Abstract
Sickle cell disease (SCD) is a group of inherited disorders caused by mutations in HBB, which encodes haemoglobin subunit β. The incidence is estimated to be between 300,000 and 400,000 neonates globally each year, the majority in sub-Saharan Africa. Haemoglobin molecules that include mutant sickle β-globin subunits can polymerize; erythrocytes that contain mostly haemoglobin polymers assume a sickled form and are prone to haemolysis. Other pathophysiological mechanisms that contribute to the SCD phenotype are vaso-occlusion and activation of the immune system. SCD is characterized by a remarkable phenotypic complexity. Common acute complications are acute pain events, acute chest syndrome and stroke; chronic complications (including chronic kidney disease) can damage all organs. Hydroxycarbamide, blood transfusions and haematopoietic stem cell transplantation can reduce the severity of the disease. Early diagnosis is crucial to improve survival, and universal newborn screening programmes have been implemented in some countries but are challenging in low-income, high-burden settings.
Similar content being viewed by others
Introduction
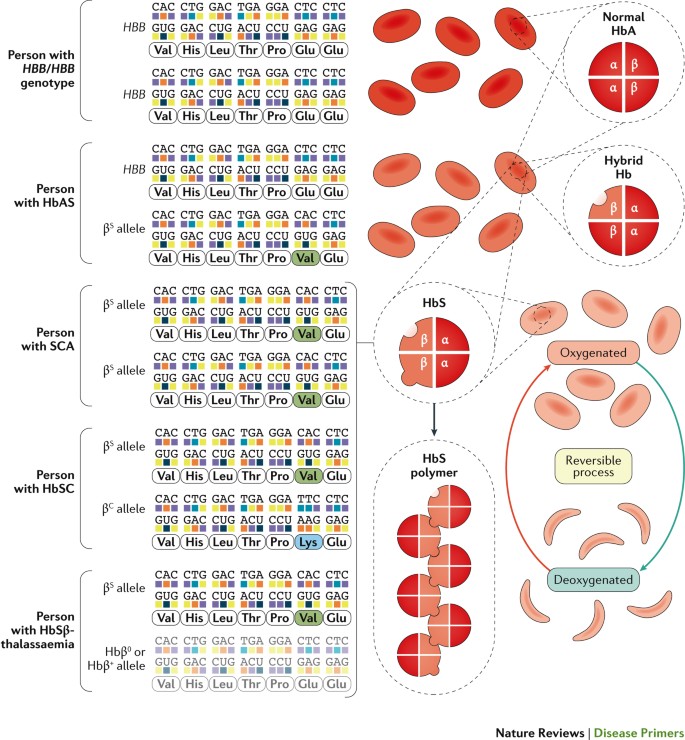
Sickle cell disease (SCD) is an umbrella term that defines a group of inherited diseases (including sickle cell anaemia (SCA), HbSC and HbSβ-thalassaemia, see below) characterized by mutations in the gene encoding the haemoglobin subunit β (HBB) (Fig. 1). Haemoglobin (Hb) is a tetrameric protein composed of different combinations of globin subunits; each globin subunit is associated with the cofactor haem, which can carry a molecule of oxygen. Hb is expressed by red blood cells, both reticulocytes (immature red blood cells) and erythrocytes (mature red blood cells). Several genes encode different types of globin proteins, and their various tetrameric combinations generate multiple types of Hb, which are normally expressed at different stages of life — embryonic, fetal and adult. Hb A (HbA), the most abundant (>90%) form of adult Hb, comprises two α-globin subunits (encoded by the duplicated HBA1 and HBA2 genes) and two β-globin subunits. A single nucleotide substitution in HBB results in the sickle Hb (HbS) allele βS; the mutant protein generated from the βS allele is the sickle β-globin subunit and has an amino acid substitution. Under conditions of deoxygenation (that is, when the Hb is not bound to oxygen), Hb tetramers that include two of these mutant sickle β-globin subunits (that is, HbS) can polymerize and cause the erythrocytes to assume a crescent or sickled shape from which the disease takes its name. Hb tetramers with one sickle β-globin subunit can also polymerize, albeit not as efficiently as HbS. Sickle erythrocytes can lead to recurrent vaso-occlusive episodes that are the hallmark of SCD.
Normal haemoglobin A (HbA) is formed by two α-globin subunits and two β-globin subunits, the latter of which are encoded by HBB. The sickle Hb (HbS) allele, βS, is an HBB allele in which an adenine-to-thymine substitution results in the replacement of glutamic acid with valine at position 6 in the mature β-globin chain. Sickle cell disease (SCD) occurs when both HBB alleles are mutated and at least one of them is the βS allele. Deoxygenated (not bound to oxygen) HbS can polymerize, and HbS polymers can stiffen the erythrocyte. Individuals with one βS allele have the sickle cell trait (HbAS) but not SCD; individuals with sickle cell anaemia (SCA), the most common SCD genotype, have two βS alleles (βS/βS). Other relatively common SCD genotypes are possible. Individuals with the HbSC genotype have one βS allele and one HBB allele with a different nucleotide substitution (HBB Glu6Lys, or βC allele) that generates another structural variant of Hb, HbC. The βC allele is mostly prevalent in West Africa or in individuals with ancestry from this region16. HbSC disease is a condition with generally milder haemolytic anaemia and less frequent acute and chronic complications than SCA, although retinopathy and osteonecrosis (also known as bone infarction, in which bone tissue is lost owing to interruption of the blood flow) are common occurrences259. The βS allele combined with a null HBB allele (Hbβ0) that results in no protein translation causes HbSβ0-thalassaemia, a clinical syndrome indistinguishable from SCA except for the presence of microcytosis (a condition in which erythrocytes are abnormally small)260. The βS allele combined with a hypomorphic HBB allele (Hbβ+; with a decreased amount of normal β-globin protein) results in HbSβ+-thalassaemia, a clinical syndrome generally milder than SCA owing to low-level expression of normal HbA. Severe and moderate forms of HbSβ-thalassaemia are most prevalent in the eastern Mediterranean region and parts of India, whereas mild forms are common in populations of African ancestry. Rarely seen compound heterozygous SCD genotypes include HbS combined with HbD, HbE, HbOArab or Hb Lepore (not shown)261.
SCD is inherited as an autosomal codominant trait1; individuals who are heterozygous for the βS allele carry the sickle cell trait (HbAS) but do not have SCD, whereas individuals who are homozygous for the βS allele have SCA. SCA, the most common form of SCD, is a lifelong disease characterized by chronic haemolytic anaemia, unpredictable episodes of pain and widespread organ damage. There is a wide variability in the clinical severity of SCA, as well as in the life expectancy2. Genetic and genome-wide association studies have consistently found that high levels of fetal Hb (HbF; the heterodimeric combination of two α-globin proteins and two γ-globin proteins (encoded by HBG1 and HBG2))3 and the co-inheritance of α-thalassaemia (which is caused by mutations in HBA1 and HBA2) are associated, on average, with milder SCD phenotypes2. However, these two biomarkers explain only a small fraction of the observed phenotypic variability.
Since the 1980s, a rapidly expanding body of knowledge has promoted a better understanding of SCD, particularly in high-income countries4,5. In the United States, research funding increased exponentially, awareness and education programmes expanded, counselling programmes were improved and universal newborn screening programmes now ensure early diagnosis and intervention. Specific research and training programmes led to a cadre of knowledgeable health professionals working in this field, improved patient management, prevention of complications and extension of life expectancy.
In this Primer, we focus on SCA and aim to balance such remarkable advances with the key major challenges remaining worldwide to improve the prevention and management of this chronic disease and ultimately to discover an affordable cure.
Epidemiology
Natural history
There is rather little information on the natural history of SCD (which is relevant for SCD prevention and control), especially in areas of high prevalence. The main sources of information are the Jamaican Cohort Study of Sickle Cell Disease, which was initiated in 1973 and followed up all individuals with SCD detected among 100,000 consecutive deliveries in Kingston, Jamaica6, and, in the United States, the Cooperative Study of Sickle Cell Disease (CSSCD; 1978–1998), which gathered data on growth and development, disease complications, clinical studies and epidemiology on >3,000 individuals with SCD7. Since the discontinuation of the CSSCD, the ongoing natural history of SCD in the United States can be gleaned from a few single-institution ongoing registries, screening populations of clinical trial cohorts and administrative health data sets.
Several cohort studies in high-income and middle-income countries have demonstrated that the clinical course of SCD has substantially changed since the 1970s in both children and adults. Survival similar to that of healthy children has been reported in children with SCA in the United States and the United Kingdom8. Adults with SCD in high-income countries can now expect to live well into their sixties, and a median survival of 67 years has been reported for patients with SCD at one London hospital9; nevertheless, survival is still much lower than that of the general population of London. As childhood mortality of SCD has fallen, the transition from paediatric to adult patterns of lifestyle and medical care delivery is increasingly important. For example, in the United States, there is a declining workforce of adult haematologists who are trained specifically in SCD, which means that adults with SCD are treated by primary care physicians or by haematologists–oncologists who are minimally experienced in SCD. There are limited data available about the survival of individuals with SCD in sub-Saharan Africa and India. Data from African studies indicate a childhood SCA mortality (before 5 years of age) of 50–90%10.
Distribution
The geographical distribution of the βS allele is mainly driven by two factors: the endemicity of malaria and population movements. The overlap between the geographical distribution of the βS allele and malaria endemicity in sub-Saharan Africa led in the 1950s to the hypothesis that individuals with HbAS might be protected against Plasmodium falciparum malaria11. There is now clear evidence that HbAS provides a remarkable protection against severe P. falciparum malaria12 (in fact, individuals with HbAS are 90% less likely to experience severe malaria than individuals with only normal Hb), which explains the high frequencies of the βS allele observed across sub-Saharan Africa and parts of the Mediterranean, the Middle East and India13. Population movements, including the slave trade, have led to a much wider distribution of the βS allele, particularly in North America and Western Europe14. Detailed mapping of the βS allele frequency has highlighted that geographical heterogeneities in the prevalence of inherited Hb disorders can occur over short distances15.
Prevalence and incidence
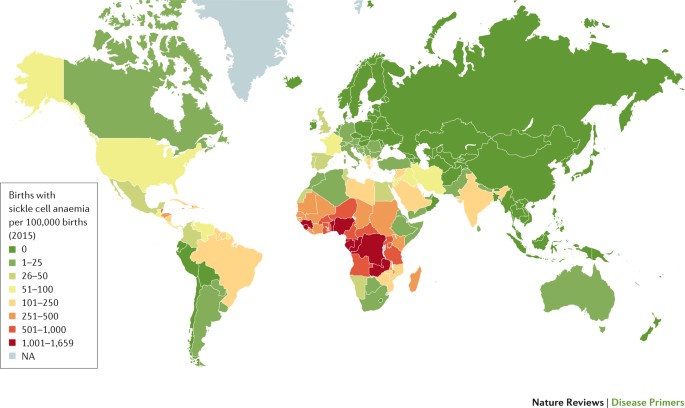
The incidence of births with SCA in sub-Saharan Africa was estimated to be ∼230,000 in 2010, which corresponds to ∼75% of births with SCA worldwide14 (Fig. 2). In addition, West Africa has the highest incidence of HbSC disease, the second most common type of SCD16 (Fig. 1). Over the next 40 years, these numbers are predicted to increase, particularly in sub-Saharan Africa17. The 2010 estimates reported that there were >3.5 million newborn infants with HbAS in sub-Saharan Africa, who could benefit from a potent protection from severe P. falciparum malaria and its associated mortality13. To date, no African country has implemented a national screening programme for SCD18. Even in countries where universal screening programmes have been in place for >10 years (for example, the United Kingdom), estimating the prevalence, incidence and burden of disease remains challenging19,20. In the past 20 years, ∼40,000 confirmed cases of SCD were identified in 76 million newborn babies, with >1.1 million newborn babies with the HbAS genotype in the United States21. Thus, 1 in every 1,941 neonates had SCD, and 1 in every 67 was heterozygous for the βS allele.
Estimated numbers of births with sickle cell anaemia per 100,000 births per country in 2015. Estimates are derived from prevalence data published in Ref. 14. Birth data for 2015–2020 were extracted from the 2017 Revision of the United Nations World Population Prospects database. NA, not applicable.
The incidence of SCD varies by state, race and ethnicity22,23. Among African Americans, ∼1 in 360 newborn babies has SCD. Substantial demographical changes have resulted in a more-diverse population at risk and a high prevalence of SCD in immigrant populations. Newborn screening studies for SCD in the state of New York document the marked effect of immigration on the frequency of neonates with SCD24, as most of them have foreign-born mothers.
The incidence of SCD in newborn babies varies substantially among the states in Brazil, reflecting the ethnic heterogeneity of the Brazilian population. In 2014, the incidence of SCD was ∼1 in 650 newborn babies screened in the state of Bahia, 1 in 1,300 in the state of Rio de Janeiro and 1 in 13,500 in the state of Santa Catarina25. Nationwide, in 2016, 1,071 newborn babies had SCD and >60,000 were heterozygous for the βS allele (F.F.C., unpublished observations). There are an estimated 30,000 individuals with SCD in the whole country. The prevalence of the βS allele in Brazil varies from 1.2% to 10.9%, depending on the region, whereas the prevalence of the βC allele is reported to be between 0.15% and 7.4%25–30. The number of all-age individuals affected by SCA globally is currently unknown and cannot be estimated reliably owing to the paucity of epidemiological data, in particular mortality data, in areas of high prevalence.
Disease severity
The variability in the clinical severity of SCA can partly be explained by genetic modifiers, including factors that affect HbF level and co-inheritance of α-thalassaemia (see below)31,32. For example, the Arab–India haplotype (a haplotype is a set of DNA polymorphisms that are inherited together), which is found in an area extending from the eastern coast of Saudi Arabia and East Africa to India, is considered to be associated with a phenotype milder than that associated with the four African haplotypes (Benin, Bantu, Cameroon and Senegal haplotypes), and, within India, this phenotype could be milder in the tribal populations than in the non-tribal populations33 owing to a higher level of HbF32. However, evidence suggests that the range of severity of SCD in India is wider than previously thought34. Environmental factors (such as the home environment, socio-economic status, nutrition and access to care) also influence the severity of the disease; however, apart from malaria, their role has rarely been investigated35,36. Although some complications are more frequent in some regions than in others (for example, leg ulcers are common in tropical regions but are relatively rare in temperate climates37, whereas priapism (persistent and painful erection) is common in patients of African ancestry but rarer in those of Indian ancestry38), these geographical differences have never been comprehensively and rigorously documented.
Disease burden
It has been estimated that 50–90% of children with SCA who live in sub-Saharan Africa die by 5 years of age10. Most of these children die from infections, including invasive pneumococcal disease and malaria39,40. Owing to the limited data across most areas of high prevalence, it is difficult to precisely assess the future health and economic burden of SCD. As low-income and middle-income countries go through epidemiological transition (that is, changing patterns of population age distributions, mortality, fertility, life expectancy and causes of death, largely driven by public health improvements), which involves substantial reductions in infant mortality that enable SCA diagnoses and treatment, and international migrations contribute to further expand the distribution of the βS allele, the health burden of this disease will increase41. Demographical projections estimated that the annual number of newborn babies with SCA worldwide will exceed 400,000 by 2050 (Ref. 17).
Mechanisms/pathophysiology
The landmark complication associated with SCA is the vaso-occlusive pain crisis. Although vaso-occlusion is a complex phenomenon, HbS polymerization is the essential pathophysiological occurrence in SCA42–44. HbS polymerization changes the shape and physical properties of erythrocytes, resulting in haemolytic anaemia and blockage of blood flow, particularly in small (and some large) vessels, which can damage any organ. HbS polymerization can also occur in reticulocytes, which account for ∼20% of the red blood cells in individuals with SCA. Direct and indirect consequences of haemolysis play a part in modifying the course and complications of SCD. Furthermore, HbS polymers lead to other abnormalities at the cellular level that contribute to the overall pathophysiological mechanism of SCD. The several variant genotypes of SCD (double heterozygous states or SCA with modifying genes) share a common pathophysiology as described in this section. The variants provide nuanced phenotypic differences or reduced severity (Fig. 1).
