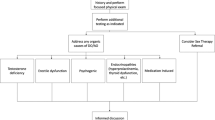
Key Points
-
Endometriosis is characterized by the presence of ectopic endometrium causing pain, infertility or lesion progression; it affects ∼5% of women of reproductive age, with a prevalence peak between 25 years and 35 years of age
-
Interaction of the number and amount of menstrual flows with genetic and environmental factors seems to determine the likelihood of development as well as the phenotypic manifestation of the disease
-
Although pain can be managed via pharmacological inhibition of ovulation and menstruation, lesions are not eradicated; surgery is generally associated with pain relief, but its benefit is often temporary
-
Medical therapy for infertility is inefficacious, whereas laparoscopic elimination of endometriotic lesions and adnexal adhesions increases the chances of conception moderately; in vitro fertilization is a valid alternative to surgery
-
Endometriosis is associated with a 50% increase in the risk of ovarian cancer; preventive interventions are possible, but screening of patients with endometriosis for ovarian cancer is presently not justified
-
Primary prevention of endometriosis is not currently feasible; treatment should be tailored to fit individual needs, and a shared decision-making approach between patient and clinician is encouraged
Abstract
Endometriosis is defined as the presence of endometrial-type mucosa outside the uterine cavity. Of the proposed pathogenic theories (retrograde menstruation, coelomic metaplasia and Müllerian remnants), none explain all the different types of endometriosis. According to the most convincing model, the retrograde menstruation hypothesis, endometrial fragments reaching the pelvis via transtubal retrograde flow, implant onto the peritoneum and abdominal organs, proliferate and cause chronic inflammation with formation of adhesions. The number and amount of menstrual flows together with genetic and environmental factors determines the degree of phenotypic expression of the disease. Endometriosis is estrogen-dependent, manifests during reproductive years and is associated with pain and infertility. Dysmenorrhoea, deep dyspareunia, dyschezia and dysuria are the most frequently reported symptoms. Standard diagnosis is carried out by direct visualization and histologic examination of lesions. Pain can be treated by excising peritoneal implants, deep nodules and ovarian cysts, or inducing lesion suppression by abolishing ovulation and menstruation through hormonal manipulation with progestins, oral contraceptives and gonadotropin-releasing hormone agonists. Medical therapy is symptomatic, not cytoreductive; surgery is associated with high recurrence rates. Although lesion eradication is considered a fertility-enhancing procedure, the benefit on reproductive performance is moderate. Assisted reproductive technologies constitute a valid alternative. Endometriosis is associated with a 50% increase in the risk of epithelial ovarian cancer, but preventive interventions are feasible.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Giudice, L. C. Clinical practice. Endometriosis. N. Engl. J. Med. 362, 2389–2398 (2010).
Eskenazi, B. & Warner, M. L. Epidemiology of endometriosis. Obstet. Gynecol. Clin. North Am. 24, 235–258 (1997).
Missmer, S. A. & Cramer, D. W. The epidemiology of endometriosis. Obstet. Gynecol. Clin. North Am. 30, 1–19 (2003).
Parazzini, F., Vercellini, P. & Pelucchi, C. in Endometriosis science and practice (eds Giudice, L. C., Johannes, L. H. & Healy, D. L.) 19–26 (Wiley-Blackwell Publishing, 2012).
Viganò, P., Parazzini, F., Somigliana, E. & Vercellini, P. Endometriosis: epidemiology and aetiological factors. Best Pract. Res. Clin. Obstet. Gynaecol. 18, 177–200 (2004).
Holt, V. L. & Weiss, N. S. Recommendations for the design of epidemiologic studies of endometriosis. Epidemiology 11, 654–659 (2000).
Zondervan, K. T., Cardon, L. R. & Kennedy, S. H. What makes a good case–control study? Design issues for complex traits such as endometriosis. Hum. Reprod. 17, 1415–1423 (2002).
Gylfason, J. T. et al. Pelvic endometriosis diagnosed in an entire nation over 20 years. Am. J. Epidemiol. 172, 237–243 (2013).
Janssen, E. B., Rijkers, A. C. M., Hoppenbrouwers, K., Meuleman, C. & D'Hooghe, T. M. Prevalence of endometriosis diagnosed by laparoscopy in adolescents with dysmenorrhea or chronic pelvic pain: a systematic review. Hum Reprod Update 19, 570–582 (2013).
Simoens, S. et al. The burden of endometriosis: costs and quality of life of women with endometriosis and treated in referral centres. Hum. Reprod. 27, 1292–1299 (2012).
Nnoaham, K. E. et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil. Steril. 96, 366–373 (2011).
Mirkin, D., Murphy-Barron, C. & Iwasaki, K. Actuarial analysis of private payer administrative claims data for women with endometriosis. J. Manag. Care Pharm. 13, 262–272 (2007).
Levy, A. R. et al. Economic burden of surgically confirmed endometriosis in Canada. J. Obstet. Gynaecol. Can. 33, 830–837 (2011).
Burney, R. O. & Giudice, L. C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 98, 511–519 (2012).
Cramer, D. W. et al. The relation of endometriosis to menstrual characteristics, smoking, and exercise. JAMA 255, 1904–1908 (1986).
Frisch, R. E., Wyshak, G., Albert, L. S. & Sober, A. J. Dysplastic nevi, cutaneous melanoma, and gynecologic disorders. Int. J. Dermatol. 31, 331–335 (1992).
Darrow, S. L. et al. Menstrual cycle characteristics and the risk of endometriosis. Epidemiology 4, 135–142 (1993).
