Key Points
-
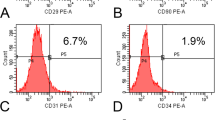
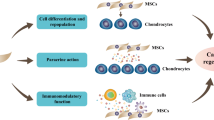
Stromal cells and/or stem cells can be isolated from different tissues on the basis of plastic adherence and surface-antigen profiles, thereby providing opportunities for bone regeneration
-
The regenerative potential of therapies that are based on adipose-tissue-derived and bone-marrow-derived mesenchymal stromal or stem cells is being tested clinically for the treatment of craniofacial bone defects, tibial non-unions and osteonecrosis of the femoral head
-
Although most approaches in this area use autologous cells, allogeneic sources that include commercially available allograft cell-based products are being investigated
-
Widespread use of cell-based products requires the development and standardization of guidelines and protocols for the shipment and storage of cell therapeutics
-
Despite strong clinical data, which indicates enhanced regenerative outcomes following stromal-cell or stem-cell transplantation, further insight is needed into the mechanisms of action of these strategies
-
Opportunities exist to develop technologies that improve cell survival, morphogenesis and functionality to advance cell therapy as standard care for the treatment of bone defects
Abstract
Stem-cell-mediated bone repair has been used in clinical trials for the regeneration of large craniomaxillofacial defects, to slow the process of bone degeneration in patients with osteonecrosis of the femoral head and for prophylactic treatment of distal tibial fractures. Successful regenerative outcomes in these investigations have provided a solid foundation for wider use of stromal cells in skeletal repair therapy. However, employing stromal cells to facilitate or enhance bone repair is far from being adopted into clinical practice. Scientific, technical, practical and regulatory obstacles prevent the widespread therapeutic use of stromal cells. Ironically, one of the major challenges lies in the limited understanding of the mechanisms via which transplanted cells mediate regeneration. Animal models have been used to provide insight, but these models largely fail to reproduce the nuances of human diseases and bone defects. Consequently, the development of targeted approaches to optimize cell-mediated outcomes is difficult. In this Review, we highlight the successes and challenges reported in several clinical trials that involved the use of bone-marrow-derived mesenchymal or adipose-tissue-derived stromal cells. We identify several obstacles blocking the mainstream use of stromal cells to enhance skeletal repair and highlight technological innovations or areas in which novel techniques might be particularly fruitful in continuing to advance the field of skeletal regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Raggatt, L. J. et al. Fracture healing via periosteal callus formation requires macrophages for both initiation and progression of early endochondral ossification. Am. J. Pathol. 184, 3192–3204 (2014).
Das, A., Segar, C. E., Hughley, B. B., Bowers, D. T. & Botchwey, E. A. The promotion of mandibular defect healing by the targeting of S1P receptors and the recruitment of alternatively activated macrophages. Biomaterials 34, 9853–9862 (2013).
Kuroda, R. et al. Clinical impact of circulating CD34-positive cells on bone regeneration and healing. Tissue Eng. Part B Rev. 20, 190–199 (2014).
Hutton, D. L. & Grayson, W. L. Stem cell-based approaches to engineering vascularized bone. Curr. Opin. Chem. Eng. 3, 75–82 (2014).
Neovius, E. & Engstrand, T. Craniofacial reconstruction with bone and biomaterials: review over the last 11 years. J. Plast. Reconstr. Aesthet. Surg. 63, 1615–1623 (2010).
Casserbette, M., Murray, A. B., Closs, E. I., Erfle, V. & Schmidt, J. Bone-formation by osteoblast-like cells in a 3-dimensional cell-culture. Calcif. Tissue Int. 46, 46–56 (1990).
Dupont, K. M. et al. Human stem cell delivery for treatment of large segmental bone defects. Proc. Natl Acad. Sci. USA 107, 3305–3310 (2010).
Friedenstein, A. J. Precursor cells of mechanocytes. Int. Rev. Cytol. 47, 327–359 (1976).
Friedenstein, A. J. & Lalykina, K. S. Thymus cells are inducible to osteogenesis. Eur. J. Immunol. 2, 602–603 (1972).
Friedenstein, A. J., Petrakova, K. V., Kurolesova, A. I. & Frolova, G. P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 6, 230–247 (1968).
