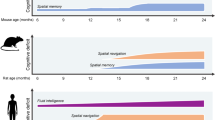
Key Points
-
Undercarboxylated osteocalcin regulates anxiety and cognition in adult mice
-
Osteocalcin is necessary and sufficient to correct age-related decline in cognitive function in mice
-
Mouse maternal osteocalcin contributes to brain development and the acquisition of cognitive function in the fetus starting at embryonic day (E) 14.5
-
Probable G protein-coupled receptor 158 (GPR158) regulates the role of osteocalcin in anxiety and cognition in the mouse brain
Abstract
A remarkable, unexpected aspect of the bone-derived hormone osteocalcin is that it is necessary for both brain development and brain function in the mouse, as its absence results in a profound deficit in spatial learning and memory and an exacerbation of anxiety-like behaviour. The regulation of cognitive function by osteocalcin, together with the fact that its circulating levels decrease in midlife compared with adolescence in all species tested, raised the prospect that osteocalcin might be an anti-geronic hormone that could prevent age-related cognitive decline. As presented in this Review, recent data indicate that this is indeed the case and that osteocalcin is necessary for the anti-geronic activity recently ascribed to the plasma of young wild-type mice. The diversity and amplitude of the functions of osteocalcin in the brain, during development and postnatally, had long called for the identification of its receptor in the brain, which was also recently achieved. This Review presents our current understanding of the biology of osteocalcin in the brain, highlighting the bony vertebrate specificity of the regulation of cognitive function and pointing toward where therapeutic opportunities might exist.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lee, N. K. et al. Endocrine regulation of energy metabolism by the skeleton. Cell 130, 456–469 (2007). This study is the first demonstration that osteocalcin acts as a hormone on pancreatic functions.
Mera, P. et al. Osteocalcin signaling in myofibers is necessary and sufficient for optimum adaptation to exercise. Cell Metab. 23, 1078–1092 (2016). This study establishes that osteocalcin is necessary and sufficient to promote exercise capacity.
Das, S. K., Sharma, N. K. & Elbein, S. C. Analysis of osteocalcin as a candidate gene for type 2 diabetes (T2D) and intermediate traits in Caucasians and African Americans. Dis. Markers 28, 281–286 (2010).
De Toni, L. et al. Polymorphism rs2274911 of GPRC6A as a novel risk factor for testis failure. J. Clin. Endocrinol. Metab. 101, 953–961 (2016).
De Toni, L. et al. Osteocalcin and sex hormone binding globulin compete on a specific binding site of GPRC6A. Endocrinology 157, 4473–4486 (2016).
Di Nisio, A. et al. The rs2274911 polymorphism in GPRC6A gene is associated with insulin resistance in normal weight and obese subjects. Clin. Endocrinol. 86, 185–191 (2017).
Gao, J. et al. The PLC/PKC/Ras/MEK/Kv channel pathway is involved in uncarboxylated osteocalcin-regulated insulin secretion in rats. Peptides 86, 72–79 (2016).
Gao, J. et al. Inhibition of voltage-gated potassium channels mediates uncarboxylated osteocalcin-regulated insulin secretion in rat pancreatic beta cells. Eur. J. Pharmacol. 777, 41–48 (2016).
Korostishevsky, M. et al. Significant association between body composition phenotypes and the osteocalcin genomic region in normative human population. Bone 51, 688–694 (2012).
Kover, K. et al. Osteocalcin protects pancreatic beta cell function and survival under high glucose conditions. Biochem. Biophys. Res. Commun. 462, 21–26 (2015).
Kunutsor, S. K., Apekey, T. A. & Laukkanen, J. A. Association of serum total osteocalcin with type 2 diabetes and intermediate metabolic phenotypes: systematic review and meta-analysis of observational evidence. Eur. J. Epidemiol. 30, 599–614 (2015).
Sabek, O. M. et al. Osteocalcin effect on human beta-cells mass and function. Endocrinology 156, 3137–3146 (2015).
Wei, J., Hanna, T., Suda, N., Karsenty, G. & Ducy, P. Osteocalcin promotes beta-cell proliferation during development and adulthood through Gprc6a. Diabetes 63, 1021–1031 (2014).
Karsenty, G. & Olson, E. N. Bone and muscle endocrine functions: unexpected paradigms of inter-organ communication. Cell 164, 1248–1256 (2016).
Khrimian, L. et al. Gpr158 mediates osteocalcin's regulation of cognition. J. Exp. Med. 214, 2859–2873 (2017). This paper provides genetic, biochemical and molecular evidence that GPR158 is the receptor for osteocalcin in the brain and that it mediates the ability of osteocalcin to reverse age-related cognitive decline.
Ducy, P. et al. Increased bone formation in osteocalcin-deficient mice. Nature 382, 448–452 (1996).
