Abstract
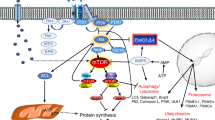
During the past decade, skeletal muscle has been identified as a secretory organ. Accordingly, we have suggested that cytokines and other peptides that are produced, expressed and released by muscle fibres and exert either autocrine, paracrine or endocrine effects should be classified as myokines. The finding that the muscle secretome consists of several hundred secreted peptides provides a conceptual basis and a whole new paradigm for understanding how muscles communicate with other organs, such as adipose tissue, liver, pancreas, bones and brain. However, some myokines exert their effects within the muscle itself. Thus, myostatin, LIF, IL-6 and IL-7 are involved in muscle hypertrophy and myogenesis, whereas BDNF and IL-6 are involved in AMPK-mediated fat oxidation. IL-6 also appears to have systemic effects on the liver, adipose tissue and the immune system, and mediates crosstalk between intestinal L cells and pancreatic islets. Other myokines include the osteogenic factors IGF-1 and FGF-2; FSTL-1, which improves the endothelial function of the vascular system; and the PGC-1α-dependent myokine irisin, which drives brown-fat-like development. Studies in the past few years suggest the existence of yet unidentified factors, secreted from muscle cells, which may influence cancer cell growth and pancreas function. Many proteins produced by skeletal muscle are dependent upon contraction; therefore, physical inactivity probably leads to an altered myokine response, which could provide a potential mechanism for the association between sedentary behaviour and many chronic diseases.
Key Points
-
Myokines are cytokines or other peptides that are produced, expressed and released by muscle fibres
-
Myokines may exert autocrine, paracrine or endocrine effects
-
Myokines may balance and counteract the effects of adipokines
-
The muscle–cell secretome consists of several hundred secreted products
-
Identified myokines include myostatin, LIF, IL-6, IL-7, BDNF, IGF-1, FGF-2, FSTL-1 and irisin
-
Myokines may mediate protective effects of muscular exercise, with regard to diseases associated with a physically inactive lifestyle
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cook, K. S. et al. Adipsin: a circulating serine protease homolog secreted by adipose tissue and sciatic nerve. Science 237, 402–405 (1987).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994).
Scherer, P. E. Adipose tissue: from lipid storage compartment to endocrine organ. Diabetes 55, 1537–1545 (2006).
Shetty, S., Kusminski, C. M. & Scherer, P. E. Adiponectin in health and disease: evaluation of adiponectin-targeted drug development strategies. Trends Pharmacol. Sci. 30, 234–239 (2009).
Olsen, R. H., Krogh-Madsen, R., Thomsen, C., Booth, F. W. & Pedersen, B. K. Metabolic responses to reduced daily steps in healthy nonexercising men. JAMA 299, 1261–1263 (2008).
Krogh-Madsen, R. et al. A 2-wk reduction of ambulatory activity attenuates peripheral insulin sensitivity. J. Appl. Physiol. 108, 1034–1040 (2010).
Booth, F. W., Chakravarthy, M. V., Gordon, S. E. & Spangenburg, E. E. Waging war on physical inactivity: using modern molecular ammunition against an ancient enemy. J. Appl. Physiol. 93, 3–30 (2002).
Tuomilehto, J. et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 344, 1343–1350 (2001).
Nocon, M. et al. Association of physical activity with all-cause and cardiovascular mortality: a systematic review and meta-analysis. Eur. J. Cardiovasc. Prev. Rehabil. 15, 239–246 (2008).
Wolin, K. Y., Yan, Y., Colditz, G. A. & Lee, I. M. Physical activity and colon cancer prevention: a meta-analysis. Br. J. Cancer 100, 611–616 (2009).
Monninkhof, E. M. et al. Physical activity and breast cancer: a systematic review. Epidemiology 18, 137–157 (2007).
Borer, K. T. Physical activity in the prevention and amelioration of osteoporosis in women: interaction of mechanical, hormonal and dietary factors. Sports Med. 35, 779–830 (2005).
Goldstein, M. S. Humoral nature of the hypoglycemic factor of muscular work. Diabetes 10, 232–234 (1961).
Pedersen, B. K. et al. Searching for the exercise factor: is IL-6 a candidate? J. Muscle Res. Cell Motil. 24, 113–119 (2003).