Owen, M. & Friedenstein, A. J. Stromal stem cells: marrow-derived osteogenic precursors. Ciba Found. Symp. 136, 42–60 (1988).
Ashton, B. A. et al. Formation of bone and cartilage by marrow stromal cells in diffusion chambers in vivo. Clin. Orthop. Relat. Res. 151, 294–307 (1980).
Allen, T. D. & Dexter, T. M. Cellular interrelationships during in vitro granulopoiesis. Differentiation 6, 191–194 (1976).
Dexter, T. M., Allen, T. D. & Lajtha, L. G. Conditions controlling the proliferation of haemopoietic stem cells in vitro. J. Cell. Physiol. 91, 335–344 (1977).
Lanotte, M., Scott, D., Dexter, T. M. & Allen, T. D. Clonal preadipocyte cell lines with different phenotypes derived from murine marrow stroma: factors influencing growth and adipogenesis in vitro. J. Cell. Physiol. 111, 177–186 (1982).
Hunt, P. et al. A single bone marrow-derived stromal cell type supports the in vitro growth of early lymphoid and myeloid cells. Cell 48, 997–1007 (1987).
Pietrangeli, C. E., Hayashi, S. & Kincade, P. W. Stromal cell lines which support lymphocyte growth: characterization, sensitivity to radiation and responsiveness to growth factors. Eur. J. Immunol. 18, 863–872 (1988).
Whitlock, C. A., Tidmarsh, G. F., Muller-Sieburg, C. & Weissman, I. L. Bone marrow stromal cell lines with lymphopoietic activity express high levels of a pre-B neoplasia-associated molecule. Cell 48, 1009–1021 (1987).
Namen, A. E. et al. Stimulation of B-cell progenitors by cloned murine interleukin-7. Nature 333, 571–573 (1988).
Lee, G., Namen, A. E., Gillis, S. & Kincade, P. W. Recombinant interleukin-7 supports the growth of normal B lymphocyte precursors. Curr. Top. Microbiol. Immunol. 141, 16–18 (1988).
Paul, S. R. et al. Molecular cloning of a cDNA encoding interleukin 11, a stromal cell-derived lymphopoietic and hematopoietic cytokine. Proc. Natl Acad. Sci. USA 87, 7512–7516 (1990).
Caplan, A. I. Mesenchymal stem cells. J. Orthop. Res. 9, 641–650 (1991).
Wakitani, S., Saito, T. & Caplan, A. I. Myogenic cells derived from rat bone marrow mesenchymal stem cells exposed to 5-azacytidine. Muscle Nerve 18, 1417–1426 (1995).
Haynesworth, S. E., Baber, M. A. & Caplan, A. I. Cell surface antigens on human marrow-derived mesenchymal cells are detected by monoclonal antibodies. Bone 13, 69–80 (1992).
Simmons, P. J. & Torok-Storb, B. Identification of stromal cell precursors in human bone marrow by a novel monoclonal antibody, STRO-1. Blood 78, 55–62 (1991).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Mendicino, M., Bailey, A. M., Wonnacott, K., Puri, R. K. & Bauer, S. R. MSC-based product characterization for clinical trials: an FDA perspective. Cell Stem Cell 14, 141–145 (2014).
Lo Surdo, J. & Bauer, S. R. Quantitative approaches to detect donor and passage differences in adipogenic potential and clonogenicity in human bone marrow-derived mesenchymal stem cells. Tissue Eng. Part C Methods 18, 877–889 (2012).
Lo Surdo, J. L., Millis, B. A. & Bauer, S. R. Automated microscopy as a quantitative method to measure differences in adipogenic differentiation in preparations of human mesenchymal stromal cells. Cytotherapy 15, 1527–1540 (2013).
Mindaye, S. T., Ra, M., Lo Surdo, J., Bauer, S. R. & Alterman, M. A. Improved proteomic profiling of the cell surface of culture-expanded human bone marrow multipotent stromal cells. J. Proteomics 78, 1–14 (2013).
Mindaye, S. T., Ra, M., Lo Surdo, J. L., Bauer, S. R. & Alterman, M. A. Global proteomic signature of undifferentiated human bone marrow stromal cells: evidence for donor-to-donor proteome heterogeneity. Stem Cell Res. 11, 793–805 (2013).
