being more prevalent in areas of low recombination, but are also concentrated in small regions called ROH islands.
-
Quantitative traits related to stature and cognition have been robustly associated with ROH burden, implying recessive variants contribute to their genetic architecture. Case–control analyses of ROH, on the other hand, appear more easily confounded by socioeconomic or cultural factors.
-
Both megacohorts and special populations are now being used to investigate diverse aspects of the scope and mechanism of inbreeding depression in humans.
Abstract
Long runs of homozygosity (ROH) arise when identical haplotypes are inherited from each parent and thus a long tract of genotypes is homozygous. Cousin marriage or inbreeding gives rise to such autozygosity; however, genome-wide data reveal that ROH are universally common in human genomes even among outbred individuals. The number and length of ROH reflect individual demographic history, while the homozygosity burden can be used to investigate the genetic architecture of complex disease. We discuss how to identify ROH in genome-wide microarray and sequence data, their distribution in human populations and their application to the understanding of inbreeding depression and disease risk.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cavalli-Sforza, L. L. & Bodmer, W. The Genetics of Human Populations (W. H. Freeman & Co Ltd, 1978).
Bittles, A. H. & Black, M. L. Consanguinity, human evolution, and complex diseases. Proc. Natl Acad. Sci. USA 107, 1779–1786 (2010).
Broman, K. W. & Weber, J. L. Long homozygous chromosomal segments in reference families from the centre d'Etude du polymorphisme humain. Am. J. Hum. Genet. 65, 1493–1500 (1999). This seminal study is the first to identify long ROH, showing they are common in humans.
Jones, C. M. R. M. Atlas of World Population History. (Facts On File, 1978).
Biraben, J.-N. An essay concerning mankind's demographic evolution. J. Hum. Evol. 9, 655–663 (1980).
Gunderson, R. C. Connecting Your Pedigree Into Royal, Noble and Medieval Families. (Genealogical Society of Utah, 1980).
Keller, M. C., Visscher, P. M. & Goddard, M. E. Quantification of inbreeding due to distant ancestors and its detection using dense single nucleotide polymorphism data. Genetics 189, 237–249 (2011). This paper shows that F ROH is the preferred genomic inbreeding measure and that sample sizes in the tens of thousands will be needed to detect inbreeding depression in humans.
Donnelly, K. P. The probability that related individuals share some section of genome identical by descent. Theor. Popul. Biol. 23, 34–63 (1983).
Rohde, D. L., Olson, S. & Chang, J. T. Modelling the recent common ancestry of all living humans. Nature 431, 562–566 (2004).
Garrod, A. E. The incidence of alkaptonuria: a study in chemical individuality. Lancet Infect. Dis. 2, 1616–1620 (1902).
Hoffman, J. I. et al. High-throughput sequencing reveals inbreeding depression in a natural population. Proc. Natl Acad. Sci. USA 111, 3775–3780 (2014).
Huisman, J., Kruuk, L. E., Ellis, P. A., Clutton-Brock, T. & Pemberton, J. M. Inbreeding depression across the lifespan in a wild mammal population. Proc. Natl Acad. Sci. USA 113, 3585–3590 (2016). Using a well-studied wild deer population from Scotland with genomic data, this paper finds effects of homozygosity on offspring survival, birth weight, juvenile survival and other components of fitness.
Charlesworth, D. & Willis, J. H. The genetics of inbreeding depression. Nat. Rev. Genet. 10, 783–796 (2009).
Szpiech, Z. A. et al. Long runs of homozygosity are enriched for deleterious variation. Am. J. Hum. Genet. 93, 90–102 (2013).
Alsalem, A. B., Halees, A. S., Anazi, S., Alshamekh, S. & Alkuraya, F. S. Autozygome sequencing expands the horizon of human knockout research and provides novel insights into human phenotypic variation. PLoS Genet. 9, e1004030 (2013).
Xue, Y. et al. Mountain gorilla genomes reveal the impact of long-term population decline and inbreeding. Science 348, 242–245 (2015).