Kjaer, M. et al. Hormonal and metabolic responses to electrically induced cycling during epidural anesthesia in humans. J. Appl. Physiol. 80, 2156–2162 (1996).
Mohr, T. et al. Long-term adaptation to electrically induced cycle training in severe spinal cord injured individuals. Spinal Cord 35, 1–16 (1997).
Bortoluzzi, S., Scannapieco, P., Cestaro, A., Danieli, G. A. & Schiaffino, S. Computational reconstruction of the human skeletal muscle secretome. Proteins 62, 776–792 (2006).
Yoon, J. H. et al. Comparative proteomic analysis of the insulin-induced L6 myotube secretome. Proteomics 9, 51–60 (2009).
Henningsen, J., Rigbolt, K. T., Blagoev, B., Pedersen, B. K. & Kratchmarova, I. Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol. Cell. Proteomics 9, 2482–2496 (2010).
Long, A., Donelson, R. & Fung, T. Does it matter which exercise? A randomized control trial of exercise for low back pain. Spine (Phila. Pa. 1976) 29, 2593–2602 (2004).
Pedersen, B. K. The diseasome of physical inactivity–and the role of myokines in muscle-fat cross talk. J. Physiol. 587, 5559–5568 (2009).
Walsh, K. Adipokines, myokines and cardiovascular disease. Circ. J. 73, 13–18 (2009).
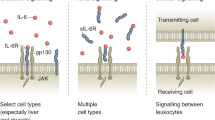
Pedersen, B. K. & Febbraio, M. A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 88, 1379–1406 (2008).
Pedersen, B. K., Akerström, T. C., Nielsen, A. R. & Fischer, C. P. Role of myokines in exercise and metabolism. J. Appl. Physiol. 103, 1093–1098 (2007).
Pedersen, B. K. The anti-inflammatory effect of exercise: its role in diabetes and cardiovascular disease control. Essays Biochem. 42, 105–117 (2006).
McPherron, A. C., Lawler, A. M. & Lee, S. J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387, 83–90 (1997).
Rodgers, B. D. & Garikipati, D. K. Clinical, agricultural, and evolutionary biology of myostatin: a comparative review. Endocr. Rev. 29, 513–534 (2008).
Allen, D. L. et al. Myostatin, activin receptor IIb, and follistatin-like-3 gene expression are altered in adipose tissue and skeletal muscle of obese mice. Am. J. Physiol. Endocrinol. Metab. 294, E918–E927 (2008).
Feldman, B. J., Streeper, R. S., Farese, R. V. Jr & Yamamoto, K. R. Myostatin modulates adipogenesis to generate adipocytes with favorable metabolic effects. Proc. Natl Acad. Sci. USA 103, 15675–15680 (2006).
Guo, T. et al. Myostatin inhibition in muscle, but not adipose tissue, decreases fat mass and improves insulin sensitivity. PLoS ONE 4, e4937 (2009).
Zhao, B., Wall, R. J. & Yang, J. Transgenic expression of myostatin propeptide prevents diet-induced obesity and insulin resistance. Biochem. Biophys. Res. Commun. 337, 248–255 (2005).
Lin, J. et al. Myostatin knockout in mice increases myogenesis and decreases adipogenesis. Biochem. Biophys. Res. Commun. 291, 701–706 (2002).
McPherron, A. C. & Lee, S. J. Suppression of body fat accumulation in myostatin-deficient mice. J. Clin. Invest. 109, 595–601 (2002).
Allen, D. L., Hittel, D. S. & McPherron, A. C. Expression and function of myostatin in obesity, diabetes, and exercise adaptation. Med. Sci. Sports Exerc. 43, 1828–1835 (2011).
Hittel, D. S., Berggren, J. R., Shearer, J., Boyle, K. & Houmard, J. A. Increased secretion and expression of myostatin in skeletal muscle from extremely obese women. Diabetes 58, 30–38 (2009).
Hansen, J. et al. Exercise induces a marked increase in plasma follistatin: evidence that follistatin is a contraction-induced hepatokine. Endocrinology 152, 164–171 (2011).
Fischer, C. P. Interleukin-6 in acute exercise and training: what is the biological relevance? Exerc. Immunol. Rev. 12, 6–33 (2006).