Bianco, P. et al. The meaning, the sense and the significance: translating the science of mesenchymal stem cells into medicine. Nat. Med. 19, 35–42 (2013).
Robey, P. G., Kuznetsov, S. A., Riminucci, M. & Bianco, P. Bone marrow stromal cell assays: in vitro and in vivo. Methods Mol. Biol. 1130, 279–293 (2014).
Zuk, P. A. et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 7, 211–228 (2001).
De Coppi, P. et al. Isolation of amniotic stem cell lines with potential for therapy. Nat. Biotechnol. 25, 100–106 (2007).
Mitchell, K. E. et al. Matrix cells from Wharton's jelly form neurons and glia. Stem Cells 21, 50–60 (2003).
Troyer, D. L. & Weiss, M. L. Wharton's jelly-derived cells are a primitive stromal cell population. Stem Cells 26, 591–599 (2008).
Gronthos, S., Mankani, M., Brahim, J., Robey, P. G. & Shi, S. Postnatal human dental pulp stem cells (DPSCS) in vitro and in vivo. Proc. Natl Acad. Sci. USA 97, 13625–13630 (2000).
Usas, A. & Huard, J. Muscle-derived stem cells for tissue engineering and regenerative therapy. Biomaterials 28, 5401–5406 (2007).
Feisst, V., Brooks, A. E., Chen, C. J. & Dunbar, P. R. Characterization of mesenchymal progenitor cell populations directly derived from human dermis. Stem Cells Dev. 23, 631–642 (2014).
Erices, A., Conget, P. & Minguell, J. J. Mesenchymal progenitor cells in human umbilical cord blood. Br. J. Haematol. 109, 235–242 (2000).
Kern, S., Eichler, H., Stoeve, J., Kluter, H. & Bieback, K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 24, 1294–1301 (2006).
Qu-Petersen, Z. et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J. Cell Biol. 157, 851–864 (2002).
Zuk, P. A. et al. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 13, 4279–4295 (2002).
Gronthos, S. et al. Surface protein characterization of human adipose tissue-derived stromal cells. J. Cell. Physiol. 189, 54–63 (2001).
Crisan, M. et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3, 301–313 (2008).
Pachon-Pena, G. et al. Stromal stem cells from adipose tissue and bone marrow of age-matched female donors display distinct immunophenotypic profiles. J. Cell. Physiol. 226, 843–851 (2011).
Bourin, P. et al. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 15, 641–648 (2013).
Halvorsen, Y. D. et al. Extracellular matrix mineralization and osteoblast gene expression by human adipose tissue-derived stromal cells. Tissue Eng. 7, 729–741 (2001).
Hicok, K. C. et al. Human adipose-derived adult stem cells produce osteoid in vivo. Tissue Eng. 10, 371–380 (2004).
Justesen, J., Pedersen, S. B., Stenderup, K. & Kassem, M. Subcutaneous adipocytes can differentiate into bone-forming cells in vitro and in vivo. Tissue Eng. 10, 381–391 (2004).
Mesimaki, K. et al. Novel maxillary reconstruction with ectopic bone formation by GMP adipose stem cells. Int. J. Oral Maxillofac. Surg. 38, 201–209 (2009).
Levi, B. et al. Human adipose derived stromal cells heal critical size mouse calvarial defects. PLoS ONE 5, e11177 (2010).
Sandor, G. K. et al. Adipose stem cells used to reconstruct 13 cases with cranio-maxillofacial hard-tissue defects. Stem Cells Transl Med. 3, 530–540 (2014).
Levi, B. et al. Differences in osteogenic differentiation of adipose-derived stromal cells from murine, canine, and human sources in vitro and in vivo. Plast. Reconstr. Surg. 128, 373–386 (2011).
de Girolamo, L., Sartori, M. F., Albisetti, W. & Brini, A. T. Osteogenic differentiation of human adipose-derived stem cells: comparison of two different inductive media. J. Tissue Eng. Regen. Med. 1, 154–157 (2007).
Im, G. I., Shin, Y. W. & Lee, K. B. Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthritis Cartilage 13, 845–853 (2005).