Moen, M. H. & Magnus, P. The familial risk of endometriosis. Acta Obstet. Gynecol. Scand. 72, 560–564 (1993).
McCann, S. E., Freudenheim, J. L., Darrow, S. L., Batt, R. E. & Zielezny, M. A. Endometriosis and body fat distribution. Obstet. Gynecol. 82, 545–549 (1993).
Parazzini, F. et al. Pelvic endometriosis: reproductive and menstrual risk factors at different stages in Lombardy, northern Italy. J. Epidemiol. Commun. Health 49, 61–64 (1995).
Sangi-Haghpeykar, H. & Poindexter, A. N. 3rd Epidemiology of endometriosis among parous women. Obstet. Gynecol. 85, 983–992 (1995).
Hornstein, M. D. et al. Association between endometriosis, dysplastic naevi and history of melanoma in women of reproductive age. Hum. Reprod. 12, 143–145 (1997).
Signorello, L. B., Harlow, B. L., Cramer, D. W., Spiegelman, D. & Hill, J. A. Epidemiologic determinants of endometriosis: a hospital-based case–control study. Ann. Epidemiol. 7, 267–274 (1997).
Bérubé, S., Marcoux. S. & Maheux, R. Characteristics related to the prevalence of minimal or mild endometriosis in infertile women. Canadian Collaborative Group on Endometriosis. Epidemiology 9, 504–510 (1998).
Stefansson, H. et al. Genetic factors contribute to the risk of developing endometriosis. Hum. Reprod. 17, 555–559 (2002).
Hemmings, R. et al. Evaluation of risk factors associated with endometriosis. Fertil. Steril. 81, 1513–1521 (2004).
Missmer, S. A. et al. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am. J. Epidemiol. 160, 784–796 (2004).
Ferrero, S., Anserini, P., Remorgida, V. & Ragni, N. Body mass index in endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 121, 94–98 (2005).
Hediger, M. L., Hartnett, H. J. & Louis, G. M. Association of endometriosis with body size and figure. Fertil. Steril. 84, 1366–1374 (2005).
Kvaskoff. M., Mesrine. S., Clavel-Chapel, F. & Boutron-Ruault, M. C. Endometriosis risk in relation to naevi, freckles and skin sensitivity to sun exposure: the French E3N cohort. Int. J. Epidemiol. 38, 1143–1153 (2009).
Somigliana, E. et al. 'Here comes the sun': pigmentary traits and sun habits in women with endometriosis. Hum. Reprod. 25, 728–733 (2010).
Vitonis, A. F., Hankinson, S. E., Hornstein, M. D. & Missmer, S. A. Adult physical activity and endometriosis risk. Epidemiology 21, 16–23 (2010).
Viganò, P. et al. Principles of phenomics in endometriosis. Hum. Reprod. Update. 18, 248–259 (2012).
Parazzini, F., Viganò, P., Candiani, M. & Fedele L. Diet and endometriosis risk: a literature review. Reprod. Biomed. Online 26, 323–336 (2013).
Lafay Pillet, M. C. et al. Deep infiltrating endometriosis is associated with markedly lower body mass index: a 476 case–control study. Hum. Reprod. 27, 265–272 (2012).
Shah, D. K., Correia, K. F., Vitonis, A. F. & Missmer, S. A. Body size and endometriosis: results from 20 years of follow-up within the Nurses' Health Study II prospective cohort. Hum. Reprod. 28, 1783–1792 (2013).
Parazzini F. et al. A metaanalysis on alcohol consumption and risk of endometriosis. Am. J. Obstet. Gynecol. 209, 106.e1–10 (2013).
Noble, L. S., Simpson, E. R., Johns, A. & Bulun, S. E. Aromatase expression in endometriosis. J. Clin. Endocrinol. Metab. 81, 174–179 (1996).
Noble, L. S. et al. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J. Clin. Endocrinol. Metab. 82, 600–606 (1997).
Hornung, D. et al. Immunolocalization and regulation of the chemokine RANTES in human endometrial and endometriosis tissues and cells. J. Clin. Endocrinol. Metab. 82, 1621–1628 (1997).
Zeitoun, K. et al. Deficient 17β-hydroxysteroid dehydrogenase type 2 expression in endometriosis: failure to metabolize 17β-estradiol. J. Clin. Endocrinol. Metab. 83, 4474–4480 (1998).
Chung, H. W. et al. Matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-3 mRNA expression in ectopic and eutopic endometrium in women with endometriosis: a rationale for endometriotic invasiveness. Fertil. Steril. 75, 152–159 (2001).
Chung, H. W. et al. Matrix metalloproteinase-2, membranous type 1 matrix metalloproteinase, and tissue inhibitor of metalloproteinase-2 expression in ectopic and eutopic endometrium. Fertil. Steril. 78, 787–795 (2002).
Defrère, S. et al. Iron overload enhances epithelial cell proliferation in endometriotic lesions induced in a murine model. Hum. Reprod. 21, 2810–2816 (2006).
Xue, Q. et al. Promoter methylation regulates estrogen receptor 2 in human endometrium and endometriosis. Biol. Reprod. 77, 681–687 (2007).
González-Ramos, R. et al. Nuclear factor-kappa B is constitutively activated in peritoneal endometriosis. Mol. Hum. Reprod. 13, 503–509 (2007).
Burney, R. O. et al. Gene expression analysis of endometrium reveals progesterone resistance and candidate susceptibility genes in women with endometriosis. Endocrinology 148, 3814–3826 (2007).