De Rossi, M., Bernasconi, P., Baggi, F., de Waal Malefyt, R. & Mantegazza, R. Cytokines and chemokines are both expressed by human myoblasts: possible relevance for the immune pathogenesis of muscle inflammation. Int. Immunol. 12, 1329–1335 (2000).
Bartoccioni, E., Michaelis, D. & Hohlfeld, R. Constitutive and cytokine-induced production of interleukin-6 by human myoblasts. Immunol. Lett. 42, 135–138 (1994).
Keller, C., Hellsten, Y., Steensberg, A. & Pedersen, B. K. Differential regulation of IL-6 and TNF-alpha via calcineurin in human skeletal muscle cells. Cytokine 36, 141–147 (2006).
Serrano, A. L., Baeza-Raja, B., Perdiguero, E., Jardí, M. & Muñoz-Cánoves, P. Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab. 7, 33–44 (2008).
Haugen, F. et al. IL-7 is expressed and secreted by human skeletal muscle cells. Am. J. Physiol. Cell Physiol. 298, C807–C816 (2010).
Green, C. J., Pedersen, M., Pedersen, B. K. & Scheele, C. Elevated NF-κB activation is conserved in human myocytes cultured from obese type 2 diabetic patients and attenuated by AMP-activated protein kinase. Diabetes 60, 2810–2819 (2011).
Nieman, D. C. et al. Influence of mode and carbohydrate on the cytokine response to heavy exertion. Med. Sci. Sports Exerc. 30, 671–678 (1998).
Nehlsen-Cannarella, S. L. et al. Carbohydrate and the cytokine response to 2.5 h of running. J. Appl. Physiol. 82, 1662–1667 (1997).
Ullum, H. et al. Bicycle exercise enhances plasma IL-6 but does not change IL-1 alpha, IL-1 beta, IL-6, or TNF-alpha pre-mRNA in BMNC. J. Appl. Physiol. 77, 93–97 (1994).
Starkie, R. L., Angus, D. J., Rolland, J., Hargreaves, M. & Febbraio, M. Effect of prolonged, submaximal exercise and carbohydrate ingestion on monocyte intracellular cytokine production in humans. J. Physiol. 528, 647–655 (2000).
Starkie, R. L., Rolland, J., Angus, D. J., Anderson, M. J. & Febbraio, M. A. Circulating monocyes are not the source of elevations in plasma IL-6 and TNF-alpha levels after prolonged running. Am. J. Physiol. Cell Physiol. 280, C769–C774 (2001).
Febbraio, M. A. et al. Hepatosplanchnic clearance of interleukin-6 in humans during exercise. Am. J. Physiol. Endocrinol. Metab. 285, E397–E402 (2003).
Keller, C. et al. Transcriptional activation of the IL-6 gene in human contracting skeletal muscle: influence of muscle glycogen content. FASEB J. 15, 2748–2750 (2001).
Steensberg, A. et al. IL-6 and TNF-alpha expression in, and release from, contracting human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 283, E1272–E1278 (2002).
Hiscock, N., Chan, M. H., Bisucci, T., Darby, I. A. & Febbraio, M. A. Skeletal myocytes are a source of interleukin-6 mRNA expression and protein release during contraction: evidence of fiber type specificity. FASEB J. 18, 992–994 (2004).
Rosendal, L. et al. Increase in interstitial interleukin-6 of human skeletal muscle with repetitive low-force exercise. J. Appl. Physiol. 98, 477–481 (2005).
Steensberg, A. et al. Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J. Physiol. 529, 237–242 (2000).
Keller, C. et al. Effect of exercise, training, and glycogen availability on IL-6 receptor expression in human skeletal muscle. J. Appl. Physiol. 99, 2075–2079 (2005).
Steensberg, A. et al. Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J. Physiol. 537, 633–639 (2001).
Pedersen, B. K. Muscular IL-6 and its role as an energy sensor. Med. Sci. Sports Exerc. 44, 392–396 (2012).
Ruderman, N. B. et al. Interleukin-6 regulation of AMP-activated protein kinase. Potential role in the systemic response to exercise and prevention of the metabolic syndrome. Diabetes 55 (Suppl. 2), S48–S54 (2006).
