Abstract
Snakebite envenoming is a neglected tropical disease that kills >100,000 people and maims >400,000 people every year. Impoverished populations living in the rural tropics are particularly vulnerable; snakebite envenoming perpetuates the cycle of poverty. Snake venoms are complex mixtures of proteins that exert a wide range of toxic actions. The high variability in snake venom composition is responsible for the various clinical manifestations in envenomings, ranging from local tissue damage to potentially life-threatening systemic effects. Intravenous administration of antivenom is the only specific treatment to counteract envenoming. Analgesics, ventilator support, fluid therapy, haemodialysis and antibiotic therapy are also used. Novel therapeutic alternatives based on recombinant antibody technologies and new toxin inhibitors are being explored. Confronting snakebite envenoming at a global level demands the implementation of an integrated intervention strategy involving the WHO, the research community, antivenom manufacturers, regulatory agencies, national and regional health authorities, professional health organizations, international funding agencies, advocacy groups and civil society institutions.
Similar content being viewed by others
Introduction
Snakebite envenoming is a neglected tropical disease resulting from the injection of a highly specialized toxic secretion — venom — by a venomous snake into humans, usually under accidental circumstances. Venom is injected through the snake's fangs, which are modified teeth connected via a duct to a venom gland (Fig. 1a). The composition of snake venoms shows high complexity and diversity1, resulting in a variable biochemical and toxicological profile that determines a wide range of clinical manifestations. Some toxins in venom provoke local tissue damage, often resulting in permanent sequelae, whereas others induce systemic effects, including neurotoxic manifestations (leading to, for example, respiratory paralysis), bleeding, acute kidney injury, rhabdomyolysis (that is, a generalized breakdown of muscle fibres), cardiotoxicity, autonomic hyperactivity or thrombosis. Venoms from snakes of the family Viperidae (viperids) cause local effects and systemic manifestations associated with bleeding, coagulopathies and hypovolaemic shock2. Venoms from snakes of the family Elapidae (elapids) predominantly induce neurotoxic manifestations, such as neuromuscular paralysis2.
Schematic illustration of the venom system in a snake of the family Viperidae (viperids; part a). Venom is synthesized and stored in a specialized gland. When a bite occurs, venom is expelled through the action of a compressor muscle that surrounds the venom gland, and is delivered by a duct to the fangs through which it is injected into the tissues of an individual. Snake species responsible for the highest mortality owing to snakebite envenoming are Echis ocellatus (West African saw-scaled viper; family Viperidae; part b) and Bitis arietans (puff adder; family Viperidae; part c) in Africa, Naja naja (cobra; family Elapidae; part d), Bungarus caeruleus (common krait; family Elapidae; part e), Daboia russelii (Western Russell's viper; family Viperidae; part f) in Asia, Bothrops atrox (common lancehead; family Viperidae; part g) and Bothrops asper (terciopelo; family Viperidae; part h) in the Americas and Oxyuranus scutellatus (Papuan taipan; family Elapidae; part i) in Oceania. Many other snake species are also capable of inducing life-threatening envenomings. Images b–h courtesy of D.A.W., University of Oxford, UK, and image i courtesy of D.J.W., University of Melbourne, Victoria, Australia.
The superfamily Colubroidea — or advanced snakes — comprise >2,500 species with a wide geographical distribution and an extended evolutionary history. This superfamily includes all venomous snakes classified in the taxon Caenophidia, order Squamata, suborder Serpentes. The most dangerous species are classified within the families Viperidae (true vipers and pit vipers) and Elapidae (elapids; for example, cobras, kraits, mambas and sea snakes)3,4 (Fig. 1b–i). In addition, some species of the families Lamprophiidae (lamprophiids; subfamily Atractaspidinae; for example, burrowing asps or stiletto snakes) and several subfamilies of non-front-fanged colubroid snakes are also capable of inflicting envenomings2.
Because snakes are ectothermic, they are abundant in warmer climates, restricting the hyperendemic regions for snakebites mostly to tropical countries of the developing world (especially to some African, Asian, Latin American and Oceanic countries)5–7. In those countries, contact between snakes and humans is relatively common, particularly in the rainy season when human agricultural activity coincides with the snakes’ breeding season. Epidemiological evidence gathered from hospital records underscores the high burden of snakebite envenomings, which is considerable in terms of mortality and sequelae5,8. Evidence from community-based surveys in some countries suggests that the actual toll is even higher than estimates from hospital-based statistics (see, for example, Ref. 6). By contrast, inhabitants of higher-income countries of North America and especially Europe have far less exposure to venomous snakes and are generally unaware of the scale of the public health problem posed by snakebites elsewhere. Consequently, snakebite envenoming has historically received little attention from funding bodies, public health authorities, the pharmaceutical industry and health advocacy groups, thereby impairing the development of effective interventions to reduce the social impact of snakebites9,10.
This Primer summarizes the main issues of snakebite envenoming, including the epidemiology, the composition of snake venoms, and the pathophysiology, clinical manifestations, prevention and clinical management of snakebite envenomings. The global efforts being carried out to reduce the impact of this pathology are described, together with future trends to better understand and confront this neglected tropical disease.
Epidemiology
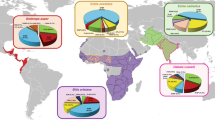
Snakebite envenoming is a major public health problem in the developing world (Fig. 2). It is an important cause of morbidity and mortality, especially in the impoverished areas of the warmer tropics and subtropics, such as sub-Saharan Africa, South to Southeast Asia, Papua New Guinea and Latin America6,7,11. Snakebite envenoming occurs in at least 1.8–2.7 million people worldwide per year, with combined upper estimates of mortality ranging from 81,410 to 137,880 deaths5,12. At least 46,000 of these deaths occur in India alone6. In sub-Saharan Africa, where data are fragmented, mortality estimates range from 7,000 to 32,000 deaths per year5,7,12, but are probably underestimated given that, in West Africa alone, annual mortality has been estimated at 3,557–5,450 deaths13.
Data shown constitute a rough approximation of the estimated range of snakebite envenomings and deaths given that, in many countries, reliable information on snakebite morbidity and mortality is lacking, resulting in underreported data of this neglected tropical condition. The highest impact of snakebite envenomings occurs in Asia, sub-Saharan Africa, Latin America and parts of Oceania. Based on estimates from Refs 5,12.
Snakebites disproportionately affect lower socioeconomic segments of society, people with poorly constructed housing and those with limited access to education and health care11,13. Countries with low gross domestic product, low Human Development Index (a composite statistic of life expectancy, education and income) and low health care expenditure are the most affected11. The disease pushes poor people further into poverty by virtue of high treatment costs, enforced borrowing and loss of income11. Indeed, bites and fatalities are most common in people 10–40 years of age, who comprise the most productive members of rural communities14. A higher case fatality rate is observed in those <5 years of age15. For example, in India, the proportion of all deaths from snakebite envenoming was highest at 5–14 years of age6. Children are exposed to snakes while helping in agricultural duties, playing or placing their hands in rodents’ burrows.
Snakebite envenoming is an occupational and environmental disease of the young and of agricultural workers. The specific populations at risk differ between countries. For example, tea pickers are at risk in southern India and Sri Lanka, rubber tappers in Liberia, Thailand, Malaysia, other Southeast Asian countries and Brazil, and sugar cane workers in South Africa, Saint Lucia and Martinique16–19. In Myanmar, snakebite envenoming is the fifth leading cause of death, particularly affecting rice paddy farmers20. Fishermen who use hand nets and lines in warmer tropical seas are also at risk, as are families of agricultural workers. Pregnant women are a highly vulnerable group, and snakebite envenoming has been recognized in Nigeria and Sri Lanka as an important cause of abortion and antepartum haemorrhage, as well as maternal and fetal loss21.
Snakebite envenoming is also an environmental hazard of indigenous nomadic peoples, hunter-gatherers, tribes, firewood collectors and indigents (extremely poor individuals). This risk has been documented in South America19, Africa (for example, the Hadza hunter-gatherers of Tanzania, the African Bushmen of the Kalahari Desert or southern Africa, the nomadic Fulani and Turkana pastoralists (sheep or cattle farmers) of the savanna in West Africa and Kenya)22, India and Sri Lanka. Envenoming has been an important cause of death among the indigenous communities of Australia, of the coastal lowlands of New Guinea23 and of the Amazon (the Yanomami and Waorani ethnic groups)18,19.
A proportion of individuals develop chronic morbidity, disability and psychological sequelae following snakebite envenoming, including amputations, post-traumatic stress disorder, blindness, maternal and fetal loss, contractures (that is, permanent shortening of a muscle or joint that often leads to deformity or rigidity), chronic infections and malignant ulcers7,21,24–26. At least 6,000 amputations owing to snakebite envenoming occur annually in sub-Saharan Africa alone7. Even when chronic disability is not factored in, the burden of premature death alone as a consequence of snakebite envenoming in India is estimated at 2.97 million disability-adjusted life years, whereas the global burden is conservatively estimated at 6.07 million disability-adjusted life years6,12,13. The global burden of snakebite envenoming is more than twice the estimate suggested for contributions grouped under ‘venomous animal contact’ in the 2013 Global Burden of Disease study27, underscoring a notorious underreporting of snakebites in official records. When both premature deaths and disability from snakebites are factored for 16 countries in West Africa, the combined burden surpasses the worldwide burdens for other neglected tropical diseases, such as Buruli ulcer, echinococcosis, leprosy, trachoma, yaws, yellow fever and podoconiosis. The burden of snakebites in sub-Saharan Africa is also higher than the burden of trypanosomiasis, leishmaniasis and onchocerciasis13.
Mechanisms/pathophysiology
Snake venom
Evolution of snake venoms. Venoms are used for predatory and defensive purposes and have evolved independently in a wide phylogenetic range of organisms, including snakes, spiders, scorpions and jellyfish28. Venom represents a trophic adaptive trait that is crucial for the foraging success of venomous snakes, and, as such, has had key roles in the organismal ecology and evolution of advanced snakes29. As a result of rapid evolution under Darwinian positive selection, venoms comprise protein mixtures of varying complexity that act individually or as an integrated phenotype to injure or kill the prey or accidental victim. Despite being traits of moderate genetic complexity in terms of the number of genes that encode toxins, within-species and between-species venom variability seems to be a common feature at all taxonomic levels30. The mechanisms that generated such biodiversity remain largely elusive, although genomic reorganizations and other alterations in genes encoding toxins and toxin expression patterns might be involved31–35. Like eyes, fins and wings, which have evolved independently in several different lineages, animal venoms have also been shaped by convergent structural and functional evolution36. Convergent evolution has repeatedly selected a restricted set of genes encoding proteins that contain specific structural motifs as templates for neofunctionalization (the process by which a gene acquires a new function after a gene duplication event) as venom toxins in different taxa37. Gene duplication, evolutionary divergence and post-translational quaternary associations in homomer or heteromer multiprotein complexes38 add to the complexity of snake venoms, by generating mixtures of proteins that belong to a handful of multigenic protein families; some of these protein families exhibit remarkable intrafamily variability39.