Scott, E. M. et al. Characterization of Greater Middle Eastern genetic variation for enhanced disease gene discovery. Nat. Genet. 48, 1071–1076 (2016).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Pemberton, T. J. et al. Genomic patterns of homozygosity in worldwide human populations. Am. J. Hum. Genet. 91, 275–292 (2012).
Joshi, P. K. et al. Directional dominance on stature and cognition in diverse human populations. Nature 523, 459–462 (2015). This paper is the largest study of ROH to date and found robust evidence for inbreeding effects on cognition and height-related traits in many populations across the world.
Gusev, A. et al. Whole population, genome-wide mapping of hidden relatedness. Genom. Res. 19, 318–326 (2009).
Browning, S. R. & Browning, B. L. High-resolution detection of identity by descent in unrelated individuals. Am. J. Hum. Genet. 86, 526–539 (2010).
Howrigan, D. P., Simonson, M. A. & Keller, M. C. Detecting autozygosity through runs of homozygosity: a comparison of three autozygosity detection algorithms. BMC Genomics 12, 460 (2011). This paper uses both simulated and real data to show that PLINK outperformed other software for the detection of ROH.
Szpiech, Z. A., Blant, A. & Pemberton, T. J. GARLIC: Genomic Autozygosity Regions Likelihood-based Inference and Classification. Bioinformatics 33, 2059–2062 (2017).
Zhuang, Z., Gusev, A., Cho, J. & Pe'er, I. Detecting identity by descent and homozygosity mapping in whole-exome sequencing data. PLoS ONE 7, e47618 (2012).
Pippucci, T., Magi, A., Gialluisi, A. & Romeo, G. Detection of runs of homozygosity from whole exome sequencing data: state of the art and perspectives for clinical, population and epidemiological studies. Hum. Hered. 77, 63–72 (2014).
Magi, A. et al. H3M2: detection of runs of homozygosity from whole-exome sequencing data. Bioinformatics 30, 2852–2859 (2014).
Narasimhan, V. et al. BCFtools/RoH: a hidden Markov model approach for detecting autozygosity from next-generation sequencing data. Bioinformatics 32, 1749–1751 (2016).
Gibson, J., Morton, N. E. & Collins, A. Extended tracts of homozygosity in outbred human populations. Hum. Mol. Genet. 15, 789–795 (2006). This seminal paper demonstrates that ROH are ubiquitous in human populations.
Simon-Sanchez, J. et al. Genome-wide SNP assay reveals structural genomic variation, extended homozygosity and cell-line induced alterations in normal individuals. Hum. Mol. Genet. 16, 1–14 (2007).
Lencz, T. et al. Runs of homozygosity reveal highly penetrant recessive loci in schizophrenia. Proc. Natl Acad. Sci. USA 104, 19942–19947 (2007).
Li, L. H. et al. Long contiguous stretches of homozygosity in the human genome. Hum. Mutat. 27, 1115–1121 (2006).
International HapMap, C. et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 449, 851–861 (2007).
Curtis, D. Extended homozygosity is not usually due to cytogenetic abnormality. BMC Genet. 8, 67 (2007).
McQuillan, R. et al. Runs of homozygosity in European populations. Am. J. Hum. Genet. 83, 359–372 (2008). Using well-studied isolate populations, this paper shows a strong correlation of genomic and pedigree inbreeding coefficients and that outbred individuals could harbour ROH up to 4 Mb in length.
Wright, S. Coefficients of Inbreeding and relationship. Amer. Naturalist 56, 330–338 (1922).
Woods, C. G. et al. Quantification of homozygosity in consanguineous individuals with autosomal recessive disease. Am. J. Hum. Genet. 78, 889–896 (2006).
Kirin, M. et al. Genomic runs of homozygosity record population history and consanguinity. PLoS ONE 5, e13996 (2010). This survey of ROH across different populations, continents and demographic histories allows classification of populations into four major groups in terms of their ROH burden.
Karafet, T. M. et al. Extensive genome-wide autozygosity in the population isolates of Daghestan. Europ. J. Hum. Genet. 23, 1405–1412 (2015).