Pedersen, B. K. et al. The metabolic role of IL-6 produced during exercise: is IL-6 an exercise factor? Proc. Nutr. Soc. 63, 263–267 (2004).
Hoene, M. & Weigert, C. The role of interleukin-6 in insulin resistance, body fat distribution and energy balance. Obes. Rev. 9, 20–29 (2008).
Febbraio, M. A. et al. Glucose ingestion attenuates interleukin-6 release from contracting skeletal muscle in humans. J. Physiol. 549, 607–612 (2003).
Phillips, S. M. et al. Effects of training duration on substrate turnover and oxidation during exercise. J. Appl. Physiol. 81, 2182–2191 (1996).
Fischer, C. P. et al. Endurance training reduces the contraction-induced interleukin-6 mRNA expression in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 287, E1189–E1194 (2004).
Carey, A. L. et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes 55, 2688–2697 (2006).
Bruce, C. R. & Dyck, D. J. Cytokine regulation of skeletal muscle fatty acid metabolism: effect of interleukin-6 and tumor necrosis factor-alpha. Am. J. Physiol. Endocrinol. Metab. 287, E616–E621 (2004).
Petersen, E. W. et al. Acute IL-6 treatment increases fatty acid turnover in elderly humans in vivo and in tissue culture in vitro. Am. J. Physiol. 288, E155–E162 (2005).
van Hall, G. et al. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 88, 3005–3010 (2003).
Kahn, B. B., Alquier, T., Carling, D. & Hardie, D. G. AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 1, 15–25 (2005).
Minokoshi, Y. et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 415, 339–343 (2002).
Watt, M. J. et al. CNTF reverses obesity-induced insulin resistance by activating skeletal muscle AMPK. Nat. Med. 12, 541–548 (2006).
Steinberg, G. R., Rush, J. W. & Dyck, D. J. AMPK expression and phosphorylation are increased in rodent muscle after chronic leptin treatment. Am. J. Physiol. Endocrinol. Metab. 284, E648–E654 (2003).
Steinberg, G. R., Watt, M. J. & Febbraio, M. A. Cytokine Regulation of AMPK signalling. Front. Biosci. 14, 1902–1916 (2009).
Wallenius, V. et al. Interleukin-6-deficient mice develop mature-onset obesity. Nat. Med. 8, 75–79 (2002).
Febbraio, M. A., Hiscock, N., Sacchetti, M., Fischer, C. P. & Pedersen, B. K. Interleukin-6 is a novel factor mediating glucose homeostasis during skeletal muscle contraction. Diabetes 53, 1643–1648 (2004).
Steensberg, A. et al. Acute interleukin-6 administration does not impair muscle glucose uptake or whole-body glucose disposal in healthy humans. J. Physiol. 548, 631–638 (2003).
Lyngsø, D., Simonsen, L. & Bülow, J. Interleukin-6 production in human subcutaneous abdominal adipose tissue: the effect of exercise. J. Physiol. 543, 373–378 (2002).
Wolsk, E., Mygind, H., Grøndahl, T. S., Pedersen, B. K. & van Hall, G. IL-6 selectively stimulates fat metabolism in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 299, E832–E840 (2010).
Nielsen, S. & Pedersen, B. K. Skeletal muscle as an immunogenic organ. Curr. Opin. Pharmacol. 8, 346–351 (2008).
Petersen, A. M. & Pedersen, B. K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 98, 1154–1162 (2005).
Schindler, R. et al. Correlations and interactions in the production of interleukin-6 (IL-6), IL-1, and tumor necrosis factor (TNF) in human blood mononuclear cells: IL-6 suppresses IL-1 and TNF. Blood 75, 40–47 (1990).
Mizuhara, H. et al. T cell activation-associated hepatic injury: mediation by tumor necrosis factors and protection by interleukin 6. J. Exp. Med. 179, 1529–1537 (1994).
Starkie, R., Ostrowski, S. R., Jauffred, S., Febbraio, M. & Pedersen, B. K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-alpha production in humans. FASEB J. 17, 884–886 (2003).
Steensberg, A., Fischer, C. P., Keller, C., Møller, K. & Pedersen, B. K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Endocrinol. Metab. 285, E433–E437 (2003).