Revealing the spatial and temporal distribution of variations in venom composition within and between species is essential to understand the evolutionary processes and the ecological constraints that moulded snake venoms to their present-day variability and to define the phylogeographical boundaries of species. Insights into the selective pressures that resulted in local adaptation and species-level divergence in venoms can shed light on the mutual relationship between evolutionary and clinical toxinology: toxins that have the highest prey incapacitation activity are often also the most medically important molecules in the context of a human envenoming. Thus, identifying the molecular basis of venomous snake adaptation to their natural ecosystems may assist in the identification of those toxins that must be neutralized to reverse the effects of venom, thereby guiding the rational development of next-generation snakebite therapeutics40.
Analysis of snake venom composition. The growing interest in different aspects of venom biology has catalysed the development of ‘-omics’ methodologies aimed at the qualitative and quantitative characterization of venom toxins, including the proteomics of venoms (that is, venomics; Box 1). In particular, the combination of next-generation transcriptomics41,42 and proteomics workflows has demonstrated unparalleled capabilities for venom characterization in unprecedented detail. The relative distribution of the main types of toxic components in viperid and elapid venoms is summarized in Fig. 3.
The graph highlights the ranges of protein levels (expressed as % of the total venom proteome) and the distinct distribution of the most abundant toxin families across different venoms of snake species from the families Viperidae (subfamilies Viperinae and Crotalinae) and Elapidae (subfamilies Elapinae and Hydrophiinae). Bars are colour-coded according to the most relevant biological activities of the corresponding toxin family36,40. Colour gradients indicate concentration dependency of the biological effect (same colour) or different effects (multiple colours; that is, some toxins may exert one effect at low doses and another effect at high doses). The crystallographic or nuclear magnetic resonance structures of some members of each protein family are also shown. More information on the crystal structures shown and their source can be found in Supplementary information S2 (box). 3FTx, three-finger toxin (Protein Data Bank accession ID (PDB ID): 1IJC); CRISP, cysteine-rich secretory protein (PDB ID: 3MZ8); CTL, C-type lectin-like protein (PDB ID: 1IXX); DTx, dendrotoxin (PDB ID: 1DTX); LAO, ʟ-amino acid oxidase (PDB ID: 2IID); Myo, low molecular mass myotoxin (PDB ID: 4GV5); PLA2, phospholipase A2 (PDB ID: 1TGM for the monomer and PDB ID: 3R0L for the dimer); SVMP, snake venom metalloproteinase (PDB ID: 3DSL for class PIII and PDB ID: 1ND1 for class PI); SVSP, snake venom serine proteinase (PDB ID: 1OP0).
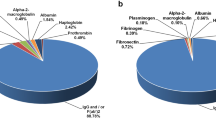
A straightforward translational application of the body of knowledge gained through venomics is the analysis of the immune reactivity of antivenoms (also known as antivenins, antivenenes, anti-snakebites or anti-snake venom sera) against venoms, a field coined ‘antivenomics’ (Fig. 4). Antivenomics is a proteomics-based protocol to quantify the extent of cross-reactivity of antivenoms against homologous and heterologous venoms43. The combination of antivenomics and in vivo neutralization tests constitute a powerful toolbox for evaluating the preclinical efficacy of an antivenom44.
Whole venom is applied to an immunoaffinity column packed with antivenom antibodies that are immobilized onto Sepharose beads. After eluting the non-immunoretained venom components, the immunoretained proteins are eluted. Comparison of the reversed-phase high-performance liquid chromatographs of the components of whole venom, the immunoretained fraction and the non-immunoretained fraction can provide qualitative and quantitative information on the set of toxins that bear antivenom-recognized epitopes and those toxins that exhibit poor immunoreactivity.
Mechanism of action
Snakes inject their venom through a specialized delivery system, which includes a set of fangs located in the frontal region of the maxillary bones in viperids (Fig. 1a), elapids and lamprophiids, whereas fangs have a posterior location in non-front-fanged colubroids. Depending on the size of the fangs, venom is injected either subcutaneously or intramuscularly. Once delivered, some venom toxins exert local pathological effects in neighbouring tissues, whereas others are distributed systemically through the lymphatic system and blood vessels, enabling toxins to act at various organs2. Activities of main toxin families are shown in Box 2, whereas the potential consequences of snakebite envenoming are shown in Fig. 5.
Venoms exert a wide range of toxic activities in the body, and the predominant deleterious actions depend on the composition of the venom. Elapid venoms, and some viperid venoms, induce neuromuscular paralysis. Most viperid venoms, and some elapid venoms, inflict prominent local tissue damage. Viperid venoms cause systemic haemorrhage, which, together with increased vascular permeability, can lead to cardiovascular shock. Viperid and some elapid and some non-front-fanged colubroid venoms act at various levels of the coagulation cascade and on platelets, thereby affecting haemostasis. Some venoms cause generalized muscle breakdown (that is, rhabdomyolysis). Acute kidney injury often develops in envenomings, owing to a multifactorial pathogenesis. 3FTx, three-finger toxin; PLA2, phospholipase A2; SVMP, snake venom metalloproteinase; SVSP, snake venom serine proteinase.
Local tissue damage. Most viperid and some elapid venoms induce local tissue damage. Myonecrosis is primarily due to the action of myotoxic phospholipases A2 (PLA2s) that are present in these venoms, which bind to and disrupt the integrity of the plasma membrane of muscle fibres45,46. For some PLA2s, disruption of plasma membranes is secondary to hydrolysis of membrane phospholipids, whereas in the case of catalytically inactive PLA2 homologues, sarcolemmal damage occurs through hydrophobic interactions47. Calcium influx into the cytosol occurs following membrane perturbation, causing myofilament hypercontraction, mitochondrial dysfunction and other degenerative events, leading to irreversible muscle cell damage46,47. Small basic myotoxic peptides that are present in some rattlesnake venoms (family Viperidae) also induce muscle contracture and can cause necrosis2. Muscle fibres are also affected by ischaemia as a result of vascular alterations and from increased pressure in muscles as a consequence of oedema48. Skeletal muscle regeneration requires the removal of necrotic debris by phagocytic cells, and depends on an intact blood supply and innervation to be successful. In viperid venoms, which affect muscle fibres and damage the vasculature and nerves, skeletal muscle regeneration is impaired, often resulting in permanent sequelae48,49.
In addition to myonecrosis, blood vessel integrity is also affected. Snake venom metalloproteinases (SVMPs) in viperid venoms hydrolyse key components of the basement membrane of capillaries, particularly type IV collagen, causing weakening of the mechanical stability of microvessels. As a consequence, the haemodynamic biophysical forces operating in the circulation cause distension and, eventually, disruption of the capillary wall, resulting in extravasation50. SVMP-induced microvascular damage can also be a consequence of the disruption of endothelial cell–cell adhesions51. SVMPs and hyaluronidases hydrolyse extracellular matrix components, including various types of collagens, hyaluronic acid and proteoglycans, affecting the structure and function of not only microvessels but also other tissue components, thereby playing a part in venom-induced local tissue damage52.
SVMPs also induce skin damage by degrading the dermal–epidermal interface, with the consequent formation of blisters53. Some cobra venoms (Naja spp.; family Elapidae) induce extensive cutaneous necrosis owing to the action of cytotoxins of the three-finger toxin family54, so named for having a common structure of three loops extending from a central core, which destabilize plasma membranes of various cell types in different tissues through a non-enzymatic mechanism55. Locally acting toxins (that is, PLA2s and SVMPs) also affect intramuscular nerves49 and vascular components, such as lymphatic vessels, arterioles and venules56.
An extensive local inflammatory process develops in envenomed tissue, with the synthesis and release of eicosanoids, nitric oxide, bradykinin, complement anaphylatoxins, histamine and cytokines, the activation of resident macrophages and other cell types, and the recruitment of leukocytes57. This inflammatory milieu induces an increased vascular permeability with the formation of an exudate that, in addition to plasma proteins, contains intracellular and extracellular protein fragments, chemokines, cytokines and damage-associated molecular patterns, which are likely to potentiate the inflammation and, possibly, contribute to further tissue damage58.
Some mediators also induce pain57. Catalytically inactive PLA2 homologues excite pain-related sensory neurons via the release of ATP and activation of purinergic receptors59.
Neurotoxicity. Snake venoms of most elapid species, and of some viperid species, contain neurotoxins that induce a descending flaccid neuromuscular paralysis, which can involve the life-threatening blockade of bulbar (muscles of the mouth and throat that are responsible for speech and swallowing) and respiratory muscles. Two main types of neurotoxins are found in snake venoms: α-neurotoxins and β-neurotoxins.
α-Neurotoxins belong to the three-finger toxin family and exert their action postsynaptically at neuromuscular junctions60. They bind with high affinity to the cholinergic receptor at the motor end plate in muscle fibres, thereby inhibiting the binding of acetylcholine and provoking flaccid paralysis60.
By contrast, β-neurotoxins are typically PLA2s that act at the presynaptic nerve terminal of neuromuscular junctions61. For example, the receptor for β-bungarotoxin, from the krait Bungarus multicinctus (family Elapidae), is a voltage-gated potassium channel62. Upon binding to their targets, neurotoxic PLA2s induce enzymatic hydrolysis of phospholipids at the nerve terminal plasma membrane, which causes neurotoxicity63. Indeed, the generation of lysophospholipids and fatty acids in the membrane cause biophysical changes that lead to the fusion of synaptic vesicles to the membrane and the exocytosis of the ready-to-release pool of vesicles46. Furthermore, membrane permeability to ions is increased, with the consequent depolarization and influx of calcium, resulting in exocytosis of the reserve pool of vesicles61. Consequently, presynaptic vesicles are depleted, and intracellular degenerative events ensue, including mitochondrial alterations, ending up in the destruction of nerve terminals64,65. These events explain the prolonged and severe paralysis observed in patients. Some neurotoxic PLA2s can also act intracellularly after entering the cytosol by endocytosis or through the damaged plasma membrane62. Within the nerve terminal, PLA2s cause further degenerative events in mitochondria66.
Other neurotoxins are dendrotoxins and fasciculins, which are present in the venoms of the African mambas (Dendroaspis spp.; family Elapidae). Dendrotoxins block voltage-gated potassium channels at the presynaptic nerve terminal67. Fasciculins, which also belong to the three-finger toxin family, are inhibitors of acetylcholinesterase68. The combined action of these neurotoxins results in excitatory effects and fasciculations (involuntary contractions of small groups of muscle fibres). Some cysteine-rich secretory proteins in venoms induce paralysis of smooth muscle2.
