Abstract
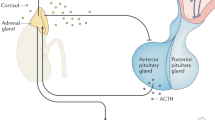
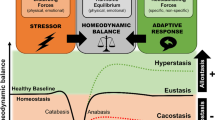
All organisms must maintain a complex dynamic equilibrium, or homeostasis, which is constantly challenged by internal or external adverse forces termed stressors. Stress occurs when homeostasis is threatened or perceived to be so; homeostasis is re-established by various physiological and behavioral adaptive responses. Neuroendocrine hormones have major roles in the regulation of both basal homeostasis and responses to threats, and are involved in the pathogenesis of diseases characterized by dyshomeostasis or cacostasis. The stress response is mediated by the stress system, partly located in the central nervous system and partly in peripheral organs. The central, greatly interconnected effectors of this system include the hypothalamic hormones arginine vasopressin, corticotropin-releasing hormone and pro-opiomelanocortin-derived peptides, and the locus ceruleus and autonomic norepinephrine centers in the brainstem. Targets of these effectors include the executive and/or cognitive, reward and fear systems, the wake–sleep centers of the brain, the growth, reproductive and thyroid hormone axes, and the gastrointestinal, cardiorespiratory, metabolic, and immune systems. Optimal basal activity and responsiveness of the stress system is essential for a sense of well-being, successful performance of tasks, and appropriate social interactions. By contrast, excessive or inadequate basal activity and responsiveness of this system might impair development, growth and body composition, and lead to a host of behavioral and somatic pathological conditions.
Key Points
-
Stress occurs when homeostasis is threatened or perceived to be so
-
The stress response is mediated by the stress system, which is located in both the central nervous system and peripheral organs
-
The main central effectors of the stress system are highly interconnected, and include hypothalamic corticotropin-releasing hormone and brainstem-derived norepinephrine
-
Malfunction of the stress system is associated with behavioral and somatic disorders
-
Stress is a major contributor to psychosocial and physical pathological conditions in humans
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Raisman, G. An urge to explain the incomprehensible: Geoffrey Harris and the discovery of the neural control of the pituitary gland. Ann. Rev. Neurosci. 20, 533–566 (1997).
Chrousos, G. P. & Gold, P. W. The concepts of stress and stress system disorders: overview of physical and behavioral homeostasis. JAMA 267, 1244–1252 (1992).
Charmandari, E., Tsigos, C. & Chrousos, G. P. Neuroendocrinology of stress. Ann. Rev. Physiol. 67, 259–284 (2005).
Chrousos, G. P., Loriaux, D. L. & Gold, P. W. (eds) Mechanisms of Physical and Emotional Stress (Advances in Experimental Medicine and Biology, Vol. 245) (Plenum Press, New York, 1988).
Chrousos, G. P. et al. (eds) Stress: Basic Mechanisms and Clinical Implications (Annals of the New York Academy of Sciences, Vol. 771) (New York Academy of Sciences, New York, 1996).
Chrousos, G. P. The hypothalamic–pituitary–adrenal axis and immune-mediated inflammation. N. Engl. J. Med. 332, 1351–1362 (1995).
Chrousos, G. P. 1997 Hans Selye memorial lecture: stressors, stress and neuroendocrine integration of the adaptive response. Ann. NY Acad. Sci. 851, 311–335 (1998).
Chrousos, G. P. The stress response and immune function: clinical implications; the 1999 Novera H. Spector lecture. Ann. NY Acad. Sci. 917, 38–67 (2000).
Karalis, C. et al. Autocrine or paracrine inflammatory actions of corticotropin releasing hormone in vivo . Science 254, 421–423 (1991).
Papanicolaou, D. A., Wilder, R. L., Manolagas, S. C. & Chrousos, G. P. The pathophysiologic roles of interleukin-6 in humans. Ann. Intern. Med. 128, 127–137 (1998).
Vale, W., Spiess, J., Rivier, C. & Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and β-endorphin. Science 213, 1394–1397 (1981).
Chrousos, G. P. Organization and integration of the endocrine system: the sleep and wakefulness perspective. Sleep Med. Clin. 2, 125–145 (2007).
Makino, S. et al. Psychological stress increased corticotropin-releasing hormone mRNA and content in the central nucleus of the amygdala but not in the hypothalamic paraventricular nucleus in the rat. Brain Res. 850, 136–143 (1999).
LeDoux, J. E. Emotion and the amygdala. In The Amygdala: Neurobiological Aspects of Emotion, Memory, and Mental Dysfunction (Ed. Aggleton, J. P.) 339–351 (Wiley-Liss, New York, 1992).
Morgan, M., Romanski, L. & LeDoux, J. Extinction of emotional learning: contribution of medial prefrontal cortex. Neurosci. Lett. 163, 109–113 (1993).