Erythrocyte morphology
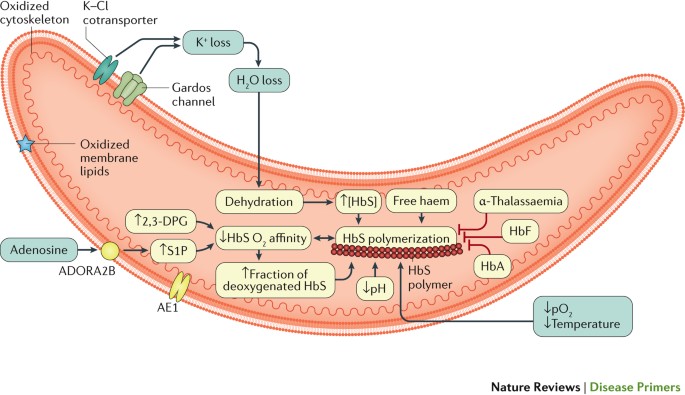
HbS oxygen affinity and polymerization. HbS has reduced oxygen affinity compared with HbA. Reduced HbS oxygen affinity exacerbates HbS polymerization, which in turn further reduces HbS oxygen affinity45 (Fig. 3). HbS oxygen affinity is further reduced by 2,3-diphosphoglycerate (2,3-DPG), which is a glycolytic intermediate that is physiologically present at very high levels in sickle erythrocytes and, through interaction with deoxygenated β-globin subunits, reduces Hb oxygen affinity46. At any partial pressure of oxygen (pO2), low HbS oxygen affinity kinetically favours an increase in the fraction of deoxygenated HbS (which is the tense conformation (T-state) that readily polymerizes), which in turn promotes HbS polymerization and the formation of sickle erythrocytes. Initial reports indicate that sickle erythrocytes have increased sphingosine kinase activity, leading to high levels of sphingosine-1-phosphate, which also decreases HbS oxygen affinity47. Sphingosine kinase is activated by increased levels of plasma adenosine (resulting from the hydrolysis of adenosine nucleotides that are released from erythrocytes during haemolysis) via the erythrocyte adenosine receptor A2b48,49.
Long polymers of sickle haemoglobin (HbS) align into fibres, which then align into parallel rods. The polymer has a helical structure with 14 HbS molecules in each section42,55,262. The polymerization of HbS depends on many factors, including the HbS concentration, partial pressure of oxygen (pO2), temperature, pH, 2,3-diphosphoglycerate (2,3-DPG) concentration and the presence of different Hb molecules263–265. The basic concept of HbS polymerization kinetics is the double nucleation mechanism. Before any polymer is detected, there is a latency period (delay time) in which deoxygenated HbS molecules form a small nucleus, which is followed by rapid polymer growth and formation266,267. Free cytoplasmic haem can increase the attraction of the HbS molecules and the speed of nucleation and polymer formation268. Cation homeostasis is abnormal in sickle erythrocytes, leading to the dehydration of cells. Potassium loss occurs via the intermediate conductance calcium-activated potassium channel protein 4 (also known as the putative Gardos channel) and K–Cl cotransporter 1 (KCC1), KCC3 and/or KCC4 (Refs 269,270). Plasma adenosine can also reprogramme the metabolism of the erythrocyte, altering sphingosine-1-phosphate (S1P). ADORA2B, adenosine receptor A2b; AE1, band 3 anion transport protein; HbA, haemoglobin A; HbF, fetal haemoglobin.
HbS polymerization correlates exponentially with the concentration of HbS within the erythrocyte and also with the composition of other haemoglobins that variably participate in polymers50. In α-thalassaemia, reduced production of α-globin subunits favours the formation of unstable βS tetramers (formed by four sickle β-globin subunits), which are proteolyzed, leaving a lower HbS concentration that slows HbS polymerization and haemolysis. Abnormal cation homeostasis (described in the following section) in sickle erythrocytes leads to cell dehydration, which results in increased HbS concentration and polymerization (Fig. 3). As the polymer fibres extend, they deform the erythrocytes and interfere with their flexibility and rheological properties (that is, how they flow), eventually resulting in vaso-occlusion51. This impaired blood flow rheology is worsened by erythrocyte aggregation, especially in individuals with SCD and high haematocrit (the percentage of blood volume composed of erythrocytes)51. Repeated episodes of HbS polymerization and erythrocyte sickling in conditions of low pO2 and unsickling in conditions of high pO2 can lead to severe alterations in the membrane structure and function (see below) and abnormal calcium compartmentalization. Membrane deformation and erythrocyte dehydration eventually result in the formation of an irreversibly sickled cell — a sickle erythrocyte that can no longer revert to its natural shape52–55.
Altered erythrocyte membrane biology. HbS polymerization directly or indirectly alters the typical lipid bilayer and proteins of the erythrocyte membrane, which leads to reduced cellular hydration, increased haemolysis and abnormal interactions with other blood cells and contributes to early erythrocyte apoptosis55–58 (Fig. 4). Several membrane ion channels are dysfunctional, including the erythroid K–Cl cotransporter 1 (KCC1; encoded by SLC12A4), KCC3 (encoded by SLC12A6) and KCC4 (encoded by SLC12A7), the putative Gardos channel (encoded by KCNN4) and Psickle, the polymerization-induced membrane permeability, most likely mediated by piezo-type mechanosensitive ion channel component 1 (PIEZO1), resulting in reduced cellular hydration. In a subpopulation of sickle erythrocytes, phosphatidylserine (which is usually confined to the inner layer of the membrane) is exposed on the erythrocyte surface. Circulating phosphatidylserine-exposing erythrocytes have a role in many important pathophysiological events, including increased haemolysis, endothelial activation, interaction between erythrocytes, white blood cells and platelets and activation of coagulation pathways59,60. HbS polymers and HbS oxidation (see below) also affect membrane proteins that have structural functions, especially the band 3 anion transport protein, and these changes lead to membrane microvesiculation and the release of erythrocyte microparticles61,62. These submicron, unilamellar vesicles are shed from the plasma membrane under cellular stress to the membrane and cytoskeleton. In SCD, they are derived in large numbers from erythrocytes63 but also from platelets, monocytes and endothelial cells. Microvesicles possess cell-surface markers, cytoplasmic proteins and microRNAs derived from their cell of origin and can affect coagulation, adhesion, inflammation and endothelial function64,65. By contrast, exosomes originate from the endosomal system66 and have been less studied in SCD.
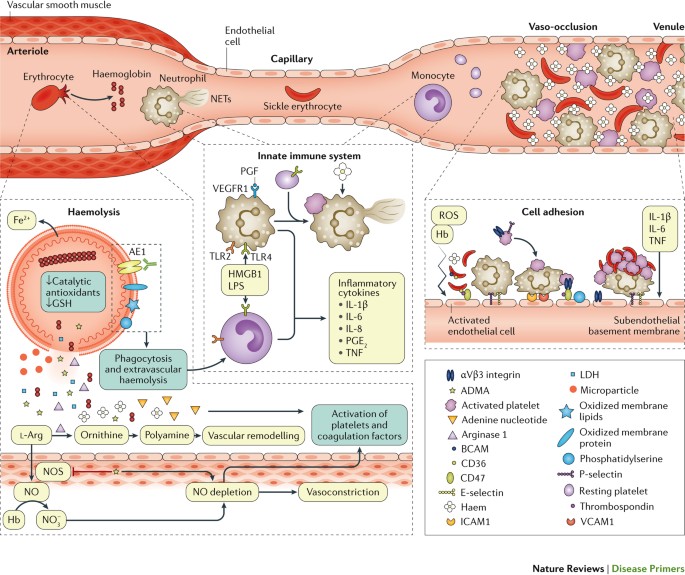
Damage and dysfunction of the erythrocyte membrane caused by sickle haemoglobin (HbS) polymerization lead to haemolysis. Oxidized membrane proteins reveal antigens that bind to existing antibodies, and membranes expose phosphatidylserine; both mechanisms promote phagocytosis of erythrocytes by macrophages, a pathway of extravascular haemolysis. Intravascular haemolysis releases the contents of erythrocytes into the plasma. Hb scavenges nitric oxide (NO), arginase 1 depletes the ʟ-arginine substrate of NO synthase (NOS), and asymmetric dimethylarginine (ADMA) inhibits NOS. Reactive oxygen species (ROS) further deplete NO, leading to vasoconstriction and vascular remodelling, especially in the lung. Adenine nucleotides and NO deficiency promote platelet activation and activation of blood clotting proteins. Haem and other danger-associated molecular pattern (DAMP) molecules activate the innate immune system. Ligand-bound Toll-like receptor 4 (TLR4) and TLR2 activate monocytes and macrophages to release inflammatory cytokines, which promote an inflammatory state and activation of endothelial cells. TLR4 activation on platelets promotes their adhesion to neutrophils, which in turn release DNA to form neutrophil extracellular traps (NETs). Circulating blood cells adhere to each other and to the activated endothelium, contributing and potentially even initiating vaso-occlusion. In postcapillary venules, activated endothelial cells that express P-selectin and E-selectin can bind rolling neutrophils. Activated platelets and adhesive sickle erythrocytes can adhere to circulating or endothelium-bound neutrophils and form aggregates. Sickle erythrocytes might also bind directly to the activated endothelium. The figure shows only some examples of the complex and redundant receptor–ligand interactions involved in the adhesion of circulating cells to the damaged endothelium and exposed subendothelium. AE1, band 3 anion transport protein; BCAM, basal cell adhesion molecule; GSH, glutathione; HMGB1, high mobility group protein B1; ICAM1, intercellular adhesion molecule 1; LDH, lactate dehydrogenase; LPS, lipopolysaccharide; PGE2, prostaglandin E2; PGF, placenta growth factor; TNF, tumour necrosis factor; VCAM1, vascular cell adhesion protein 1; VEGFR1, vascular endothelial growth factor receptor 1.
Haemolysis
Sickle erythrocytes are highly unstable, with a lifespan that is reduced by ≥75%65,67. Haemolysis is thought to occur principally via extravascular phagocytosis by macrophages, but a substantial fraction (roughly one-third) occurs through intravascular haemolysis68 (Fig. 4). It has been hypothesized that the rate of intravascular haemolysis in SCD is insufficient to produce a clinical phenotype, including pulmonary hypertension69, the most serious consequence of intravascular haemolysis. However, the epidemiological, biochemical, genetic and physiological data supporting a link between intravascular haemolysis and vasculopathy continue to expand70.
Oxidative stress. Haemolysis is both a cause and an effect of oxidative stress. The substantial levels of oxidative stress in sickle erythrocytes enhance HbS auto-oxidation, which could contribute to the damage of the cell membrane, premature erythrocyte ageing and haemolysis65. In addition to the accelerated auto-oxidation of HbS, oxygen radicals result from increased expression of oxidases, especially xanthine dehydrogenase and xanthine oxidase, and reduced NADPH oxidase71,72, extracellular haem and Hb in plasma and probably also from recurrent ischaemia–reperfusion of tissues. Cytoskeletal proteins and membrane lipids become oxidized, and this chronic severe oxidative stress in sickle erythrocytes depletes the levels of catalytic antioxidants65 such as superoxide dismutase, peroxiredoxin 2 and peroxiredoxin 4 (Refs 46,73). This issue is worsened by depletion of the endogenous reductant glutathione46,74; impaired antioxidant capacity probably contributes to haemolysis.
Free plasma Hb and haem. Extracellular Hb (in plasma or in microparticles64,65) and haem in plasma promote severe oxidative stress, especially to blood vessels and blood cells65. Continuous auto-oxidation of extracellular Hb produces superoxide, which dismutates into hydrogen peroxide (H2O2), a source for additional potent oxidative species, including the ferryl ion, which promotes vasoconstriction65. Extracellular Hb scavenges nitric oxide (NO; which is generated by NO synthase (NOS) in endothelial cells and promotes vasodilation) ∼1,000-fold more rapidly than cytoplasmic Hb, thereby decreasing NO bioavailability75. This decreased bioavailability of NO results in vascular dysfunction, indicated by impaired vasodilatory response to NO donors, activation of endothelial cells (producing cell-surface expression of endothelial adhesion molecules and detected by elaboration of soluble ectodomains of the adhesion molecules into plasma) and haemostatic activation of platelets, indicated by cell-surface expression of P-selectin (which mediates the interaction between activated platelets and leukocytes) and activated αIIbβ3 integrin70. Markers of haemolytic severity (such as low Hb or high serum lactate dehydrogenase) predict the clinical risk of developing vascular disease complications (see below).
Disruption of arginine metabolism. Intravascular haemolysis releases two factors that interfere with NOS activity. The enzyme arginase 1 competes with NOS for L-arginine, the substrate required for NO production by NOS76. Arginase 1 converts L-arginine into ornithine, which fuels the synthesis of polyamines, which in turn facilitate cell proliferation77, potentially of vascular cells, probably promoting vascular remodelling. Asymmetric dimethylarginine (ADMA) is an endogenous NOS inhibitor and a proteolytic product of proteins methylated on arginine; ADMA is abundant in erythrocytes and is also released during haemolysis78. Both ADMA and depletion of L-arginine by arginase 1 could contribute to uncoupling of NOS, which then produces reactive oxygen species (ROS) instead of NO79,80.
Plasma lipids. Individuals with SCA often have a form of dyslipidaemia that is associated with vasculopathy: triglyceride levels are high and correlate with haemolytic severity81. Although total cholesterol levels are generally low in individuals with SCA, the levels of apolipoprotein A-I (which promotes hepatic cholesterol catabolism and NOS activity) are particularly low, especially during vaso-occlusive pain crises and in association with markers of pulmonary hypertension and endothelial dysfunction82. Genetic variants of apolipoprotein L1 have been associated with renal disease in SCA83.
Innate immune system activation
Plasma haem and Hb act as danger-associated molecular patterns (DAMPs) to activate the innate immune system and heighten the adhesiveness of circulating blood cells to each other and to the endothelium, thereby triggering vaso-occlusion70 (Fig. 4). Haem activates neutrophils to release DNA as neutrophil extracellular traps (NETs) that increase platelet activation and thrombosis and promote pulmonary vaso-occlusion84 and release of placenta growth factor (PGF) from erythroblasts (nucleated precursors of erythrocytes). PGF is a ligand for vascular endothelial growth factor receptor 1 on endothelial cells and macrophages, promoting release of endothelin 1 (a vasoconstrictor), which contributes to pulmonary hypertension85. Toll-like receptor 4 (TLR4) is highly expressed in immune cells in SCD, and tissue damage and platelet activation release high mobility group protein B1 (HMGB1), a high-affinity TLR4 ligand. TLR4 also binds lipopolysaccharide (LPS) derived from Gram-negative bacteria, which could explain why infections promote vaso-occlusive crises in individuals with SCA. TLR4 ligands activate monocytes and macrophages to release inflammatory cytokines, which promote an inflammatory state and activate the adhesiveness of neutrophils, platelets and endothelial cells. Finally, increased intracellular iron from turnover of haemolyzed and transfused erythrocytes is associated with markedly increased expression in the peripheral blood mononuclear cells of several components of the inflammasome pathway86.
Cell adhesion and vaso-occlusion
Endothelium activation. Vaso-occlusion in SCA is a complex phenomenon in which interactions between erythrocytes and endothelial cells, leukocytes and platelets play a central part (Fig. 4). Endothelial cells are probably activated by direct contact of sickle erythrocytes, free haem and Hb and hypoxia-induced ROS87. Reduced NO bioavailability could induce the expression of adhesion molecules and production of endothelin 1. The increased expression of endothelial adhesion molecules (such as vascular cell adhesion protein 1 (VCAM1)88,89, intercellular adhesion molecule 1 (ICAM1)90, P-selectin, E-selectin, leukocyte surface antigen CD47 and αVβ3 integrin) and exposed heparin sulfate proteoglycans and phosphatidylserine are responsible for erythrocyte and leukocyte adhesion89. Activated endothelial cells also produce inflammatory mediators, such as IL-1β, IL-6 and tumour necrosis factor (TNF), which lead to a chronic inflammatory state.