Cardiovascular and haemostatic disturbances. Systemic haemorrhage occurs in envenomings by viperids and by some species of non-front-fanged colubroids, and can also develop in envenomings by Australian elapids. In viperid venoms, the main toxins responsible for systemic haemorrhage are SVMPs, especially those of the class PIII. These toxins have a multi-domain structure containing exosites (molecular sites distinct from the active catalytic site that serve as secondary binding sites) that enable them to target the microvasculature50,69. Bleeding can occur in different organs with several pathophysiological consequences. For example, intracranial haemorrhage has been described in envenomings, causing ischaemia, stroke and neurological sequelae70,71. The mechanism of action of systemically acting haemorrhagic SVMPs is likely to be similar to that described for local haemorrhage, that is, cleavage of key substrates at the basement membrane of capillaries and at cell–cell junctions, resulting in the mechanical weakening of the microvessel wall and extravasation50,51.
Snake venoms affect haemostasis in various ways. Many viperid venoms, and some elapid and non-front-fanged colubroid venoms, contain enzymes that promote coagulation; these enzymes are either SVMPs or snake venom serine proteinases that act in the coagulation cascade, such as thrombin-like enzymes or activators of coagulation factor V, factor X or prothrombin72,73. Some venom enzymes also hydrolyse fibrinogen and fibrin73. In addition, SVMPs release tissue factor74 and affect endothelial function in various ways. Although these procoagulant components can cause intravascular coagulation, in the majority of cases they induce a consumption coagulopathy, which results in defibrinogenation and incoagulability that are reflected in the alteration of blood clotting tests70. This condition may contribute to systemic bleeding, especially in venoms containing haemorrhagic toxins that disrupt the integrity of blood vessels2,70. Some Australian elapid venoms, which lack haemorrhagic SVMPs but cause coagulopathy secondary to the action of serine proteinase prothrombin activators, often induce systemic bleeding70.
Many snake venoms affect platelets. SVMP-mediated microvascular damage and C-type lectin-like proteins contribute to the decrease in platelet numbers75. Moreover, disintegrins, C-type lectin-like proteins, snake venom serine proteinases and some SVMPs impair platelet aggregation by blocking platelet receptors or by interacting with von Willebrand factor73,76,77. Thrombocytopenia has been associated with an increased risk of systemic bleeding in envenomings by haemorrhagic venoms. By contrast, the venoms of two endemic Caribbean viperid species induce severe thrombosis, which leads to infarcts in the lungs, brain and heart, despite not being directly procoagulant78. Thrombosis is probably dependent on SVMP-induced systemic endothelial dysfunction. Acute pituitary insufficiency secondary to thrombi formation and focal haemorrhage in the anterior pituitary glands occurs in bites by some viperids79.
Venom-induced systemic bleeding is one of the leading causes of the haemodynamic disturbances experienced by patients envenomed by viperids, which may progress to cardiovascular shock2. In these envenomings, hypovolaemia also results from an increase in vascular permeability, including systemic plasma leakage. This effect is induced by snake venom serine proteinases that release bradykinin and also by the action of many vasoactive endogenous inflammatory mediators. In addition, viperid venoms contain bradykinin-potentiating peptides, some of which inhibit angiotensin-converting enzyme and contribute to haemodynamic alterations80.
Hyponatraemia, possibly caused by venom natriuretic factor, may play a part in cardiovascular disturbances in some envenomings81. A direct cardiotoxic effect might also add to the multifactorial setting of haemodynamic disturbances; sarafotoxins are responsible for cardiotoxicity in atractaspid venoms2. Sepsis has been described in individuals with snakebite envenoming as a consequence of infection, further contributing to cardiovascular dysfunction and shock.
Acute kidney injury. Some viperid and some elapid snakebite envenomings can lead to acute kidney injury82. Depending on the type of venom, the following mechanisms have been associated with the pathogenesis of renal damage: ischaemia secondary to decreased renal blood flow that results from haemodynamic alterations caused by systemic bleeding and vascular leakage; proteolytic degradation of the glomerular basement membrane by SVMPs; deposition of microthrombi in the renal microvasculature (that is, thrombotic microangiopathy), which might also cause haemolysis; direct cytotoxic action of venom components, such as cytotoxic PLA2s, in renal tubular cells; and in the cases of venoms inducing systemic myotoxicity (that is, rhabdomyolysis), accumulation of large amounts of myoglobin in renal tubules, with consequent toxicity82,83.
Rhabdomyolysis. Envenomings by sea snakes, some Australian terrestrial elapids and some viperid species are associated with rhabdomyolysis2. This effect is due to the action of myotoxic PLA2s at the systemic level as a result of the binding of these toxins to receptors in muscle fibres. Myotoxins disrupt the integrity of the plasma membrane of muscle cells, as described for locally acting myotoxins, causing calcium influx and cellular degeneration47. Thus, large amounts of muscle cytosolic proteins, such as creatine kinase and myoglobin, are released. Deposition of myoglobin in the renal tubules may contribute to acute kidney injury82.
Diagnosis, screening and prevention
Snakebite envenomings are emergencies that are clinically challenging owing to their potentially rapid lethality. Uncertainties about species identity and the quantity of venom injected and its composition, which can vary with the age of the snake and within species throughout its geographical range, complicate decision making84. Most snakebites are managed by nurses or health assistants in district and rural hospitals, clinics, dispensaries and health posts. In some cases, referral to a provincial tertiary hospital with specialists, intensive care units and laboratories might be possible.
Clinical presentation
Individuals with snakebite envenoming present with local and systemic symptoms of envenoming, as well as anxiety and symptoms associated with the treatment they received before arrival to the hospital or health care post. Fear can cause misleading symptoms, such as vomiting, sweating, tachycardia, acroparaesthesia (abnormal sensation in the extremities), carpopedal spasm (tetany causing painful cramps of the hands, wrists and feet), tachypnoea and hyperventilation leading to syncope, and functional neurological disorders. Widely practiced traditional first aid treatments for snakebites include tight bands or tourniquets85, local incisions, ingestion of emetic herbs or topical application of herbs, or application of ice, suction, fire or electric shocks at the site of the bite. Not only is the effectiveness of these traditional treatments not proven but they can also result in treatment-associated comorbidities even in the absence of envenoming, such as a painful, swollen, ischaemic or even gangrenous limb, bleeding or infections.
The specific clinical manifestations associated with bites of viperids, elapids or non-front-fanged colubroid snakes are described below.
Viperids. The classic syndrome associated with bites from viperids (including Viperinae (true Old World vipers and adders) and Crotalinae (Asian pit vipers, mamushis, habus and New World rattlesnakes, moccasins, bushmasters and lanceheads)) consists of local and systemic effects. Local effects in the bitten limb include immediate radiating pain; rapidly extending tender swelling with hot inflammatory erythema, which usually becomes evident within 2 hours of the bite; signs of lymphangitis (inflammation of the lymph vessels, which presents as red lines on the skin); prolonged bleeding from fang puncture wounds; bullae (blistering); ecchymosis (bruising); tender regional lymph node enlargement; and superficial soft tissue and muscle necrosis and secondary infection (cellulitis or abscess)19,86,87 (Fig. 6a,b). Systemic effects include early syncope and collapse with transient loss of vision and consciousness; hypotension and shock; cardiac tachyarrhythmia or bradyarrhythmia; severe bleeding diathesis, spontaneous systemic bleeding from the nose, gums (Fig. 6c), respiratory, gastrointestinal and genitourinary tracts and sites of recent trauma or healing wounds, and subarachnoid, cerebral, and antepartum or postpartum haemorrhages leading to abortion and fetal death19,86,87.
Swelling and blistering following a bite on the dorsum of the foot by a jararaca (Bothrops jararaca; family Viperidae) in Brazil (part a). Swelling, blistering and gangrene of the hand, which required amputation, and extensive ecchymoses (discoloration of the skin owing to bleeding under the skin) following a bite of the Malayan pit viper (Calloselasma rhodostoma; family Viperidae) in Thailand (part b). Bleeding gums, a cardinal sign of failure of haemostasis, in a patient bitten by a West African saw-scaled viper (Echis ocellatus; family Viperidae) in Nigeria (part c). Bilateral conjunctival oedema (chemosis), which indicates a generalized increase in capillary permeability, following a bite by Eastern Russell's viper (Daboia siamensis; family Viperidae) in Myanmar (part d). Bilateral ptosis (paralysis of both upper eyelids; part e) and external ophthalmoplegia (paralysis of the eye muscles; the patient cannot look to the right; part f), in patients bitten by Papuan taipans (Oxyuranus scutellatus; family Elapidae) in Papua New Guinea. Extensive necrosis of skin and subcutaneous tissue following a bite on the elbow by a black-necked spitting cobra (Naja nigricollis; family Elapidae) in Nigeria (part g). Images courtesy of D.A.W., University of Oxford, UK.
Variant syndromes consist of symptoms in addition to those described above and are associated with envenoming by particular species. These symptoms include early ‘anaphylactic’ (autonomic) symptoms (for example, urticaria, angioedema, shock, sweating, vomiting and diarrhoea); acute kidney injury; generalized increase in capillary permeability, such as chemosis (Fig. 6d); acute or chronic pituitary failure; neuromyotoxicity; and in situ arterial thrombosis (causing ischaemic infarcts in the brain, kidney, lungs, heart or elsewhere). Envenoming by some rattlesnakes (Crotalus spp.) causes neuromyotoxic symptoms, characterized by fasciculations, in North America and elapid-like descending paralysis with rhabdomyolysis and acute kidney injury in South America19. Envenoming by bushmasters (Lachesis spp.) can cause dramatic early autonomic symptoms, sometimes with vasovagal features, and severe local envenoming19. A review of the species causing these symptoms is beyond the scope of this Primer, but additional information is provided in Supplementary information S1 (box) and reviewed elsewhere19,86,87.
Elapids. Bites of elapids (including cobras, kraits, mambas, coral snakes, Oceanic venomous snakes and sea snakes) are associated with the classic neurotoxic syndrome, which is characterized by flaccid paralysis that is first evident as bilateral ptosis and external ophthalmoplegia (Fig. 6e,f), sometimes with dilated pupils. Drowsiness is occasionally observed as a symptom, but snake venom toxins are not thought to cross the blood–brain barrier, so this finding is difficult to explain. Paralysis descends to involve muscles innervated by lower cranial nerves, as well as neck flexors and bulbar, respiratory, trunk and limb muscles. Other symptoms are pooling of secretions in the pharynx, loss of the gag reflex, dyspnoea, declining ventilatory capacity, paradoxical abdominal respiration, use of accessory muscles and cyanosis, which are ominous signs of impending bulbar and respiratory paralysis. Paralysis is sometimes reversible following treatment with acetylcholinesterase inhibitors or specific antivenoms, and recovers over time in all cases, provided that respiration is adequately supported19,23,86–89. Local symptoms include absent-to-moderate pain, paraesthesia and local swelling, without blistering or necrosis.