Mezzavilla, M. et al. Increased rate of deleterious variants in long runs of homozygosity of an inbred population from Qatar. Hum. Hered. 79, 14–19 (2015).
Narasimhan, V. M. et al. Health and population effects of rare gene knockouts in adult humans with related parents. Science 352, 474–477 (2016). This first large survey of gene knockouts in a consanguineous population describes homozygous loss of function for hundreds of genes.
Jones, E. R. et al. Upper Palaeolithic genomes reveal deep roots of modern Eurasians. Nat. Commun. 6, 8912 (2015).
Waldman, Y. Y. et al. The genetic history of Cochin Jews from India. Hum. Genet. 135, 1127–1143 (2016).
Gilbert, E., Carmi, S., Ennis, S., Wilson, J. F. & Cavalleri, G. L. Genomic insights into the population structure and history of the Irish Travellers. Sci. Rep. 7, 42187 (2017).
Esko, T. et al. Genetic characterization of northeastern Italian population isolates in the context of broader European genetic diversity. Europ. J. Hum. Genet. 21, 659–665 (2013).
Bryc, K. et al. Colloquium paper: genome-wide patterns of population structure and admixture among Hispanic/Latino populations. Proc. Natl Acad. Sci. USA 107 (Suppl. 2), 8954–8961 (2010).
Abdellaoui, A. et al. Association between autozygosity and major depression: stratification due to religious assortment. Behav. Genet. 43, 455–467 (2013).
Abdellaoui, A. et al. Educational attainment influences levels of homozygosity through migration and assortative mating. PLoS ONE 10, e0118935 (2015). This study is a great example of how confounding effects, such as assortative mating, can influence ROH analyses.
Nalls, M. A. et al. Measures of autozygosity in decline: globalization, urbanization, and its implications for medical genetics. PLoS Genet. 5, e1000415 (2009).
Higasa, K. et al. Evaluation of haplotype inference using definitive haplotype data obtained from complete hydatidiform moles, and its significance for the analyses of positively selected regions. PLoS Genet. 5, e1000468 (2009).
Papenhausen, P. et al. UPD detection using homozygosity profiling with a SNP genotyping microarray. Am. J. Med. Genet. A 155A, 757–768 (2011).
Schaaf, C. P. et al. Identification of incestuous parental relationships by SNP-based DNA microarrays. Lancet 377, 555–556 (2011).
Sund, K. L. et al. Regions of homozygosity identified by SNP microarray analysis aid in the diagnosis of autosomal recessive disease and incidentally detect parental blood relationships. Genet. Med. 15, 70–78 (2013).
Grote, L. et al. Variability in laboratory reporting practices for regions of homozygosity indicating parental relatedness as identified by SNP microarray testing. Genet. Med. 14, 971–976 (2012).
Hawass, Z. et al. Ancestry and pathology in King Tutankhamun's family. J. Am. Med. Assoc. 303, 638–647 (2010).
Leutenegger, A. L., Sahbatou, M., Gazal, S., Cann, H. & Genin, E. Consanguinity around the world: what do the genomic data of the HGDP-CEPH diversity panel tell us? Europ. J. Hum. Genet. 19, 583–587 (2011).
Jalkh, N. et al. Genome-wide inbreeding estimation within Lebanese communities using SNP arrays. Europ. J. Hum. Genet. 23, 1364–1369 (2015).
Curtis, D., Vine, A. E. & Knight, J. Study of regions of extended homozygosity provides a powerful method to explore haplotype structure of human populations. Ann. Hum. Genet. 72, 261–278 (2008).
Nothnagel, M. et al. Genomic and geographic distribution of SNP-defined runs of homozygosity in Europeans. Hum. Mol. Genet. 19, 2927–2935 (2010). This is the first study to perform in-depth analysis of ROH islands, regions of the genome where a high proportion of people are homozygous.