Ferron, M. et al. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 142, 296–308 (2010). This paper demonstrates that uncarboxylated and undercarboxylated osteocalcin are the active forms of the hormone.
Oury, F. et al. Endocrine regulation of male fertility by the skeleton. Cell 144, 796–809 (2011). This study shows that osteocalcin regulates male fertility and importantly identifies its first receptor, GPRC6A.
Chamouni, A. & Oury, F. Reciprocal interaction between bone and gonads. Arch. Biochem. Biophys. 561, 147–153 (2014).
Oury, F. et al. Osteocalcin regulates murine and human fertility through a pancreas-bone-testis axis. J. Clin. Invest. 123, 2421–2433 (2013).
Yoshikawa, Y. et al. Genetic evidence points to an osteocalcin-independent influence of osteoblasts on energy metabolism. J. Bone Miner. Res. 26, 2012–2025 (2011).
Frye, C. A., Edinger, K. & Sumida, K. Androgen administration to aged male mice increases anti-anxiety behavior and enhances cognitive performance. Neuropsychopharmacology 33, 1049–1061 (2008).
Aikey, J. L., Nyby, J. G., Anmuth, D. M. & James, P. J. Testosterone rapidly reduces anxiety in male house mice (Mus musculus). Horm. Behav. 42, 448–460 (2002).
Oury, F. et al. Maternal and offspring pools of osteocalcin influence brain development and functions. Cell 155, 228–241 (2013). This paper provides the first in vivo evidence that osteocalcin is necessary for brain development and cognition and that it is necessary to prevent anxiety.
Pi, M. et al. GPRC6A null mice exhibit osteopenia, feminization and metabolic syndrome. PLoS ONE 3, e3858 (2008).
Pi, M., Wu, Y. & Quarles, L. D. GPRC6A mediates responses to osteocalcin in beta-cells in vitro and pancreas in vivo. J. Bone Miner. Res. 26, 1680–1683 (2011).
Nakazawa, K. et al. Requirement for hippocampal CA3 NMDA receptors in associative memory recall. Science 297, 211–218 (2002).
Valenstein, E. et al. Retrosplenial amnesia. Brain 110, 1631–1646 (1987).
To¨rk, I., Tracey, D. J., Paxinos, G. & Stone, J. Neurotransmitters in the Human Brain (Plenum Press, 1995).
Ende, G. Proton magnetic resonance spectroscopy: relevance of glutamate and GABA to neuropsychology. Neuropsychol. Rev. 25, 315–325 (2015).
Daubner, S. C., Le, T. & Wang, S. Tyrosine hydroxylase and regulation of dopamine synthesis. Arch. Biochem. Biophys. 508, 1–12 (2011).
Wang, S., Lasagna, M., Daubner, S. C., Reinhart, G. D. & Fitzpatrick, P. F. Fluorescence spectroscopy as a probe of the effect of phosphorylation at serine 40 of tyrosine hydroxylase on the conformation of its regulatory domain. Biochemistry 50, 2364–2370 (2011).
Walther, D. J. et al. Synthesis of serotonin by a second tryptophan hydroxylase isoform. Science 299, 76 (2003).
Tochitani, S. & Kondo, S. Immunoreactivity for GABA, GAD65, GAD67 and bestrophin-1 in the meninges and the choroid plexus: implications for non-neuronal sources for GABA in the developing mouse brain. PLoS ONE 8, e56901 (2013).
Beckstead, R. M., Domesick, V. B. & Nauta, W. J. Efferent connections of the substantia nigra and ventral tegmental area in the rat. Brain Res. 175, 191–217 (1979).
Lira, A. et al. Altered depression-related behaviors and functional changes in the dorsal raphe nucleus of serotonin transporter-deficient mice. Biol. Psychiatry 54, 960–971 (2003).
Ducy, P., Zhang, R., Geoffroy, V., Ridall, A. L. & Karsenty, G. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell 89, 747–754 (1997).
Wei, J. et al. Glucose uptake and Runx2 synergize to orchestrate osteoblast differentiation and bone formation. Cell 161, 1576–1591 (2015).
Tole, S., Christian, C. & Grove, E. A. Early specification and autonomous development of cortical fields in the mouse hippocampus. Development 124, 4959–4970 (1997).
Bradburn, S. et al. Association between osteocalcin and cognitive performance in healthy older adults. Age Ageing 45, 844–849 (2016).
Puig, J. et al. Lower serum osteocalcin concentrations are associated with brain microstructural changes and worse cognitive performance. Clin. Endocrinol. 84, 756–763 (2016).
Barker, D. J. Maternal nutrition, fetal nutrition, and disease in later life. Nutrition 13, 807–813 (1997).