Erythrocytes. Sickle erythrocytes are more adhesive to endothelial cells than normal erythrocytes87,91. Many adhesion molecules (the most important include α4β1 integrin (also known as very late antigen 4 (VLA4), which is reticulocyte-specific), platelet glycoprotein 4 (also known as CD36) and basal cell adhesion molecule (BCAM)) are overexpressed by sickle red blood cells and mediate the adhesion to the endothelium92. Interestingly, reticulocytes and deformable erythrocytes (that is, erythrocytes that have not become permanently sickled) are substantially more adhesive than the irreversible and dense sickle erythrocytes93.
Leukocytes. High baseline leukocyte numbers are associated with increased morbidity and mortality in SCA94,95. Many studies in mouse models of SCA indicate that neutrophils have an important role in vaso-occlusion; neutrophils adhere to the endothelium and sickle erythrocytes could bind to these cells, thereby reducing blood flow and promoting vaso-occlusion96. Indeed, neutrophils are in an activated state in SCA and have increased expression of αMβ2 integrin with enhanced adhesion to endothelial and subendothelial proteins (such as fibronectin)97. Selectins produced by activated endothelium have an important role in the initial binding of neutrophils to the vascular wall96.
Platelets. Platelets play an important part in the pathophysiology of SCA and are in an activated state96, with high levels of P-selectin and activated αIIbβ3 integrin. Moreover, several biological markers of activated platelets are increased in SCA (for example, platelet microparticles64, thrombospondin93, platelet factor 4 (also known as CXC-chemokine ligand 4 (CXCL4)) and β-thromboglobulin). Platelets are found in the circulating heterocellular aggregates of neutrophils and red blood cells (mainly reticulocytes) in the blood from individuals with SCA, and their adhesion to these aggregates is mediated in part through P-selectin98. These data strongly suggest that platelets have a role in the formation of these aggregates. Platelets could also act as accessory cells of the innate immune system by releasing cytokines99.
Diagnosis, screening and prevention
Diagnostic opportunities
The goals and methods of diagnosis of SCD vary with the age of the person. In general, there are four overlapping testing periods: preconception, prenatal, neonatal and post-neonatal. Preconception testing is designed to identify asymptomatic potential parents whose offspring would be at risk of SCD. Laboratory techniques used for preconception testing are routine basic methods of protein chemistry that enable separation of Hb species according to their protein structure, including Hb electrophoresis, high-performance liquid chromatography (HPLC) and isoelectric focusing100. Prenatal diagnosis is a generally safe but invasive procedure and is offered during early pregnancy to couples who tested positive at preconception screening. It requires fetal DNA samples obtained from chorionic villus analysis performed at 9 weeks of gestation100. Non-invasive prenatal diagnosis techniques are being developed but are still investigational. These new techniques can detect fetal DNA in maternal circulation by as early as 4 weeks of gestation. Some couples who test positive at preconception screening might opt for in vitro fertilization with pre-implantation genetic diagnosis, if available, to genetically identify at-risk embryos before embryo transfer occurs101.
Newborn screening. Newborn screening for SCD is performed at birth before symptoms occur, using Hb protein analysis methodologies. Two types of newborn screening programmes have been used: selective screening of infants of high-risk parents (targeted screening) and universal screening. Universal screening is generally more cost-effective, identifies more newborn babies with disease and prevents more deaths17,102. In areas without newborn screening programmes, the initial diagnosis of SCD occurs at approximately 21 months of age103. For many individuals with SCD, the initial presentation is a fatal infection or acute splenic sequestration crisis103. Early diagnosis accompanied by penicillin prophylaxis and family education reduces the mortality in the first 5 years of life from 25% to <3%103,104. Similar positive results are found in low-income countries105,106.
Post-neonatal testing. The requirement of post-neonatal testing for SCD is influenced by several factors that affect the general population's knowledge of their SCD status. These factors include the regional success of neonatal screening programmes, immigration of at-risk patients not previously tested and access to neonatal results in older patients107. HbAS is a benign condition and not a disease, but it is also a risk factor for uncommon serious complications107. Thus, knowledge of one's own HbAS status is important in the prevention of rare serious complications and in family planning.
HbAS can also be detected by newborn screening programmes; however, HbAS detection is not the primary objective, and many programmes do not provide this information or offer associated counselling. Individuals who wish to have children should be screened to discover heterozygous genotypes that could be important in genetic counselling. HbAS screening enables informed decisions concerning preconception counselling and prenatal diagnosis.
Routine fitness training does not increase the risk of mortality for individuals with HbAS. However, there is a concern of increased risk of rhabdomyolysis (rapid destruction of skeletal muscle) and sudden death during intense, prolonged physical activity; this risk can be mitigated by proper training108. In some regions, these observations have resulted in voluntary or mandatory screening of athletes for HbAS107. There are rare and specific complications of HbAS that should prompt HbAS testing. These include haematuria (blood in the urine), hyphema (blood inside the eye's anterior chamber) and renal medullary carcinoma, a rare malignancy. HbAS could be a risk factor for chronic kidney disease and pulmonary embolism109.
Newborn screening programmes
Newborn screening programmes for SCD are now in place in several European countries, the United States (Box 1), India, Africa (Box 2) and Brazil (Box 3).
Screening in Europe. Newborn screening programmes for SCD in the United Kingdom became universal in 2006 (Ref. 110); the primary aim of the programme is to diagnose SCD, but if a baby has HbAS, the parents are provided with specific informational materials. In France, screening for SCD has been in place since 2000 but is restricted to newborn babies whose parents both originate from SCD-endemic regions111. In Spain, universal screening has been recommended for regions with a high annual birth rate and SCD prevalence (for example, Catalonia and Madrid), whereas targeted screening is recommended for regions with a low annual birth rate and SCD prevalence112. Screening programmes are also present in Italy113 and Germany114.
Screening in the United States. In the United States, statewide newborn screening originated in New York state in 1975 (Box 1), and by 2007, all states had universal screening programmes21. In the United States, HPLC and isoelectric focusing are the predominant screening methods21,100. Confirmation of the diagnosis by DNA analyses to detect Hb variants is commonly used but is not standardized among states. A major gap in these programmes is the lack of follow-up and the variability of statewide education programmes115. The identification of substantial clinical morbidity occasionally associated with individuals with HbAS has not yet resulted in routine counselling and genetic testing of family members of newborn babies with HbAS107.
Screening in India. The population of India consists of >2,000 different ethnic groups, most of which have practised endogamy (the custom of marrying only within the limits of the local community) over centuries. Thus, although the βS allele has been detected in many ethnic groups, its prevalence has been enriched in some. The at-risk population consists of several hundreds of millions of individuals, predominantly belonging to historically disadvantaged groups116. Screening efforts have focused on groups with a high prevalence of the βS allele and areas with large numbers of these at-risk populations. Screening typically consists of an Hb solubility test (a screening test that does not distinguish HbAS from disease) at the point of care with further testing of initial positive samples by HPLC analysis at a reference centre. Screening programmes also include education, testing and genetic counselling. In many hospitals, such services are also offered to the relatives of patients diagnosed with SCD and in the prenatal setting to mothers either previously diagnosed with HbAS or belonging to an at-risk ethnic group. Pilot projects of newborn screening programmes for SCD have been implemented in the states of Gujarat, Maharashtra and Chhattisgarh105,106,117–120, which resulted in detailed data on the prevalence of HbAS in various populations, with ranges of 2–40%. There is considerable regional variation in the implementation of follow-up approaches such as comprehensive care, penicillin prophylaxis and immunization against pneumococcus.
Screening in Africa. No country in sub-Saharan Africa has implemented a universal newborn screening programme for any disease121. However, a few countries in sub-Saharan Africa have developed pilot newborn screening programmes for SCD. Among these, Ghana's National Newborn Screening Programme for SCD, launched in 2010 following a 15-year pilot study, is the most developed122 (Box 2). Other countries in Africa where small-scale or pilot newborn screening programmes for SCD have been conducted or are ongoing include Angola123, Benin124, Burkina Faso125, Burundi126, Democratic Republic of the Congo127, Nigeria128, Rwanda126, Senegal129, Tanzania130 and Uganda131. Screening followed by penicillin prophylaxis can reduce early mortality from pneumococcal bacteraemia103,104. Nevertheless, current and future numbers of individuals with SCA or HbAS make the scalability of the interventions implemented in high-income, low-burden countries (such as universal newborn screening programmes) in low-resource settings challenging. There is no mandatory or large-scale preconception screening programme for adults who wish to have children in any African country. However, several churches require couples to be screened for SCD-related conditions as a prerequisite for marriage approval. Such screening often involves inexpensive but inconclusive ‘sickling’ and solubility tests, which cannot identify individuals with the βC allele or β-thalassaemia, conditions that, although not characterized by the presence of HbS, are of genetic counselling relevance. There are very few much-needed certified genetic counsellors to support the screening programmes. The Sickle Cell Foundation of Ghana launched the first Sickle Cell Genetic Counsellor Training and Certification Programme in June 2015 (Box 2).
Phenotypes in sickle cell disease
There is great phenotypic variability among individuals with SCD. Some variability shows a specific geographical distribution and is associated with known or suspected genetic variants132. However, some complications cluster together epidemiologically in subphenotypes, at times united by a common biomarker that suggests a mechanism, such as a particularly low Hb level with a high reticulocyte count or high serum lactate dehydrogenase level, implying more-intense haemolysis. These phenotypes are not mutually exclusive, exist often as a spectrum, can overlap, are probably due to independent genetic modifiers of the underlying mechanisms and might change with ageing.
Vaso-occlusive subphenotype. This SCA subphenotype is characterized by a higher haematocrit than that observed in individuals with other SCA phenotypes; a higher haematocrit promotes higher blood viscosity. Individuals with this phenotype are predisposed to frequent vaso-occlusive pain crises, acute chest syndrome (that is, a vaso-occlusive crisis of the pulmonary vasculature) and osteonecrosis. Co-inheritance of α-thalassaemia reduces haemolysis (by reducing the intracellular concentration of HbS, which slows HbS polymerization and haemolysis) but promotes higher haematocrit133.
Haemolysis and vasculopathy subphenotype. This phenotype is characterized by a lower haematocrit than that found in individuals with the vaso-occlusive subphenotype accompanied by higher levels of serum lactate dehydrogenase and bilirubin, which indicate more-severe haemolytic anaemia. Individuals in this group are at risk of ischaemic stroke, pulmonary hypertension, leg ulceration, gallstones, priapism and possibly nephropathy134. Decreased NO bioavailability, haem exposure and haem turnover are associated with these vasculopathic complications. The severe anaemia also promotes high cardiac output as a compensatory mechanism, and this excessive blood flow has been suggested to promote vasculopathy in the kidney and potentially in other organs.
High HbF subphenotype. Persistent expression of HbF in the range of 10–25% of total Hb owing to genetic variants generally reduces the clinical severity of SCA3,135. However, not all individuals with the common, uneven cellular distribution of HbF (heterocellular distribution) have a mild phenotype. Expression levels of 25–50% of HbF in every erythrocyte (pancellular distribution) lead to nearly complete amelioration of SCA, with rare clinical symptoms and no anaemia136, a finding that could prompt the development of drugs that can induce ‘globin switching’ (that is, the preferential expression of HBG1 and HBG2).
Pain subphenotypes. Individuals with pain-sensitive or pain-protective phenotypes experience pain differently, potentially owing to altered neurophysiology of pain sensation pathways. One example of a genetic modifier of pain is GCH1, which is associated with pain sensitivity in healthy individuals, and a variant of GCH1 is associated with frequency of severe pain in SCA137. Quantitative sensory testing of pain sensitivity is being used to functionally characterize these phenotypes in SCA138.
Management
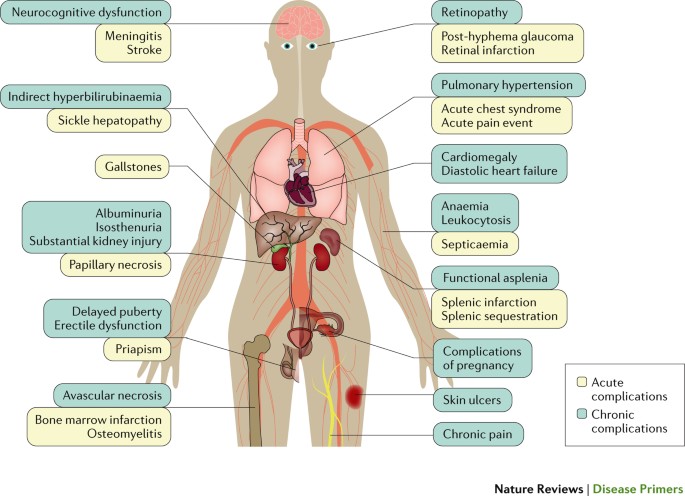
SCD is a complex, multisystem condition characterized by acute and chronic complications (Fig. 5). Advances in general medical care, early diagnosis and comprehensive treatment have led to substantial improvements in the life expectancy of individuals with SCA in high-income countries8,9 as almost all patients survive beyond 18 years of age139. However, even with the best of care, life expectancy is still reduced by ∼30 years, routine and emergency care for individuals with SCD have great financial costs, the quality of life often deteriorates during adulthood and the social and psychological effects of SCD on affected individuals and their families remain underappreciated140. Furthermore, most of these advances have not reached low-income countries141.
Acute complications bring the individual with sickle cell disease (SCD) to immediate medical attention; pain is the most common acute complication. As individuals with SCD age, chronic complications produce organ dysfunction that can contribute to earlier death. Complications of pregnancy include pre-eclampsia, intrauterine growth restriction, preterm delivery and perinatal mortality.
Therapies
Three therapies modify the disease course of SCA: hydroxycarbamide, erythrocyte transfusion and haematopoietic stem cell transplantation142.
Hydroxycarbamide. Hydroxycarbamide (alternatively known in some countries as hydroxyurea), a ribonucleotide reductase inhibitor, has multiple physiological effects, including increasing HbF expression (in most individuals with SCA143) and decreasing leukocyte count. It was approved by the US FDA in 1998 and by the European Medicines Agency (EMA) in 2007 for the treatment of SCD. The drug significantly reduces the incidence of SCA vaso-occlusive crises, hospitalizations and mortality in high-income countries (with studies ongoing in low-resource countries) with an excellent safety profile144, although some patients do not have a beneficial response, usually because of limitations of adherence to treatment145 but possibly sometimes for pharmacogenomic reasons146. Hydroxycarbamide is underutilized because of health-care infrastructure deficiencies in both low-resource and high-resource countries and disproportionate perceptions of carcinogenicity, teratogenicity and reduced fertility, which have not been problems thus far in follow-up studies143,147,148; however, hydroxycarbamide use is increasing. Snapshots from various cohorts over the years show that in high-resource countries, at specialized SCD clinics, up to 63% of patients with SCA may be on hydroxycarbamide149, but the percentage is near zero in most African countries150. Because of very favourable clinical trial results in infants and toddlers151, hydroxycarbamide is prescribed with increasing frequency to children with SCA — up to 45% in multinational SCD centres152. Although there is still limited evidence on whether hydroxycarbamide improves survival and prevents SCD complications in low-income countries153, various studies, including the Realizing Effectiveness Across Continents with Hydroxyurea (REACH) trial, are currently underway and should address knowledge gaps about treatment options for SCA in sub-Saharan Africa150.
Erythrocyte transfusion. This therapy improves microvascular flow by decreasing the number of circulating sickle erythrocytes and is associated with decreased endothelial injury and inflammatory damage154,155. Chronic transfusion therapy, prescribed in high-resource countries primarily to the roughly 10% of patients with SCA at high risk of stroke, can ameliorate and prevent stroke and vaso-occlusive crises156; however, several potential adverse effects, including iron overload, alloimmunization (an immune response to foreign antigens that are present in the donor's blood) and haemolytic transfusion reactions, limit its potential benefits. The availability of oral iron-chelating drugs since 2005 has reduced the adverse effects of iron overload. In countries with limited testing of blood products for infectious agents, there are substantial risks of transmission of blood-borne infections, such as hepatitis B, hepatitis C, HIV infection, West Nile virus infection and others. Transfusion protocols with extended erythrocyte matching that includes the erythrocyte antigens Kell, C, E and Jkb and iron-chelation therapy guidelines improve the safety of this therapy156. Systematic genotyping of blood groups for the patient has been proposed to reduce alloimmunization157.