Bukulmez, O., Hardy, D. B., Carr, B. R., Word, R. A. & Mendelson, C. R. Inflammatory status influences aromatase and steroid receptor expression in endometriosis. Endocrinology 149, 1190–1204 (2008).
Defrère, S. et al. Potential involvement of iron in the pathogenesis of peritoneal endometriosis. Mol. Hum. Reprod. 14, 377–385 (2008).
Yu, J. et al. Combination of estrogen and dioxin is involved in the pathogenesis of endometriosis by promoting chemokine secretion and invasion of endometrial stromal cells. Hum. Reprod. 23, 1614–1626 (2008).
Smuc, T. et al. Disturbed estrogen and progesterone action in ovarian endometriosis. Mol. Cell. Endocrinol. 301, 59–64 (2009).
Lousse, J. C. et al. Iron storage is significantly increased in peritoneal macrophages of endometriosis patients and correlates with iron overload in peritoneal fluid. Fertil. Steril. 91, 1668–1675 (2009).
Bulun, S. E. Endometriosis. N. Engl. J. Med. 360, 268–279 (2009).
Bacci, M. et al. Macrophages are alternatively activated in patients with endometriosis and required for growth and vascularization of lesions in a mouse model of disease. Am. J. Pathol. 175, 547–556 (2009).
Aghajanova, L., Velarde, M. C. & Giudice, L. C. The progesterone receptor coactivator Hic-5 is involved in the pathophysiology of endometriosis. Endocrinology 369–371, 3863–3870 (2009).
González-Ramos, R. et al. Involvement of the nuclear factor-κB pathway in the pathogenesis of endometriosis. Fertil. Steril. 94, 1985–1994 (2010).
Capobianco, A. et al. Proangiogenic Tie2(+) macrophages infiltrate human and murine endometriotic lesions and dictate their growth in a mouse model of the disease. Am. J. Pathol. 179, 2651–2659 (2011).
Yin, X., Pavone, M. E., Lu, Z., Wei, J. & Kim, J. J. Increased activation of the PI3K/AKT pathway compromises decidualization of stromal cells from endometriosis. J. Clin. Endocrinol. Metab. 97, E35–E43 (2012).
Khanjani, S., Al-Sabbagh, M. K., Fusi, L. & Brosens, L. in Endometriosis Science and Practice (eds Giudice, L. C., Johannes, L. H. & Healy, D. L.) 145–152 (Wiley-Blackwell Publishing, 2012).
Capobianco, A. & Rovere-Querini, P. Endometriosis, a disease of the macrophage. Front. Immunol. 4, 1–13 (2013).
Reis, F. M., Petraglia, F. & Taylor, R. N. Endometriosis: hormone regulation and clinical consequences of chemotaxis and apoptosis. Hum. Reprod. Update 19, 406–418 (2013).
Vercellini, P. et al. Post-operative endometriosis recurrence: a plea for prevention based on pathogenetic, epidemiological and clinical evidence. Reprod. Biomed. Online 21, 259–265 (2010).
Nyholt, D. R. et al. Genome-association meta-analysis identifies new endometriosis risk loci. Nat. Genet. 44, 1355–1359 (2012).
Pagliardini, L. et al. An Italian association study and meta-analysis with previous GWAS confirm WNT4, CDKN2BAS and FN1 as the first identified susceptibility loci for endometriosis. J. Med. Genet. 50, 43–46 (2013).
Bulun, S. E. et al. Role of estrogen receptor-β in endometriosis. Semin. Reprod. Med. 30, 39–45 (2012).
Aghajanova, L., Hamilton, A., Kwintkiewicz, J., Vo, K. C. & Guidice, L. C. Steroidogenic enzyme and key decidualization marker dysregulation in endometrial stromal cells from women with versus without endometriosis. Biol. Reprod. 80, 105–114 (2009).
Wei, Q., St Clair, J. B., Fu, T., Stratton, P. & Nieman, L. K. Reduced expression of biomarkers associated with the implantation window in women with endometriosis. Fertil. Steril. 91, 1686–1691 (2009).
Monsivais, D. et al. Activated glucocorticoid and eicosanoid pathways in endometriosis. Fertil. Steril. 98, 117–125 (2012).
Kyama, C. M. et al. Endometrial and peritoneal expression of aromatase, cytokines, and adhesion factors in women with endometriosis. Fertil Steril. 89, 301–310 (2008).
Lousse, J. C. et al. Increased activation of nuclear factor-kappa B (NF-kappaB) in isolated peritoneal macrophages of patients with endometriosis. Fertil. Steril. 90, 217–220 (2008).
Hagemann, T. et al. “Re-educating” tumor-associated macrophages by targeting NF-kappaB. J. Exp. Med. 205, 1261–1268 (2008).
Recalcati, S. et al. Differential regulation of iron homeostasis during human macrophage polarized activation. Eur. J. Immunol. 40, 824–835 (2010).
Nisolle, M. & Donnez, J. Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil. Steril. 68, 585–596 (1997).
Vercellini, P. et al. Deep endometriosis: definition, pathogenesis, and clinical management. J. Am. Assoc. Gynecol. Laparosc. 11, 153–161 (2004).
Brosens, I. A., Puttemans, P. J. & Deprest, J. The endoscopic localization of endometrial implants in the ovarian chocolate cyst. Fertil. Steril. 61, 1034–1038 (1994).
Brosens, I. A., Van Ballaer, P., Puttemans, P. & Deprest, J. Reconstruction of the ovary containing large endometriomas by an extraovarian endosurgical technique. Fertil. Steril. 66, 517–521 (1996).