Rubio, N. & Sanz-Rodriguez, F. Induction of the CXCL1 (KC) chemokine in mouse astrocytes by infection with the murine encephalomyelitis virus of Theiler. Virology 358, 98–108 (2007).
Lira, S. A. et al. Expression of the chemokine N51/KC in the thymus and epidermis of transgenic mice results in marked infiltration of a single class of inflammatory cells. J. Exp. Med. 180, 2039–2048 (1994).
Keane, M. P. et al. The CXC chemokines, IL-8 and IP-10, regulate angiogenic activity in idiopathic pulmonary fibrosis. J. Immunol. 159, 1437–1443 (1997).
Belperio, J. A. et al. CXC chemokines in angiogenesis. J. Leukoc. Biol. 68, 1–8 (2000).
Addison, C. L. et al. The CXC chemokine receptor 2, CXCR2, is the putative receptor for ELR+ CXC chemokine-induced angiogenic activity. J. Immunol. 165, 5269–5277 (2000).
Tseng, Y. L., Wu, M. H., Yang, H. C., Wang, C. Y. & Lin, C. F. Autocrine IL-6 regulates GRO-alpha production in thymic epithelial cells. Cytokine 51, 195–201 (2010).
Pedersen, L. et al. Exercise-induced liver CXCL-1 expression is linked to muscle derived interleukin-6 expression. J. Physiol. 589, 1409–1420 (2011).
Nieman, D. C. et al. Muscle cytokine mRNA changes after 2.5 h of cycling: influence of carbohydrate. Med. Sci. Sports Exerc. 37, 1283–1290 (2005).
Hilton, D. J., Nicola, N. A. & Metcalf, D. Purification of a murine leukemia inhibitory factor from Krebs ascites cells. Anal. Biochem. 173, 359–367 (1988).
Heinrich, P. C., Behrmann, I., Müller-Newen, G., Schaper, F. & Graeve, L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem. J. 334, 297–314 (1998).
Metcalf, D. The unsolved enigmas of leukemia inhibitory factor. Stem Cells 21, 5–14 (2003).
Broholm, C. & Pedersen, B. K. Leukaemia inhibitory factor—an exercise-induced myokine. Exerc. Immunol. Rev. 16, 77–85 (2010).
Broholm, C. et al. Exercise induces expression of leukaemia inhibitory factor in human skeletal muscle. J. Physiol. 586, 2195–2201 (2008).
Nielsen, A. R. & Pedersen, B. K. The biological roles of exercise-induced cytokines: IL-6, IL-8, and IL-15. Appl. Physiol. Nutr. Metab. 32, 833–839 (2007).
Carbó, N. et al. Interleukin-15 mediates reciprocal regulation of adipose and muscle mass: a potential role in body weight control. Biochim. Biophys. Acta 1526, 17–24 (2001).
Quinn, L. S., Strait-Bodey, L., Anderson, B. G., Argilés, J. M. & Havel, P. J. Interleukin-15 stimulates adiponectin secretion by 3T3-L1 adipocytes: evidence for a skeletal muscle-to-fat signaling pathway. Cell Biol. Int. 29, 449–457 (2005).
Nielsen, A. R. et al. Association between IL-15 and obesity: IL-15 as a potential regulator of fat mass. J. Clin. Endocrinol. Metab. 93, 4486–4493 (2008).
Izumiya, Y. et al. Fast/Glycolytic muscle fiber growth reduces fat mass and improves metabolic parameters in obese mice. Cell Metab. 7, 159–172 (2008).
Ouchi, N. et al. Follistatin-like 1, a secreted muscle protein, promotes endothelial cell function and revascularization in ischemic tissue through a nitric-oxide synthase-dependent mechanism. J. Biol. Chem. 283, 32802–32811 (2008).
Oshima, Y. et al. Follistatin-like 1 is an Akt-regulated cardioprotective factor that is secreted by the heart. Circulation 117, 3099–3108 (2008).
Izumiya, Y. et al. FGF21 is an Akt-regulated myokine. FEBS Lett. 582, 3805–3810 (2008).
Hojman, P. et al. Fibroblast growth factor-21 is induced in human skeletal muscles by hyperinsulinemia. Diabetes 58, 2797–2801 (2009).