Haematopoietic stem cell transplantation. Haematopoietic stem cell transplantation in SCA is curative and should be considered in symptomatic patients with a human leukocyte antigen (HLA)-matched family donor. Worldwide, it is estimated that nearly 2,000 individuals with SCA have undergone allogeneic haematopoietic stem cell transplantation; the survival exceeds 90% in US and European studies158,159. In pooled registry data, the average rate of both acute and chronic graft-versus-host disease has been 14% and is generally lower with newer approaches158, and the rate of graft failure has been 2%159. Early results with experimental reduced-intensity conditioning regimens (pretransplantation chemotherapy to ablate or suppress the recipient's bone marrow) are very encouraging160. However, most patients do not have an HLA-matched related donor. Experimental use of expanded donor pools (haploidentical donors (who share 50% of the HLA antigens with the recipient) and unrelated HLA-matched donors) can increase the probability of cure but also increase the rate of graft rejection and mortality, which seem to improve with ongoing research161. Although haematopoietic stem cell transplantation from the bone marrow of a healthy HLA-matched donor can cure SCA, this therapy is limited by the paucity of suitable donors and is available only in high-income countries162.
Management of acute complications
The principles of management of acute complications in SCA (Fig. 5) include the need for early diagnosis, consideration of other non-SCD-related causes and rapid initiation of treatment. The use of standardized protocols for common complications improves outcomes.
Acute pain. Acute pain events usually affecting the extremities, chest and back are the most common cause of hospitalization for individuals with SCA. However, the majority of such events are managed at home with NSAIDs or non-prescription oral opioid analgesics without the involvement of the health care provider. The pathophysiology and natural history of acute pain events are complex, and treatment is suboptimal163. Individual personalized protocols for outpatient and inpatient pain management improve quality of life and decrease hospital admissions164–166. The treatment is guided by the severity of pain, which is generally self-reported using pain severity scales. When home management with oral analgesics, hydration and rest is ineffective, rapid triage with timely administration of opioids is recommended. Initial treatment in a day unit compared with an emergency room drastically decreases hospitalization167. Initiation of treatment for emergency room patients with SCD is often markedly delayed, with patients with SCD waiting 25–50% longer than patients without SCD with similar pain acuity168. In some programmes, innovative emergency room treatment protocols for patients with SCD using standardized time-specific dosing protocols and intranasal fentanyl (an opioid) have substantially reduced time to treatment; similar approaches should be adopted universally164,165. Once an individual is hospitalized, a standardized protocol using patient-controlled analgesia devices is indicated. These intravenous infusion pumps enable patient self-medication and, in general, result in improved analgesic control and less analgesic use169. Incentive spirometry, a simple device that prevents atelectasis (the complete or partial collapse of a lung), with close monitoring of the patient's levels of sedation, hydration and oxygenation improves outcomes. Although intensive analgesia is important for effective medical management of pain in SCD, in some countries, opioids are unavailable owing to resource limitations or are not prescribed or consumed owing to stigma170. Vaso-occlusive crises can sometimes result in sudden unexpected death3,171. The precise aetiology of sudden death in such cases is unclear, although autopsy often shows histopathological evidence of pulmonary arterial hypertension171.
Acute chest syndrome. Acute chest syndrome is the second most frequent reason for hospitalization and a leading cause of death in individuals with SCD — it is often linked to and follows an acute pain event172. The severity of acute chest syndrome increases with age. In adults, >10% of cases are fatal or complicated by neurological events and multi-organ failure173. The initial pulmonary injury is multifactorial, including infection, pulmonary fat embolism, pulmonary infarction and pulmonary embolism174. The presence of underlying, often undetected bronchoreactive lung disease can increase the frequency and severity of acute chest syndrome events175. Early chest X-ray imaging tests and oxygen monitoring of patients with any pulmonary symptoms are necessary. Hospitalization with broad-spectrum antibiotics, bronchodilators, oxygen supplementation and red cell transfusions is often indicated176. Exchange transfusions (in which the patient's blood is replaced by donor blood) and steroids, which decrease acute inflammation, could modify a severe or rapidly deteriorating event176,177. Exchange transfusion is the most effective method of lowering the level of HbS below 30% of the total Hb without raising the total Hb level above 10 g dl−1 (Ref. 178). However, delayed transfusion reactions can complicate transfusion therapy and present as a hyperhaemolytic episode in which the transfused cells and the patient's own red blood cells are destroyed179. Steroids often provide benefit but are associated with an ∼25% risk of mild or severe complications (in particular, there is a high rate of recurrence of acute chest syndrome once the steroids are stopped); thus, their use is usually limited to life-threatening acute chest syndrome events180.
Acute stroke. An acute stroke, including ischaemic and haemorrhagic events, is a medical emergency. Children with SCA have a 300-fold higher risk of acute stroke than other children without SCD, and by 45 years of age, one in four adults with SCA has had a stroke181. In the United States, 25% of individuals with SCA develop an overt stroke, and another 35% have non-focal central nervous system injury181–183. Ischaemic stroke is usually caused by occlusion of a large cerebral artery and can occur as a complication of acute chest syndrome (defined above) or independently, and can manifest with transient ischaemic attack, sudden weakness or loss of consciousness. Prompt evaluation (including MRI of patients with subtler presentations) is indicated. Rapid exchange transfusion is the standard treatment. In addition, chronic transfusion decreases secondary stroke recurrence178. The importance of subsequent monthly chronic transfusion to prevent secondary stroke has been re-affirmed by the Stroke With Transfusions Changing to Hydroxyurea (SWiTCH) study184.
Intracranial haemorrhage or haemorrhagic stroke account for 3–30% of acute neurological events and have a 25–50% acute mortality185. Clinically, these patients present with severe headache or loss of consciousness without hemiparesis. Imaging with angiography could reveal a surgically treatable aneurysm. Individuals with moyamoya vasculopathy, which is a prominent collateral circulation around occluded arteries of the circle of Willis that is frequent in individuals with SCD, are at high risk of intracranial bleeding. When this pathology is electively detected, indirect revascularization using encephaloduroarteriosynangiosis (a surgical procedure that implants the superficial temporal artery to the brain surface, increasing blood flow to the ischaemic area) is often considered to decrease bleeding risk and improve oxygenation186,187.
Acute anaemic events. Over half of patients with SCD will experience an acute anaemic event, which can be fatal, at some point in their life. The most common types of anaemic events are splenic sequestration crisis, aplastic crisis (temporary absence of erythropoiesis) and hyperhaemolytic crisis. Acute splenic sequestration crisis is characterized by rapid swelling of the spleen and hypovolemia with a sudden fall in Hb levels. As many as 30% of young children experience acute sequestration events, which are a leading cause of infant mortality. Early detection is crucial, and transfusion followed by elective splenectomy is usually required188. Nonsurgical supportive care can be successful, and, when necessary, transfusion with extended red blood cell antigen-matched erythrocyte units and selective use of immunosuppressive therapy are indicated.
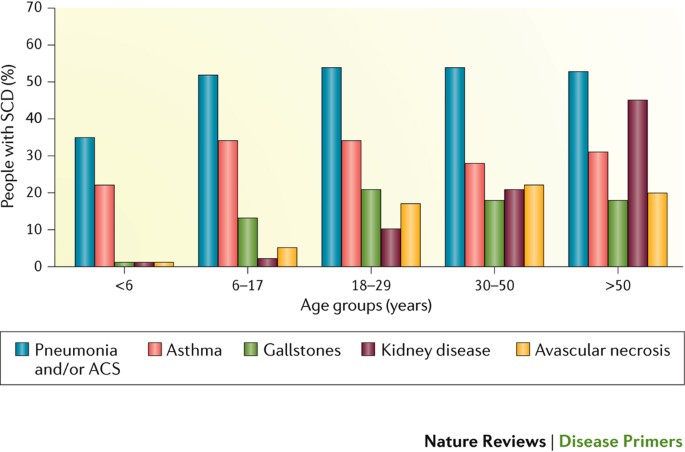
Cholelithiasis. Cholelithiasis (gallstones) results from the chronic accelerated rate of erythrocyte destruction in individuals with SCD. Haem is metabolized to bilirubin, which in the bile can form insoluble calcium bilirubinate, which in turn precipitates as a pigment and forms gallstones. Of note, a variant of UGT1A1 (which encodes a protein involved in bilirubin processing) increases bilirubin metabolism and, therefore, the formation of gallstones in individuals with SCD189. By adulthood (Fig. 6), 20% of patients with SCD have acute complications from gallstones, which can promote cholecystitis (inflammation of the gall bladder) and often necessitate cholecystectomy (surgical removal of the gallbladder)190. By contrast, individuals with SCD who also inherit α-thalassemia have reduced haemolysis, bilirubin production and gallstone formation189.
Development of clinical complications in 5,100 individuals with sickle cell disease (SCD) identified in the California Hemoglobinopathy Surveillance Program271. ACS, acute chest syndrome.
Long-term management
Improved management of acute complications is associated with a longer survival. As individuals with SCD age, chronic problems resulting from cumulative organ injury can lead to severe morbidity191 (Figs 5,6). Chronic pain is common; the Pain in Sickle Cell Epidemiology Study (PiSCES) found that adults with SCD have pain on 55% of days192, and pain, in general, is a poorly managed complication of SCD193. Individuals with SCD and recurrent pain have altered brain network connectivity, which affects their response to treatment194. Chronic pain requires a multidisciplinary team familiar with neuropathic pain tolerance, withdrawal symptoms and hyperanalgesia syndrome193. Hydroxycarbamide, selective use of chronic transfusions in severe cases and long-acting opioids are useful components of a multidisciplinary pain management approach.
Avascular necrosis of the hip is a common cause of chronic pain that eventually develops in many individuals with SCD195; in >20% of hospitalizations, symptoms are related to avascular necrosis. Although core decompression (in which a small core of bone is removed from the damaged area, lowering the bone marrow pressure and stimulating healthy bone regrowth), physiatry (rehabilitation) therapy and analgesics are temporarily helpful, total hip replacement is often required.
Chronic kidney disease is relatively common in older individuals with SCD and is thought to have a poor prognosis in these individuals compared with individuals without SCD196. This worse outcome could in part be due to delayed access to dialysis and renal transplant for individuals with SCD, as they might not be considered good candidates for these therapies. Of note, individuals with SCD who receive a timely renal transplantation have an outcome comparable with that of individuals without SCD who receive a transplant197,198.
Although screening for brain injury with annual transcranial Doppler (TCD) screening and/or MRI and chronic transfusion therapy for high-risk patients decrease the frequency and severity of stroke complications, patients continue to have progressive neurocognitive injury and require close observation and long-term therapy182. In addition, implementation of multidisciplinary plans for management of other common chronic complications of SCD (for example, cardiopulmonary dysfunction, priapism and leg ulcers) improves the quality of life of these patients as they age199,200.
Prevention of complications
Preventive strategies have changed the long-term outcome in SCD more than any other approach. Prevention of life-threatening infections and stroke has drastically reduced childhood mortality in SCD; generalized screening of individuals with SCD for risk factors and early evidence of disease enable the implementation of treatment that can reduce morbidity. Screening for pulmonary, renal and systemic hypertension, retinopathy and damage to other organs is indicated201. Detailed generalized screening recommendations for SCD are available202,203.
Prevention of infection. Until the 1990s, in the United States, up to 30% of young children with SCA died from infections, predominantly due to encapsulated bacteria104, owing to a common childhood deficiency of immune response to polysaccharide antigens204 and exacerbated in SCA by impaired clearance of blood-borne bacteria as a result of functional asplenia104. The introduction of prophylactic penicillin treatment decreased the incidence of pneumococcal bacteraemia associated with impaired splenic function by 85%104. Prophylactic penicillin has remained safe and beneficial in patients up to at least 5 years of age. The universal use of pneumococcal and other standard vaccinations has further lowered infectious disease mortality. The first conjugated pneumococcal vaccine decreased the rate of pneumococcal bacteraemia in children of <3 years of age by 93.4% and added protection to the large cohort of individuals with SCD who have suboptimal compliance with prophylactic penicillin therapy205. Long-term penicillin prophylaxis has raised concerns about the development of penicillin-resistant pneumococcal colonization and disease206, especially in low-income countries, although the benefit-to-risk ratio of prophylaxis is still high. The pneumococcal conjugate vaccine PCV13 and pneumococcal polysaccharide vaccine PPSV23 (Ref. 207) can prevent infection by most, but not all, serotypes.
Prevention of central nervous system injury. Cerebral vascular injury and neuro-ischaemic damage are a leading cause of death and morbidity in children and adults with SCA. The complications of these events are largely irreversible and mandate universal prevention and screening policies. TCD screening to detect increased vascular velocity can contribute to identifying children at high risk of stroke, which can be largely prevented by initiating transfusion therapy208. The landmark Stroke Prevention Trial in Sickle Cell Anaemia (STOP) study demonstrated that neurologically healthy children with elevated TCD measurements (vascular velocity >200 cm s−1) are at high risk of stroke, and chronic monthly transfusions reduced the rate of strokes from ∼11% to 1%208. These findings suggest that all children with SCA should be screened annually with TCD. The STOP II study found that discontinuing these preventive transfusions was not safe and that transfusion therapy for an indefinite period of time might be necessary209.
Nevertheless, chronic transfusion therapy for primary stroke prevention is associated with substantial complications and is not available in many low-income countries. Hydroxycarbamide therapy has been associated with decreased TCD vascular velocity210. The TCD With Transfusions Changing to Hydroxyurea (TWiTCH) trial determined that hydroxycarbamide therapy at maximum dosing was non-inferior to blood transfusions for primary stroke prevention in children with non-severe vasculopathy on MRI findings and who had been receiving transfusions for ≥1 year211. The Stroke Prevention Study in Nigeria (SPIN) provided pilot evidence that TCD screening followed by fixed-dose hydroxycarbamide therapy is feasible and has the potential to prevent strokes in low-resource areas212. Global TCD screening of all children with SCA is a major public health priority.
TCD screening does not detect silent infarction involving small-vessel disease, which is a major cause of neurocognitive impairment in SCD. The Silent Cerebral Infarct Transfusion Multi-Centre Clinical Trial (SIT) used MRI to screen children who had normal TCD measurements and no neurological symptoms213. Children with small non-focal cerebral infarctions (detected by MRI) were randomly assigned to receive transfusion or observation. Children in the transfusion group had a 59% relative risk reduction for stroke. Whether all children should be screened with MRI remains debated. However, all individuals with soft (subtle) neurological signs or neurocognitive changes (such as sudden unexplained decline in school or work performance) should undergo MRI screening, and those with silent infarction should be offered transfusion therapy. Neurocognitive testing, where available, is a useful tool in identifying individuals who have non-focal ischaemic cerebral injury, which can progress with age and is common in adults with no neurological symptoms182.
Prevention of pulmonary complications. Pulmonary disease is a leading cause of morbidity and mortality in individuals with SCD3,191,214. Asthma is an independent predictor of mortality in this population215,216. Unrecognized bronchoreactive lung disease is common in paediatric patients and increases the severity and frequency of acute chest syndrome events. Many adults have undetected, restrictive chronic lung disease, which is a risk factor of pulmonary failure and myocardial injury217. Incorporating respiratory symptom questionnaires and routine spirometry into outpatient management is indicated. Pulmonary hypertension or an elevation in the tricuspid regurgitant jet velocity (TRV), which is a marker of pulmonary hypertension, are also independent predictors of mortality. Individuals with TRV of ≥3 cm s−1 have a tenfold increased mortality compared with individuals with normal TRV214. The American Thoracic Society recommends that all adults with SCA undergo serial echocardiography every 1–3 years to detect pulmonary hypertension203.