Morgan, M. & LeDoux, J. E. Differential contribution of dorsal and ventral medial prefrontal cortex to the acquisition and extinction of conditioned fear. Behav. Neurosci. 109, 681–688 (1995).
Sullivan, R. M. & Gratton, A. Lateralized effects of medial prefrontal cortex lesions on neuroendocrine and autonomic stress responses in rats. J. Neurosci. 19, 2834–2840 (1999).
Fuster, J. M. The prefrontal cortex. An update: time is of the essence. Neuron 30, 319–333 (2001).
Kalivas, P. W. & Volkow, N. D. The neural basis of addiction: a pathology of motivation and choice. Am. J. Psychiatry 162, 1403–1413 (2005).
McEwen, B. S. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol. Rev. 87, 873–904 (2007).
Vgontzas, A. N. et al. Sleep deprivation effects on the activity of the hypothalamic–pituitary–adrenal and growth axes: potential clinical implications. Clin. Endocrinol. (Oxf.), 51, 205–215 (1999).
Vgontzas, A. N. et al. Circadian interleukin-6 secretion and quality and depth of sleep. J. Clin. Endocrinol. Metab. 84, 2603–2607 (1999).
Vgontzas, A. N. et al. Impaired nighttime sleep is associated with elevated plasma IL-6 and cortisol levels in healthy old vs. young adults: physiologic and therapeutic implications. J. Clin. Endocrinol. Metab. 88, 2087–2095 (2003).
Vgontzas, A. et al. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J. Clin. Endocrinol. Metab. 89, 2119–2126 (2004).
Vgontzas, A. N. et al. Daytime napping after a night of sleep loss decreases sleepiness, improves performance, and causes beneficial changes in cortisol and interleukin-6 secretion. Am. J. Physiol. Endocrinol. Metab. 292, E253–E261 (2007).
Vgontzas, A. N. & Chrousos, G. P. Sleep, the hypothalamic–pituitary–adrenal axis, and cytokines: multiple interactions and disturbances in sleep disorders. Endocrinol. Metab. Clin. North Am. 31, 15–36 (2002).
Chrousos, G. P., Torpy, D. & Gold, P. W. Interactions between the hypothalamic–pituitary–adrenal axis and the female reproductive system: clinical implications. Ann. Intern. Med. 129, 229–240 (1998).
Taché, Y. & Bonaz, B. Corticotropin-releasing factor receptors and stress-related alterations of gut motor function. J. Clin. Invest. 117, 33–40 (2007).
Elenkov, I. J., Papanicolaou, D. A., Wilder, R. L. & Chrousos, G. P. Modulatory effects of glucocorticoids and catecholamines on human interleukin-12 and interleukin-10 production: clinical implications. Proc. Assoc. Am. Phys. 108, 374–381 (1996).
Elenkov, I. J. & Chrousos, G. P. Stress hormones, TH1/TH2-patterns, pro/anti- inflammatory cytokines and susceptibility to disease. Trends Endocrinol. Metab. 10, 359–368 (1999).
Elenkov, I. J. et al. Low versus high baseline epinephrine output shapes opposite innate cytokine profiles: presence of Lewis- and Fischer-like neurohormonal-immune phenotypes in humans. J. Immunol. 181, 1737–1745 (2008).
Theoharides, T. C. et al. Corticotropin-releasing hormone induces skin mast cell degranulation and increased vascular permeability, a possible explanation for its proinflammatory effects. Endocrinology 139, 403–413 (1998).
Theoharides, T. C. et al. Stress-induced intracranial mast cell degranulation. A corticotropin-releasing hormone-mediated effect. Endocrinology 136, 5745–5750 (1995).
Franchimont, D., Kino, T., Galon, J., Meduri, G. U. & Chrousos, G. P. Glucocorticoids and inflammation revisited. NIH Clinical Staff Conference. Neuroimmunomodulation 10, 247–260 (2003).
Chrousos, G. P. & Kino, T. Intracellular glucocorticoid signaling: a formerly simple system turns stochastic. Sci. STKE 304, pe48 (2005).
Chrousos, G. P. & Kino, T. Glucocorticoid action networks and complex psychiatric and/or somatic disorders. Stress 10, 213–219 (2007).
Levine, S. The pituitary–adrenal system and the developing brain. Prog. Brain Res. 32, 79–85 (1970).
Newport, D. J., Stowe, Z. N. & Nemeroff, C. B. Parental depression: animal models of an adverse life event. Am. J. Psychiatry 159, 1265–1283 (2002).
Szyf, M., Weaver, I. C., Champagne, F. A., Diorio, J. & Meaney, M. J. Maternal programming of steroid receptor expression in the rat. Front. Neuroendocrinol. 26, 139–162 (2005).