Lander, E. S. & Botstein, D. Homozygosity mapping — a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Rudan, I., Campbell, H., Carothers, A. D., Hastie, N. D. & Wright, A. F. Contribution of consanguinuity to polygenic and multifactorial diseases. Nat. Genet. 38, 1224–1225 (2006).
Keller, M. C. et al. Runs of homozygosity implicate autozygosity as a schizophrenia risk factor. PLoS Genet. 8, e1002656 (2012).
Johnson, E. C. et al. No reliable association between runs of homozygosity and schizophrenia in a well-powered replication study. PLoS Genet. 12, e1006343 (2016).
Panagiotou, O. A., Willer, C. J., Hirschhorn, J. N. & Ioannidis, J. P. The power of meta-analysis in genome-wide association studies. Annu. Rev. Genom. Hum. Genet. 14, 441–465 (2013).
Power, R. A. et al. A recessive genetic model and runs of homozygosity in major depressive disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 165B, 157–166 (2014).
McQuillan, R. et al. Evidence of inbreeding depression on human height. PLoS Genet. 8, e1002655 (2012).
Howrigan, D. P. et al. Genome-wide autozygosity is associated with lower general cognitive ability. Mol. Psychiatry 21, 837–843 (2016).
Morton, N. E. Effects of inbreeding on IQ and mental retardation. Proc. Natl Acad. Sci. USA 75, 3906–3908 (1978).
Schull, W. J. Inbreeding and maternal effects in the Japanese. Eugen. Q. 9, 14–22 (1962).
Rudan, I. et al. Quantifying the increase in average human heterozygosity due to urbanisation. Europ. J. Hum. Genet. 16, 1097–1102 (2008).
Yang, H. C., Chang, L. C., Liang, Y. J., Lin, C. H. & Wang, P. L. A genome-wide homozygosity association study identifies runs of homozygosity associated with rheumatoid arthritis in the human major histocompatibility complex. PLoS ONE 7, e34840 (2012).
Yang, T. L. et al. Genome-wide survey of runs of homozygosity identifies recessive loci for bone mineral density in Caucasian and Chinese populations. J. Bone Miner. Res. 30, 2119–2126 (2015).
Kaiser, V. B. et al. Homozygous loss-of-function variants in European cosmopolitan and isolate populations. Hum. Mol. Genet. 24, 5464–5474 (2015).
Lim, E. T. et al. Distribution and medical impact of loss-of-function variants in the Finnish founder population. PLoS Genet. 10, e1004494 (2014).
Saleheen, D. et al. Human knockouts and phenotypic analysis in a cohort with a high rate of consanguinity. Nature 544, 235–239 (2017).
Sudlow, C. et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Gaziano, J. M. et al. Million Veteran Program: a mega-biobank to study genetic influences on health and disease. J. Clin. Epidemiol. 70, 214–223 (2016).
Nagai, A. et al. Overview of the BioBank Japan project: study design and profile. J. Epidemiol. 27, S2–S8 (2017).
Chen, Z. et al. China Kadoorie Biobank of 0.5 million people: survey methods, baseline characteristics and long-term follow-up. Int. J. Epidemiol. 40, 1652–1666 (2011).
Ebel, E. R. & Phillips, P. C. Intrinsic differences between males and females determine sex-specific consequences of inbreeding. BMC Evol. Biol. 16, 36 (2016).
Darwin, C. R. The Variation of Animals and Plants Under Domestication. (John Murray, 1868).
Darwin, C. R. The Effects of Cross and Self Fertilisation in the Vegetable Kingdom. (John Murray, 1876).
Berra, T. M. Darwin & His Children: His Other Legacy. (Oxford Univ. Press, 2013).
Berra, T. M., Alvarez, G. & Ceballos, F. C. Was the Darwin/Wedgwood dynasty adversely affected by consanguinity? Bioscience 60, 376–383 (2010).
Alvarez, G., Ceballos, F. C. & Berra, T. M. Darwin was right: inbreeding depression on male fertility in the Darwin family. Biol. J. Linn. Soc. 114, 474–483 (2015).