Variant syndromes consist of severe local envenoming with immediate radiating pain and rapidly extending tender swelling; blistering; superficial, patchy (the presence of ‘skip lesions’) soft tissue necrosis (Fig. 6g) and secondary infection; tender regional lymph node enlargement; autonomic overactivity and fasciculations; severe abdominal pain that resembles renal or biliary colic (which gradually increases in intensity); excruciating pain radiating up the bitten limb; acute kidney injury associated with rhabdomyolysis; hyponatraemia; spontaneous bleeding and coagulopathy; and microangiopathic haemolysis. Venom released by spitting elapids into an individual's eyes can lead to ophthalmia, resulting in intensely painful chemical conjunctivitis with lacrimation and swelling of the eyelids, risk of corneal ulceration, anterior uveitis and secondary infection leading to permanent blindness (see Supplementary information S1 (box))19,86,87.
Non-front-fanged colubroids. Bites from non-front-fanged colubroid snakes (including the African boomslang (Dispholidus typus), vine snakes (Thelotornis spp.), Asian keelbacks (Rhabdophis spp.) and South American racers (Philodryas spp.)) are associated with slowly or late evolving ecchymoses, systemic bleeding, coagulopathy and acute kidney injury with minimal local envenoming. Fatalities from envenoming by the African and Asian species have occurred, but South American non-front-fanged colubroid snakes seem to be less dangerous. Mild local envenoming can be caused by bites of many non-front-fanged colubroid species, some of which are kept as pets in Western countries (for example, hognose snakes (Heterodon spp.)) (see Supplementary information S1 (box))90,91.
Clinical time course. After bites by vipers (family Viperidae) and some cobras (family Elapidae), local swelling is usually detectable within 2–4 hours and can extend rapidly to reach its peak on the second or third day. Blistering appears within 2–12 hours, and tissue necrosis becomes obvious within 1 day of the bite. Sloughing of necrotic tissue and secondary infections, including osteomyelitis (infection of underlying bone), develop during subsequent weeks or month. Complete resolution of swelling and restoration of normal function in the bitten limb may take weeks. Vomiting or syncope within minutes of the bite may indicate systemic envenoming. Coagulopathy and bleeding develop within a few hours and can persist for ≥2 weeks in untreated individuals. Neurotoxic signs can progress to generalized flaccid paralysis and respiratory arrest within 30 minutes to a few hours. Patients with neurotoxic envenoming usually recover within a few days with assisted ventilation, but some may need respiratory support for as long as 10 weeks19,86,87.
Diagnosis
To deduce the nature and severity of envenoming, a sequential clinical history of symptoms must be obtained and signs of envenoming rapidly elicited (Box 3), so that appropriate, urgent, life-saving treatment can be given. Rapid clinical assessment must include vital signs, measurement of postural blood pressure to exclude hypovolaemia, formal testing for ptosis and signs of more-advanced paralysis especially causing respiratory failure, and examination for spontaneous systemic bleeding.
Patients usually know that they have been bitten, except those who experience painless nocturnal bites by kraits (Bungarus spp., family Elapidae) while asleep92,93. Differential diagnoses of snakebites include bites by arthropods (for example, spiders), lizards, rodents or fish; stings (for example, by hymenopterans, scorpions or centipedes); or punctures by plant spines or thorns, nails, splinters or other sharp objects. Definite snakebites that result in negligible or no symptoms may have been ‘dry bites’ (that is, transcutaneous bites without envenoming94) inflicted by venomous snakes or bites by non-venomous species. If the dead snake, or a photograph of it, is available, an expert herpetologist can identify the species. Otherwise, descriptions by individuals with a snakebite or bystanders can be helpful, supported by recognition of evolving characteristic patterns (syndromes) of symptoms95 and aided by biochemical measurements and imaging.
Laboratory and other investigations. Laboratory investigations can help to identify systemic envenoming and aid the management of snakebites. Peripheral neutrophil leukocytosis, which indicates a general inflammatory response, confirms systemic envenoming. A low haematocrit (the volume percentage of red blood cells in blood) value reflects severe haemorrhage, whereas a high haematocrit value reflects haemoconcentration from the leakage of plasma into the tissues as a result of increased capillary permeability. Severe thrombocytopenia is associated with severe bleeding diathesis and sometimes with microangiopathic haemolysis, which is diagnosed by the presence of schistocytes in a blood film, causing acute kidney injury. Incoagulable blood is a cardinal sign of systemic envenoming by viperids, Oceanic elapids and non-front-fanged colubroid snakes. The simple 20-minute whole-blood clotting test involves placing a few millilitres of venous blood in a new, clean, dry, glass vessel, leaving it undisturbed at room temperature for 20 minutes, and then tipping it once to see if it has clotted96,97. Lack of clotting indicates severe consumption coagulopathy or an anticoagulant venom87. Laboratory tests, such as prothrombin and activated partial thromboplastin times, fibrin degradation products and D-dimer, are more-sensitive indices of disseminated intravascular coagulation and fibrinolysis. Levels of creatine kinase of >10,000 units per litre indicate severe rhabdomyolysis. Blood urea or serum creatinine and potassium concentrations should be measured in patients who are at risk of acute kidney injury. Urine should be tested on admission for the presence of haemoglobin, myoglobin, other proteins and blood.
Electrocardiographic abnormalities include sinus bradycardia, ST–T changes, and varying degrees of atrioventricular block or evidence of myocardial ischaemia. Myocardial infarction can occur secondary to shock in patients with pre-existing coronary artery disease. Echocardiography can detect pericardial effusion and myocardial dysfunction, and bleeding into the pleural and peritoneal cavities. The use of wound ultrasonography has also been advocated to detect tissue damage98. CT and MRI are increasingly available for assessing intracranial haemorrhages and infarcts.
Detection of venom. Detection and quantification of venom antigens in body fluids of individuals with snakebite envenoming, using enzyme immunoassays99–102 provides retrospective confirmation of species diagnosis, predicts prognosis and is one measure of the effectiveness of antivenom treatment. High concentrations of venom antigens (that is, from wound swabs or wound aspirates) can be detected within 15–30 minutes, but commercial venom detection kits are available only in Australia (produced by Seqirus)103. Venom detection kits are highly sensitive but insufficiently specific to distinguish between venoms of closely related species. Detection of venom in a wound swab does not prove that the patient has been envenomed and is not, on its own, an indication for antivenom treatment. For retrospective species diagnosis, including forensic cases, tissue around the fang punctures, wound and blister aspirate, and serum and urine samples should be stored for enzyme immunoassays. For determining the identity of the biting species, highly specific methods are being developed, such as the detection of venom gland mRNA by reverse-transcription PCR104 or snake-derived DNA in bite-wound swabs105.
Prevention
The most effective method of preventing snakebites is through education directed at high-risk communities, and designed and driven from within those communities86,87. A full range of media should be used, including radio, TV, mobile phone apps, social media, posters, puppet and drama performances and village-based public meetings. Awareness of snakebite envenoming must be increased, together with advice on safer walking, working106 and sleeping107. Transport of individuals with a snakebite to clinics where they can receive medical care can be improved, even in areas that are inaccessible to conventional ambulances, for example, using boats or volunteer village-based motorcyclists108. Wasting time by visiting traditional therapists should be tactfully but firmly discouraged.
Management
First aid
Immediate first aid after a bite should be done by the affected individual or bystanders. Important elements are reassurance, immobilization of the whole body, especially the bitten limb (to reduce dissemination of venom through veins and the lymphatic system), removal of rings and tight objects around the bitten limb and application of a pressure pad or pressure bandage over the bite wound109,110. The patient must be transported rapidly and preferably passively to the nearest place providing medical care. Pain should be controlled using paracetamol (also known as acetaminophen) or opioids, but not using aspirin or NSAIDs because of the risk of exaggerating bleeding problems. Minimizing the risk of fatal shock and upper respiratory obstruction (through bulbar paralysis or fluid aspiration) during transit is achieved by placing the patient in the recovery position and inserting an oropharyngeal airway (a tube to maintain the airway). Ineffective and damaging traditional treatments, such as incisions, suction and tight tourniquets, must be discouraged. In suspected cases of neurotoxic envenoming by cobras, death adders (Acanthophis spp.), some Latin American coral snakes (Micrurus spp.) and other elapids whose venoms act mainly on postsynaptic receptors of the neuromuscular junction, administration of atropine (an antimuscarinic) and neostigmine (an acetylcholinesterase inhibitor) to improve neuromuscular transmission has been suggested as first aid111.
Hospital management
Patients who claim to have been bitten by a snake should be admitted for a minimum of 24 hours and be clinically assessed as described above. An intravenous line and the necessary resources for immediate resuscitation should be in place before an in situ compression bandage or tourniquet is removed, as this may precipitate dramatic deterioration112. In patients who are breathless and centrally cyanosed (the lips, tongue and mucosae are blue because the blood is poorly oxygenated), the airway should be restored and oxygen given by any possible means. If the patient is in shock, the foot of the bed should be raised immediately and intravenous fluid infused. Pain is variable but might be very severe and should be treated appropriately, as described above.
Patients who initially present without evidence of envenoming can deteriorate rapidly and unpredictably over minutes or hours. Published severity scores usually based on arbitrary criteria are, therefore, inherently unreliable or even potentially dangerous. However, several studies have shown that admission levels of venom antigenaemia (that is, the concentration of venom antigens in serum or plasma detected by enzyme immunoassays) were of prognostic value113,114. Patients with snakebite envenoming should be carefully observed and their blood pressure, pulse rate, level of consciousness, the presence or absence of ptosis and spontaneous bleeding, the extent and magnitude of local swelling, and urine output should be monitored. If clinical compartment syndrome (that is, marked swelling of muscles contained in a tight fascial compartment that might jeopardize the blood supply) is suspected, intra-compartmental pressure should be monitored. Assessing the level of consciousness of patients with neurotoxic envenoming can be difficult because their generalized flaccid paralysis makes the commonly used Glasgow Coma Scale misleading. For example, a patient may not be able to open their eyes, speak or obey commands, but if cardiorespiratory support is adequate and their paralysed upper eyelids are raised, they may be found to be fully conscious and able to signal ‘yes’ or ‘no’ in response to simple questions by flexing a finger or toe. After resuscitation and attempted species diagnosis, the most crucial management decision is whether the patient requires antivenom.