Champagne, F. A. et al. Maternal care associated with methylation of the estrogen receptor-α1b promoter and estrogen receptor-α expression in the medial preoptic area of female offspring. Endocrinology 147, 2909–2915 (2006).
Gold, P. W., Goodwin, F. & Chrousos, G. P. Clinical and biochemical manifestations of depression: relationship to the neurobiology of stress (Part I). N. Engl. J. Med. 319, 348–353 (1988).
Gold, P. W., Goodwin, F. & Chrousos, G. P. Clinical and biochemical manifestations of depression: relationship to the neurobiology of stress (Part 2). N. Engl. J. Med. 319, 413–420 (1988).
Gold, P. W. et al. Cardiac implications of increased arterial entry and reversible 24-h central and peripheral norepinephrine levels in melancholia. Proc. Natl Acad. Sci. USA 102, 8303–8308 (2005).
Wong, M.-L. et al. Pronounced and sustained central hypernoradrenergic function in major depression with melancholic features: relation to hypercortisolism and corticotropin releasing hormone. Proc. Natl Acad. Sci. USA 97, 325–330 (2000).
Alesci, S. et al. Major depression is associated with significant diurnal elevations in plasma IL-6 levels, a shift of its circadian rhythm, and loss of physiologic complexity in its secretion: clinical implications. J. Clin. Endocrinol. Metab. 90, 2522–2530 (2005).
Vgontzas, A. et al. Chronic insomnia is associated with nyctohemeral activation of the hypothalamic–pituitary–adrenal axis. J. Clin. Endocrinol. Metab. 86, 3787–3794 (2001).
Vgontzas, A. N. et al. Chronic insomnia is associated with a shift of IL-6 and TNFα secretion from nighttime to daytime. Metabolism 29, 1252–1261 (2002).
Charmandari, E., Kino, T., Souvatzoglou, E. & Chrousos, G. P. Pediatric stress: hormonal mediators and human development. Hormone Res. 59, 161–179 (2003).
Chrousos, G. P. The role of stress and the hypothalamic–pituitary–adrenal axis in the pathogenesis of the metabolic syndrome: neuro-endocrine and target tissue-related causes. Int. J. Obes. (London) 24, S50f–S55f (2000).
Chrousos, G. P. & Tsigos, C. (Eds) Annals of the New York Academy of Sciences, Stress, Obesity, and Metabolic Syndrome, Vol. 1083 (Wiley Blackwell, Hoboken, 2006).
Vgontzas, A. N. et al. Elevation of plasma cytokines in disorders of excessive daytime sleepiness: role of sleep disturbance and obesity. J. Clin. Endocrinol. Metab. 82, 1313–1316 (1997).
Vgontzas, A. N. et al. Sleep apnea and daytime sleepiness and fatigue: relations with visceral obesity, insulin resistance, and hypercytokinemia, J . Clin. Endocrinol. Metab. 85, 1151–1158 (2000).
De Bellis, M. D. et al. Hypothalamic–pituitary–adrenal dysregulation in sexually abused girls. J. Clin. Endocrinol. Metab. 78, 249–255.
Pervanidou, P. et al. The natural history of neuroendocrine changes in pediatric post-traumatic stress disorder (PTSD) after motor vehicle accidents: progressive divergence of noradrenaline and cortisol concentrations over time. Biol. Psychiatry 62, 1095–1102 (2007).
Pervanidou, P. et al. Elevated morning serum interleukin (IL)-6 or evening salivary cortisol concentrations predict posttraumatic stress disorder in children and adolescents six months after a motor vehicle accident. Psychoneuroendocrinology 32, 991–999 (2007).
Grundy, D. et al. Fundamentals of neurogastroenterology: basic science. Gastroenterology 130, 1391–1411 (2006).
Habib, K. E. et al. Oral administration of a CRH receptor antagonist significantly attenuates behavioral, neuroendocrine, and autonomic responses to stress in primates. Proc. Natl Acad. Sci. USA 10, 1073–1079 (2000).
Webster, E. L. et al. Corticotropin-releasing hormone (CRH) antagonist attenuates adjuvant-induced arthritis: evidence supporting major role for CRH in peripheral inflammation. J. Rheumatol. 29, 1252–1261 (2002).
Gabry, K. E. et al. Marked suppression of gastric ulcerogenesis and intestinal responses to stress by a novel class of drugs. Mol. Psychiatry 7, 474–483 (2002).
Grammatopoulos, D. & Chrousos, G. P. Structural and signalling diversity of corticotropin-releasing hormone (CRH) and related peptides and their receptors: potential clinical applications of CRH receptor antagonists. Trends Endocrinol. Metab. 13, 436–444 (2002).