Ober, C., Hyslop, T. & Hauck, W. W. Inbreeding effects on fertility in humans: evidence for reproductive compensation. Am. J. Hum. Genet. 64, 225–231 (1999).
Bittles, A. H. & Neel, J. V. The costs of human inbreeding and their implications for variations at the DNA level. Nat. Genet. 8, 117–121 (1994).
Ceballos, F. C. & Alvarez, G. Royal dynasties as human inbreeding laboratories: the Habsburgs. Heredity 111, 114–121 (2013).
Alvarez, G. & Ceballos, F. C. Royal inbreeding and the extinction of lineages of the Habsburg dynasty. Hum. Hered. 80, 62–68 (2015).
Alvarez, G., Ceballos, F. C. & Quinteiro, C. The role of inbreeding in the extinction of a European royal dynasty. PLoS ONE 4, e5174 (2009).
Cassidy, L. M. et al. Neolithic and Bronze Age migration to Ireland and establishment of the insular Atlantic genome. Proc. Natl Acad. Sci. USA 113, 368–373 (2016).
Gamba, C. et al. Genome flux and stasis in a five millennium transect of European prehistory. Nat. Commun. 5, 5257 (2014).
Kılınç, G. M. et al. The demographic development of the first farmers in Anatolia. Curr. Biol. 26, 2659–2666 (2016).
Prüfer, K. et al. The complete genome sequence of a Neanderthal from the Altai Mountains. Nature 505, 43–49 (2014).
Kuhlwilm, M. et al. Ancient gene flow from early modern humans into Eastern Neanderthals. Nature 530, 429–433 (2016).
Prado-Martinez, J. et al. Great ape genetic diversity and population history. Nature 499, 471–475 (2013).
Callaway, E. DNA mutation clock proves tough to set. Nature 519, 139–140 (2015).
Kong, A. et al. Rate of de novo mutations and the importance of father's age to disease risk. Nature 488, 471–475 (2012).
Campbell, C. D. et al. Estimating the human mutation rate using autozygosity in a founder population. Nat. Genet. 44, 1277–1281 (2012).
Alkuraya, F. S. Autozygome decoded. Genet. Med. 12, 765–771 (2010).
Narasimhan, V. M. et al. in 65th Annual Meeting of The American Society of Human Genetics PgmNr 353 (Baltimore, MD, 2015).
Lipson, M. et al. Calibrating the human mutation rate via ancestral recombination density in diploid genomes. PLoS Genet. 11, e1005550 (2015).
Palamara, P. F., Lencz, T., Darvasi, A. & Pe'er, I. Length distributions of identity by descent reveal fine-scale demographic history. Am. J. Hum. Genet. 91, 809–822 (2012).
The 1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012).
Behar, D. M. et al. The genome-wide structure of the Jewish people. Nature 466, 238–U112 (2010).
Busby, G. B. J. et al. The role of recent admixture in forming the contemporary west Eurasian genomic landscape. Curr. Biol. 25, 2518–2526 (2015).
Henn, B. M. et al. Hunter-gatherer genomic diversity suggests a southern African origin for modern humans. Proc. Natl Acad. Sci. USA 108, 5154–5162 (2011).
Li, J. Z. et al. Worldwide human relationships inferred from genome-wide patterns of variation. Science 319, 1100–1104 (2008).
Hellenthal, G. et al. A genetic atlas of human admixture history. Science 343, 747–751 (2014).
Metspalu, M. et al. Shared and unique components of human population structure and genome-wide signals of positive selection in South Asia. Am. J. Hum. Genet. 89, 731–744 (2011).
Pagani, L. et al. Ethiopian genetic diversity reveals linguistic stratification and complex influences on the Ethiopian gene pool. Am. J. Hum. Genet. 91, 83–96 (2012).
Rasmussen, M. et al. Ancient human genome sequence of an extinct Palaeo-Eskimo. Nature 463, 757–762 (2010).
Schlebusch, C. M. et al. Genomic variation in seven Khoe-San groups reveals adaptation and complex African history. Science 338, 374–379 (2012).