Prevention of renal complications. One-third of individuals with SCA develop chronic kidney disease, and up to 18% of individuals with SCA require dialysis or renal transplantation218. Proteinuria is strongly associated with progressive disease; serial urinary screening for proteinuria accompanied with treatment with angiotensin-converting enzyme inhibitors (which correct the proteinuria) could lower the risk of chronic kidney disease201. Mild systemic hypertension (120–139/80–90 mmHg) increases the risk of stroke, pulmonary hypertension, nephropathy, mortality and hospitalization in SCD219,220, and early diagnosis and treatment are beneficial220,221. Asymptomatic proliferative retinopathy can occur in up to 43% of individuals with HbSC disease and in 14% of individuals with SCA222; if untreated, asymptomatic proliferative retinopathy results in loss of visual acuity223.
Comorbidities
Individuals with SCD are prone to other unrelated diseases that can modify each individual's clinical course. Very common (in at least one-third of individuals with SCD) comorbidities identified using screening questionnaires are depression and anxiety224,225. Depression and anxiety are associated with greater sensitivity to pain226 and greater health care utilization227. Depression is also linked to sleep disturbance228 and, in general, might be under-recognized and undertreated in individuals with SCD. Asthma is common: it occurs in at least 25% of children with SCD and is associated with an increased incidence of acute pain events, acute chest syndrome and early death175,215,216. Venous thrombosis has been reported in up to 25% of individuals with SCD and could be due to the commonly observed activation of the haemostatic system229.
Quality of life
Generic health-related quality-of-life (HRQOL) instruments (for example, the 36-Item Short Form Health Survey (SF-36) for adults and the Pediatric Quality of Life Inventory (PedsQL) for children)230,231 measure physical, emotional and social functioning and enable the comparison of individuals with SCD with healthy individuals. Disease-specific measures, such as the PedsQL Sickle Cell Disease module for children with SCD, have better specificity for detecting differences within a population of individuals with SCD and are designed to detect changes in HRQOL over time232.
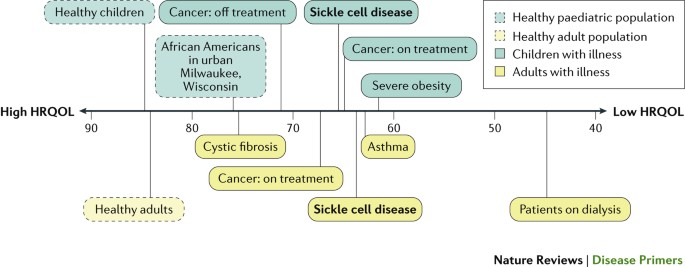
Both adults and children with SCD have substantially impaired baseline HRQOL199,233 (Fig. 7). Compared with healthy individuals, individuals with SCD have impaired HRQOL in nearly every domain, especially within the areas of pain, fatigue and physical functioning234,235. Adolescents and adults report poor sleep quality, moderate levels of fatigue and that sleep quality mediates the relationship between pain and fatigue236. The baseline physical functioning HRQOL domain of many individuals with SCD is worse than or comparable with that of individuals with other chronic diseases, such as cancer, cystic fibrosis or obesity237.
Physical functioning scores measured using the 36-Item Short Form Health Survey (SF-36) and the Pediatric Quality of Life Inventory (PedsQL) generic core scales in healthy individuals and individuals with chronic disease237,272. Scores range from 100, representing the best health-related quality of life (HRQOL), to 0. Specific areas represented in physical functioning scores include the ability to perform all types of physical activities, such as running, walking for a short distance, lifting heavy objects and bathing without help.
Acute complications, such as an acute vaso-occlusive pain crisis, are significantly associated with worse HRQOL than at baseline238. Children with SCD report substantial problems with physical functioning, pain and sleep during and immediately following vaso-occlusive crises239. Daily pain can affect the ability to attend school or work240,241 and is predictive of worse HRQOL in adults with SCD242. Nearly one-third of adults with SCD report pain almost every day, and over half of the individuals with SCD have pain 50% of the time241.
Effect of treatment on health-related quality of life
Adults with SCD who had a favourable response to hydroxycarbamide had better general health and reduced pain than those who received placebo or who had a low response to treatment243. Similar results were observed in children with SCD who received hydroxycarbamide244 or chronic red blood cell transfusion therapy245. As more experimental drugs for individuals with SCD are tested in clinical trials, it is imperative to measure the effect of these new therapies on individuals’ HRQOL.
Outlook
The wide implementation of affordable interventions, including neonatal diagnosis, penicillin prophylaxis and vaccination (which led to substantial reductions in mortality among children with SCA of <5 years of age in high-income countries), could prolong the lives of ∼5 million newborn babies with SCA by 2050 (Ref. 17). Similarly, large-scale screening and treatment programmes could save the lives of up to 10 million newborn babies with SCA globally, most of them in sub-Saharan Africa17,40.
Screening
Screening for SCD and related conditions is essential in Africa, where the incidence is highest. However, the implementation of universal newborn screening programmes remains a major economical and public health challenge. African communities and governments should also develop culturally acceptable programmes for screening adults for family planning purposes. The development of new, accurate and affordable rapid diagnostic tests would offer a long-awaited point-of-care screening option for low-income and middle-income countries. Clinical validation of such tests showed that they can reliably detect the βS and βC alleles with high specificity and sensitivity246. These tests could be used as a large-scale first screening step before confirmation of diagnosis by HPLC or isoelectric focusing, which will be necessary to identify individuals who also have thalassaemia or other Hb variants.
Treatment
In the short term, the identification of ways to enhance the use of proven therapies, such as hydroxycarbamide and haematopoietic stem cell transplantation, is the quickest route to improve management. Nevertheless, questions remain about the long-term effectiveness of hydroxycarbamide, ways to improve adherence to hydroxycarbamide therapy and possible development of antibacterial resistance in children with SCD under long-term penicillin prophylaxis. Owing to the complexity of SCD and the range of possible complications, a multidrug approach will probably be used by health care providers. However, drug development is a time-consuming process; thus, multidrug treatments will probably be available only in the mid-term or long term. Future work to understand the HRQOL of individuals with SCD over time and outside of the medical system and the effect of therapy on HRQOL is needed to provide tailored care and maximize HRQOL247.
Gene therapy has been considered a promising cure for SCD since the mid-1990s. Lentiviral vectors have been developed to insert γ-globin or modified β-globin genes that have been engineered to reduce sickling into haematopoietic stem cells; these vectors are now in clinical trials248 and have yielded a promising initial result249. Newer gene editing approaches based on zinc-finger nucleases and transcription activator-like effector nucleases have been designed and tested for proof of principle in SCD250. The development of CRISPR techniques, which enable the precise replacement of a specific region of DNA, is another promising gene therapy approach for SCD, currently tested only in mice251 and cultured human cells252 until the multiyear regulatory process is cleared for human trials. However, many ethical issues need to be resolved before these techniques can be used in humans: long-term follow-up trials will be needed to confirm the safety and sustainability of these techniques, and the accessibility of gene therapy in high-burden, low-income areas needs to be addressed. Although some of these current gene therapy strategies are potentially curative, many of them only aim to ameliorate disease severity.
New drugs. In the United States, the decision of the FDA Division of Hematology Products to consider the development of new SCD treatments as a top priority and grant orphan drug status or ‘fast-track’ designation to several drugs and biological products has facilitated investments from pharmaceutical companies. Many products that target one or more of the mechanisms that contribute to the disease process (for example, by boosting HbF levels or countering oxidative stress) are currently in phase II or phase III trials253 (Table 1). A large clinical trial of an anti-platelet agent, prasugrel, failed to significantly reduce vaso-occlusive crisis episodes in children with SCA152, but P-selectin blocking approaches are promising to prevent149 and to reduce the duration and severity254 of vaso-occlusive crisis episodes. Enrolment in SCD trials remains challenging: a systematic review of 174 SCD interventional trials closed to enrolment showed that 57% of them terminated owing to low enrolment255. However, the recent completion of a series of large, multicentre, multinational clinical trials demonstrates that the community of patients with SCD and health care providers is eager to collaborate with the pharmaceutical industry to find effective new treatments149,150,152,254,256. The prospects for new treatments in SCD have never looked better.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
How to cite this Primer
Kato, G. J. et al. Sickle cell disease. Nat. Rev. Dis. Primers 4, 18010 (2018).
Related links
References
Neel, J. V. The inheritance of sickle cell anemia. Science 110, 64–66 (1949).
Steinberg, M. H. & Sebastiani, P. Genetic modifiers of sickle cell disease. Am. J. Hematol. 87, 795–803 (2012).
Platt, O. S. et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. N. Engl. J. Med. 330, 1639–1644 (1994). This landmark natural history study established life expectancy and risk factors for mortality for SCD in the United States.
Piel, F. B., Steinberg, M. H. & Rees, D. C. Sickle cell disease. N. Engl. J. Med. 376, 1561–1573 (2017).
Ware, R. E., de Montalembert, M., Tshilolo, L. & Abboud, M. R. Sickle cell disease. Lancet 390, 311–323 (2017).
Serjeant, G. R. & Serjeant, B. E. Management of sickle cell disease; lessons from the Jamaican Cohort Study. Blood Rev. 7, 137–145 (1993).
Bonds, D. R. Three decades of innovation in the management of sickle cell disease: the road to understanding the sickle cell disease clinical phenotype. Blood Rev. 19, 99–110 (2005).
Quinn, C. T., Rogers, Z. R., McCavit, T. L. & Buchanan, G. R. Improved survival of children and adolescents with sickle cell disease. Blood 115, 3447–3452 (2010).
Gardner, K. et al. Survival in adults with sickle cell disease in a high-income setting. Blood 128, 1436–1438 (2016).
Grosse, S. D. et al. Sickle cell disease in Africa: a neglected cause of early childhood mortality. Am. J. Prev. Med. 41, S398–405 (2011).
Allison, A. C. Protection afforded by sickle-cell trait against subtertian malarial infection. BMJ 1, 290–294 (1954).
Luzzatto, L. Sickle cell anaemia and malaria. Mediterr. J. Hematol. Infect. Dis. 4, e2012065 (2012).
Piel, F. B. et al. Global distribution of the sickle cell gene and geographical confirmation of the malaria hypothesis. Nat. Commun. 1, 104 (2010).
Piel, F. B. et al. Global epidemiology of sickle haemoglobin in neonates: a contemporary geostatistical model-based map and population estimates. Lancet 381, 142–151 (2013).
Weatherall, D. J. The importance of micromapping the gene frequencies for the common inherited disorders of haemoglobin. Br. J. Haematol. 149, 635–637 (2010).
Piel, F. B. et al. The distribution of haemoglobin C and its prevalence in newborns in Africa. Sci. Rep. 3, 1671 (2013).
Piel, F. B., Hay, S. I., Gupta, S., Weatherall, D. J. & Williams, T. N. Global burden of sickle cell anaemia in children under five, 2010-2050: modelling based on demographics, excess mortality, and interventions. PLoS Med. 10, e1001484 (2013). This study places the disease burden of SCA into a global perspective.
Diallo, D. A. & Guindo, A. Sickle cell disease in sub-Saharan Africa: stakes and strategies for control of the disease. Curr. Opin. Hematol. 21, 210–214 (2014).
Therrell, B. L. Jr., Lloyd-Puryear, M. A., Eckman, J. R. & Mann, M. Y. Newborn screening for sickle cell diseases in the United States: a review of data spanning 2 decades. Semin. Perinatol. 39, 238–251 (2015).
Charlton, M. NHS Sickle Cell and Thalassaemia Screening Programme Data Report 2015/16: Trends and Performance Analysis (Public Health England, 2017).
Benson, J. M. & Therrell, B. L. Jr. History and current status of newborn screening for hemoglobinopathies. Semin. Perinatol. 34, 134–144 (2010).
Feuchtbaum, L., Carter, J., Dowray, S., Currier, R. J. & Lorey, F. Birth prevalence of disorders detectable through newborn screening by race/ethnicity. Genet. Med. 14, 937–945 (2012).
Ojodu, J. et al. Incidence of sickle cell trait — United States, 2010. MMWR Morb. Mortal. Wkly Rep. 63, 1155–1158 (2014).
Wang, Y. et al. Sickle cell disease incidence among newborns in New York State by maternal race/ethnicity and nativity. Genet. Med. 15, 222–228 (2013).
Ministry of Health Brazil. Sickle Cell disease: what you should know about genetic inheritance, 2014 [Portuguese]. Ministério da Saúdehttp://bvsms.saude.gov.br/bvs/publicacoes/doenca_falciforme_deve_saber_sobre_heranca.pdf (2014).
Silva, W. S. et al. Screening for structural hemoglobin variants in Bahia, Brazil. Int. J. Environ. Res. Publ. Health 13, 225 (2016).
Lobo, C. L. et al. Newborn screening program for hemoglobinopathies in Rio de Janeiro, Brazil. Pediatr. Blood Cancer 61, 34–39 (2014).
Braga, J. A., Verissimo, M. P., Saad, S. T., Cancado, R. D. & Loggetto, S. R. Guidelines on neonatal screening and painful vaso-occlusive crisis in sickle cell disease: Associacao Brasileira de Hematologia, Hemoterapia e Terapia Celular: Project guidelines: Associacao Medica Brasileira - 2016. Rev. Bras. Hematol. Hemoter. 38, 147–157 (2016).
Lervolino, L. G. et al. Prevalence of sickle cell disease and sickle cell trait in national neonatal screening studies. Rev. Bras. Hematol. Hemoter. 33, 49–54 (2011).
Brandelise, S. et al. Newborn screening for sickle cell disease in Brazil: the Campinas experience. Clin. Lab. Haematol. 26, 15–19 (2004).
Steinberg, M. H. Predicting clinical severity in sickle cell anaemia. Br. J. Haematol. 129, 465–481 (2005).
Ngo, D. et al. Fetal hemoglobin in sickle cell anemia: genetic studies of the Arab-Indian haplotype. Blood Cells Mol. Dis. 51, 22–26 (2013).
Italia, K. et al. Variable phenotypes of sickle cell disease in India with the Arab-Indian haplotype. Br. J. Haematol. 168, 156–159 (2015).
Mukherjee, M. B. et al. Clinical, hematologic and molecular variability of sickle cell-beta thalassemia in western India. Indian J. Hum. Genet. 16, 154–158 (2010).
Jones, S. et al. Windy weather and low humidity are associated with an increased number of hospital admissions for acute pain and sickle cell disease in an urban environment with a maritime temperate climate. Br. J. Haematol. 131, 530–533 (2005).
Tewari, S., Brousse, V., Piel, F. B., Menzel, S. & Rees, D. C. Environmental determinants of severity in sickle cell disease. Haematologica 100, 1108–1116 (2015).
Minniti, C. P., Eckman, J., Sebastiani, P., Steinberg, M. H. & Ballas, S. K. Leg ulcers in sickle cell disease. Am. J. Hematol. 85, 831–833 (2010).
Dash, B. P. & Kar, B. C. Priapism is rare in sickle cell disease in India. J. Assoc. Physicians India 48, 255 (2000).
McAuley, C. F. et al. High mortality from Plasmodium falciparum malaria in children living with sickle cell anemia on the coast of Kenya. Blood 116, 1663–1668 (2010).
Williams, T. N. et al. Bacteraemia in Kenyan children with sickle-cell anaemia: a retrospective cohort and case-control study. Lancet 374, 1364–1370 (2009).
Weatherall, D. J. The inherited diseases of hemoglobin are an emerging global health burden. Blood 115, 4331–4336 (2010).
Steinberg, M. H. Pathophysiology of sickle cell disease. Baillieres Clin. Haematol. 11, 163–184 (1998).
Pawliuk, R. et al. Correction of sickle cell disease in transgenic mouse models by gene therapy. Science 294, 2368–2371 (2001).
Vekilov, P. G. Sickle-cell haemoglobin polymerization: is it the primary pathogenic event of sickle-cell anaemia? Br. J. Haematol. 139, 173–184 (2007).
Seakins, M., Gibbs, W. N., Milner, P. F. & Bertles, J. F. Erythrocyte Hb-S concentration. An important factor in the low oxygen affinity of blood in sickle cell anemia. J. Clin. Invest. 52, 422–432 (1973).