Hayashi, O., Katsube, Y., Hirose, M., Ohgushi, H. & Ito, H. Comparison of osteogenic ability of rat mesenchymal stem cells from bone marrow, periosteum, and adipose tissue. Calcif. Tissue Int. 82, 238–247 (2008).
Wolff, J. et al. GMP-level adipose stem cells combined with computer-aided manufacturing to reconstruct mandibular ameloblastoma resection defects: experience with three cases. Ann. Maxillofac. Surg. 3, 114–125 (2013).
Eilertsen, K. J., Floyd, Z. & Gimble, J. M. The epigenetics of adult (somatic) stem cells. Crit. Rev. Eukaryot. Gene Expr. 18, 189–206 (2008).
Li, H. et al. Sustained release of bone morphogenetic protein 2 via coacervate improves the osteogenic potential of muscle-derived stem cells. Stem Cells Transl Med. 2, 667–677 (2013).
Usas, A. et al. Skeletal muscle-derived stem cells: implications for cell-mediated therapies. Medicina 47, 469–479 (2011).
Gao, X. et al. Role of donor and host cells in muscle-derived stem cell-mediated bone repair: differentiation vs. paracrine effects. FASEB J. 28, 3792–3809 (2014).
Studeny, M. et al. Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J. Natl Cancer Inst. 96, 1593–1603 (2004).
Katakowski, M. et al. Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett. 335, 201–204 (2013).
Xin, H. et al. Targeted delivery of CX3CL1 to multiple lung tumors by mesenchymal stem cells. Stem Cells 25, 1618–1626 (2007).
Loebinger, M. R., Eddaoudi, A., Davies, D. & Janes, S. M. Mesenchymal stem cell delivery of trail can eliminate metastatic cancer. Cancer Res. 69, 4134–4142 (2009).
Belmar-Lopez, C. et al. Tissue-derived mesenchymal stromal cells used as vehicles for anti-tumor therapy exert different in vivo effects on migration capacity and tumor growth. BMC Med. 11, 139 (2013).
Dwyer, R. M. et al. Mesenchymal stem cell-mediated delivery of the sodium iodide symporter supports radionuclide imaging and treatment of breast cancer. Stem Cells 29, 1149–1157 (2011).
Shah, K. Mesenchymal stem cells engineered for cancer therapy. Adv. Drug Deliv. Rev. 64, 739–748 (2012).
Zhao, J. et al. Stem cell-mediated delivery of SPIO-loaded gold nanoparticles for the theranosis of liver injury and hepatocellular carcinoma. Nanotechnology 25, 5101–5101 (2014).
Zhang, T. et al. Bone marrow-derived mesenchymal stem cells promote growth and angiogenesis of breast and prostate tumors. Stem Cell Res. Ther. 4, 70 (2014).
Luo, J. et al. Infiltrating bone marrow mesenchymal stem cells increase prostate cancer stem cell population and metastatic ability via secreting cytokines to suppress androgen receptor signaling. Oncogene 33, 2768–2778 (2013).
Warnke, P. H. et al. Growth and transplantation of a custom vascularised bone graft in a man. Lancet 364, 766–770 (2004).
Mesimaki, K. et al. Novel maxillary reconstruction with ectopic bone formation by GMP adipose stem cells. Int. J. Oral Maxillofac. Surg. 38, 201–209 (2009).
Sandor, G. K. et al. Adipose stem cell tissue-engineered construct used to treat large anterior mandibular defect: a case report and review of the clinical application of good manufacturing practice-level adipose stem cells for bone regeneration. J. Oral Maxillofac. Surg. 71, 938–950 (2013).
Lendeckel, S. et al. Autologous stem cells (adipose) and fibrin glue used to treat widespread traumatic calvarial defects: case report. J. Craniomaxillofac. Surg. 32, 370–373 (2004).
Bose, S., Roy, M. & Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 30, 546–554 (2012).
Shrivats, A. R., McDermott, M. C. & Hollinger, J. O. Bone tissue engineering: state of the union. Drug Discov. Today 19, 781–786 (2014).