Hodoglugil, U. & Mahley, R. W. Turkish population structure and genetic ancestry reveal relatedness among Eurasian populations. Ann. Hum. Genet. 76, 128–141 (2012).
Yunusbayev, B. et al. The Caucasus as an asymmetric semipermeable barrier to ancient human migrations. Mol. Biol. Evol. 29, 359–365 (2012).
Vine, A. E. et al. No evidence for excess runs of homozygosity in bipolar disorder. Psychiatr. Genet. 19, 165–170 (2009).
Nalls, M. A. et al. Extended tracts of homozygosity identify novel candidate genes associated with late-onset Alzheimer's disease. Neurogenetics 10, 183–190 (2009).
Ghani, M. et al. Association of long runs of homozygosity with Alzheimer disease among african american individuals. JAMA Neurol. 72, 1313–1323 (2015).
Chahrour, M. H. et al. Whole-Exome sequencing and homozygosity analysis implicate depolarization-regulated neuronal genes in autism. PLoS Genet. 8, 236–244 (2012).
Casey, J. P. et al. A novel approach of homozygous haplotype sharing identifies candidate genes in autism spectrum disorder. Hum. Genet. 131, 565–579 (2012).
Lin, P. I. et al. Runs of homozygosity associated with speech delay in autism in a taiwanese han population: evidence for the recessive model. PLoS ONE 8, e72056 (2013).
Gamsiz, E. D. et al. Intellectual disability is associated with increased runs of homozygosity in simplex autism. Am. J. Hum. Genet. 93, 103–109 (2013).
Gandin, I. et al. Excess of runs of homozygosity is associated with severe cognitive impairment in intellectual disability. Genet. Med. 17, 396–399 (2015).
Iourov, I. Y., Vorsanova, S. G., Korostelev, S. A., Zelenova, M. A. & Yurov, Y. B. Long contiguous stretches of homozygosity spanning shortly the imprinted loci are associated with intellectual disability, autism and/or epilepsy. Mol. Cytogenet. 8, 77 (2015).
Melhem, N. M. et al. Characterizing runs of homozygosity and their impact on risk for psychosis in a population isolate. Am. J. Med. Genet. B Neuropsychiatr. Genet. 165B, 521–530 (2014).
Bacolod, M. D. et al. The signatures of autozygosity among patients with colorectal cancer. Cancer Res. 68, 2610–2621 (2008).
Spain, S. L. et al. Colorectal cancer risk is not associated with increased levels of homozygosity in a population from the United Kingdom. Cancer Res. 69, 7422–7429 (2009).
Siraj, A. K. et al. Colorectal cancer risk is not associated with increased levels of homozygosity in Saudi Arabia. Genet. Med. 14, 720–728 (2012).
Hosking, F. J. et al. Genome-wide homozygosity signatures and childhood acute lymphoblastic leukemia risk. Blood 115, 4472–4477 (2010).
Enciso-Mora, V., Hosking, F. J. & Houlston, R. S. Risk of breast and prostate cancer is not associated with increased homozygosity in outbred populations. Europ. J. Hum. Genet. 18, 909–914 (2010).
Orloff, M. S., Zhang, L., Bebek, G. & Eng, C. Integrative genomic analysis reveals extended germline homozygosity with lung cancer risk in the PLCO cohort. PLoS ONE 7, e31975 (2012).
Thomsen, H. et al. Inbreeding and homozygosity in breast cancer survival. Sci. Rep. 5, 16467 (2015).
Thomsen, H. et al. Runs of homozygosity and inbreeding in thyroid cancer. BMC Cancer 16, 227 (2016).
McLaughlin, R. L. et al. Homozygosity mapping in an Irish ALS case-control cohort describes local demographic phenomena and points towards potential recessive risk loci. Genomics 105, 237–241 (2015).
McWhirter, R. E., McQuillan, R., Visser, E., Counsell, C. & Wilson, J. F. Genome-wide homozygosity and multiple sclerosis in Orkney and Shetland Islanders. Europ. J. Hum. Genet. 20, 198–202 (2012).