Jain, S. & Dalton, M. E. Chocolate cysts from ovarian follicles. Fertil. Steril. 72, 852–856 (1999).
Vercellini, P. et al. “Blood On The Tracks” from corpora lutea to endometriosis. BJOG 116, 366–371 (2009).
Bricou, A., Batt, R. E. & Chapron, C. Peritoneal fluid flow influences anatomical distribution of endometriotic lesions: why Sampson seems to be right. Eur. J. Obstet. Gynecol. Reprod. Biol. 138, 127–134 (2008).
Vercellini, P. Endometriosis: What a pain it is. Semin. Reprod. Endocrinol. 15, 251–261 (1997).
Vercellini, P. et al. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis on 1000 patients. Hum. Reprod. 22, 266–271 (2007).
Ballard, K. D., Seaman, H. E., de Vries, C. S. & Wright, J. T. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case–control study—Part 1. BJOG 115, 1382–1391 (2008).
Fauconnier, A. & Chapron, C. Endometriosis and pelvic pain: epidemiological evidence of the relationship and implications. Hum. Reprod. Update 11, 595–606 (2005).
Fauconnier, A. et al. Relation between pain symptoms and the anatomic location of deep infiltrating endometriosis. Fertil. Steril. 78, 719–726 (2002).
Ferrero, S. et al. Quality of sex life in women with endometriosis and deep dyspareunia. Fertil. Steril. 83, 573–579 (2005).
Howard, F. M. Endometriosis and mechanisms of pelvic pain. J. Minim. Invasive Gynecol. 16, 540–550 (2009).
Anaf, V. et al. Relationship between endometriotic foci and nerves in rectovaginal endometriotic nodules. Hum. Reprod. 15, 1744–1750 (2000).
Anaf, V. et al. Hyperalgesia, nerve infiltration and nerve growth factor expression in deep adenomyotic nodules, peritoneal and ovarian endometriosis. Hum. Reprod. 17, 1895–1900 (2002).
Anaf, V. et al. Pain, mast cells, and nerves in peritoneal, ovarian, and deep infiltrating endometriosis. Fertil. Steril. 86, 1336–1343 (2006).
Giamberardino, M. A. et al. Modulation of pain and hyperalgesia from the urinary tract by algogenic conditions of the reproductive organs in women. Neurosci. Lett. 304, 61–64 (2001).
Bajaj, P., Bajaj, P., Madsen, H. & Arendt-Nielsen, L. Endometriosis is associated with central sensitization: a psychophysical controlled study. J. Pain 4, 372–380 (2003).
Berkley, K. J., Rapkin, A. J. & Papka, R. E. The pains of endometriosis. Science 308, 1587–1589 (2005).
Stratton, P. & Berkley, K. J. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum. Reprod. Update 17, 327–346 (2011).
Somigliana, E. et al. Non-invasive diagnosis of endometriosis: the goal or own goal? Hum. Reprod. 25, 1863–1868 (2010).
Eskenazi, B. et al. Validation study of nonsurgical diagnosis of endometriosis. Fertil. Steril. 76, 929–935 (2001).
Royal College of Obstetricians and Gynaecologists. The investigation and management of endometriosis. Green-top Guideline No. 24. October 2006 [online] (2006).
Johnson, N. P. & Hummelshoj, L. World Endometriosis Society Montpellier Consortium. Consensus on current management of endometriosis. Hum. Reprod. 28, 1552–1568 (2013).
Abrao, M. S. et al. Comparison between clinical examination, transvaginal sonography and magnetic resonance imaging for the diagnosis of deep endometriosis. Hum. Reprod. 22, 3092–3097 (2007).
Piketty, M. et al. Preoperative work-up for patients with deeply infiltrating endometriosis: transvaginal ultrasonography must definitely be the first-line imaging examination. Hum. Reprod. 24, 602–607 (2009).
Savelli, L. Transvaginal sonography for the assessment of ovarian and pelvic endometriosis: how deep is our understanding? Ultrasound Obstet. Gynecol. 33, 497–501 (2009).
Vercellini, P. et al. Surgery for deep endometriosis: a pathogenesis-oriented approach. Gynecol. Obstet. Invest. 68, 88–103 (2009).
Practice Committee of the American Society for Reproductive Medicine. Treatment of pelvic pain associated with endometriosis. Fertil. Steril. 90 (Suppl.), S260–S269 (2008).
[No authors listed] Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil. Steril. 67, 817–821 (1997).
Berlanda, N. et al. Role of surgery in endometriosis-associated subfertility. Semin. Reprod. Med. 31, 133–143 (2013).
Jacobs, T. Z., Duffy, J. M., Barlow, D., Koninckx, P. R. & Garry, R. Laparoscopic surgery for pelvic pain associated with endometriosis. Cochrane Database of Systematic Reviews, Issue 4. Art. No.: CD001300 http://dx.doi.org/10.1002/14651858.CD001300.pub2.
Vercellini, P. et al. The effect of surgery for symptomatic endometriosis: the other side of the story. Hum. Reprod. Update 15, 177–188 (2009).
Vercellini, P. et al. Repetitive surgery for recurrent symptomatic endometriosis: what to do? Eur. J. Obstet. Gynecol. Reprod. Biol. 146, 15–21 (2009).
Wright, J., Lotfallah, H., Jones, K. & Lovell, D. A randomized trial of excision versus ablation for mild endometriosis. Fertil. Steril. 83, 1830–1836 (2005).