Bhumiratana, S. & Vunjak-Novakovic, G. Personalized human bone grafts for reconstructing head and face. Stem Cells Transl Med. 1, 64–69 (2012).
Tevlin, R. et al. Biomaterials for craniofacial bone engineering. J. Dent. Res. 93, 1187–1195 (2014).
Liebergall, M. et al. Stem cell-based therapy for prevention of delayed fracture union: a randomized and prospective preliminary study. Mol. Ther. 21, 1631–1638 (2013).
Gangji, V., De Maertelaer, V. & Hauzeur, J.-P. Autologous bone marrow cell implantation in the treatment of non-traumatic osteonecrosis of the femoral head: five year follow-up of a prospective controlled study. Bone 49, 1005–1009 (2011).
Gangji, V. et al. Treatment of osteonecrosis of the femoral head with implantation of autologous bone-marrow cells—a pilot study. J. Bone Joint Surg. Am. 86–A, 1153–1160 (2004).
Mao, Q. et al. The efficacy of targeted intraarterial delivery of concentrated autologous bone marrow containing mononuclear cells in the treatment of osteonecrosis of the femoral head: a five year follow-up study. Bone 57, 509–516 (2013).
Zhao, D. et al. Treatment of early stage osteonecrosis of the femoral head with autologous implantation of bone marrow-derived and cultured mesenchymal stem cells. Bone 50, 325–330 (2012).
Levi, B. et al. Human adipose derived stromal cells heal critical size mouse calvarial defects. PLoS ONE 5, e11177 (2010).
Levi, B. et al. Dura mater stimulates human adipose-derived stromal cells to undergo bone formation in mouse calvarial defects. Stem Cells 29, 1241–1255 (2011).
Cowan, C. M. et al. Adipose-derived adult stromal cells heal critical-size mouse calvarial defects. Nat. Biotechnol. 22, 560–567 (2004).
Boerckel, J. D., Uhrig, B. A., Willett, N. J., Huebsch, N. & Guldberg, R. E. Mechanical regulation of vascular growth and tissue regeneration in vivo. Proc. Natl Acad. Sci. USA 108, E674–E680 (2011).
Caplan, A. I. & Dennis, J. E. Mesenchymal stem cells as trophic mediators. J. Cell. Biochem. 98, 1076–1084 (2006).
Dong, F. & Caplan, A. I. Cell transplantation as an initiator of endogenous stem cell-based tissue repair. Curr. Opin. Organ Transplant. 17, 670–674 (2012).
Eshkar-Oren, I. et al. The forming limb skeleton serves as a signaling center for limb vasculature patterning via regulation of Vegf. Development 136, 1263–1272 (2009).
Okuyama, H. et al. Expression of vascular endothelial growth factor receptor 1 in bone marrow-derived mesenchymal cells is dependent on hypoxia-inducible factor 1. J. Biol. Chem. 281, 15554–15563 (2006).
Kubo, S. et al. Blocking vascular endothelial growth factor with soluble Flt-1 improves the chondrogenic potential of mouse skeletal muscle-derived stem cells. Arthritis Rheum. 60, 155–165 (2009).
Reichert, J. C. et al. A tissue engineering solution for segmental defect regeneration in load-bearing long bones. Sci. Transl Med. 4, 141ra93 (2012).
Springer, I. N. et al. Bone graft versus BMP-7 in a critical size defect—cranioplasty in a growing infant model. Bone 37, 563–569 (2005).
Wilson, S. M. et al. Adipose-derived mesenchymal stem cells enhance healing of mandibular defects in the ramus of swine. J. Oral Maxillofac. Surg. 70, E193–E203 (2012).
Au, P., Tam, J., Fukumura, D. & Jain, R. K. Bone marrow-derived mesenchymal stem cells facilitate engineering of long-lasting functional vasculature. Blood 111, 4551–4558 (2008).
Correia, C. et al. In vitro model of vascularized bone: synergizing vasculogenesis and osteogenesis. PLoS ONE 6, e28352 (2011).
Correia, C. et al. Human adipose-derived cells can serve as a single-cell source for the in vitro cultivation of vascularized bone grafts. J. Tissue Eng. Regen. Med. 8, 629–639 (2012).