Pedersen, B. K. et al. Role of exercise-induced brain-derived neurotrophic factor production in the regulation of energy homeostasis in mammals. Exp. Physiol. 94, 1153–1160 (2009).
Mortensen, O. H. et al. Calprotectin is released from human skeletal muscle tissue during exercise. J. Physiol. 586, 3551–3562 (2008).
Hojman, P. et al. Erythropoietin over-expression protects against diet-induced obesity in mice through increased fat oxidation in muscles. PLoS ONE 4, e5894 (2009).
Horsley, V., Jansen, K. M., Mills, S. T. & Pavlath, G. K. IL-4 acts as a myoblast recruitment factor during mammalian muscle growth. Cell 113, 483–494 (2003).
Hamrick, M. W. A role for myokines in muscle-bone interactions. Exerc. Sport Sci. Rev. 39, 43–47 (2011).
Hamrick, M. W., McNeil, P. L. & Patterson, S. L. Role of muscle-derived growth factors in bone formation. J. Musculoskelet. Neuronal Interact. 10, 64–70 (2010).
Boström, P. et al. A PGC1-α dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 481, 463–468 (2012).
Henningsen, J., Pedersen, B. K. & Kratchmarova, I. Quantitative analysis of the secretion of the MCP family of chemokines by muscle cells. Mol. Biosyst. 7, 311–321 (2011).
Norheim, F. et al. Proteomic identification of secreted proteins from human skeletal muscle cells and expression in response to strength training. Am. J. Physiol. Endocrinol. Metab. 301, E1013–E1021 (2011).
Sadagurski, M. et al. Human IL6 enhances leptin action in mice. Diabetologia 53, 525–535 (2010).
Wunderlich, F. T. et al. Interleukin-6 signaling in liver-parenchymal cells suppresses hepatic inflammation and improves systemic insulin action. Cell Metab. 12, 237–249 (2010).
Watt, M. J., Hevener, A., Lancaster, G. I. & Febbraio, M. A. Ciliary neurotrophic factor prevents acute lipid-induced insulin resistance by attenuating ceramide accumulation and phosphorylation of c-Jun N-terminal kinase in peripheral tissues. Endocrinology 147, 2077–2085 (2006).
Ettinger, M. P. et al. Recombinant variant of ciliary neurotrophic factor for weight loss in obese adults: a randomized, dose-ranging study. JAMA 289, 1826–1832 (2003).
Bouzakri, K. et al. Bimodal effect on pancreatic [beta]-cells of secretory products from normal or insulin-resistant human skeletal muscle. Diabetes 60, 1111–1121 (2011).
Ellingsgaard, H. et al. Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat. Med. 17, 1481–1489 (2011).
Food, Nutrition, Physical Activity and the Prevention of Cancer. World Cancer Research Fund and American Institute of Cancer Research. Ref. Type: Report (2007).
Hojman, P. et al. Exercise-induced muscle-derived cytokines inhibit mammary cancer cell growth. Am. J. Physiol. Endocrinol. Metab. 301, E504–E510 (2011).
Acknowledgements
B. K. Pedersen is supported by a grant from the Danish National Research Foundation (#02-512-55). M. A. Febbraio is supported by grants from the National Health and Medical Research Council (NHMRC), The Diabetes Australia Research Trust and the Victorian Government Operational Infrastructure Support Program.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Pedersen, B., Febbraio, M. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol 8, 457–465 (2012). https://doi.org/10.1038/nrendo.2012.49
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2012.49
This article is cited by
-
Identification of circRNA-associated ceRNA networks in the longissimus dorsi of yak under different feeding systems
BMC Veterinary Research (2024)
-
Impact of skeletal muscle mass on postoperative complications in oral cancer surgery
Maxillofacial Plastic and Reconstructive Surgery (2024)
-
Exercise mimetics: a novel strategy to combat neuroinflammation and Alzheimer’s disease
Journal of Neuroinflammation (2024)
-
Predictive markers for infections after extracorporeal shockwave lithotripsy in patients with kidney stone based on a large prospective cohort
World Journal of Urology (2024)
-
Characterization of Gold-Enhanced Titania: Boosting Cell Proliferation and Combating Bacterial Infestation
Tissue Engineering and Regenerative Medicine (2024)