Rogers, S. C. et al. Sickle hemoglobin disturbs normal coupling among erythrocyte O2 content, glycolysis, and antioxidant capacity. Blood 121, 1651–1662 (2013).
Zhang, Y. et al. Elevated sphingosine-1-phosphate promotes sickling and sickle cell disease progression. J. Clin. Invest. 124, 2750–2761 (2014).
Sun, K. et al. Sphingosine-1-phosphate promotes erythrocyte glycolysis and oxygen release for adaptation to high-altitude hypoxia. Nat. Commun. 7, 12086 (2016).
Sun, K. et al. Elevated adenosine signaling via adenosine A2B receptor induces normal and sickle erythrocyte sphingosine kinase 1 activity. Blood 125, 1643–1652 (2015).
Noguchi, C. T. & Schechter, A. N. Sickle haemoglobin polymerization in solution and in cells. Annu. Rev. Biophys. Biophys. Chem. 14, 239–263 (1985)
Connes, P. et al. The role of blood rheology in sickle cell disease. Blood Rev. 30, 111–118 (2016).
Smith, C. M., Krivit, W. & White, J. G. The irreversibly sickled cell. Am. J. Pediatr. Hematol. Oncol. 4, 307–315 (1982).
Evans, E. A. & Mohandas, N. Membrane-associated sickle hemoglobin: a major determinant of sickle erythrocyte rigidity. Blood 70, 1443–1449 (1987).
Nash, G. B., Johnson, C. S. & Meiselman, H. J. Rheologic impairment of sickle RBCs induced by repetitive cycles of deoxygenation-reoxygenation. Blood 72, 539–545 (1988).
Kuypers, F. A. Hemoglobin s polymerization and red cell membrane changes. Hematol. Oncol. Clin. North Am. 28, 155–179 (2014).
Allan, D. & Raval, P. Some morphological consequences of uncoupling the lipid bilayer from the plasma membrane skeleton in intact erythrocytes. Biomed. Biochim. Acta 42, S11–16 (1983).
Blumenfeld, N., Zachowski, A., Galacteros, F., Beuzard, Y. & Devaux, P. F. Transmembrane mobility of phospholipids in sickle erythrocytes: effect of deoxygenation on diffusion and asymmetry. Blood 77, 849–854 (1991).
Fadok, V. A., de Cathelineau, A., Daleke, D. L., Henson, P. M. & Bratton, D. L. Loss of phospholipid asymmetry and surface exposure of phosphatidylserine is required for phagocytosis of apoptotic cells by macrophages and fibroblasts. J. Biol. Chem. 276, 1071–1077 (2001).
Kuypers, F. A. Membrane lipid alterations in hemoglobinopathies. Hematol. Am. Soc. Hematol. Educ. Program 2007, 68–73 (2007).
Kuypers, F. A. & de Jong, K. The role of phosphatidylserine in recognition and removal of erythrocytes. Cell. Mol. Biol. 50, 147–158 (2004).
Piccin, A., Murphy, W. G. & Smith, O. P. Circulating microparticles: pathophysiology and clinical implications. Blood Rev. 21, 157–171 (2007).
Westerman, M. et al. Microvesicles in haemoglobinopathies offer insights into mechanisms of hypercoagulability, haemolysis and the effects of therapy. Br. J. Haematol. 142, 126–135 (2008).
Westerman, M. & Porter, J. B. Red blood cell-derived microparticles: an overview. Blood Cells Mol. Dis. 59, 134–139 (2016).
Hebbel, R. P. & Key, N. S. Microparticles in sickle cell anaemia: promise and pitfalls. Br. J. Haematol. 174, 16–29 (2016).
Alayash, A. I. Oxidative pathways in the sickle cell and beyond. Blood Cells Mol. Dis.https://doi.org/10.1016/j.bcmd.2017.05.009 (2017).
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol.https://doi.org/10.1038/nrm.2017.125 (2018).
Quinn, C. T. et al. Biochemical surrogate markers of hemolysis do not correlate with directly measured erythrocyte survival in sickle cell anemia. Am. J. Hematol. 91, 1195–1201 (2016).
Crosby, W. H. The metabolism of hemoglobin and bile pigment in hemolytic disease. Am. J. Med. 18, 112–122 (1955).
Bunn, H. F. et al. Pulmonary hypertension and nitric oxide depletion in sickle cell disease. Blood 116, 687–692 (2010).
Kato, G. J., Steinberg, M. H. & Gladwin, M. T. Intravascular hemolysis and the pathophysiology of sickle cell disease. J. Clin. Invest. 127, 750–760 (2017). This is a comprehensive review of the contribution of haemolysis to SCD pathophysiology.
Wood, K. C. & Granger, D. N. Sickle cell disease: role of reactive oxygen and nitrogen metabolites. Clin. Exp. Pharmacol. Physiol. 34, 926–932 (2007).
Aslan, M. & Freeman, B. A. Redox-dependent impairment of vascular function in sickle cell disease. Free Radic. Biol. Med. 43, 1469–1483 (2007).
Cho, C. S. et al. Hydroxyurea-induced expression of glutathione peroxidase 1 in red blood cells of individuals with sickle cell anemia. Antioxid. Redox Signal. 13, 1–11 (2010).
Morris, C. R. et al. Erythrocyte glutamine depletion, altered redox environment, and pulmonary hypertension in sickle cell disease. Blood 111, 402–410 (2008).
Reiter, C. D. et al. Cell-free hemoglobin limits nitric oxide bioavailability in sickle-cell disease. Nat. Med. 8, 1383–1389 (2002).
Morris, C. R. et al. Dysregulated arginine metabolism, hemolysis-associated pulmonary hypertension, and mortality in sickle cell disease. JAMA 294, 81–90 (2005).
Miller-Fleming, L., Olin-Sandoval, V., Campbell, K. & Ralser, M. Remaining mysteries of molecular biology: the role of polyamines in the cell. J. Mol. Biol. 427, 3389–3406 (2015).
Landburg, P. P. et al. Plasma asymmetric dimethylarginine concentrations in sickle cell disease are related to the hemolytic phenotype. Blood Cells Mol. Dis. 44, 229–232 (2010).
Antoniades, C. et al. Association of plasma asymmetrical dimethylarginine (ADMA) with elevated vascular superoxide production and endothelial nitric oxide synthase uncoupling: implications for endothelial function in human atherosclerosis. Eur. Heart J. 30, 1142–1150 (2009).
Luo, S., Lei, H., Qin, H. & Xia, Y. Molecular mechanisms of endothelial NO synthase uncoupling. Curr. Pharm. Des. 20, 3548–3553 (2014).
Zorca, S. et al. Lipid levels in sickle-cell disease associated with haemolytic severity, vascular dysfunction and pulmonary hypertension. Br. J. Haematol. 149, 436–445 (2010).
Tumblin, A. et al. Apolipoprotein A-I and serum amyloid A plasma levels are biomarkers of acute painful episodes in patients with sickle cell disease. Haematologica 95, 1467–1472 (2010).
Saraf, S. L. et al. APOL1, alpha-thalassemia, and BCL11A variants as a genetic risk profile for progression of chronic kidney disease in sickle cell anemia. Haematologica 102, e1–e6 (2017).
Gladwin, M. T. & Ofori-Acquah, S. F. Erythroid DAMPs drive inflammation in SCD. Blood 123, 3689–3690 (2014).
Wang, X. et al. Heme-bound iron activates placenta growth factor in erythroid cells via erythroid Kruppel-like factor. Blood 124, 946–954 (2014).
van Beers, E. J. et al. Iron, inflammation, and early death in adults with sickle cell disease. Circ. Res. 116, 298–306 (2015).
Hebbel, R. P. Adhesive interactions of sickle erythrocytes with endothelium. J. Clin. Invest. 100, S83–86 (1997).
Kaul, D. K. et al. Monoclonal antibodies to alphaVbeta3 (7E3 and LM609) inhibit sickle red blood cell-endothelium interactions induced by platelet-activating factor. Blood 95, 368–374 (2000).
Setty, B. N. & Stuart, M. J. Vascular cell adhesion molecule-1 is involved in mediating hypoxia-induced sickle red blood cell adherence to endothelium: potential role in sickle cell disease. Blood 88, 2311–2320 (1996).
Hines, P. C. et al. Novel epinephrine and cyclic AMP-mediated activation of BCAM/Lu-dependent sickle (SS) RBC adhesion. Blood 101, 3281–3287 (2003).
Wagner, M. C., Eckman, J. R. & Wick, T. M. Sickle cell adhesion depends on hemodynamics and endothelial activation. J. Lab Clin. Med. 144, 260–268 (2004).
Murphy, M. M. et al. Role of Rap1 in promoting sickle red blood cell adhesion to laminin via BCAM/LU. Blood 105, 3322–3329 (2005).
Sugihara, K., Sugihara, T., Mohandas, N. & Hebbel, R. P. Thrombospondin mediates adherence of CD36+ sickle reticulocytes to endothelial cells. Blood 80, 2634–2642 (1992).
Miller, S. T. et al. Prediction of adverse outcomes in children with sickle cell disease. N. Engl. J. Med. 342, 83–89 (2000).
Elmariah, H. et al. Factors associated with survival in a contemporary adult sickle cell disease cohort. Am. J. Hematol. 89, 530–535 (2014).
Zhang, D., Xu, C., Manwani, D. & Frenette, P. S. Neutrophils, platelets, and inflammatory pathways at the nexus of sickle cell disease pathophysiology. Blood 127, 801–809 (2016). This is an updated review of the principal adhesive pathways involved in SCD vaso-occlusion.
Canalli, A. A. et al. Participation of Mac-1, LFA-1 and VLA-4 integrins in the in vitro adhesion of sickle cell disease neutrophils to endothelial layers, and reversal of adhesion by simvastatin. Haematologica 96, 526–533 (2011).
Dominical, V. M. et al. Prominent role of platelets in the formation of circulating neutrophil-red cell heterocellular aggregates in sickle cell anemia. Haematologica 99, e214–e217 (2014).
Davila, J. et al. A novel inflammatory role for platelets in sickle cell disease. Platelets 26, 726–729 (2015).
Hoppe, C. C. Prenatal and newborn screening for hemoglobinopathies. Int. J. Lab. Hematol. 35, 297–305 (2013).
Traeger-Synodinos, J. Pre-implantation genetic diagnosis. Best Pract. Res. Clin. Obstet. Gynaecol. 39, 74–88 (2017).
Robitaille, N., Delvin, E. E. & Hume, H. A. Newborn screening for sickle cell disease: A 1988–2003 Quebec experience. Paediatr. Child Health 11, 223–227 (2006).
Vichinsky, E., Hurst, D., Earles, A., Kleman, K. & Lubin, B. Newborn screening for sickle cell disease: effect on mortality. Pediatrics 81, 749–755 (1988).
Gaston, M. H. et al. Prophylaxis with oral penicillin in children with sickle cell anemia. A randomized trial. N. Engl. J. Med. 314, 1593–1599 (1986). This study provides proof that penicillin prophylaxis reduces mortality, which marked a turning point for life expectancy in SCA.
Kate, S. & Lingojwar, D. Epidemiology of sickle cell disorder in the state of Maharashtra. Int. J. Hum. Genet. 2, 161–167 (2002).
Patra, P. K., Khodiar, P. K., Hambleton, I. R. & Serjeant, G. R. The Chhattisgarh state screening programme for the sickle cell gene: a cost-effective approach to a public health problem. J. Commun. Genet. 6, 361–368 (2015).
Naik, R. P. & Haywood, C. Jr. Sickle cell trait diagnosis: clinical and social implications. Hematol. Am. Soc. Hematol. Educ. Program 2015, 160–167 (2015).
Nelson, D. A. et al. Sickle cell trait, rhabdomyolysis, and mortality among U. S. army soldiers. N. Engl. J. Med. 375, 435–442 (2016).
Key, N. S., Connes, P. & Derebail, V. K. Negative health implications of sickle cell trait in high income countries: from the football field to the laboratory. Br. J. Haematol. 170, 5–14 (2015).
Streetly, A., Latinovic, R. & Henthorn, J. Positive screening and carrier results for the England-wide universal newborn sickle cell screening programme by ethnicity and area for 2005–2007. J. Clin. Pathol. 63, 626–629 (2010).
Thuret, I. et al. Neonatal screening for sickle cell disease in France: evaluation of the selective process. J. Clin. Pathol. 63, 548–551 (2010).
Manu Pereira, M. & Corrons, J. L. Neonatal haemoglobinopathy screening in Spain. J. Clin. Pathol. 62, 22–25 (2009).
Colombatti, R. et al. Organizing national responses for rare blood disorders: the Italian experience with sickle cell disease in childhood. Orphanet J. Rare Dis. 8, 169 (2013).
Kunz, J. B. et al. Significant prevalence of sickle cell disease in Southwest Germany: results from a birth cohort study indicate the necessity for newborn screening. Ann. Hematol. 95, 397–402 (2016).
Minkovitz, C. S., Grason, H., Ruderman, M. & Casella, J. F. Newborn screening programs and sickle cell disease: a public health services and systems approach. Am. J. Prev. Med. 51, S39–S47 (2016).
Colah, R. B., Mukherjee, M. B., Martin, S. & Ghosh, K. Sickle cell disease in tribal populations in India. Indian J. Med. Res. 141, 509–515 (2015).
Chandrashekar, V. & Soni, M. Hemoglobin disorders in South India. ISRN Hematol. 2011, 748939 (2011).
Italia, K. et al. Hydroxyurea in sickle cell disease — a study of clinico-pharmacological efficacy in the Indian haplotype. Blood Cells Mol. Dis. 42, 25–31 (2009).
Kamble, M. & Chatruvedi, P. Epidemiology of sickle cell disease in a rural hospital of central India. Indian Pediatr. 37, 391–396 (2000).
Shukla, R. N. & Solanki, B. R. Sickle-cell trait in Central India. Lancet 1, 297–298 (1958).
Therrell, B. L. et al. Current status of newborn screening worldwide: 2015. Semin. Perinatol. 39, 171–187 (2015).
Ohene-Frempong, K., Oduro, J., Tetteh, H. & Nkrumah, F. Screening newborns for sickle cell disease in Ghana. Pediatrics 121 (Suppl. 2), S120–S121(2008).
McGann, P. T. et al. A prospective newborn screening and treatment program for sickle cell anemia in Luanda, Angola. Am. J. Hematol. 88, 984–989 (2013).
Rahimy, M. C., Gangbo, A., Ahouignan, G. & Alihonou, E. Newborn screening for sickle cell disease in the Republic of Benin. J. Clin. Pathol. 62, 46–48 (2009).
Kafando, E. et al. Neonatal haemoglobinopathy screening in Burkina Faso. J. Clin. Pathol. 62, 39–41 (2009).
Mutesa, L. et al. Neonatal screening for sickle cell disease in Central Africa: a study of 1825 newborns with a new enzyme-linked immunosorbent assay test. J. Med. Screen. 14, 113–116 (2007).
Tshilolo, L. et al. Neonatal screening for sickle cell anaemia in the Democratic Republic of the Congo: experience from a pioneer project on 31 204 newborns. J. Clin. Pathol. 62, 35–38 (2009).
Odunvbun, M. E., Okolo, A. A. & Rahimy, C. M. Newborn screening for sickle cell disease in a Nigerian hospital. Publ. Health 122, 1111–1116 (2008).
Mbodj, M. et al. Sickle cell disease neonatal screening. First evaluation [French]. Dakar Med. 48, 202–205 (2003).
Makani, J. et al. Health policy for sickle cell disease in Africa: experience from Tanzania on interventions to reduce under-five mortality. Trop. Med. Int. Health 20, 184–187 (2015).
Ndeezi, G. et al. Burden of sickle cell trait and disease in the Uganda Sickle Surveillance Study (US3): a cross-sectional study. Lancet Global Health 4, e195–e200 (2016).
Serjeant, G. R. The natural history of sickle cell disease. Cold Spring Harb. Perspect. Med. 3, a011783 (2013).
