Abstract
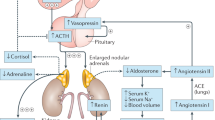
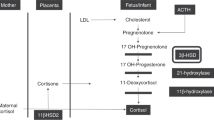
Congenital adrenal hyperplasia (CAH) caused by steroid 21-hydroxylase deficiency occurs in 1:16,000–1:20,000 births. If not promptly diagnosed and treated, CAH can cause death in early infancy from shock, hyponatremia and hyperkalemia. Affected girls usually have ambiguous genitalia but boys appear normal; therefore, newborn babies are commonly screened for CAH in the US and many other countries. By identifying babies with severe, salt-wasting CAH before they develop adrenal crises, screening reduces morbidity and mortality, particularly among affected boys. Diagnosis is based on elevated levels of 17-hydroxyprogesterone, the preferred substrate for steroid 21-hydroxylase. Initial testing usually involves dissociation-enhanced lanthanide fluorescence immunoassay that has a low positive predictive value (about 1%), which leads to many follow-up evaluations that have negative results. The positive predictive value might be improved by second-tier screening using DNA-based methods or liquid chromatography followed by tandem mass spectrometry, but these methods are not widely adopted. Cost estimates for such screening range from US$20,000 to $300,000 per life-year saved. In babies with markedly abnormal screen results, levels of serum electrolytes and 17-hydroxyprogesterone should be immediately determined, but the most reliable way to diagnose CAH is measurement of levels of steroid precursors after stimulation with cosyntropin.
Key Points
-
Neonatal screening for congenital adrenal hyperplasia (CAH) caused by steroid 21-hydroxylase deficiency has been widely adopted in the US and many other countries
-
Neonatal screening for CAH reduces morbidity and mortality, particularly among boys, by identifying infants with the severe, salt-wasting form of CAH before they develop adrenal crises
-
Initial testing usually consists of an immunoassay for 17-hydroxyprogesterone levels; this assay has a low positive predictive value (approximately 1%), which results in many follow-up evaluations that have negative results
-
The positive predictive value might be improved by second-tier screening using DNA-based methods or liquid chromatography followed by tandem mass spectrometry, but these methods are not widely adopted yet
-
In infants with markedly abnormal test results, clinicians should immediately determine serum electrolyte and 17-hydroxyprogesterone levels and start treatment with hydrocortisone and fludrocortisone pending the results of hormonal tests
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
White, P. C. & Speiser, P. W. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Endocr. Rev. 21, 245–291 (2000).
Speiser, P. W. & White, P. C. Congenital adrenal hyperplasia. N. Engl. J. Med. 349, 776–788 (2003).
Joint LWPES/ESPE CAH Working Group. Consensus statement on 21-hydroxylase deficiency from the Lawson Wilkins Pediatric Endocrine Society and the European Society for Paediatric Endocrinology. J. Clin. Endocrinol. Metab. 87, 4048–4053 (2002).
Pang, S. & Shook, M. K. Current status of neonatal screening for congenital adrenal hyperplasia. Curr. Opin. Pediatr. 9, 419–423 (1997).
Therrell, B. L. Newborn screening for congenital adrenal hyperplasia. Endocrinol. Metab. Clin. North Am. 30, 15–30 (2001).
van der Kamp, H. J. & Wit, J. M. Neonatal screening for congenital adrenal hyperplasia. Eur. J. Endocrinol. 151 (Suppl. 3), U71–U75 (2004).
Loeber, J. G. Neonatal screening in Europe; the situation in 2004. J. Inherit. Metab. Dis. 30, 430–438 (2007).
Pang, S., Hotchkiss, J., Drash, A. L., Levine, L. S. & New, M. I. Microfilter paper method for 17α-hydroxyprogesterone radioimmunoassay: its application for rapid screening for congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 45, 1003–1008 (1977).
National Newborn Screening Information System 2009. Disorder Report for Congenital Adrenal Hyperplasia (CAH) [online] (2009).
Gonzalez, R. R., Mäentausta, O., Solyom, J. & Vihko, R. Direct solid-phase time-resolved fluoroimmunoassay of 17α-hydroxyprogesterone in serum and dried blood spots on filter paper. Clin. Chem. 36, 1667–1672 (1990).
Olgemoller, B., Roscher, A. A., Liebl, B. & Fingerhut, R. Screening for congenital adrenal hyperplasia: adjustment of 17-hydroxyprogesterone cut-off values to both age and birth weight markedly improves the predictive value. J. Clin. Endocrinol. Metab. 88, 5790–5794 (2003).