Tsigkou, O. et al. Engineered vascularized bone grafts. Proc. Natl Acad. Sci. USA 107, 3311–3316 (2010).
Rivron, N. C. et al. Sonic hedgehog-activated engineered blood vessels enhance bone tissue formation. Proc. Natl Acad. Sci. USA 109, 4413–4418 (2012).
Fuchs, S., Hofmann, A. & Kirkpatrick, C. J. Microvessel-like structures from outgrowth endothelial cells from human peripheral blood in 2-dimensional and 3-dimensional co-cultures with osteoblastic lineage cells. Tissue Eng. 13, 2577–2588 (2007).
Martineau, L. & Doillon, C. J. Angiogenic response of endothelial cells seeded dispersed versus on beads in fibrin gels. Angiogenesis 10, 269–277 (2007).
Laib, A. M. et al. Spheroid-based human endothelial cell microvessel formation in vivo. Nat. Protoc. 4, 1202–1215 (2009).
Alajati, A. et al. Spheroid-based engineering of a human vasculature in mice. Nat. Methods 5, 439–445 (2008).
Hutton, D. L., Moore, E. M., Gimble, J. & Grayson, W. L. Platelet-derived growth factor and spatiotemporal cues induce development of vascularized bone tissue by adipose-derived stem cells. Tissue Eng. Part A 19, 2076–2086 (2013).
Mehta, M., Schmidt-Bleek, K., Duda, G. N. & Mooney, D. J. Biomaterial delivery of morphogens to mimic the natural healing cascade in bone. Adv. Drug Deliv. Rev. 64, 1257–1276 (2012).
Kaigler, D., Silva, E. A. & Mooney, D. J. Guided bone regeneration using injectable vascular endothelial growth factor delivery gel. J. Periodontol. 84, 230–238 (2013).
Martino, M. M. et al. Engineering the growth factor microenvironment with fibronectin domains to promote wound and bone tissue healing. Sci. Transl Med. 3, 100ra89 (2011).
Shah, N. J. et al. Tunable dual growth factor delivery from polyelectrolyte multilayer films. Biomaterials 32, 6183–6193 (2011).
Shah, N. J. et al. Surface-mediated bone tissue morphogenesis from tunable nanolayered implant coatings. Sci. Transl Med. 5, 191ra183 (2013).
Spiess, B. D. Perfluorocarbon emulsions as a promising technology: a review of tissue and vascular gas dynamics. J. Appl. Physiol. 106, 1444–1452 (2009).
Kimelman-Bleich, N. et al. The use of a synthetic oxygen carrier-enriched hydrogel to enhance mesenchymal stem cell-based bone formation in vivo. Biomaterials 30, 4639–4648 (2009).
Kelly, B. D. et al. Cell type-specific regulation of angiogenic growth factor gene expression and induction of angiogenesis in nonischemic tissue by a constitutively active form of hypoxia-inducible factor 1. Circ. Res. 93, 1074–1081 (2003).
Bosch-Marce, M. et al. Effects of aging and hypoxia-inducible factor-1 activity on angiogenic cell mobilization and recovery of perfusion after limb ischemia. Circ. Res. 101, 1310–1318 (2007).
Wang, Y. et al. The hypoxia-inducible factor a pathway couples angiogenesis to osteogenesis during skeletal development. J. Clin. Invest. 117, 1616–1626 (2007).
Wang, Y., Wan, C., Gilbert, S. R. & Clemens, T. L. Oxygen sensing and osteogenesis. Ann. NY Acad. Sci. 1117, 1–11 (2007).
Schipani, E., Maes, C., Carmeliet, G. & Semenza, G. L. Regulation of osteogenesis-angiogenesis coupling by HIFs and VEGF. J. Bone Miner. Res. 24, 1347–1353 (2009).
Hu, X. et al. Leptin signaling is required for augmented therapeutic properties of mesenchymal stem cells conferred by hypoxia preconditioning. Stem Cells 32, 2702–2713 (2014).
Jiang, Q. et al. Remote ischemic postconditioning enhances cell retention in the myocardium after intravenous administration of bone marrow mesenchymal stromal cells. J. Mol. Cell. Cardiol. 56, 1–7 (2013).