Antivenom. Antivenom is the only effective specific antidote for the systemic effects of snakebite envenoming115. Antivenom comprises concentrated immunoglobulins of horse, sheep or other large domesticated animals such as camels that have been hyper-immunized with one or more venoms over periods of months to years. Worldwide, most antivenom manufacturers refine the whole IgG extracted from the animals’ plasma by enzyme digestion with pepsin to produce F(ab′)2 fragments, under the assumption that removal of the Fc moiety from the antigen-binding (Fab) fragment reduces the risk of adverse reactions. Other manufacturers use papain to produce smaller Fab fragments to improve safety and increase the speed of distribution throughout the body, but with the disadvantage of rapid renal clearance of the antivenom so that recurrent envenoming becomes a problem116. Some antivenoms comprise whole IgG molecules that are usually purified by caprylic acid precipitation115. Antivenom antibodies can be extracted by affinity column purification, increasing safety but also cost.
Polyvalent (polyspecific) antivenoms are raised against the venoms of the most medically important snake species in a particular geographical area. Examples are the Indian antivenoms that are effective against the ‘big four’ national species: Naja naja (family Elapidae), Bungarus caeruleus (family Elapidae), Daboia russelii (family Viperidae) and Echis carinatus (family Viperidae). By contrast, monovalent (monospecific) antivenoms are raised against the venom of a single species, for example, European ViperaTab (Flynn Pharma) is effective against venom from Vipera berus (family Viperidae).
Antivenoms have proved to be effective against many of the lethal and damaging effects of venoms for more than a century. Antivenom administration can reverse anti-haemostasis, hypotension and postsynaptic neurotoxicity, and, if given early, prevent or limit presynaptic neurotoxicity, rhabdomyolysis and local tissue necrosis19,86,87,115. In the management of snakebites, the most important clinical decision is whether to give antivenom. Antivenoms are highly specific and, therefore, will neutralize only the venoms used in their production, together with those of a few related species; thus, in a particular case of snakebite, an appropriate antivenom must be selected, based on identification of the snake responsible for the envenoming. In addition, antivenoms are costly, often scarce, poorly distributed in areas where they are most needed and may require cold-chain for transport and storage117. Over the past few decades, several major antivenom manufacturers (such as Syntex, Behringwerke and Sanofi Pasteur) have stopped production, mainly for commercial reasons, creating serious shortages of antivenom in the countries that they previously supplied, especially in Africa118. Only a minority of patients who are bitten by a snake fulfil the criteria for antivenom use (Table 1).
Dosage is the same for adults and children. The initial dose would ideally be based on clinical trial data119–123, but as these data are rarely available, the manufacturer's estimate of neutralizing potency, based on rodent median effective dose, is usually the guide. Dose is increased according to clinical estimates of severity of envenoming and is repeated in the face of deteriorating neurotoxic or cardiovascular signs after 1–2 hours or persistence of incoagulable blood after 6 hours. Administration is always intravenous, over 10–60 minutes. Patients must be closely observed for early anaphylactic and pyrogenic reactions, especially during the first 2 hours after starting antivenom treatment. The incidence and severity of these dose-related early adverse antivenom reactions can be reduced by prophylactic adrenaline administration124. If reactions do occur, they should be treated at the earliest sign (often itching and the appearance of urticarial plaques, restlessness, nausea, tachycardia or tachypnoea) with adrenaline by intramuscular injection19,86,87,119–123.
Additional supportive treatment. Organ and system failures caused by envenoming must be detected and treated (Table 2). Supraglottal or endotracheal intubation and assisted ventilation either manually or by a ventilator are vital in cases of bulbar and respiratory paralysis87. Especially in individuals bitten by species with predominantly postsynaptic neurotoxins, such as cobras (Naja spp.; family Elapidae), death adders (Acanthophis spp.; family Elapidae) and some coral snakes (Micrurus spp.; family Elapidae), acetylcholinesterase inhibitors such as neostigmine, given with atropine, may improve neuromuscular transmission at least temporarily, but they are no substitute for antivenom111,125. Short-acting acetylcholinesterase inhibitors (for example, edrophonium) or the ice pack test (whereby an ice pack is applied to one upper eyelid in a patient with bilateral ptosis; the ice lowers the local temperature and inhibits endogenous acetylcholinesterase) may be used to predict response to acetylcholinesterase inhibition126. Hypotension and shock that persist after antivenom treatment are treated with cautious fluid volume repletion and vasopressor drugs, such as dopamine19,20. If acute kidney injury progresses despite conservative management, renal replacement therapy is needed.
A tetanus toxoid booster should be given in all cases, but prophylactic antibiotics are not justified127. However, if the wound has been incised or there are signs of tissue necrosis, wound infection or local abscess formation, a broad-spectrum antibiotic should be given. Surgical debridement (removal of necrotic tissue) and skin grafting may be needed in some cases, and some gangrenous digits or limbs might require amputation. Painful, tender, tensely swollen, cold, cyanosed and apparently pulseless snake-bitten limbs often appear to fulfil criteria for compartment syndrome (for example, anterior tibial compartment), tempting surgeons to undertake fasciotomy (a surgical procedure to improve circulation by incising fascial compartments). Fasciotomy is rarely justified, as intra-compartmental pressure usually remains within normal limits, and fasciotomy in patients whose anti-haemostasis has not been corrected by adequate doses of antivenom, has proved to be catastrophic. Unnecessary fasciotomy prolongs hospital stay and contributes to long-term morbidity128.
Rehabilitation is a rare luxury for individuals with snakebite envenoming but is essential for helping to restore function to the bitten limb, especially in children and agricultural workers, and to ameliorate the chronic physical handicap that blights the lives of many survivors of snakebite.
Hospital discharge and follow-up
No patient with snakebite envenoming should be discharged back to the same environment where the bite occurred without receiving practical advice, preferably in the form of a printed leaflet, about reducing the risk of further bites. At follow-up, patients should be checked for late antivenom-related serum sickness 5–15 days after treatment, and chronic physical and psychological sequelae of envenoming13,26. Persisting sequelae after snakebite are common, and include tissue loss, amputations, contractures, arthrodeses (fusion fixation of a joint), septic arthritis, hypertrophic and keloid scars, tendon damage, complications of fasciotomies, chronic skin ulcers and osteomyelitis leading to malignant Marjolin ulcers2,19,24,25,78,82,129. Visible and functional defects may lead to social stigmatization. Acute kidney injury may lead to chronic renal failure and panhypopituitarism associated with Russell's viper (family Viperidae) envenoming and to arrested puberty, amenorrhoea and infertility. Persisting neurotoxic effects include mydriasis (pupil dilatation) and loss of olfaction. Cerebral haemorrhages or thromboses may result in chronic neurological deficits, and severe presynaptic neurotoxicity may lead to increased risk of developing late poliomyelitis-like syndrome. Many survivors of snakebite complain of chronic or recurrent symptoms in the bitten limb and attribute a wide variety of physical and mental problems to that frightening and traumatic life event.
Quality of life
Snakebite envenomings occur mostly in impoverished settings11, affecting underserved rural populations that, quite often, lack the appropriate resources to confront this neglected disease. Snakebite alters the lives of affected individuals, but also families and communities because this disease largely affects people who are devoted to agricultural or pastoralist activities whose economic performance, and that of their dependents, relies on their good health. The death or incapacitation of someone responsible for the basic economic support of their family can devastate the socioeconomic sustainability and interpersonal stability of many people. Moreover, where medical treatment and especially antivenom is not provided free of charge, the economic cost of snakebite envenoming can be catastrophic — creating debt, forcing asset liquidation and driving families further into the cycle of poverty130,131.
Although many snakebites are fatal, far more leave behind individuals who experience horrifically debilitating disfigurement and long-term disability. Without timely access to health systems with adequate resources and capacity, the consequences of snakebite envenoming may include various types of sequelae2,7,129,132,133. Where health systems falter, affected people as well as their families and communities must deal with these consequences on their own often with severe implications, often expanding the toll of a single snakebite envenoming.
This disease also exerts a heavy psychological impact, a phenomenon that has not received attention in medical research until recently26,134. The lack of effective diagnosis and treatment, and poor follow-up of affected people, even when they are treated in health facilities, often results in psychological consequences that remain untreated, thus affecting patients and their families in ways that go beyond the biomedical aspects of the disease26,134.
Rolling back the impact of snakebite envenoming on quality of life requires concerted and coordinated efforts spanning entire health systems. Snakebite envenoming is a ‘tool-ready’ disease, in the sense that effective therapy (that is, antivenom) exists, and other cross-cutting interventions are available to strengthen health systems, empower communities and encourage policy change. Implementing an effective action plan to control snakebite envenoming will lead to a situation in which individuals with snakebite envenoming are treated rapidly with safe, effective antivenoms by trained health staff, followed up with ancillary treatment, together with psychological, social and economic support after hospital discharge. Public and private organizations, as well as community-based groups, should follow-up and help people affected by sequelae of snakebites.
Outlook
Diagnosis
Deciding on when to start antivenom treatment can be difficult, particularly for inexperienced physicians, as signs of envenoming and their time of onset vary by snake genus, the amount of venom injected, the site of injection and the age and health of the individual. In addition, current clinical guidelines recommend withholding antivenom administration until symptoms of systemic envenoming are detected in patients with a snakebite. Rapid, affordable, point-of-care (bedside) diagnostic kits providing physicians in rural hospitals with information that enables earlier detection of envenoming and identification of the biting species are urgently needed, to enable earlier treatment with antivenom and anticipate likely clinical course and the need for supportive therapy (for example, ventilation).
An experimental lateral flow assay has been developed to identify envenoming by two Indian snakes; the assay uses antivenom to detect circulating venom proteins135. The near-ubiquitous delivery of PLA2s into the circulation of patients with a snakebite has been suggested as a marker to detect systemic envenoming136. A PCR-based approach has also been reported as a possible diagnostic tool for the detection of venom DNA in and around the bite site, but may be less useful for the detection of circulating venom as a marker of systemic envenoming105.
Management of systemic effects
Antivenom has been the primary treatment of systemic snake envenoming for more than a century. Although life-saving, antivenoms still have therapeutic limitations137,138. Conventional antivenom has limited efficacy against some effects of envenoming, such as local tissue damage, and, when treatment is delayed, presynaptic neurotoxicity. In addition, only 10–15% of IgG in a vial of antivenom is specific against venom proteins, because the animals used for antivenom manufacture already have mature immune systems, and hyper-immunization with venom is unable to engender higher antivenom IgG titres. Finally, the greater the phylogenetic disparity between the snakes whose venoms are used for immunization, the greater the number of IgG specificities generated in the venom-immunized animals. This means that the proportion of total IgG targeting the venom of any one snake is small and, consequently, more vials are needed to achieve clinical cure. This creates a therapeutic paradox because each extra vial of antivenom increases both the risk of potentially severe antivenom-induced adverse effects and the treatment cost. Thus, highly efficient, polyspecific antivenoms are needed, without compromising safety and affordability.