Varness, T. S., Allen, D. B. & Hoffman, G. L. Newborn screening for congenital adrenal hyperplasia has reduced sensitivity in girls. J. Pediatr. 147, 493–498 (2005).
al Saedi, S., Dean, H., Dent, W., Stockl, E. & Cronin, C. Screening for congenital adrenal hyperplasia: the Delfia screening test overestimates serum 17-hydroxyprogesterone in preterm infants. Pediatrics 97, 100–102 (1996).
Allen, D. B. et al. Improved precision of newborn screening for congenital adrenal hyperplasia using weight-adjusted criteria for 17-hydroxyprogesterone levels. J. Pediatr. 130, 128–133 (1997).
Gruñeiro-Papendieck, L. et al. Neonatal screening program for congenital adrenal hyperplasia: adjustments to the recall protocol. Horm. Res. 55, 271–277 (2001).
Ohkubo, S., Shimozawa, K., Matsumoto, M. & Kitagawa, T. Analysis of blood spot 17α-hydroxyprogesterone concentration in premature infants—proposal for cutoff limits in screening congenital adrenal hyperplasia. Acta Paediatr. Jpn 34, 126–133 (1992).
Steigert, M., Schoenle, E. J., Biason-Lauber, A. & Torresani, T. High reliability of neonatal screening for congenital adrenal hyperplasia in Switzerland. J. Clin. Endocrinol. Metab. 87, 4106–4110 (2002).
van der Kamp, H. J. et al. Cutoff levels of 17-α-hydroxyprogesterone in neonatal screening for congenital adrenal hyperplasia should be based on gestational age rather than on birth weight. J. Clin. Endocrinol. Metab. 90, 3904–3907 (2005).
van der Kamp, H. J. et al. Newborn screening for congenital adrenal hyperplasia in The Netherlands. Pediatrics 108, 1320–1324 (2001).
Nomura, S. Immature adrenal steroidogenesis in preterm infants. Early Hum. Dev. 49, 225–233 (1997).
Wong, T., Shackleton, C. H., Covey, T. R. & Ellis, G. Identification of the steroids in neonatal plasma that interfere with 17α-hydroxyprogesterone radioimmunoassays. Clin. Chem. 38, 1830–1837 (1992).
Lange-Kubini, K., Zachmann, M., Kempken, B. & Torresani, T. 15-β-hydroxylated steroids may be diagnostically misleading in confirming congenital adrenal hyperplasia suspected by a newborn screening programme. Eur. J. Pediatr. 155, 928–931 (1996).
Crowther, C. A. & Harding, J. E. Repeat doses of prenatal corticosteroids for women at risk of preterm birth for preventing neonatal respiratory disease. Cochrane Database of Systematic Reviews Issue 3. Art. No.: CD003935. doi:10.1002/14651858.CD003935.pub2 (2007).
Gatelais, F. et al. Effect of single and multiple courses of prenatal corticosteroids on 17-hydroxyprogesterone levels: implication for neonatal screening of congenital adrenal hyperplasia. Pediatr. Res. 56, 701–705 (2004).
King, J. L. et al. Antenatal corticosteroids and newborn screening for congenital adrenal hyperplasia. Arch. Pediatr. Adolesc. Med. 155, 1038–1042 (2001).
Fitness, J. et al. Genotyping of CYP21, linked chromosome 6p markers, and a sex-specific gene in neonatal screening for congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 84, 960–966 (1999).
Baumgartner-Parzer, S. M., Nowotny, P., Heinze, G., Waldhäusl, W. & Vierhapper, H. Carrier frequency of congenital adrenal hyperplasia (21-hydroxylase deficiency) in a middle European population. J. Clin. Endocrinol. Metab. 90, 775–778 (2005).
Kosel, S. et al. Rapid second-tier molecular genetic analysis for congenital adrenal hyperplasia attributable to steroid 21-hydroxylase deficiency. Clin. Chem. 51, 298–304 (2005).
Speiser, P. W. et al. High frequency of nonclassical steroid 21-hydroxylase deficiency. Am. J. Hum. Genet. 37, 650–667 (1985).
Tajima, T., Fujieda, K., Nakae, J., Mikami, A. & Cutler, G. B. Jr. Mutations of the CYP21 gene in nonclassical steroid 21-hydroxylase deficiency in Japan. Endocr. J. 45, 493–497 (1998).