Carlier, A. et al. Mosaic: a multiscale model of osteogenesis and sprouting angiogenesis with lateral inhibition of endothelial cells. PLoS Comput. Biol. 8, e10022724 (2012).
Xie, C. et al. Structural bone allograft combined with genetically engineered mesenchymal stem cells as a novel platform for bone tissue engineering. Tissue Eng. 13, 435–445 (2007).
Huang, C., Tang, M., Yehling, E. & Zhang, X. Overexpressing sonic hedgehog peptide restores periosteal bone formation in a murine bone allograft transplantation model. Mol. Ther. 22, 430–439 (2014).
Rosen, V. Harnessing the parathyroid hormone, Wnt, and bone morphogenetic protein signaling cascades for successful bone tissue engineering. Tissue Eng. Part B Rev. 17, 475–479 (2011).
Bleich, N. K. et al. Gene therapy approaches to regenerating bone. Adv. Drug Deliv. Rev. 64, 1320–1330 (2012).
Hung, B. P., Babalola, O. M. & Bonassar, L. J. Quantitative characterization of mesenchymal stem cell adhesion to the articular cartilage surface. J. Biomed. Mater. Res. A. 101, 3592–3598 (2013).
Vangsness, C. T. et al. Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy. J. Bone Joint Surg. Am. 96, 90–98 (2014).
Sart, S., Tsai, A. C., Li, Y. & Ma, T. Three-dimensional aggregates of mesenchymal stem cells: cellular mechanisms, biological properties, and applications. Tissue Eng. Part B 20, 365–380 (2014).
Sart, S., Ma, T. & Li, Y. Preconditioning stem cells for in vivo delivery. Biores. Open Access 3, 137–149 (2014).
Harrison, B. S., Eberli, D., Lee, S. J., Atala, A. & Yoo, J. J. Oxygen producing biomaterials for tissue regeneration. Biomaterials 28, 4628–4634 (2007).
Oh, S. H., Ward, C. L., Atala, A., Yoo, J. J. & Harrison, B. S. Oxygen generating scaffolds for enhancing engineered tissue survival. Biomaterials 30, 757–762 (2009).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2011).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2011).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2008).
US National Library of Medicine. ClinicalTrials.gov[online], (2010).
US National Library of Medicine. ClinicalTrials.gov[online], (2011).
US National Library of Medicine. ClinicalTrials.gov[online], (2009).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
Acknowledgements
The authors acknowledge funding from the Maryland Stem Cell Research Fund (2014-MSCRFI-0699), NSF CAREER award (CBET 1350554), Johns Hopkins University Centre for Musculoskeletal Research, and the American Society for Bone and Mineral Research (2013CEA13). The authors also thank D. Hutton for assistance with the artwork.
Author information
Authors and Affiliations
Contributions
W.L.G., B.P.H. and J.M.G. researched data for the article and wrote the article. W.L.G., B.A.B., E.M., T.F. and J.M.G. made substantial contributions to discussions of the content. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.M.G. is co-founder, co-owner and Chief Scientific Officer of LaCell, a biotechnology company focusing on the clinical translation of stromal-cell and stem-cell science. The other authors declare no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Grayson, W., Bunnell, B., Martin, E. et al. Stromal cells and stem cells in clinical bone regeneration. Nat Rev Endocrinol 11, 140–150 (2015). https://doi.org/10.1038/nrendo.2014.234
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2014.234
This article is cited by
-
Therapeutic application of regeneration-associated cells: a novel source of regenerative medicine
Stem Cell Research & Therapy (2023)
-
Cell unit-inspired natural nano-based biomaterials as versatile building blocks for bone/cartilage regeneration
Journal of Nanobiotechnology (2023)
-
Exosomal Lnc NEAT1 from endothelial cells promote bone regeneration by regulating macrophage polarization via DDX3X/NLRP3 axis
Journal of Nanobiotechnology (2023)
-
Bone marrow-derived dedifferentiated fat cells exhibit similar phenotype as bone marrow mesenchymal stem cells with high osteogenic differentiation and bone regeneration ability
Journal of Orthopaedic Surgery and Research (2023)
-
Harnessing electromagnetic fields to assist bone tissue engineering
Stem Cell Research & Therapy (2023)