Binzer, S. et al. High inbreeding in the Faroe Islands does not appear to constitute a risk factor for multiple sclerosis. Mult. Scler. 21, 996–1002 (2015).
Christofidou, P. et al. Runs of homozygosity: association with coronary artery disease and gene expression in monocytes and macrophages. Am. J. Hum. Genet. 97, 228–237 (2015).
Kuningas, M. et al. Runs of homozygosity do not influence survival to old age. PLoS ONE 6, e22580 (2011).
Yang, T. L. et al. Runs of homozygosity identify a recessive locus 12q21.31 for human adult height. J. Clin. Endocrinol. Metab. 95, 3777–3782 (2010).
Power, R. A., Nagoshi, C., DeFries, J. C., Wellcome Trust Case Control Consortium 2 & Plomin, R. Genome-wide estimates of inbreeding in unrelated individuals and their association with cognitive ability. Europ. J. Hum. Genet. 22, 386–390 (2014).
Verweij, K. J. et al. The association of genotype-based inbreeding coefficient with a range of physical and psychological human traits. PLoS ONE 9, e103102 (2014).
Acknowledgements
This work was supported by the Medical Research Council Human Genetics Unit quinquennial programme grant 'QTL in Health and Disease.' F.C.C. is supported by the South African National Research Foundation (NRF), and M.R. holds a South African Research Chair in Genomics and Bioinformatics of African populations hosted by the University of the Witwatersrand, funded by the Department of Science and Technology and administered by the NRF. The authors thank T. Gonzalez for help with figures and G. Alvarez, R. Vilas, O. Polašek, T. Esko, A. Wright, H. Campbell and C. Haley for helpful discussions and comments on the manuscript.
Author information
Authors and Affiliations
Contributions
F.C.C. and J.F.W. researched data for the article. F.C.C., P.K.J., D.W.C. and J.F.W. wrote the manuscript. All authors contributed to reviewing and editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
FURTHER INFORMATION
Glossary
- Consanguinity
-
Mating among relatives, for example, first or second cousins. Literally 'of the same blood'.
- Endogamy
-
Marriage within the population or community.
- Runs of homozygosity
-
(ROH). Contiguous regions of the genome where an individual is homozygous across all sites. This arises if the haplotypes transmitted from the mother and father are identical, having in turn been inherited from a common ancestor at some point in the past. It is important to note that this notion does not rely on a known pedigree and does not require an (arbitrary) baseline population (the first generation of ancestors or founders in a pedigree). However, ROH in practice are required to have an (arbitrary) minimum size, depending on the density of genotypes available, to distinguish identity-by-descent from chance.
- Autozygous
-
Also known as homozygosity-by-descent; homozygosity arising at a locus owing to identity-by-descent.
- Effective population size
-
(Ne). The size of an idealized population that would show the same amount of genetic drift or inbreeding, often thought of as the number of breeding individuals and usually lower than the census population size.
- Demographic histories
-
The histories of the changes in population size; for example, populations may be large or small, of constant size, or expanding or contracting; may undergo bottlenecks (severe declines in population size) or founder events (establishment of populations by a limited number of ancestors); may be substructured geographically; or may admix with one another.
- Inbreeding depression
-
The reduction in evolutionary fitness of a population or individual due to the presence of increased homozygosity arising from inbreeding. Values of traits related to fitness, such as fertility, are reduced.
- Genetic architecture
-
The makeup of the genetic basis of a trait, in particular whether there are few or many causal loci, whether the causal variants are rare or common or have small or large effect sizes and the degree to which dominance plays a part.
- Haplotype
-
A set of alleles on a chromosome or chromosomal segment inherited from one parent — often a series of alleles at neighbouring loci that are strongly statistically associated due to lack of recombination. Certain haplotypes may become common in the population owing to natural selection or drift until broken down over time by recombination.
- Admixed
-
Genetic admixture occurs when mating begins between two previously separate populations and individuals within the new population have a mix of haplotypes from each parental population.
- Inbreeding loops
-
Also known as pedigree loops; the connection in a pedigree between the maternal and paternal ancestors of an individual. The closed loops show how the same haplotypes could pass down both sides of families.