Contoreggi, C., Rice, K. C. & Chrousos, G. P. Non-peptide corticotropin-releasing hormone receptor type 1 antagonists and their applications in psychosomatic disorders. Neuroendocrinology 80, 111–123 (2004).
Zoumakis, E., Grammatopoulos, D. & Chrousos, G. Corticotropin-releasing hormone antagonists. Eur. J. Endocrinol. 155 (Suppl. 1), S85–S90 (2006).
Clauw, D. J. & Chrousos, G. P. Chronic pain and fatigue syndromes: overlapping clinical and neuroendocrine features and potential pathogenic mechanisms. Neuroimmunomodulation 4, 134–153 (1997).
Magiakou, M. A. et al. Hypothalamic corticotropin releasing hormone suppression during the postpartum period: implications for the increase of psychiatric manifestations during this time. J. Clin. Endocrinol. Metab. 81, 1912–1917 (1996).
Elenkov, I. J. et al. Interleukin 12, tumor necrosis factor-α and hormonal changes during late pregnancy and early postpartum: implications for autoimmune disease activity during these times. J. Clin. Endocrinol. Metab. 86, 4933–4938 (2001).
Gold, P. W. & Chrousos, G. P. The endocrinology of melancholic and atypical depression: relation to neurocircuitry and somatic consequences. Proc. Assoc. Am. Physicians 111, 22–34 (1999).
Gold, P. W., Gabry, K. E., Yasuda, M. R. & Chrousos, G. P. Divergent endocrine abnormalities in melancholic and atypical depression: clinical and pathophysiologic implications. Endocrinol. Metab. Clin. North Am. 31, 37–62 (2002).
Chrousos, G. The glucocorticoid receptor gene, longevity, and the highly prevalent complex disorders of western societies. Am. J. Med. 117, 204–207 (2004).
Brown, G. R. & Anderson, B. Psychiatric morbidity in adult inpatients with childhood histories of sexual and physical abuse. Am. J. Psychiatry 148, 55–61 (1991).
Smith, G. D., Hart, C., Blane, D. & Hole, D. Adverse socioeconomic conditions in childhood and cause specific adult mortality: prospective observation study. BMJ 316, 1631–1635 (1998).
Felitti, V. J. et al. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. Am. J. Prevent. Med. 14, 245–258 (1998).
Repetti, R. L., Taylor, S. E. & Seeman, T. E. Risky families: family social environments and the mental and physical health of offspring. Psych. Bull. 128, 330–366 (2002).
Banks, J., Marmot, M., Oldfield Z. & Smith, J. P. Disease and disadvantage in the United States and in England. JAMA 295, 2037–2045 (2006).
Flegal, K. M., Graubard, B. I., Williamson, D. F. & Gail, M. H. Excess deaths associated with underweight, overweight, and obesity. JAMA 298, 1861–1867 (2005).
Warren, J. Presocratics: Natural Philosophers before Socrates (University of California, Berkeley, 2007).
Cannon, W. B. The Wisdom of the Body edn 2 (W. W. Norton, New York, 1939).
Cannon, W. B. The Way of an Investigator (Hafner, New York, 1968).
Wolfe, E. L., Barger, A. C. & Benison, S. Walter B. Cannon: Science and Society (Harvard University Press, Cambridge, 2000).
Selye, H. A syndrome produced by diverse nocuous agents. J. Neuropsychiatry Clin. Neurosci. 138, 230–231 (1936).
Selye, H. The Stress of Life (McGraw-Hill, New York, 1956).
Landau, L. D., Pitaevskii, L. P., Lifshitz, E. M. & Kosevich, A. M. Theory of Elasticity edn 3 (Butterworth-Heinemann, Oxford, 1986).
Acknowledgements
This review is partially based on the Geoffrey Harris Memorial Lecture given by the author at the 10th European Congress of Endocrinology, 3–7 May 2008, Berlin, Germany.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Chrousos, G. Stress and disorders of the stress system. Nat Rev Endocrinol 5, 374–381 (2009). https://doi.org/10.1038/nrendo.2009.106
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2009.106
This article is cited by
-
Habitual salt preference worsens blood pressure in hospitalized hypertensive patients with omicron infection under epidemic-related stress
BMC Public Health (2024)
-
Hypertension and urologic chronic pelvic pain syndrome: An analysis of MAPP-I data
BMC Urology (2024)
-
The cortisol awakening response in a 3 month clinical trial of service dogs for veterans with posttraumatic stress disorder
Scientific Reports (2024)
-
The effects of acute exercise on stress reactivity assessed via a multidimensional approach: a systematic review
Journal of Behavioral Medicine (2024)
-
Sleep quality in relation to perceived psychological stress in patients with type 2 diabetes and in age- and sex-matched control individuals
Acta Diabetologica (2024)