Hales, C. N. & Barker, D. J. The thrifty phenotype hypothesis. Br. Med. Bull. 60, 5–20 (2001).
Broad, K. D. & Keverne, E. B. Placental protection of the fetal brain during short-term food deprivation. Proc. Natl Acad. Sci. USA 108, 15237–15241 (2011).
Lawlor, D. A., Relton, C., Sattar, N. & Nelson, S. M. Maternal adiposity — a determinant of perinatal and offspring outcomes? Nat. Rev. Endocrinol. 8, 679–688 (2012).
Van den Bergh, B. R., Mulder, E. J., Mennes, M. & Glover, V. Antenatal maternal anxiety and stress and the neurobehavioural development of the fetus and child: links and possible mechanisms. A review. Neurosci. Biobehav. Rev. 29, 237–258 (2005).
Wadhwa, P. D., Sandman, C. A. & Garite, T. J. The neurobiology of stress in human pregnancy: implications for prematurity and development of the fetal central nervous system. Prog. Brain Res. 133, 131–142 (2001).
Black, M. H., Sacks, D. A., Xiang, A. H. & Lawrence, J. M. The relative contribution of prepregnancy overweight and obesity, gestational weight gain, and IADPSG-defined gestational diabetes mellitus to fetal overgrowth. Diabetes Care 36, 56–62 (2013).
Modder, U. I. et al. Relation of age, gender, and bone mass to circulating sclerostin levels in women and men. J. Bone Miner. Res. 26, 373–379 (2011).
Ding, Q., Vaynman, S., Souda, P., Whitelegge, J. P. & Gomez-Pinilla, F. Exercise affects energy metabolism and neural plasticity-related proteins in the hippocampus as revealed by proteomic analysis. Eur. J. Neurosci. 24, 1265–1276 (2006).
Levinger, I. et al. The effect of acute exercise on undercarboxylated osteocalcin in obese men. Osteoporos. Int. 22, 1621–1626 (2011).
Schwab, P. & Scalapino, K. Exercise for bone health: rationale and prescription. Curr. Opin. Rheumatol. 23, 137–141 (2011).
van Praag, H., Shubert, T., Zhao, C. & Gage, F. H. Exercise enhances learning and hippocampal neurogenesis in aged mice. J. Neurosci. 25, 8680–8685 (2005).
Hopkins, M. E., Nitecki, R. & Bucci, D. J. Physical exercise during adolescence versus adulthood: differential effects on object recognition memory and brain-derived neurotrophic factor levels. Neuroscience 194, 84–94 (2011).
Scahill, R. I. et al. A longitudinal study of brain volume changes in normal aging using serial registered magnetic resonance imaging. Arch. Neurol. 60, 989–994 (2003).
Dall, T. M. et al. An aging population and growing disease burden will require a large and specialized health care workforce by 2025. Health Aff. 32, 2013–2020 (2013).
Grady, P. A. Advancing the health of our aging population: a lead role for nursing science. Nurs. Outlook 59, 207–209 (2011).
Finch, C. E. Evolution in health and medicine Sackler colloquium: evolution of the human lifespan and diseases of aging: roles of infection, inflammation, and nutrition. Proc. Natl Acad. Sci. USA 107 (Suppl. 1), 1718–1724 (2010).
Kramer, A. F. et al. Ageing, fitness and neurocognitive function. Nature 400, 418–419 (1999).
La Rue, A. Memory loss and aging. Distinguishing dementia from benign senescent forgetfulness and depressive pseudodementia. Psychiatr. Clin. North Am. 5, 89–103 (1982).
Small, S. A. Age-related memory decline: current concepts and future directions. Arch. Neurol. 58, 360–364 (2001).
Pavlopoulos, E. et al. Molecular mechanism for age-related memory loss: the histone-binding protein RbAp48. Sci. Transl Med. 5, 200ra115 (2013).
Chiappe, A. et al. Influence of age and sex in serum osteocalcin levels in thoroughbred horses. Arch. Physiol. Biochem. 107, 50–54 (1999).
Price, P. A. & Nishimoto, S. K. Radioimmunoassay for the vitamin K-dependent protein of bone and its discovery in plasma. Proc. Natl Acad. Sci. USA 77, 2234–2238 (1980).
Villeda, S. A. et al. Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice. Nat. Med. 20, 659–663 (2014). This landmark study establishes that age-related cognitive decline can be reversed by treating aged mice with young blood.
Hall, J., Thomas, K. L. & Everitt, B. J. Rapid and selective induction of BDNF expression in the hippocampus during contextual learning. Nat. Neurosci. 3, 533–535 (2000).
Yamada, K. & Nabeshima, T. Brain-derived neurotrophic factor/TrkB signaling in memory processes. J. Pharmacol. Sci. 91, 267–270 (2003).