The rapidly increasing availability of snake venom proteomes and venom gland transcriptomes40 have provided essential information on venom protein composition and inferred function of the proteins. Together with DNA sequence information, this allows guided development of IgGs that target only the most toxic venom proteins. One approach is to analyse transcriptome data of each toxin group expressed in snake venom (of several snake families) to identify antigenic sequence motifs (epitopes) with the greatest cross-species and cross-genera sequence conservation. These motifs are manipulated to construct a synthetic epitope string that is designed to generate multiple distinct antitoxin group IgGs that are capable of neutralizing the entire toxin group, irrespective of the snake species139–141. The intent is to pool all the antitoxin group IgGs to create a polyspecific antivenom. To address the dose–efficacy challenge, the current intent is to isolate B cells from epitope string-immunized mice, and extract, screen and manipulate the genes encoding IgG to prepare monoclonal antibodies to produce an antivenom that consists only of IgG capable of polyspecifically neutralizing the toxins present in the venoms of a defined group of snakes. Humanizing these monoclonal antibodies also offers substantial safety advantages over the current antivenoms142.
Another toxin-specific recombinant antivenom approach is based on screening bespoke high-density toxin-specific microarrays to identify the most medically important venom toxin epitopes bound by clinically effective antivenoms143. This toxin-focused approach aligned with the production of human or humanized IgGs (or fractions thereof, for example, single-chain variable fragments) using new biotechnology production processes may yield antivenoms at a cost similar to conventional antivenoms in the future144, but with a much-improved dose–efficacy. International collaborative efforts are underway in these research and development topics.
Chemical inhibitor research is also being promoted142 as an overlooked but potential source of drugs to treat snakebites. For example, a recent drug repurposing study identified a potent, broad-spectrum inhibitor of the nearly ubiquitous group of venom PLA2s145. Other efforts have been directed at the synthesis of nanoparticles, which could sequester and neutralize venom toxins146. If successful, the chemical outputs of these approaches will probably have substantial cost and logistical advantages.
A pharmacological study reported that an ointment containing a nitric oxide donor, which impedes the intrinsic lymphatic pump, delays ingress of venom proteins into the systemic circulation and improved survival of venom-injected rats147. This first aid-focused application is likely to be effective in predominantly neurotoxic venoms, but needs to be excluded for envenoming by those snakes that cause local tissue destruction.
Management of local effects
No medicinal treatment of the local tissue-destructive effects of envenoming exists. Antivenom, unless it is administered very soon after the bite, is largely ineffective in preventing the rapidly manifested destruction of tissues by venom. Chemical inhibitors have potential as a treatment for venom-induced local tissue destruction owing to their low-cost, thermostability and rapid tissue-distribution dynamics. Small peptidomimetic matrix metalloproteinase inhibitors have shown an exciting, repurposed, ability to neutralize SVMP-induced haemorrhage and dermonecrosis148. It is not inconceivable that combining inhibitors of SVMPs and PLA2s145,146,148,149 may produce an affordable, rapid, polyspecific and effective treatment of venom-induced tissue damage.
Other groups are pursuing a recombinant approach using single-domain VHH antibody fragments, based on the demonstration that an experimental VHH antivenom (prepared from the heavy-chain-only IgG3 fraction of a venom-immunized dromedary camel) proved the most dose-effective antivenom137 to neutralize various toxic effects of the West African saw-scaled viper (Echis ocellatus). The small size, thermostability and toxin specificity of camelid VHH promote them as exciting therapeutic candidates for preventing the tissue necrotic effects of envenoming for saw-scaled vipers and other snake venoms150,151.
Treatment availability and accessibility
There is a current crisis in antivenom provision in various regions, particularly in sub-Saharan Africa and parts of Asia10,138,152. The market failure of two of the most effective polyspecific antivenoms manufactured for Africa in the past118,138,153 underscores the fact that the commercial constraints associated with marketing non-subsidised conventional antivenoms of high cost but limited demand can result in important, life-threatening therapeutic gaps. It also reinforces the need to incorporate commercial realities into the design of innovative approaches to generate antivenoms with improved polyspecific efficacy, safety and affordability137,138. The research and development tasks discussed above need to be complemented by research and innovation in the public health realm that aim to improve the availability and accessibility of antivenoms, which demands research in subjects such as health economics, provision of health services and other social science-related topics. The long-term goal is to develop knowledge-based policies that ensure that people with snakebite envenomings receive safe and effective antivenoms in a timely manner. New treatments should be affordable and available to invariably remote, impoverished people with snakebite envenoming138,152. In addition, innovative treatment and diagnostic research need to be complemented with equally important research on ways to ensure that affected communities can better access effective health care, and indeed, on affordable and appropriate means by which they can reduce their likelihood of being bitten.
Venoms as a source of therapy
Some of the targets of toxins are also dysregulated in several human diseases, such as thrombotic disorders, vascular pathologies, neurodegenerative diseases and inflammatory conditions. The study of venom toxins is of growing interest for the pharmacological and biotechnological communities, as venoms are increasingly recognized as a rich source for lead compounds that can drive the development of biotechnological tools and novel biotherapeutics154. Determining the molecular landscape of snake venoms represents the necessary first step towards these basic and applied goals.
Global efforts to reduce impact
For decades, a major barrier to effectively reducing the worldwide impact of snakebite envenoming has been the lack of inclusion of this disease on the global public health agenda. The absence of appropriate prioritization and resourcing of control efforts has seen snakebite envenoming fail to receive proper attention by international and national health authorities, fall off research priority lists and public health agendas, and lose the interest of the pharmaceutical industry. However, the decision of the WHO in June 2017 to include snakebite envenoming in the category A of its list of neglected tropical diseases is an important step forward in the global struggle to reduce the impact of this pathology. This is an essential advance that is necessary to raise the profile of snakebite envenoming in the eyes of Member State governments, donors and other stakeholders, and to empower the WHO to provide the leadership that is needed to coordinate a global control programme. The WHO is the only organization with the political, operational and policy reach to drive an integrated global strategy involving multiple actors and a multidisciplinary approach, and this is clearly demonstrated by the success in combating other neglected tropical diseases in the 5 years since the London Declaration on Neglected Tropical Diseases was enacted to support the WHO's 2020 Roadmap.
Inclusion of snakebite envenoming in the neglected tropical disease category will focus attention on concentrated efforts to control snakebite through an integrated and intensified disease management strategy that incorporates several key elements, ranging from prevention to improved primary treatment, rehabilitation and investment in research that can unlock new diagnostic and therapeutic pathways, or enhance surveillance and resource deployment (Box 4).
The tasks included in Box 4 are by no means an exhaustive list of all the potential interventions available to immediately bring the burden of snakebite envenoming under effective control, but it exemplifies the key approaches that will drive the process. Broadening the current focus — which revolves around improving treatment with antivenoms, measuring burden and assessing the consequences to include a more holistic strategy that begins with community engagement, empowerment and education, and spans all stages of treatment, reporting, therapeutic and diagnostic translational research, policy, training and research — is essential.
This integrated plan for controlling snakebite envenoming requires international cooperation, through a multitiered roadmap coordinated by the WHO, involving many stakeholders: the scientific and technological research community; antivenom manufacturers and developers; health regulatory agencies; public health authorities at national, regional and global levels, and health professional organizations; international health foundations and advocacy groups, such as the Global Snakebite Initiative10, Health Action International and others; and organizations of the civil society in countries with a high incidence of snakebites. Realizing such a comprehensive strategy requires considerable resources, and it is incumbent on United Nation Member States, donor organizations, snakebite experts and other actors to wholeheartedly support the leadership of the WHO as it moves forward to implement effective control programmes and projects.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
How to cite this article
Gutiérrez, J. M. et al. Snakebite envenoming. Nat. Rev. Dis. Primers 3, 17063 (2017).
References
Calvete, J. J. Proteomic tools against the neglected pathology of snake bite envenoming. Expert Rev. Proteomics 8, 739–758 (2011).
Warrell, D. A. Snake bite. Lancet 375, 77–88 (2010).
Pyron, R., Burbrink, F. T. & Wiens, J. J. A phylogeny and revised classification of Squamata, including 4161 species of lizards and snakes. BMC Evol. Biol. 13, 93 (2013).
Hsiang, A. Y. et al. The origin of snakes: revealing the ecology, behavior, and evolutionary history of early snakes using genomics, phenomics, and the fossil record. BMC Evol. Biol. 15, 87 (2015).
Chippaux, J. P. Snake-bites: appraisal of the global situation. Bull. World Health Organ. 76, 515–524 (1998).
Mohapatra, B. et al. Snakebite mortality in India: a nationally representative mortality survey. PLoS Negl. Trop. Dis. 5, e1018 (2011). This nationally representative study of snakebite mortality in India demonstrates that the magnitude of the problem in terms of mortality is much higher than previously thought.
Chippaux, J.-P. Estimate of the burden of snakebites in sub-Saharan Africa: a meta-analytic approach. Toxicon 57, 586–599 (2011).
Gutiérrez, J. M., Williams, D., Fan, H. W. & Warrell, D. A. Snakebite envenoming from a global perspective: towards an integrated approach. Toxicon 56, 1223–1235 (2010).
Gutiérrez, J. M., Theakston, R. D. G. & Warrell, D. A. Confronting the neglected problem of snake bite envenoming: the need for a global partnership. PLoS Med. 3, e150 (2006).
Williams, D. et al. The Global Snake Bite Initiative: an antidote for snake bite. Lancet 375, 89–91 (2010). This paper describes the launch of the first organization to confront snakebite envenoming from a global perspective.
Harrison, R. A., Hargreaves, A., Wagstaff, S. C., Faragher, B. & Lalloo, D. G. Snake envenoming: a disease of poverty. PLoS Negl. Trop. Dis. 3, e569 (2009). This study highlights the relationship between snakebite envenoming and poverty.
Kasturiratne, A. et al. The global burden of snakebite a literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 5, e218 (2008).
Habib, A. G. et al. Snakebite is under appreciated: appraisal of burden from West Africa. PLoS Negl. Trop. Dis. 9, e0004088 (2015). This study analyses the impact of snakebite envenoming in terms of disability-adjusted life years in 16 countries in West Africa.
Alirol, E., Sharma, S. K., Bawaskar, H. S., Kuch, U. & Chappuis, F. Snake bite in South Asia: a review. PLoS Negl. Trop. Dis. 4, e603 (2010).