- Population bottleneck
-
A severe decline in population size over a short time or a lesser reduction over a longer time, followed by a recovery.
- Cosmopolitan populations
-
Populations that are not isolated; typical urban populations.
- Overdominance
-
Also known as heterozygote advantage; overdominance occurs if the heterozygote trait value (phenotype) is outside the range of the trait values of the two homozygotes.
- Balancing selection
-
When two or more alleles are favoured by natural selection rather than one, for example, when the heterozygote is fitter than either homozygote.
- Dominance
-
Dominance is present at a genetic locus when the effect of one copy of an allele gives rise to a trait or phenotypic value that, rather than being halfway between the values for the two homozygotes, is nearer the trait value for a carrier of two copies of the allele. In this situation, the other allele is recessive.
- Directional dominance
-
Directional dominance occurs when the dominance effect across all causal loci in the genome has a trend in one direction, that is, to raise or lower the trait, rather than the individual dominance effects at loci cancelling each other out.
- Identity-by-descent
-
(IBD). The inheritance of an identical haplotype from both parents owing to it having been passed without recombination from a common ancestor in the baseline population.
- Inbreeding coefficient
-
The probability, denoted F, of inheriting two alleles identical-by-descent at an autosomal locus in the presence of consanguinity. F is one-sixteenth for first-cousin offspring, one-sixty-fourth for second cousins and one-eighth for the progeny of avuncular or double first-cousin matings.
- Genomic inbreeding coefficient
-
FROH; the proportion of the genome that is in ROH. F and FROH have been shown to be highly correlated.
- Avuncular union
-
Marriage or mating between an uncle and niece or aunt and nephew.
- Confounding
-
Literally, confusion. Statistical confounding arises when the association between a proposed explanatory variable and an outcome is distorted by the presence of a third variable associating with both. Unless all confounding can be excluded, causal inferences cannot be made from observational associations.
- Darwinian fitness
-
The expected relative contribution of an individual or allele to the next generation of the population. It is the ability of an organism of a particular genotype to survive and leave viable offspring in its particular environment, captured in the phrase 'the survival of the fittest', although reproduction of the fittest might be more apt.
- Panmixia
-
Random mating rather than mating structured by geography, ethnicity, socioeconomic status or other factors.
- Gene conversion
-
A mechanism of recombination where one DNA sequence is replaced by a highly homologous one, leaving the sequences identical. In mammals, gene conversion tracts are usually short, between 200 bp and 1 kb.
- Heterosis
-
Also called hybrid vigour; the propensity when inbred lines of, for example, maize or domesticated animals are crossed to result in hybrids that are fitter than either parent. The trait values that were reduced by inbreeding depression increase after outbreeding.
- Outbreeding depression
-
When the offspring of distantly related mates are less fit than the parents; for example, if one homozygote has the highest fitness, outbreeding will usually increase the number of heterozygotes and thus reduce fitness.
Rights and permissions
About this article
Cite this article
Ceballos, F., Joshi, P., Clark, D. et al. Runs of homozygosity: windows into population history and trait architecture. Nat Rev Genet 19, 220–234 (2018). https://doi.org/10.1038/nrg.2017.109
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg.2017.109
This article is cited by
-
Genome-wide diversity, population structure and signatures of inbreeding in the African buffalo in Mozambique
BMC Ecology and Evolution (2024)
-
Multilocus pathogenic variants contribute to intrafamilial clinical heterogeneity: a retrospective study of sibling pairs with neurodevelopmental disorders
BMC Medical Genomics (2024)
-
Inbreeding depression is associated with recent homozygous-by-descent segments in Belgian Blue beef cattle
Genetics Selection Evolution (2024)
-
Investigating the genetic makeup of the major histocompatibility complex (MHC) in the United Arab Emirates population through next-generation sequencing
Scientific Reports (2024)
-
Constructing telomere-to-telomere diploid genome by polishing haploid nanopore-based assembly
Nature Methods (2024)