Dean, C. et al. Synaptotagmin-IV modulates synaptic function and long-term potentiation by regulating BDNF release. Nat. Neurosci. 12, 767–776 (2009).
Gao, J. et al. A novel pathway regulates memory and plasticity via SIRT1 and miR-134. Nature 466, 1105–1109 (2010).
Zala, D. et al. Vesicular glycolysis provides on-board energy for fast axonal transport. Cell 152, 479–491 (2013).
Chun, L., Zhang, W. H. & Liu, J. F. Structure and ligand recognition of class C GPCRs. Acta Pharmacol. Sin. 33, 312–323 (2012).
Rondard, P., Goudet, C., Kniazeff, J., Pin, J. P. & Prezeau, L. The complexity of their activation mechanism opens new possibilities for the modulation of mGlu and GABAB class C G protein-coupled receptors. Neuropharmacology 60, 82–92 (2011).
Brauner-Osborne, H., Wellendorph, P. & Jensen, A. A. Structure, pharmacology and therapeutic prospects of family C G-protein coupled receptors. Curr. Drug Targets 8, 169–184 (2007).
Luo, J., Liu, Z., Liu, J. & Eugene, C. Y. Distribution pattern of GPRC6A mRNA in mouse tissue by in situ hybridization. Zhong Nan Da Xue Xue Bao Yi Xue Ban 35, 1–10 (2010).
Wellendorph, P. & Brauner-Osborne, H. Molecular cloning, expression, and sequence analysis of GPRC6A, a novel family C G-protein-coupled receptor. Gene 335, 37–46 (2004).
Dragunow, M. & Robertson, H. A. Kindling stimulation induces c-fos protein(s) in granule cells of the rat dentate gyrus. Nature 329, 441–442 (1987).
Sagar, S. M., Sharp, F. R. & Curran, T. Expression of c-fos protein in brain: metabolic mapping at the cellular level. Science 240, 1328–1331 (1988).
Burgess, G. M. et al. The second messenger linking receptor activation to internal Ca release in liver. Nature 309, 63–66 (1984).
Nishizuka, Y. Studies and perspectives of protein kinase C. Science 233, 305–312 (1986).
Kandel, E. R. The molecular biology of memory storage: a dialogue between genes and synapses. Science 294, 1030–1038 (2001).
Wigstrom, H. & Gustafsson, B. On long-lasting potentiation in the hippocampus: a proposed mechanism for its dependence on coincident pre- and postsynaptic activity. Acta Physiol. Scand. 123, 519–522 (1985).
Barco, A., Bailey, C. H. & Kandel, E. R. Common molecular mechanisms in explicit and implicit memory. J. Neurochem. 97, 1520–1533 (2006).
Hommel, J. D., Sears, R. M., Georgescu, D., Simmons, D. L. & DiLeone, R. J. Local gene knockdown in the brain using viral-mediated RNA interference. Nat. Med. 9, 1539–1544 (2003).
Castellano, J. M. et al. Human umbilical cord plasma proteins revitalize hippocampal function in aged mice. Nature 544, 488–492 (2017).
Acknowledgements
This work was supported by 2P01 AG032959-06A1 and the Columbia Aging Center (G.K.), a 5T32DK007328-38 Endocrinology Training Grant (L.K.) from the NIH, Fondation pour la Recherche Medicale grant AJE20130928594, the Human Frontier Scientific Program–Grant ATIP-AVENIR INSERM - R14080KS - RSE15007KSA Program–INSERM, Grant AGEMED-INSERM (F.O.) and the Philippe Foundation (A.O.).
Author information
Authors and Affiliations
Contributions
A.O., L.K., G.K. and F.O. researched data for the article, contributed to discussion of the content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Obri, A., Khrimian, L., Karsenty, G. et al. Osteocalcin in the brain: from embryonic development to age-related decline in cognition. Nat Rev Endocrinol 14, 174–182 (2018). https://doi.org/10.1038/nrendo.2017.181
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2017.181
This article is cited by
-
Bidirectional mediation of bone mineral density and brain atrophy on their associations with gait variability
Scientific Reports (2024)
-
Positive associations between sex hormones, bone metabolism and cognitive impairment in Chinese oldest-old females
BMC Psychiatry (2023)
-
Neural EGFL-like 1, a craniosynostosis-related osteochondrogenic molecule, strikingly associates with neurodevelopmental pathologies
Cell & Bioscience (2023)
-
Osteocalcin ameliorates cognitive dysfunctions in a mouse model of Alzheimer’s Disease by reducing amyloid β burden and upregulating glycolysis in neuroglia
Cell Death Discovery (2023)
-
Potential association of bone mineral density loss with cognitive impairment and central and peripheral amyloid-β changes: a cross-sectional study
BMC Musculoskeletal Disorders (2022)