Sankar, J., Nabeel, R., Sankar, M. J., Priyambada, L. & Mahadevan, S. Factors affecting outcome in children with snake envenomation: a prospective observational study. Arch. Dis. Child. 98, 596–601 (2013).
Stahel, E. Epidemiological aspects of snake bites on a Liberian rubber plantation. Acta Trop. 37, 367–374 (1980).
Warrell, D. A. et al. Randomized comparative trial of three monospecific antivenoms for bites by the Malayan pit viper (Calloselasma rhodostoma) in southern Thailand: clinical and laboratory correlations. Am. J. Trop. Med. Hyg. 35, 1235–1247 (1986).
Pierini, S. V., Warrell, D. A., De Paulo, A. & Theakston, R. D. G. High incidence of bites and stings by snakes and other animals among rubber tappers and Amazonian Indians of the Juruá Valley, Acre State, Brazil. Toxicon 34, 225–236 (1996).
Warrell, D. A. in Venomous Reptiles of the Western Hemisphere (eds Campbell, J. R. & Lamar, W. W. ) 709–761 (Cornell Univ. Press, 2004).
Myint-Lwin et al. Bites by Russell's viper (Vipera russelli siamensis) in Burma: haemostatic, vascular, and renal disturbances and response to treatment. Lancet 326, 1259–1264 (1985).
Habib, A. G. et al. Envenoming after carpet viper (Echis ocellatus) bite during pregnancy: timely use of effective antivenom improves maternal and foetal outcomes. Trop. Med. Int. Health 13, 1172–1175 (2008).
Warrell, D. A. & Arnett, C. The importance of bites by the saw-scaled or carpet viper (Echis carinatus): epidemiological studies in Nigeria and a review of the world literature. Acta Trop. 33, 307–341 (1976).
Williams, D., Jensen, S., Nimorakiotakis, B. & Winkel, K. D. Venomous Bites and Stings in Papua New Guinea (Australian Venom Research Unit, 2005).
Warrell, D. A. & Ormerod, L. D. Snake venom ophthalmia and blindness caused by the spitting cobra (Naja nigricollis) in Nigeria. Am. J. Trop. Med. Hyg. 25, 525–529 (1976).
Smith, J. et al. Malignancy in chronic ulcers and scars of the leg (Marjolin's ulcer): a study of 21 patients. Skeletal Radiol. 30, 331–337 (2001).
Williams, S. S. et al. Delayed psychological morbidity associated with snakebite envenoming. PLoS Negl. Trop. Dis. 5, e1255 (2011).
GBD 2013 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990–2013: quantifying the epidemiological transition. Lancet 386, 2145–2191 (2015).
Fry, B. G. et al. The toxicogenomic multiverse: convergent recruitment of proteins into animal venoms. Annu. Rev. Genomics Hum. Genet. 10, 483–511 (2009).
Daltry, J. C., Wüster, W. & Thorpe, R. S. Diet and snake venom evolution. Nature 379, 537–540 (1996).
Chippaux, J.-P., Williams, V. & White, J. Snake venom variability: methods of study, results and interpretation. Toxicon 29, 1279–1303 (1991).
Olivera, B. M. Conus peptides: biodiversity-based discovery and exogenomics. J. Biol. Chem. 281, 31173–31177 (2006).
Durban, J. et al. Integrated ‘omics’ profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus simus. BMC Genomics 14, 234 (2013).
Casewell, N. R. et al. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc. Natl Acad. Sci. USA 111, 9205–9210 (2014).
Sanz, L. & Calvete, J. Insights into the evolution of a snake venom multi-gene family from the genomic organization of Echis ocellatus SVMP genes. Toxins (Basel) 8, E216 (2016).
Dowell, N. L. et al. The deep origin and recent loss of venom toxin genes in rattlesnakes. Curr. Biol. 26, 2434–2445 (2016).
Calvete, J. J. Venomics: integrative venom proteomics and beyond. Biochem. J. 474, 611–634 (2017). A comprehensive review of the complexity of snake venoms and the biological and medical implications of this complexity.
Reeks, T. A., Fry, B. G. & Alewood, P. F. Privileged frameworks from snake venom. Cell. Mol. Life Sci. 72, 1939–1958 (2015).
Doley, R. & Kini, R. M. Protein complexes in snake venom. Cell. Mol. Life Sci. 66, 2851–2871 (2009).
Vonk, F. J. et al. The king cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc. Natl Acad. Sci. USA 110, 20651–20656 (2013).
Calvete, J. J. Snake venomics: from the inventory of toxins to biology. Toxicon 75, 44–62 (2013).
Rokyta, D. R., Wray, K. P., McGivern, J. J. & Margres, M. J. The transcriptomic and proteomic basis for the evolution of a novel venom phenotype within the timber rattlesnake (Crotalus horridus). Toxicon 98, 34–48 (2015).
Brahma, R. K., McCleary, R. J. R., Kini, R. M. & Doley, R. Venom gland transcriptomics for identifying, cataloging, and characterizing venom proteins in snakes. Toxicon 93, 1–10 (2015).
Pla, D., Gutiérrez, J. M. & Calvete, J. J. Second generation antivenomics: comparing immunoaffinity and immunodepletion protocols. Toxicon 60, 213–214 (2012).
Gutiérrez, J. M. et al. Assessing the preclinical efficacy of antivenoms: from the lethality neutralization assay to antivenomics. Toxicon 69, 168–179 (2013).
Dixon, R. W. & Harris, J. B. Myotoxic activity of the toxic phospholipase, notexin, from the venom of the Australian tiger snake. J. Neuropathol. Exp. Neurol. 55, 1230–1237 (1996).
Montecucco, C., Gutiérrez, J. M. & Lomonte, B. Cellular pathology induced by snake venom phospholipase A2 myotoxins and neurotoxins: common aspects of their mechanisms of action. Cell. Mol. Life Sci. 65, 2897–2912 (2008). This paper reviews the mechanisms by which venom PLA2s induce myotoxicity and neurotoxicity.
Gutiérrez, J. M. & Ownby, C. L. Skeletal muscle degeneration induced by venom phospholipases A2: insights into the mechanisms of local and systemic myotoxicity. Toxicon 42, 915–931 (2003).
Gutiérrez, J. M., Rucavado, A., Chaves, F., Díaz, C. & Escalante, T. Experimental pathology of local tissue damage induced by Bothrops asper snake venom. Toxicon 54, 958–975 (2009).
Hernández, R. et al. Poor regenerative outcome after skeletal muscle necrosis induced by Bothrops asper venom: alterations in microvasculature and nerves. PLoS ONE 6, e19834 (2011).
Escalante, T., Rucavado, A., Fox, J. W. & Gutiérrez, J. M. Key events in microvascular damage induced by snake venom hemorrhagic metalloproteinases. J. Proteomics 74, 1781–1794 (2011). This review summarizes the mechanisms by which zinc-dependent SVMPs induce microvascular damage and haemorrhage.
Seo, T. et al. Haemorrhagic snake venom metalloproteases and human ADAMs cleave LRP5/6, which disrupts cell–cell adhesions in vitro and induces haemorrhage in vivo. FEBS J. 284, 1657–1671 (2017).
Gutiérrez, J., Escalante, T., Rucavado, A., Herrera, C. & Fox, J. A comprehensive view of the structural and functional alterations of extracellular matrix by snake venom metalloproteinases (SVMPs): novel perspectives on the pathophysiology of envenoming. Toxins (Basel) 8, 304 (2016).
Jiménez, N., Escalante, T., Gutiérrez, J. M. & Rucavado, A. Skin pathology induced by snake venom metalloproteinase: acute damage, revascularization, and re-epithelization in a mouse ear model. J. Invest. Dermatol. 128, 2421–2428 (2008).
Rivel, M. et al. Pathogenesis of dermonecrosis induced by venom of the spitting cobra. Naja nigricollis: an experimental study in mice. Toxicon 119, 171–179 (2016).
Dubovskii, P. V. & Utkin, Y. N. Cobra cytotoxins: structural organization and antibacterial activity. Acta Naturae 6, 11–18 (2014).
Mora, J., Mora, R., Lomonte, B. & Gutiérrez, J. M. Effects of Bothrops asper snake venom on lymphatic vessels: insights into a hidden aspect of envenomation. PLoS Negl. Trop. Dis. 2, e318 (2008).
Teixeira, C., Cury, Y., Moreira, V., Picolo, G. & Chaves, F. Inflammation induced by Bothrops asper venom. Toxicon 54, 988–997 (2009).
Rucavado, A. et al. Viperid envenomation wound exudate contributes to increased vascular permeability via a DAMPs/TLR-4 mediated pathway. Toxins (Basel) 8, 349 (2016).
Zhang, C., Medzihradszky, K. F., Sánchez, E. E., Basbaum, A. I. & Julius, D. Lys49 myotoxin from the Brazilian lancehead pit viper elicits pain through regulated ATP release. Proc. Natl Acad. Sci. USA 114, E2524–E2532 (2017).
Barber, C. M., Isbister, G. K. & Hodgson, W. C. Alpha neurotoxins. Toxicon 66, 47–58 (2013).
Rossetto, O. & Montecucco, C. Presynaptic neurotoxins with enzymatic activities. Handb. Exp. Pharmacol. 184, 129–170 (2008).
Pungercar, J. & Križaj, I. Understanding the molecular mechanism underlying the presynaptic toxicity of secreted phospholipases A2. Toxicon 50, 871–892 (2007).
Paoli, M. et al. Mass spectrometry analysis of the phospholipase A2 activity of snake pre-synaptic neurotoxins in cultured neurons. J. Neurochem. 111, 737–744 (2009).
Harris, J. B., Grubb, B. D., Maltin, C. A. & Dixon, R. The neurotoxicity of the venom phospholipases A2, notexin and taipoxin. Exp. Neurol. 161, 517–526 (2000).
Prasarnpun, S., Walsh, J. & Harris, J. B. β-Bungarotoxin-induced depletion of synaptic vesicles at the mammalian neuromuscular junction. Neuropharmacology 47, 304–314 (2004).
Rigoni, M. et al. Snake phospholipase A2 neurotoxins enter neurons, bind specifically to mitochondria, and open their transition pores. J. Biol. Chem. 283, 34013–34020 (2008).
Harvey, A. & Robertson, B. Dendrotoxins: structure–activity relationships and effects on potassium ion channels. Curr. Med. Chem. 11, 3065–3072 (2004).
Harvey, A. L. in Handbook of Venoms and Toxins of Reptiles (ed. Mackessy, S. P. ) 317–324 (CRC Press, 2010).
Fox, J. W. & Serrano, S. M. T. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon 45, 969–985 (2005).