Fertrin, K. Y. & Costa, F. F. Genomic polymorphisms in sickle cell disease: implications for clinical diversity and treatment. Expert Rev. Hematol. 3, 443–458 (2010).
Kato, G. J., Gladwin, M. T. & Steinberg, M. H. Deconstructing sickle cell disease: reappraisal of the role of hemolysis in the development of clinical subphenotypes. Blood Rev. 21, 37–47 (2007).
Lettre, G. & Bauer, D. E. Fetal haemoglobin in sickle-cell disease: from genetic epidemiology to new therapeutic strategies. Lancet 387, 2554–2564 (2016).
Steinberg, M. H., Chui, D. H., Dover, G. J., Sebastiani, P. & Alsultan, A. Fetal hemoglobin in sickle cell anemia: a glass half full? Blood 123, 481–485 (2014).
Belfer, I. et al. A GCH1 haplotype confers sex-specific susceptibility to pain crises and altered endothelial function in adults with sickle cell anemia. Am. J. Hematol. 89, 187–193 (2014).
Brandow, A. M., Stucky, C. L., Hillery, C. A., Hoffmann, R. G. & Panepinto, J. A. Patients with sickle cell disease have increased sensitivity to cold and heat. Am. J. Hematol. 88, 37–43 (2013).
Quinn, C. T., Rogers, Z. R. & Buchanan, G. R. Survival of children with sickle cell disease. Blood 103, 4023–4027 (2004).
Anie, K. A. Psychological complications in sickle cell disease. Br. J. Haematol. 129, 723–729 (2005).
Weatherall, D. J. The challenge of haemoglobinopathies in resource-poor countries. Br. J. Haematol. 154, 736–744 (2011).
Kassim, A. A. & DeBaun, M. R. The case for and against initiating either hydroxyurea therapy, blood transfusion therapy or hematopoietic stem cell transplant in asymptomatic children with sickle cell disease. Expert Opin. Pharmacother. 15, 325–336 (2014).
McGann, P. T. & Ware, R. E. Hydroxyurea therapy for sickle cell anemia. Expert Opin. Drug Saf. 14, 1749–1758 (2015).
Charache, S. et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. N. Engl. J. Med. 332, 1317–1322 (1995). This landmark trial proved that hydroxycarbamide reduces the frequency of pain episodes in SCA, leading to the approval of this drug.
Walsh, K. E. et al. Medication adherence among pediatric patients with sickle cell disease: a systematic review. Pediatrics 134, 1175–1183 (2014).
Husain, M., Hartman, A. D. & Desai, P. Pharmacogenomics of sickle cell disease: steps toward personalized medicine. Pharmgenom. Pers. Med. 10, 261–265 (2017).
Wong, T. E., Brandow, A. M., Lim, W. & Lottenberg, R. Update on the use of hydroxyurea therapy in sickle cell disease. Blood 124, 3850–3857 (2014).
DeBaun, M. R. Hydroxyurea therapy contributes to infertility in adult men with sickle cell disease: a review. Expert Rev. Hematol. 7, 767–773 (2014).
Ataga, K. I. et al. Crizanlizumab for the prevention of pain crises in sickle cell disease. N. Engl. J. Med. 376, 429–439 (2017).
McGann, P. T. et al. Hydroxyurea therapy for children with sickle cell anemia in sub-Saharan Africa: rationale and design of the REACH trial. Pediatr. Blood Cancer 63, 98–104 (2016).
Wang, W. C. et al. Hydroxycarbamide in very young children with sickle-cell anaemia: a multicentre, randomised, controlled trial (BABY HUG). Lancet 377, 1663–1672 (2011). This article presents evidence that hydroxycarbamide is effective in infants and toddlers with SCA.
Heeney, M. M. et al. A multinational trial of Prasugrel for sickle cell vaso-occlusive events. N. Engl. J. Med. 374, 625–635 (2016).
Mulaku, M. et al. Evidence review of hydroxyurea for the prevention of sickle cell complications in low-income countries. Arch. Dis. Child. 98, 908–914 (2013).
Hyacinth, H. I., Adams, R. J., Voeks, J. H., Hibbert, J. M. & Gee, B. E. Frequent red cell transfusions reduced vascular endothelial activation and thrombogenicity in children with sickle cell anemia and high stroke risk. Am. J. Hematol. 89, 47–51 (2014).
Hyacinth, H. I. et al. Effect of chronic blood transfusion on biomarkers of coagulation activation and thrombin generation in sickle cell patients at risk for stroke. PLoS ONE 10, e0134193 (2015).
Quirolo, K. & Vichinsky, E. in Rossi's Principles of Transfusion Medicine (eds Simon T. L. et al.) (Wiley-Blackwell, 2016).
Fasano, R. M. & Chou, S. T. Red blood cell antigen genotyping for sickle cell disease, thalassemia, and other transfusion complications. Transfus. Med. Rev. 30, 197–201 (2016).
Walters, M. C. et al. Indications and results of HLA-identical sibling hematopoietic cell transplantation for sickle cell disease. Biol. Blood Marrow Transplant. 22, 207–211 (2016).
Gluckman, E. et al. Sickle cell disease: an international survey of results of HLA-identical sibling hematopoietic stem cell transplantation. Blood 129, 1548–1556 (2017).
Hsieh, M. M. et al. Nonmyeloablative HLA-matched sibling allogeneic hematopoietic stem cell transplantation for severe sickle cell phenotype. JAMA 312, 48–56 (2014).
Saraf, S. L. et al. Nonmyeloablative stem cell transplantation with alemtuzumab/low-dose irradiation to cure and improve the quality of life of adults with sickle cell disease. Biol. Blood Marrow Transplant. 22, 441–448 (2016).
Gluckman, E. Allogeneic transplantation strategies including haploidentical transplantation in sickle cell disease. Hematol. Am. Soc. Hematol. Educ. Program 2013, 370–376 (2013).
Ballas, S. K., Gupta, K. & Adams-Graves, P. Sickle cell pain: a critical reappraisal. Blood 120, 3647–3656 (2012).
Treadwell, M. J. et al. A quality improvement initiative to improve emergency department care for pediatric patients with sickle cell disease. J. Clin. Outcomes Manag. 21, 62–70 (2014).
Kavanagh, P. L. et al. Improving the management of vaso-occlusive episodes in the pediatric emergency department. Pediatrics 136, e1016–1025 (2015).
Tanabe, P. et al. A randomized controlled trial comparing two vaso-occlusive episode (VOE) protocols in sickle cell disease (SCD). Am. J. Hematol. 93, 159–168 (2018).
Lanzkron, S. et al. Impact of a dedicated infusion clinic for acute management of adults with sickle cell pain crisis. Am. J. Hematol. 90, 376–380 (2015).
Haywood, C. et al. The impact of race and disease on sickle cell patient wait times in the emergency department. Am. J. Emerg. Med. 31, 651–656 (2013).
van Beers, E. J. et al. Patient-controlled analgesia versus continuous infusion of morphine during vaso-occlusive crisis in sickle cell disease, a randomized controlled trial. Am. J. Hematol. 82, 955–960 (2007).
Makani, J., Ofori-Acquah, S. F., Nnodu, O., Wonkam, A. & Ohene-Frempong, K. Sickle cell disease: new opportunities and challenges in Africa. Sci. World J. 2013, 193252 (2013).
Manci, E. A. et al. Causes of death in sickle cell disease: an autopsy study. Br. J. Haematol. 123, 359–365 (2003).
Novelli, E. M. & Gladwin, M. T. Crises in sickle cell disease. Chest 149, 1082–1093 (2016).
Vichinsky, E. P. et al. Acute chest syndrome in sickle cell disease: clinical presentation and course. Blood 89, 1787–1792 (1997).
Vichinsky, E. P. et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. N. Engl. J. Med. 342, 1855–1865 (2000). This study comprehensively establishes the causes and outcomes of acute chest syndrome.
DeBaun, M. R. & Strunk, R. C. The intersection between asthma and acute chest syndrome in children with sickle-cell anaemia. Lancet 387, 2545–2553 (2016).
Howard, J. et al. Guideline on the management of acute chest syndrome in sickle cell disease. Br. J. Haematol. 169, 492–505 (2015).
Bernini, J. C. et al. Beneficial effect of intravenous dexamethasone in children with mild to moderately severe acute chest syndrome complicating sickle cell disease. Blood 92, 3082–3089 (1998).
Kassim, A. A., Galadanci, N. A., Pruthi, S. & DeBaun, M. R. How I treat and manage strokes in sickle cell disease. Blood 125, 3401–3410 (2015).
Gardner, K., Hoppe, C., Mijovic, A. & Thein, S. L. How we treat delayed haemolytic transfusion reactions in patients with sickle cell disease. Br. J. Haematol. 170, 745–756 (2015).
Ogunlesi, F., Heeney, M. M. & Koumbourlis, A. C. Systemic corticosteroids in acute chest syndrome: friend or foe? Paediatr. Respir. Rev. 15, 24–27 (2014).
Ohene-Frempong, K. et al. Cerebrovascular accidents in sickle cell disease: rates and risk factors. Blood 91, 288–294 (1998).
Vichinsky, E. P. et al. Neuropsychological dysfunction and neuroimaging abnormalities in neurologically intact adults with sickle cell anemia. JAMA 303, 1823–1831 (2010).
DeBaun, M. R. et al. Silent cerebral infarcts: a review on a prevalent and progressive cause of neurologic injury in sickle cell anemia. Blood 119, 4587–4596 (2012).
Ware, R. E. & Helms, R. W. Stroke With Transfusions Changing to Hydroxyurea (SWiTCH). Blood 119, 3925–3932 (2012).
Strouse, J. J., Hulbert, M. L., DeBaun, M. R., Jordan, L. C. & Casella, J. F. Primary hemorrhagic stroke in children with sickle cell disease is associated with recent transfusion and use of corticosteroids. Pediatrics 118, 1916–1924 (2006).
Scott, R. M. & Smith, E. R. Moyamoya disease and moyamoya syndrome. N. Engl. J. Med. 360, 1226–1237 (2009).
Kennedy, B. C. et al. Pial synangiosis for moyamoya syndrome in children with sickle cell anemia: a comprehensive review of reported cases. Neurosurg. Focus 36, E12 (2014).
Brousse, V. et al. Acute splenic sequestration crisis in sickle cell disease: cohort study of 190 paediatric patients. Br. J. Haematol. 156, 643–648 (2012).
Vasavda, N. et al. The linear effects of alpha-thalassaemia, the UGT1A1 and HMOX1 polymorphisms on cholelithiasis in sickle cell disease. Br. J. Haematol. 138, 263–270 (2007).
Leake, P. A., Reid, M. & Plummer, J. A case series of cholecystectomy in Jamaican sickle cell disease patients — the need for a new strategy. Ann. Med. Surg. 15, 37–42 (2017).
Powars, D. R., Chan, L. S., Hiti, A., Ramicone, E. & Johnson, C. Outcome of sickle cell anemia: a 4-decade observational study of 1056 patients. Medicine 84, 363–376 (2005).
McClish, D. K. et al. Pain site frequency and location in sickle cell disease: the PiSCES project. Pain 145, 246–251 (2009).
Ballas, S. K. Update on pain management in sickle cell disease. Hemoglobin 35, 520–529 (2011).
Darbari, D. S. et al. Frequency of hospitalizations for pain and association with altered brain network connectivity in sickle cell disease. J. Pain 16, 1077–1086 (2015).
Neumayr, L. D. et al. Physical therapy alone compared with core decompression and physical therapy for femoral head osteonecrosis in sickle cell disease. Results of a multicenter study at a mean of three years after treatment. J. Bone Joint Surg. Am. 88, 2573–2582 (2006).
McClellan, A. C. et al. High one year mortality in adults with sickle cell disease and end-stage renal disease. Br. J. Haematol. 159, 360–367 (2012).
Abbott, K. C., Hypolite, I. O. & Agodoa, L. Y. Sickle cell nephropathy at end-stage renal disease in the United States: patient characteristics and survival. Clin. Nephrol. 58, 9–15 (2002).
Huang, E. et al. Improved survival among sickle cell kidney transplant recipients in the recent era. Nephrol. Dial. Transplant. 28, 1039–1046 (2013).
Dampier, C. et al. Health-related quality of life in adults with sickle cell disease (SCD): a report from the comprehensive sickle cell centers clinical trial consortium. Am. J. Hematol. 86, 203–205 (2011).
Chaturvedi, S. & DeBaun, M. R. Evolution of sickle cell disease from a life-threatening disease of children to a chronic disease of adults: the last 40 years. Am. J. Hematol. 91, 5–14 (2016).
Yawn, B. P. et al. Management of sickle cell disease: summary of the 2014 evidence-based report by expert panel members. JAMA 312, 1033–1048 (2014).
Buchanan, G. et al. Evidence-Based Management of Sickle Cell Disease: Expert Panel Report 2014 (National Heart Lung and Blood Institute, 2014). This article presents detailed evidence-based guidelines for the clinical management of individuals with SCD.
Klings, E. S. et al. An official American Thoracic Society clinical practice guideline: diagnosis, risk stratification, and management of pulmonary hypertension of sickle cell disease. Am. J. Respir. Crit. Care Med. 189, 727–740 (2014).
Butler, J. C., Breiman, R. F., Lipman, H. B., Hofmann, J. & Facklam, R. R. Serotype distribution of Streptococcus pneumoniae infections among preschool children in the United States, 1978-1994: implications for development of a conjugate vaccine. J. Infect. Dis. 171, 885–889 (1995).
Halasa, N. B. et al. Incidence of invasive pneumococcal disease among individuals with sickle cell disease before and after the introduction of the pneumococcal conjugate vaccine. Clin. Infect. Dis. 44, 1428–1433 (2007).
Cober, M. P. & Phelps, S. J. Penicillin prophylaxis in children with sickle cell disease. J. Pediatr. Pharmacol. Ther. 15, 152–159 (2010).
Obaro, S. K. & Iroh Tam, P. Y. Preventing infections in sickle cell disease: the unfinished business. Pediatr. Blood Cancer 63, 781–785 (2016).
Adams, R. J. et al. Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. N. Engl. J. Med. 339, 5–11 (1998). This study shows that ischaemic stroke can be prevented by chronic transfusion in children identified at high risk by non-invasive ultrasonography screening.
Adams, R. J., Brambilla, D. & Investigators, S. T. Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. N. Engl. J. Med. 353, 2769–2778 (2005).
Zimmerman, S. A., Schultz, W. H., Burgett, S., Mortier, N. A. & Ware, R. E. Hydroxyurea therapy lowers transcranial Doppler flow velocities in children with sickle cell anemia. Blood 110, 1043–1047 (2007).
Ware, R. E. et al. Hydroxycarbamide versus chronic transfusion for maintenance of transcranial doppler flow velocities in children with sickle cell anaemia-TCD With Transfusions Changing to Hydroxyurea (TWiTCH): a multicentre, open-label, phase 3, non-inferiority trial. Lancet 387, 661–670 (2016).
Galadanci, N. A. et al. Primary stroke prevention in Nigerian children with sickle cell disease (SPIN): challenges of conducting a feasibility trial. Pediatr. Blood Cancer 62, 395–401 (2015).
DeBaun, M. R. et al. Controlled trial of transfusions for silent cerebral infarcts in sickle cell anemia. N. Engl. J. Med. 371, 699–710 (2014).
Gladwin, M. T. et al. Pulmonary hypertension as a risk factor for death in patients with sickle cell disease. N. Engl. J. Med. 350, 886–895 (2004).
Boyd, J. H., Macklin, E. A., Strunk, R. C. & DeBaun, M. R. Asthma is associated with Increased mortality in individuals with sickle cell anemia. Haematologica 92, 1115–1118 (2007).
Glassberg, J. A. et al. Wheezing and asthma are independent risk factors for increased sickle cell disease morbidity. Br. J. Haematol. 159, 472–479 (2012).
Powars, D., Weidman, J. A., Odom-Maryon, T., Niland, J. C. & Johnson, C. Sickle cell chronic lung disease: prior morbidity and the risk of pulmonary failure. Medicine 67, 66–76 (1988).