Tusie-Luna, M. T., Speiser, P. W., Dumic, M., New, M. I. & White, P. C. A mutation (Pro30 to Leu) in CYP21 represents a potential nonclassic steroid 21-hydroxylase deficiency allele. Mol. Endocrinol. 5, 685–692 (1991).
Therrell, B. L. Jr et al. Results of screening 1.9 million Texas newborns for 21-hydroxylase-deficient congenital adrenal hyperplasia. Pediatrics 101, 583–590 (1998).
Votava, F. et al. Estimation of the false-negative rate in newborn screening for congenital adrenal hyperplasia. Eur. J. Endocrinol. 152, 869–874 (2005).
Lacey, J. M. et al. Improved specificity of newborn screening for congenital adrenal hyperplasia by second-tier steroid profiling using tandem mass spectrometry. Clin. Chem. 50, 621–625 (2004).
Rauh, M., Gröschl, M., Rascher, W. & Dörr, H. G. Automated, fast and sensitive quantification of 17 α-hydroxy-progesterone, androstenedione and testosterone by tandem mass spectrometry with on-line extraction. Steroids 71, 450–458 (2006).
Janzen, N. et al. Newborn screening for congenital adrenal hyperplasia: additional steroid profile using liquid chromatography–tandem mass spectrometry. J. Clin. Endocrinol. Metab. 92, 2581–2589 (2007).
Minutti, C. Z. et al. Steroid profiling by tandem mass spectrometry improves the positive predictive value of newborn screening for congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 89, 3687–3693 (2004).
Matern, D., Tortorelli, S., Oglesbee, D., Gavrilov, D. & Rinaldo, P. Reduction of the false-positive rate in newborn screening by implementation of MS/MS-based second-tier tests: the Mayo Clinic experience (2004–2007). J. Inherit. Metab. Dis. 30, 585–592 (2007).
Yang, Y. P., Corley, N. & Garcia-Heras, J. Reverse dot-blot hybridization as an improved tool for the molecular diagnosis of point mutations in congenital adrenal hyperplasia caused by 21-hydroxylase deficiency. Mol. Diagn. 6, 193–199 (2001).
Sorensen, K. M. et al. Multiplex ligation-dependent probe amplification technique for copy number analysis on small amounts of DNA material. Anal. Chem. doi:10.1021/ac801688c.
Olney, R. C., Mougey, E. B., Wang, J., Shulman, D. I. & Sylvester, J. E. Using real-time, quantitative PCR for rapid genotyping of the steroid 21-hydroxylase gene in a north Florida population. J. Clin. Endocrinol. Metab. 87, 735–741 (2002).
Nordenstrom, A., Thilén, A., Hagenfeldt, L., Larsson, A. & Wedell, A. Genotyping is a valuable diagnostic complement to neonatal screening for congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency. J. Clin. Endocrinol. Metab. 84, 1505–1509 (1999).
Krone, N. et al. Multiplex minisequencing of the 21-hydroxylase gene as a rapid strategy to confirm congenital adrenal hyperplasia. Clin. Chem. 48, 818–825 (2002).
Wu, Y. L. et al. Phenotypes, genotypes and disease susceptibility associated with gene copy number variations: complement C4 CNVs in European American healthy subjects and those with systemic lupus erythematosus. Cytogenet. Genome Res. 123, 131–141 (2008).
Day, D. J. et al. Identification of non-amplifying CYP21 genes when using PCR-based diagnosis of 21-hydroxylase deficiency in congenital adrenal hyperplasia (CAH) affected pedigrees. Hum. Mol. Genet. 5, 2039–2048 (1996).
New, M. I. et al. Genotyping steroid 21-hydroxylase deficiency: hormonal reference data. J. Clin. Endocrinol. Metab. 57, 320–326 (1983).
Abdu, T. A., Elhadd, T. A., Neary, R. & Clayton, R. N. Comparison of the low dose short synacthen test (1 µg), the conventional dose short synacthen test (250 µg), and the insulin tolerance test for assessment of the hypothalamo–pituitary–adrenal axis in patients with pituitary disease. J. Clin. Endocrinol. Metab. 84, 838–843 (1999).
White, P. C. The endocrinologist's approach to the intersex patient. Adv. Exp. Med. Biol. 511, 107–119 (2002).
Lee, P. A. et al. Consensus statement on management of intersex disorders. International Consensus Conference on Intersex. Pediatrics 118, e488–e500 (2006).