Healey, M., Ang, W. C. & Cheng, C. Surgical treatment of endometriosis: a prospective randomized double-blinded trial comparing excision and ablation. Fertil. Steril. 94, 2536–2540 (2010).
Radosa, M. P. et al. Coagulation versus excision of primary superficial endometriosis: a 2-year follow-up. Eur. J. Obstet. Gynecol. Reprod. Biol. 150, 195–198 (2010).
Hart, R. J., Hickey, M., Maouris, P. & Buckett, W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database of Systematic Reviews, Issue 2. Art. No.: CD004992. http://dx.doi.org/10.1002/14651858.CD004992.pub3.
Tsolakidis, D. et al. The impact on ovarian reserve after laparoscopic ovarian cystectomy versus three-stage management in patients with endometriomas: a prospective randomized study. Fertil. Steril. 94, 71–77 (2010).
Donnez, J., Lousse, J. C., Jadoul, P., Donnez, O. & Squifflet, J. Laparoscopic management of endometriomas using a combined technique of excisional (cystectomy) and ablative surgery. Fertil. Steril. 94, 28–32 (2010).
Benschop, L., Farquhar, C., van der Poel, N. & Heineman, M. J. Interventions for women with endometrioma prior to assisted reproductive technology. Cochrane Database of Systematic Reviews, Issue. 11. Art No.: CD008571. http://dx.doi.org/10.1002/14651858.CD008571.pub2.
Donnez, J. & Squifflet, J. Complications, pregnancy and recurrence in a prospective series of 500 patients operated on by the shaving technique for deep rectovaginal endometriotic nodules. Hum. Reprod. 25, 1949–1958 (2010).
Roman, H. et al. Bowel dysfunction before and after surgery for endometriosis. Am. J. Obstet. Gynecol. 209, 524–530 (2013).
Mohr, C., Nezhat, F. R., Nezhat, C. H., Seidman, D. S. & Nezhat, C. R. Fertility considerations in laparoscopic treatment of infiltrative bowel endometriosis. JSLS 9, 16–24 (2005).
Kondo, W. et al. Complications after surgery for deeply infiltrating pelvic endometriosis. BJOG 118, 292–298 (2011).
De Cicco, C. et al. Bowel resection for deep endometriosis: a systematic review. BJOG 118, 285–291 (2011).
Nezhat, C., Hajhosseini, B. & King, L. P. Laparoscopic management of bowel endometriosis: predictors of severe disease and recurrence. JSLS 15, 431–438 (2011).
Nezhat, C. et al. Robotic versus standard laparoscopy for the treatment of endometriosis. Fertil. Steril. 94, 2758–2760 (2010).
Ercoli, A. et al. Robotic treatment of colorectal endometriosis: technique, feasibility and short-term results. Hum. Reprod. 27, 722–726 (2012).
Vercellini, P. et al. Endometriosis: current and future medical therapies. Best Pract. Res. Clin. Obstet. Gynecol. 22, 275–306 (2008).
Vercellini, P. et al. Endometriosis: current therapies and new pharmacological developments. Drugs 69, 649–675 (2009).
Vercellini, P. et al. 'Waiting for Godot': a commonsense approach to the medical treatment of endometriosis. Hum. Reprod. 26, 3–13 (2011).
Razzi, S. et al. Efficacy of vaginal danazol treatment in women with recurrent deeply infiltrating endometriosis. Fertil. Steril. 88, 789–794 (2007).
Brown, J., Pan, A. & Hart, R. J. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database of Systematic Reviews, Issue 12. Art. No.: CD008475. http://dx.doi.org/10.1002/14651858.CD008475.pub2.
Vercellini, P. et al. Medical treatment for rectovaginal endometriosis: what is the evidence? Hum. Reprod. 24, 2504–2514 (2009).
Vercellini, P. et al. Treatment of symptomatic rectovaginal endometriosis with an estrogen-progestogen combination versus low-dose norethindrone acetate. Fertil. Steril. 84, 1375–1387 (2005).
Vercellini, P. et al. Surgical versus medical treatment for endometriosis-associated severe deep dyspareunia: I. Effect on pain during intercourse and patient satisfaction. Hum. Reprod. 27, 3450–3459 (2012).
Ferrero, S. et al. Letrozole combined with norethisterone acetate compared with norethisterone acetate alone in the treatment of pain symptoms caused by endometriosis. Hum. Reprod. 24, 3033–3041 (2009).
Kaser, D. J., Missmer, S. A., Berry, K. F. & Laufer, M. R. Use of norethindrone acetate alone for postoperative suppression of endometriosis symptoms. J. Pediatr. Adolesc. Gynecol. 25, 105–108 (2012).
Harada, T. et al. Dienogest is as effective as intranasal buserelin acetate for the relief of pain symptoms associated with endometriosis: a randomized, double-blind, multicenter, controlled trial. Fertil. Steril. 91, 675–681 (2009).
Strowitzki, T., Marr, J., Gerlinger, C., Faustmann, T. & Seitz, C. Dienogest is as effective as leuprolide acetate in treating the painful symptoms of endometriosis: a 24-week, randomized, multicentre, open-label trial. Hum. Reprod. 25, 633–641 (2010).
Chu, M. C., Zhang, X., Gentzschein, E., Stanczyk, F. Z. & Lobo, R. A. Formation of ethinyl estradiol in women during treatment with norethindrone acetate. J. Clin. Endocrinol. Metab. 92, 2205–2207 (2007).