Falk, R. J. et al. Prevalence and pathologic features of sickle cell nephropathy and response to inhibition of angiotensin-converting enzyme. N. Engl. J. Med. 326, 910–915 (1992).
Gordeuk, V. R. et al. Relative systemic hypertension in patients with sickle cell disease is associated with risk of pulmonary hypertension and renal insufficiency. Am. J. Hematol. 83, 15–18 (2008).
Pegelow, C. H. et al. Natural history of blood pressure in sickle cell disease: risks for stroke and death associated with relative hypertension in sickle cell anemia. Am. J. Med. 102, 171–177 (1997).
Rodgers, G. P., Walker, E. C. & Podgor, M. J. Is “relative” hypertension a risk factor for vaso-occlusive complications in sickle cell disease? Am. J. Med. Sci. 305, 150–156 (1993).
Downes, S. M. et al. Incidence and natural history of proliferative sickle cell retinopathy: observations from a cohort study. Ophthalmology 112, 1869–1875 (2005).
Moriarty, B. J., Acheson, R. W., Condon, P. I. & Serjeant, G. R. Patterns of visual loss in untreated sickle cell retinopathy. Eye 2, 330–335 (1988).
Adam, S. S. et al. Depression, quality of life, and medical resource utilization in sickle cell disease. Blood Adv. 1, 1983–1992 (2017).
McClish, D. K. et al. Comorbidity, pain, utilization, and psychosocial outcomes in older versus younger sickle cell adults: the PiSCES project. Biomed. Res. Int. 2017, 4070547 (2017).
Bakshi, N., Lukombo, I., Shnol, H., Belfer, I. & Krishnamurti, L. Psychological characteristics and pain frequency are associated with experimental pain sensitivity in pediatric patients with sickle cell disease. J. Pain 18, 1216–1228 (2017).
Jonassaint, C. R., Jones, V. L., Leong, S. & Frierson, G. M. A systematic review of the association between depression and health care utilization in children and adults with sickle cell disease. Br. J. Haematol. 174, 136–147 (2016).
Wallen, G. R. et al. Sleep disturbance, depression and pain in adults with sickle cell disease. BMC Psychiatry 14, 207 (2014).
Noubouossie, D., Key, N. S. & Ataga, K. I. Coagulation abnormalities of sickle cell disease: relationship with clinical outcomes and the effect of disease modifying therapies. Blood Rev. 30, 245–256 (2016).
Ware, J. E. Jr & Sherbourne, C. D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 30, 473–483 (1992).
Varni, J. The PedsQLTM 4.0 Measurement Model for the Pediatric Quality of Life InventoryTM Version 4.0: Administration Guidelines. PedsQLTMhttp://www.pedsql.org/pedsqladmin.html (2004).
Panepinto, J. A. et al. Determining the longitudinal validity and meaningful differences in HRQL of the PedsQL Sickle Cell Disease Module. Health Qual. Life Outcomes 15, 124 (2017).
Panepinto, J. A. & Bonner, M. Health-related quality of life in sickle cell disease: past, present, and future. Pediatr. Blood Cancer 59, 377–385 (2012).
Keller, S. D., Yang, M., Treadwell, M. J., Werner, E. M. & Hassell, K. L. Patient reports of health outcome for adults living with sickle cell disease: development and testing of the ASCQ-Me item banks. Health Qual. Life Outcomes 12, 125 (2014).
Panepinto, J. A. et al. PedsQL sickle cell disease module: feasibility, reliability, and validity. Pediatr. Blood Cancer 60, 1338–1344 (2013).
Ameringer, S., Elswick, R. K. Jr & Smith, W. Fatigue in adolescents and young adults with sickle cell disease: biological and behavioral correlates and health-related quality of life. J. Pediatr. Oncol. Nurs. 31, 6–17 (2014).
McClish, D. K. et al. Health related quality of life in sickle cell patients: the PiSCES project. Health Qual. Life Outcomes 3, 50 (2005).
Brandow, A. M., Brousseau, D. C., Pajewski, N. M. & Panepinto, J. A. Vaso-occlusive painful events in sickle cell disease: impact on child well-being. Pediatr. Blood Cancer 54, 92–97 (2010).
Brandow, A. M., Brousseau, D. C. & Panepinto, J. A. Postdischarge pain, functional limitations and impact on caregivers of children with sickle cell disease treated for painful events. Br. J. Haematol. 144, 782–788 (2009).
Dampier, C., Ely, E., Brodecki, D. & O’Neal, P. Home management of pain in sickle cell disease: a daily diary study in children and adolescents. J. Pediatr. Hematol. Oncol. 24, 643–647 (2002).
Smith, W. R. et al. Daily assessment of pain in adults with sickle cell disease. Ann. Intern. Med. 148, 94–101 (2008).
Smith, W. R. et al. Understanding pain and improving management of sickle cell disease: the PiSCES study. J. Natl Med. Assoc. 97, 183–193 (2005).
Ballas, S. K. et al. Hydroxyurea and sickle cell anemia: effect on quality of life. Health Qual. Life Outcomes 4, 59 (2006).
Thornburg, C. D., Calatroni, A. & Panepinto, J. A. Differences in health-related quality of life in children with sickle cell disease receiving hydroxyurea. J. Pediatr. Hematol. Oncol. 33, 251–254 (2011).
Beverung, L. M. et al. Health-related quality of life in children with sickle cell anemia: impact of blood transfusion therapy. Am. J. Hematol. 90, 139–143 (2015).
Kanter, J. et al. Validation of a novel point of care testing device for sickle cell disease. BMC Med. 13, 225 (2015).
Beverung, L. M., Varni, J. W. & Panepinto, J. A. Clinically meaningful interpretation of pediatric health-related quality of life in sickle cell disease. J. Pediatr. Hematol. Oncol. 37, 128–133 (2015).
Hoban, M. D., Orkin, S. H. & Bauer, D. E. Genetic treatment of a molecular disorder: gene therapy approaches to sickle cell disease. Blood 127, 839–848 (2016).
Ribeil, J. A. et al. Gene therapy in a patient with sickle cell disease. N. Engl. J. Med. 376, 848–855 (2017).
Tasan, I., Jain, S. & Zhao, H. Use of genome-editing tools to treat sickle cell disease. Hum. Genet. 135, 1011–1028 (2016).
Traxler, E. A. et al. A genome-editing strategy to treat beta-hemoglobinopathies that recapitulates a mutation associated with a benign genetic condition. Nat. Med. 22, 987–990 (2016).
Dever, D. P. et al. CRISPR/Cas9 beta-globin gene targeting in human haematopoietic stem cells. Nature 539, 384–389 (2016).
Telen, M. J. Beyond hydroxyurea: new and old drugs in the pipeline for sickle cell disease. Blood 127, 810–819 (2016).
Telen, M. J. et al. Randomized phase 2 study of GMI-1070 in SCD: reduction in time to resolution of vaso-occlusive events and decreased opioid use. Blood 125, 2656–2664 (2015).
Lebensburger, J. D. et al. Systematic review of interventional sickle cell trials registered in ClinicalTrials.gov. Clin. Trials 12, 575–583 (2015).
Niihara, Y. et al. Phase 3 study of L-glutamine therapy in sickle cell anemia and sickle β0-thalassemia subgroup analyses show consistent clinical improvement. Blood 128, 1318–1318 (2016).
Pace, B. Renaissance of Sickle Cell Disease Research in the Genome Era (Imperial College Press, 2007).
Sabarense, A. P., Lima, G. O., Silva, L. M. & Viana, M. B. Survival of children with sickle cell disease in the comprehensive newborn screening programme in Minas Gerais, Brazil. Paediatr. Int. Child Health 35, 329–332 (2015).
Gualandro, S. F., Fonseca, G. H., Yokomizo, I. K., Gualandro, D. M. & Suganuma, L. M. Cohort study of adult patients with haemoglobin SC disease: clinical characteristics and predictors of mortality. Br. J. Haematol. 171, 631–637 (2015).
Figueiredo, M. S. The compound state: Hb S/beta-thalassemia. Rev. Bras. Hematol. Hemoter. 37, 150–152 (2015).
Steinberg, M. H. in Disorders of Hemoglobin: Genetics, Pathophysiology, and Clinical Management (eds Steinberg, M. H., Forget, B. G., Higgs, D. R. & Weatherall, D. J. ) 786–810 (Cambridge Univ. Press, 2009).
Harrington, D. J., Adachi, K. & Royer, W. E. Jr. The high resolution crystal structure of deoxyhemoglobin S. J. Mol. Biol. 272, 398–407 (1997).
Eaton, W. A. & Hofrichter, J. Sickle cell hemoglobin polymerization. Adv. Protein Chem. 40, 63–279 (1990).
Briehl, R. W. & Ewert, S. Effects of pH, 2,3-diphosphoglycerate and salts on gelation of sickle cell deoxyhemoglobin. J. Mol. Biol. 80, 445–458 (1973).
Bookchin, R. M., Balazs, T. & Landau, L. C. Determinants of red cell sickling. Effects of varying pH and of increasing intracellular hemoglobin concentration by osmotic shrinkage. J. Lab Clin. Med. 87, 597–616 (1976).
Eaton, W. A., Hofrichter, J. & Ross, P. D. Editorial: Delay time of gelation: a possible determinant of clinical severity in sickle cell disease. Blood 47, 621–627 (1976).
Ferrone, F. A. The delay time in sickle cell disease after 40 years: a paradigm assessed. Am. J. Hematol. 90, 438–445 (2015).
Uzunova, V. V., Pan, W., Galkin, O. & Vekilov, P. G. Free heme and the polymerization of sickle cell hemoglobin. Biophys. J. 99, 1976–1985 (2010).
Hebbel, R. P., Boogaerts, M. A., Eaton, J. W. & Steinberg, M. H. Erythrocyte adherence to endothelium in sickle-cell anemia. A possible determinant of disease severity. N. Engl. J. Med. 302, 992–995 (1980).
Brugnara, C., de Franceschi, L. & Alper, S. L. Inhibition of Ca2+-dependent K+ transport and cell dehydration in sickle erythrocytes by clotrimazole and other imidazole derivatives. J. Clin. Invest. 92, 520–526 (1993).
Centers for Disease Control and Prevention. Registry and Surveillance System for Hemoglobinopathies (RuSH). CDChttps://www.cdc.gov/ncbddd/hemoglobinopathies/rush.html (2017).
Panepinto, J. A., Pajewski, N. M., Foerster, L. M. & Hoffmann, R. G. The performance of the PedsQL generic core scales in children with sickle cell disease. J. Pediatr. Hematol. Oncol. 30, 666–673 (2008).
[No authors listed.] FDA Briefing Document, Oncologic Drugs Advisory Committee Meeting, NDA 208587, L-glutamine, Applicant: Emmaus Medical, Inc. U.S. Food and Drug Administrationhttps://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/OncologicDrugsAdvisoryCommittee/UCM559734.pdf (2017).
Hoppe, C. C. et al. Design of the DOVE (Determining Effects of Platelet Inhibition on Vaso-Occlusive Events) trial: a global phase 3 double-blind, randomized, placebo-controlled, multicenter study of the efficacy and safety of prasugrel in pediatric patients with sickle cell anemia utilizing a dose titration strategy. Pediatr. Blood Cancer 63, 299–305 (2016).
Gibbs, W. J. & Hagemann, T. M. Purified poloxamer 188 for sickle cell vaso-occlusive crisis. Ann. Pharmacother. 38, 320–324 (2004).
Morris, C. R. et al. A randomized, placebo-controlled trial of arginine therapy for the treatment of children with sickle cell disease hospitalized with vaso-occlusive pain episodes. Haematologica 98, 1375–1382 (2013).
Du, E., Mendelsohn, L., Nichols, J. S., Dao, M. & Kato, G. J. Quantification of anti-sickling effect of Aes-103 in sickle cell disease using an in vitro microfluidic assay. Blood 124, 2699–2699 (2014).
Sins, J. W. R. et al. Effect of N-acetylcysteine on pain in daily life in patients with sickle cell disease: a randomised clinical trial. Br. J. Haematol.https://doi.org/10.1111/bjh.14809 (2017).
Brousseau, D. C. et al. A multicenter randomized controlled trial of intravenous magnesium for sickle cell pain crisis in children. Blood 126, 1651–1657 (2015).
Ware, R. E., Helms, R. W. & Investigators, S. Stroke With Transfusions Changing to Hydroxyurea (SWiTCH). Blood 119, 3925–3932 (2012).
Metcalf, B. et al. Discovery of GBT440, an orally bioavailable R-state stabilizer of sickle cell hemoglobin. ACS Med. Chem. Lett. 8, 321–326 (2017).
Gladwin, M. T. et al. Nitric oxide for inhalation in the acute treatment of sickle cell pain crisis: a randomized controlled trial. JAMA 305, 893–902 (2011).
Machado, R. F. et al. Hospitalization for pain in patients with sickle cell disease treated with sildenafil for elevated TRV and low exercise capacity. Blood 118, 855–864 (2011).
Misra, H. et al. A Phase Ib open label, randomized, safety study of SANGUINATE in patients with sickle cell anemia. Rev. Bras. Hematol. Hemoter 39, 20–27 (2017).
Telen, M. J. et al. Sevuparin binds to multiple adhesive ligands and reduces sickle red blood cell-induced vaso-occlusion. Br. J. Haematol. 175, 935–948 (2016).
Moutouh-de Parseval, L. A. et al. Pomalidomide and lenalidomide regulate erythropoiesis and fetal hemoglobin production in human CD34+ cells. J. Clin. Invest. 118, 248–258 (2008).
McArthur, J. G. et al. A novel, highly potent and selective PDE9 inhibitor for the treatment of sickle cell disease. Blood 128, 268–268 (2016).
Wambebe, C. et al. Double-blind, placebo-controlled, randomised cross-over clinical trial of NIPRISAN in patients with Sickle Cell Disorder. Phytomedicine 8, 252–261 (2001).
Conran, N. Prospects for early investigational therapies for sickle cell disease. Expert Opin. Investig. Drugs 24, 595–602 (2015).
Author information
Authors and Affiliations
Contributions
Introduction (M.H.G. and C.D.R.); Epidemiology (D.J.W. and F.B.P.); Mechanisms/pathophysiology (G.J.K. and F.F.C.); Diagnosis, screening and prevention (K.O.-F., E.P.V. and L.K.); Management (G.J.K., E.P.V. and F.B.P.); Quality of life (W.R.S. and J.A.P.); Outlook (G.J.K., F.B.P. and E.P.V.); Overview of Primer (G.J.K., F.B.P. and E.P.V.).
Corresponding author
Ethics declarations
Competing interests
G.J.K. is listed as a co-inventor on a patent application by the US NIH for the formulation of topical sodium nitrite (PCT/US2015/060015), receives research support from Bayer Pharmaceuticals and has received research support from AesRx and personal consulting fees (honoraria) from Novartis and Bioverativ outside the submitted work. The University of Pittsburgh received support for G.J.K.'s salary to serve on the steering committee for a clinical trial by Mast Therapeutics. F.B.P. reports personal fees (honoraria) from Novartis outside the submitted work. L.K., W.R.S., J.A.P., D.J.W., F.F.C. and E.V.P. declare no competing interests. Editor's note: all other authors have chosen not to declare any competing interests.
Rights and permissions
About this article
Cite this article
Kato, G., Piel, F., Reid, C. et al. Sickle cell disease. Nat Rev Dis Primers 4, 18010 (2018). https://doi.org/10.1038/nrdp.2018.10
Published:
DOI: https://doi.org/10.1038/nrdp.2018.10
This article is cited by
-
Medical educators’ perceptions of race in clinical practice
BMC Medical Education (2024)
-
Biophysical chemistry behind sickle cell anemia and the mechanism of voxelotor action
Scientific Reports (2024)
-
Evaluating sheep hemoglobins with MD simulations as an animal model for sickle cell disease
Scientific Reports (2024)
-
Base Editors-Mediated Gene Therapy in Hematopoietic Stem Cells for Hematologic Diseases
Stem Cell Reviews and Reports (2024)
-
Differential expression of adhesion molecules in sickle cell anemia and gut microbiome effect
Annals of Hematology (2024)