Flück, C. E. et al. Mutant P450 oxidoreductase causes disordered steroidogenesis with and without Antley–Bixler syndrome. Nat. Genet. 36, 228–230 (2004).
Peter, M. et al. A case of 11β-hydroxylase deficiency detected in a newborn screening program by second-tier LC–MS/MS. Horm. Res. 69, 253–256 (2008).
Kushnir, M. M. et al. Development and performance evaluation of a tandem mass spectrometry assay for 4 adrenal steroids. Clin. Chem. 52, 1559–1567 (2006).
Balsamo, A. et al. Congenital adrenal hyperplasia: neonatal mass screening compared with clinical diagnosis only in the Emilia-Romagna region of Italy, 1980–1995. Pediatrics 98, 362–367 (1996).
Thil'en, A. et al. Benefits of neonatal screening for congenital adrenal hyperplasia (21-hydroxylase deficiency) in Sweden. Pediatrics 101, E11 (1998).
Brosnan, P. G. et al. Effect of newborn screening for congenital adrenal hyperplasia. Arch. Pediatr. Adolesc. Med. 153, 1272–1278 (1999).
Strnadová, K. A. et al. Prevalence of congenital adrenal hyperplasia among sudden infant death in the Czech Republic and Austria. Eur. J. Pediatr. 166, 1–4 (2007).
Thompson, R., Seargeant, L. & Winter, J. S. Screening for congenital adrenal hyperplasia: distribution of 17α-hydroxyprogesterone concentrations in neonatal blood spot specimens. J. Pediatr. 114, 400–404 (1989).
Nordenström, A. et al. Female preponderance in congenital adrenal hyperplasia due to CYP21 deficiency in England: implications for neonatal screening. Horm. Res. 63, 22–28 (2005).
Thilen, A. & Larsson, A. Congenital adrenal hyperplasia in Sweden 1969–1986: prevalence, symptoms and age at diagnosis. Acta Paediatr. Scand. 79, 168–175 (1990).
Watson, M. S., Lloyd-Puryear, M. A., Mann, M. Y., Rinaldo, P. & Howell, R. R. (eds) Newborn Screening: Toward a Uniform Screening Panel and System. [online] (2009).
Grosse, S. D. & Van, V. G. How many deaths can be prevented by newborn screening for congenital adrenal hyperplasia? Horm. Res. 67, 284–291 (2007).
Nass, R. & Baker, S. Learning disabilities in children with congenital adrenal hyperplasia. J. Child Neurol. 6, 306–312 (1991).
Donaldson, M. D. et al. Presentation, acute illness, and learning difficulties in salt-wasting 21-hydroxylase deficiency. Arch. Dis. Child 70, 214–218 (1994).
Pang, S. Y. et al. Worldwide experience in newborn screening for classical congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Pediatrics 81, 866–874 (1988).
Carroll, A. E. & Downs, S. M. Comprehensive cost-utility analysis of newborn screening strategies. Pediatrics 117, S287–S295 (2006).
Yoo, B. K. & Grosse, S. D. The cost effectiveness of screening newborns for congenital adrenal hyperplasia. Public Health Genomics 12, 67–72 (2009).
Gurian, E. A., Kinnamon, D. D., Henry, J. J. & Waisbren, S. E. Expanded newborn screening for biochemical disorders: the effect of a false-positive result. Pediatrics 117, 1915–1921 (2006).
Prosser, L. A., Ladapo, J. A., Rusinak, D. & Waisbren, S. E. Parental tolerance of false-positive newborn screening results. Arch. Pediatr. Adolesc. Med. 162, 870–876 (2008).
Acknowledgements
Charles P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
White, P. Neonatal screening for congenital adrenal hyperplasia. Nat Rev Endocrinol 5, 490–498 (2009). https://doi.org/10.1038/nrendo.2009.148
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2009.148
This article is cited by
-
Screening neonatale della Sindrome Adreno-genitale Congenita (SAG): stato dell’arte
L'Endocrinologo (2024)
-
Differentiation of steroid isomers by steroid analogues adducted trapped ion mobility spectrometry-mass spectrometry
Analytical and Bioanalytical Chemistry (2024)
-
Clinical utility of serum 17-hydroxyprogesterone as a marker for medical therapy for male infertility: recommendations based on clinical scenarios
International Journal of Impotence Research (2023)
-
Management challenges and therapeutic advances in congenital adrenal hyperplasia
Nature Reviews Endocrinology (2022)
-
An update on male infertility and intratesticular testosterone—insight into novel serum biomarkers
International Journal of Impotence Research (2022)