Abou-Setta, A. M., Houston, B., Al-Inany, H. G. & Farguhar, C. Levonorgestrel-releasing intrauterine device (LNG-IUD) for symptomatic endometriosis following surgery. Cochrane Database of Systematic Reviews, Issue. 1. Art No.: CD005072. http://dx.doi.org/10.1002/14651858.CD005072.pub3.
De Ziegler, D., Borghese, B. & Chapron, C. Endometriosis and infertility: pathophysiology and management. Lancet 376, 730–738 (2010).
Hughes, E. et al. Ovulation suppression for endometriosis for women with subfertility. Cochrane Database of Systematic Reviews, Issue 3. Art No.: CD000155. http://dx.doi.org/10.1002/14651858.CD000155.pub2.
Jacobson, T. Z. et al. Laparoscopic surgery for subfertility associated with endometriosis. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD001398. http://dx.doi.org/10.1002/14651858.CD001398.pub2.
Vercellini, P. et al. Surgery for endometriosis-associated infertility: a pragmatic approach. Hum. Reprod. 24, 254–269 (2009).
Tanahatoe, S. J., Lambalk, C. B. & Hompes, P. G. The role of laparoscopy in intrauterine insemination: a prospective randomized reallocation study. Hum. Reprod. 20, 3225–3230 (2005).
Vercellini, P. et al. Effect of patient selection on estimate of reproductive success after surgery for rectovaginal endometriosis: literature review. Reprod. Biomed. Online 24, 389–395 (2012).
Vercellini, P. et al. Reproductive performance in infertile women with rectovaginal endometriosis: Is surgery worthwhile? Am. J. Obstet. Gynecol. 195, 1303–1310 (2006).
Moen, M. H. & Stokstad, T. A long-term follow-up study of women with asymptomatic endometriosis diagnosed incidentally at sterilization. Fertil. Steril. 78, 773–776 (2002).
Matorras, R. et al. Fertility in women with minimal endometriosis compared with normal women was assessed by means of a donor insemination program in unstimulated cycles. Am. J. Obstet. Gynecol. 203, 345.e1–6 (2010).
Tummon, I. S., Asher, L. J., Martin, J. S. & Tulandi, T. Randomized controlled trial of superovulation and insemination for infertility associated with minimal or mild endometriosis. Fertil. Steril. 68, 8–12 (1997).
Deaton, J. L. et al. A randomized, controlled trial of clomiphene citrate and intrauterine insemination in couples with unexplained infertility or surgically corrected endometriosis. Fertil. Steril. 54, 1083–1088 (1990).
Barnhart, K., Dunsmoor-Su, R. & Coutifaris, C. Effect of endometriosis on in vitro fertilization. Fertil. Steril. 277, 1148–1155 (2002).
Harb, H., Gallos, I., Chu, J., Harb, M. & Coomarasamy, A. The effect of endometriosis on in vitro fertilisation outcome: a systematic review and meta-analysis. BJOG 120, 1308–1320 (2013).
Sallam, H. N., Garcia-Velasco, J. A., Dias, S. & Arici, A. Long-term pituitary down-regulation before in vitro fertilization (IVF) for women with endometriosis. Cochrane Database of Systematic Reviews, Issue. 1. Art No.: CD004635. http://dx.doi.org/10.1002/14651858.CD004635.pub2.
De Ziegler, D. et al. Use of oral contraceptives in women with endometriosis before assisted reproduction treatment improves outcomes. Fertil. Steril. 94, 2796–2799 (2010).
Littman, E. et al. Role of laparoscopic treatment of endometriosis in women with failed in vitro fertilization cycles. Fertil. Steril. 84, 1574–1578 (2005).
Somigliana, E. et al. Surgical excision of endometriomas and ovarian reserve: a systematic review on serum antimüllerian hormone level modifications. Fertil. Steril. 98, 1531–1538 (2012).
Raffi, F., Metwally, M. & Amer, S. The impact of excision of ovarian endometrioma on ovarian reserve: a systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 97, 3146–3154 (2012).
Uncu, G. et al. Prospective assessment of the impact of endometriomas and their removal on ovarian reserve and determinants of the rate of decline in ovarian reserve. Hum. Reprod. 28, 2140–2145 (2013).
Busacca, M. et al. Postsurgical ovarian failure after laparoscopic excision of bilateral endometriomas. Am. J. Obstet. Gynecol. 195, 421–425 (2006).
Somigliana, E. et al. IVF-ICSI outcome in women operated on for bilateral endometriomas. Hum. Reprod. 23, 1526–1530 (2008).
Garcia-Velasco, J. A. & Somigliana, E. Management of endometriomas in women requiring IVF: to touch or not to touch. Hum. Reprod. 24, 496–501 (2009).
Van Voorhis, B. J. Clinical practice. In vitro fertilization. N. Engl. J. Med. 356, 379–386 (2007).
Canis, M., Mage, G., Wattiez, A., Pouly J. L. & Bruhat, M. A. The ovarian endometrioma: why is it so poorly managed? Laparoscopic treatment of large ovarian endometrioma: why such a long learning curve? Hum. Reprod. 18, 5–7 (2003).
Soares, S. R., Martinez-Varea, A., Hidalgo-Mora, J. J. & Pellicer, A. Pharmacologic therapies in endometriosis: a systematic review. Fertil. Steril. 98, 529–555 (2012).
Guo, S. W. et al. A call for more transparency of registered clinical trials on endometriosis. Hum. Reprod. 24, 1247–1254 (2009).
Soysal, S., Soysal, M. E., Ozer, S., Gul, N. & Gezgin, T. The effects of post-surgical administration of goserelin plus anastrozole compared to goserelin alone in patients with severe endometriosis: a prospective randomized trial. Hum. Reprod. 19, 160–167 (2004).
Alborzi, S., Hamedi, B., Omidvar, A., Dehbashi, S. & Alborzi, M. A comparison of the effect of short-term aromatase inhibitor (letrozole) and GnRH agonist (triptorelin) versus case control on pregnancy rate and symptom and sign recurrence after laparoscopic treatment of endometriosis. Arch. Gynecol. Obstet. 284, 105–110 (2011).
Stratton, P. et al. Return of chronic pelvic pain from endometriosis after raloxifene treatment: a randomized controlled trial. Obstet. Gynecol. 111, 88–96 (2008).
Cobellis L. et al. The treatment with a COX-2 specific inhibitor is effective in the management of pain related to endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 116, 100–102 (2004).
Balasch, J. et al. Pentoxifylline versus placebo in the treatment of infertility associated with minimal or mild endometriosis: a pilot randomized clinical trial. Hum. Reprod. 12, 2046–2050 (1997).
Alborzi, S., Ghotbi, S., Parsanezhad, M. E., Dehbashi, S. & Alborzi, M. Pentoxifylline therapy after laparoscopic surgery for different stages of endometriosis: a prospective, double-blind, randomized, placebo-controlled study. J. Minimal Invasive Gynecol. 14, 54–58 (2007).
Kamencic, H. & Thiel, J. A. Pentoxyfilline after conservative surgery for endometriosis: a randomized, controlled trial. J. Minimal Invasive Gynecol. 15, 62–66 (2008).
Acién, P. et al. Use of intraperitoneal interferon α-2b therapy after conservative surgery for endometriosis and postoperative medical treatment with depot gonadotropin-releasing hormone analog: a randomized clinical trial. Fertil. Steril. 78, 705–711 (2002).
Acién, P. et al. GnRH analogues, transvaginal ultrasound-guided drainage and intracystic injection of recombinant interleukin-2 in the treatment of endometriosis. Gynecol. Obstet. Invest. 55, 96–104 (2003).
Koninckx, P. R., Craessaerts, M., Timmerman, D. Cornillie, F. & Kennedy, S. Anti-TNF-α treatment for deep endometriosis-associated pain: a randomized placebo-controlled trial. Hum. Reprod. 23, 2017–2023 (2008).
Guo, S. W. & Evers J. L. Lack of transparency of clinical trials on endometriosis. Obstet. Gynecol. 121, 1281–1290 (2013).
Guo, S. W. Recurrence of endometriosis and its control. Hum. Reprod. Update 15, 441–461 (2009).
Berlanda, N., Vercellini, P. & Fedele, L. The outcomes of repeat surgery for recurrent symptomatic endometriosis. Curr. Opin. Obstet. Gynecol. 22, 320–325 (2010).
Vercellini, P. et al. The effect of second-line surgery on reproductive performance of women with recurrent endometriosis: a systematic review. Acta Obstet. Gynecol. Scand. 88, 1074–1082 (2009).
Kennedy, S. et al. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum. Reprod. 20, 2698–2704 (2005).
Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility: a committee opinion. Fertil. Steril. 98, 591–598 (2012).
Seracchioli, R. et al. Long-term oral contraceptive pills and postoperative pain management after laparoscopic excision of ovarian endometrioma: a randomized controlled trial. Fertil. Steril. 94, 464–471 (2010).
Seracchioli, R. et al. Post-operative use of oral contraceptive pills for prevention of anatomical relapse or symptom-recurrence after conservative surgery for endometriosis. Hum. Reprod. 24, 2729–2735 (2009).
Vercellini, P. et al. Long-term adjuvant therapy for the prevention of postoperative endometrioma recurrence: a systematic review and meta-analysis. Acta Obstet. Gynecol. Scand. 92, 8–16 (2013).
Brinton, L. A., Gridley, G., Persson, I., Baron, J. & Bergqvist, A. Cancer risk after a hospital discharge diagnosis of endometriosis. Am. J. Obstet. Gynecol 176, 572–579 (1997).
Pearce, C. L. et al. Association between endometriosis and risk of histological subtypes of ovarian cancer: a pooled analysis of case–control studies. Lancet Oncol. 13, 385–394 (2012).
Kurman, R. J. et al. Papillary tubal hyperplasia: the putative precursor of ovarian atypical proliferative (borderline) serous tumors, noninvasive implants, and endosalpingiosis. Am. J. Surg. Pathol. 35, 1605–1614 (2011).
Nezhat, F. et al. The relationship of endometriosis and ovarian malignancy: a review. Fertil. Steril. 90, 1559–1570 (2008).
Wei, J. J., Williams, J. & Bulun, S. Endometriosis and ovarian cancer: a review of clinical, pathologic, and molecular aspects. Int. J. Gynecol. Pathol. 30, 553–68 (2011).
Vercellini, P., Somigliana E., Buggio L., Bolis G. & Fedele L. Endometriosis and ovarian cancer. Lancet Oncol. 13, e188–e189 (2012).
Vercellini, P. et al. The oncofetal protein IMP3: a novel biomarker and triage tool for premalignant atypical endometriotic lesions. Fertil. Steril. 99, 1974–1979 (2013).
Viganò P., Somigliana, E., Chiodo, I., Abbiati, A. & Vercellini, P. Molecular mechanisms and biological plausibility underlying the malignant transformation of endometriosis: a critical analysis. Hum. Reprod. Update 12, 77–89 (2006).
Kobayashi, H. et al. The role of hepatocyte nuclear factor-1β in the pathogenesis of clear cell carcinoma of the ovary. Int. J. Gynecol. Cancer 19, 471–479 (2009).
Vercellini, P. et al. The “incessant menstruation” hypothesis: a mechanistic ovarian cancer model with implications for prevention. Hum. Reprod. 26, 2262–2273 (2011).
Yamada, Y. et al. Redox-active iron-induced oxidative stress in the pathogenesis of clear cell carcinoma of the ovary. Int. J. Gynecol. Cancer 21, 1200–1207 (2011).
Wiegand, K. C. et al. ARID1A mutations in endometriosis-associated ovarian carcinomas. N. Engl. J. Med. 363, 1532–1543 (2010).
Maeda, D. & Shih, I. M. Pathogenesis and the role of ARID1A mutation in endometriosis-related ovarian neoplasms. Adv. Anat. Pathol. 20, 45–52 (2013).
Tanase, Y. et al. Modulation of estrogenic action in clear cell carcinoma of the ovary (Review). Exp. Ther. Med. 3, 18–24 (2012).
Kajihara, H., Yamada, Y., Shigetomi, H. Higashiura, Y & Kobayashi, H. The dichotomy in the histogenesis of endometriosis-associated ovarian cancer: clear cell-type versus endometrioid-type adenocarcinoma. Int. J. Gynecol. Pathol. 31, 304–312 (2012).
Llauradó M. et al. Molecular bases of endometrial cancer: New roles for new actors in the diagnosis and the therapy of the disease. Mol. Cell. Endocrinol. 358, 244–255 (2012).
Herrinton, L. J., Voigt, L. F., Weiss, N. S., Beresford, S. A. A. & Wingo, PA. Risk factors for synchronous primary endometrial and ovarian cancers. Ann. Epidemiol. 11, 529–533 (2001).
Mangili, G. et al. Unraveling the two entities of endometrioid ovarian cancer: a single center clinical experience. Gynecol. Oncol. 126, 403–407 (2012).
Liu, Y., Li, J., Jin, H., Lu, Y. & Lu, X. Clinicopathological characteristics of patients with synchronous primary endometrial and ovarian cancers: A review of 43 cases. Oncol. Lett. 5, 267–270 (2013).
Modugno, F. et al. Oral contraceptive use, reproductive history, and risk of epithelial ovarian cancer in women with and without endometriosis. Am. J. Obstet. Gynecol. 191, 733–740 (2004).
Rossing, M. A., Cushing-Haugen, K. L., Wicklund, K. G., Doherty, J. A. & Weiss, N. S. Risk of epithelial ovarian cancer in relation to benign ovarian conditions and ovarian surgery. Cancer Causes Control 19, 1357–1364 (2008).
Melin, A. S. et al. Hormonal and surgical treatments for endometriosis and risk of epithelial ovarian cancer. Acta Obstet. Gynecol. Scand. 92, 546–554 (2013).
Somigliana, E. et al. Association between endometriosis and cancer: a comprehensive review and a critical analysis of clinical and epidemiological evidence. Gynecol. Oncol. 101, 331–341 (2006).
Viganò, P., Somigliana, E., Parazzini, F. & Vercellini, P. Bias versus casuality: interpreting recent evidence of association between endometriosis and ovarian cancer. Fertil. Steril. 88, 588–593 (2007).
Leyland, N. Casper, R., Laberge, P. & Singh, S. S; SOGC. Endometriosis: diagnosis and management. J. Obstet. Gynaecol. Can. 32, S1–S32 (2010).
Farquhar, C. Endometriosis. BMJ 334, 249–253 (2007).
Moynihan, R. The future of medicine lies in truly shared decision making. BMJ 346, f2789 (2013).
Fagervold, B., Jenssen, M., Hummerlshoj, L. & Moen, M. H. Life after diagnosis with endometriosis—a 15 years follow-up study. Acta Obstet. Gynecol. Scand. 88, 914–919 (2009).
De Graaff, A. A. et al. The significant effect of endometriosis on physical, mental and social wellbeing: results from an international cross-sectional survey. Hum. Reprod. 28, 2677–2685 (2013).
Culley, L. et al. The social and psychological impact of endometriosis on women's lives: a critical narrative review. Hum. Reprod. Update 19, 625–639 (2013).
Acknowledgements
D. Alberico, D. Dridi, A. M. Sanchez and C. Vercellini are greatly acknowledged for their support.
Author information
Authors and Affiliations
Contributions
P. Vercellini, P. Viganò and E. Somigliana researched data for the article, provided a substantial contribution to discussion of the content, wrote the article and reviewed/edited the manuscript before submission. L. Fedele provided a substantial contribution to discussion of the content and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Vercellini, P., Viganò, P., Somigliana, E. et al. Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol 10, 261–275 (2014). https://doi.org/10.1038/nrendo.2013.255
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2013.255
This article is cited by
-
Neutrophil to lymphocytes ratio in deep infiltrating endometriosis as a new toll for clinical management
Scientific Reports (2024)
-
Transcription factor EB-mediated autophagy affects cell migration and inhibits apoptosis to promote endometriosis
Apoptosis (2024)
-
Exploring the Effects of Endocrine-Disrupting Chemicals and miRNA Expression in the Pathogenesis of Endometriosis by Unveiling the Pathways: a Systematic Review
Reproductive Sciences (2024)
-
The effects of coagulation factors on the risk of endometriosis: a Mendelian randomization study
BMC Medicine (2023)
-
Endometriosis in an ectopic kidney: a rare case report and literature review
BMC Women's Health (2023)