White, J. Snake venoms and coagulopathy. Toxicon 45, 951–967 (2005).
Del Brutto, O. H. & Del Brutto, V. J. Neurological complications of venomous snake bites: a review. Acta Neurol. Scand. 125, 363–372 (2011).
Kini, R. M. The intriguing world of prothrombin activators from snake venom. Toxicon 45, 1133–1145 (2005).
Kini, R. & Koh, C. Metalloproteases affecting blood coagulation, fibrinolysis and platelet aggregation from snake venoms: definition and nomenclature of interaction sites. Toxins (Basel) 8, 284 (2016).
Yamashita, K. M., Alves, A. F., Barbaro, K. C. & Santoro, M. L. Bothrops jararaca venom metalloproteinases are essential for coagulopathy and increase plasma tissue factor levels during envenomation. PLoS Negl. Trop. Dis. 8, e2814 (2014).
Rucavado, A. et al. Thrombocytopenia and platelet hypoaggregation induced by Bothrops asper snake venom: toxins involved and their contribution to metalloproteinase-induced pulmonary hemorrhage. Thromb. Haemost. 94, 123–131 (2005).
Calvete, J. J. et al. Snake venom disintegrins: evolution of structure and function. Toxicon 45, 1063–1074 (2005).
Du, X. Y. & Clemetson, K. J. in Handbook of Venoms and Toxins of Reptiles (ed. Mackessy, S. P. ) 359–375 (CRC Press, 2010).
Resiere, D., Mégarbane, B., Valentino, R., Mehdaoui, H. & Thomas, L. Bothrops lanceolatus bites: guidelines for severity assessment and emergent management. Toxins (Basel) 2, 163–173 (2010).
Than-Than et al. Contribution of focal haemorrhage and microvascular fibrin deposition to fatal envenoming by Russell's viper (Vipera russelli siamensis) in Burma. Acta Trop. 46, 23–38 (1989).
Hayashi, M. A. F. & Camargo, A. C. M. The bradykinin-potentiating peptides from venom gland and brain of Bothrops jararaca contain highly site specific inhibitors of the somatic angiotensin-converting enzyme. Toxicon 45, 1163–1170 (2005).
Höjer, J., Tran Hung, H. & Warrell, D. Life-threatening hyponatremia after krait bite envenoming — a new syndrome. Clin. Toxicol. 48, 956–957 (2010).
Sitprija, V. & Sitprija, S. Renal effects and injury induced by animal toxins. Toxicon 60, 943–953 (2012).
Pinho, F. M. O., Zanetta, D. M. T. & Burdmann, E. A. Acute renal failure after Crotalus durissus snakebite: a prospective survey on 100 patients. Kidney Int. 67, 659–667 (2005).
Warrell, D. A. in Venomous Snakes. Ecology, Evolution and Snakebite (eds Thorpe, R. S., Wuster, W. & Malhotra, A. ) 189–203 (Clarendon Press, 1997).
Harris, J. B. et al. Snake bite in Chittagong division, Bangladesh: a study of bitten patients who developed no signs of systemic envenoming. Trans. R. Soc. Trop. Med. Hyg. 104, 320–327 (2010).
WHO Regional Office for Africa. Guidelines for the prevention and clinical management of snakebite in Africa. WHOhttp://apps.who.int/medicinedocs/documents/s17810en/s17810en.pdf (2010). These are the WHO guidelines for snakebite envenoming in Africa, which are to be used in the training of health staff on the correct diagnosis and management of envenomings.
WHO Regional Office for South-East Asia. Guidelines for the management of snakebites. WHOhttp://apps.searo.who.int/PDS_DOCS/B5255.pdf?ua=1 (2016).
Sutherland, S. K. & Tibballs, J. Australian Animal Toxins: The Creatures, their Toxins and Care of the Poisoned Patient2nd edn (Oxford Univ. Press, 2001).
White, J. A Clinician's Guide to Australian Venomous Bites and Stings (bioCSL, 2013).
Weinstein, S., Warrell, D. A., White, J. & Keyler, D. ‘Venomous’ Bites from Non-Venomous Snakes: A Critical Analysis of Risk and Management of ‘Colubrid’ Snake Bites (Elsevier, 2011).
Weinstein, S. A., White, J., Keyler, D. E. & Warrell, D. A. Non-front-fanged colubroid snakes: a current evidence-based analysis of medical significance. Toxicon 69, 103–113 (2013).
Warrell, D. A. et al. Severe neurotoxic envenoming by the Malayan krait Bungarus candidus (Linnaeus): response to antivenom and anticholinesterase. BMJ 286, 678–680 (1983).
Ariaratnam, C. A., Sheriff, M. H. R., Theakston, R. D. G. & Warrell, D. A. Distinctive epidemiologic and clinical features of common krait (Bungarus caeruleus) bites in Sri Lanka. Am. J. Trop. Med. Hyg. 79, 458–462 (2008).
Russell, F. E. Snake Venom Poisoning (JB Lippincott, 1980).
Ariaratnam, C. A., Sheriff, M. H. R., Arambepola, C., Theakston, R. D. G. & Warrell, D. A. Syndromic approach to treatment of snake bite in Sri Lanka based on results of a prospective national hospital-based survey of patients envenomed by identified snakes. Am. J. Trop. Med. Hyg. 81, 725–731 (2009).
Warrell, D. A. et al. Poisoning by bites of the saw-scaled or carpet viper (Echis carinatus) in Nigeria. QJ Med. 46, 33–62 (1977).
Sano-Martins, I. S. et al. Reliability of the simple 20 minute whole blood clotting test (WBCT20) as an indicator of low plasma fibrinogen concentration in patients envenomed by Bothrops snakes. Toxicon 32, 1045–1050 (1994).
Wood, D., Sartorius, B. & Hift, R. Ultrasound findings in 42 patients with cytotoxic tissue damage following bites by South African snakes. Emerg. Med. J. 33, 477–481 (2016).
Theakston, R. & Laing, G. Diagnosis of snakebite and the importance of immunological tests in venom research. Toxins (Basel) 6, 1667–1695 (2014).
Ho, M., Warrell, M. J., Warrell, D. A., Bidwell, D. & Voller, A. A critical reappraisal of the use of enzyme-linked immunosorbent assays in the study of snake bite. Toxicon 24, 211–221 (1986).
Dong, L. Immunogenicity of venoms from four common snakes in the south of Vietnam and development of ELISA kit for venom detection. J. Immunol. Methods 282, 13–31 (2003).
Kulawickrama, S. et al. Development of a sensitive enzyme immunoassay for measuring taipan venom in serum. Toxicon 55, 1510–1518 (2010).
Sutherland, S. K. Rapid venom identification: availability of kits. Med. J. Aust. 2, 602–603 (1979).
Chen, T. et al. Unmasking venom gland transcriptomes in reptile venoms. Anal. Biochem. 311, 152–156 (2002).
Sharma, S. K. et al. Use of molecular diagnostic tools for the identification of species responsible for snakebite in Nepal: a pilot study. PLoS Negl. Trop. Dis. 10, e0004620 (2016).
Tun-Pe et al. in Management of Snakebite and Research (ed. WHO) 7–11 (WHO, 2002).
Chappuis, F., Sharma, S. K., Jha, N., Loutan, L. & Bovier, P. A. Protection against snake bites by sleeping under a bed net in southeastern Nepal. Am. J. Trop. Med. Hyg. 77, 197–199 (2007).
Sharma, S. K. et al. Effectiveness of rapid transport of victims and community health education on snake bite fatalities in rural Nepal. Am. J. Trop. Med. Hyg. 89, 145–150 (2013). This study describes a successful intervention at the community level aimed at improving the access of patients with a snakebite to health facilities.
Tun-Pe, Aye-Aye-Myint, Khin-Ei-Han, Thi-Ha & Tin-Nu-Swe . Local compression pads as a first-aid measure for victims of bites by Russell's viper (Daboia russelii siamensis) in Myanmar. Trans. R. Soc. Trop. Med. Hyg. 89, 293–295 (1995).
Avau, B., Borra, V., Vandekerckhove, P. & De Buck, E. The treatment of snake bites in a first aid setting: a systematic review. PLoS Negl. Trop. Dis. 10, e0005079 (2016).
Faiz, M. A. et al. Bites by the monocled cobra, Naja kaouthia, in Chittagong division, Bangladesh: epidemiology, clinical features of envenoming and management of 70 identified cases. Am. J. Trop. Med. Hyg. 96, 876–884 (2017).
Watt, G., Theakston, R. D. G., Padre, L., Laughlin, L. W. & Tuazon, M. L. Tourniquet application after cobra bite: delay in the onset of neurotoxicity and the dangers of sudden release. Am. J. Trop. Med. Hyg. 38, 618–622 (1988).
Audebert, F., Sorkine, M. & Bon, C. Envenoming by viper bites in France: clinical gradation and biological quantification by ELISA. Toxicon 30, 599–609 (1992).
Bucher, B. et al. Clinical indicators of envenoming and serum levels of venom antigens in patients bitten by Bothrops lanceolatus in Martinique. Trans. R. Soc. Trop. Med. Hyg. 91, 186–190 (1997).
World Health Organization. Guidelines for the production, control and regulation of snake antivenom immunoglobulins. WHOhttp://www.who.int/bloodproducts/snake_antivenoms/en (2010). These guidelines provide a detailed account on the main aspects related to the production and control of antivenoms, and contain valuable information for manufacturers, researchers and regulatory agencies.
Gutiérrez, J. M., León, G. & Lomonte, B. Pharmacokinetic–pharmacodynamic relationships of immunoglobulin therapy for envenomation. Clin. Pharmacokinet. 42, 721–741 (2003).
Habib, A. G. & Warrell, D. A. Antivenom therapy of carpet viper (Echis ocellatus) envenoming: effectiveness and strategies for delivery in West Africa. Toxicon 69, 82–89 (2013).
Theakston, R. D. G. & Warrell, D. A. Crisis in snake antivenom supply for Africa. Lancet 356, 2104 (2000).
Warrell, D. A. et al. Bites by the saw-scaled or carpet viper (Echis carinatus): trial of two specific antivenoms. Br. Med. J. 4, 437–440 (1974).
Cardoso, J. L. et al. Randomized comparative trial of three antivenoms in the treatment of envenoming by lance-headed vipers (Bothrops jararaca) in São Paulo, Brazil. QJ Med. 86, 315–325 (1993).
Smalligan, R. Crotaline snake bite in the Ecuadorian Amazon: randomised double blind comparative trial of three South American polyspecific antivenoms. BMJ 329, 1120–1129 (2004).
Abubakar, S. B. et al. Pre-clinical and preliminary dose-finding and safety studies to identify candidate antivenoms for treatment of envenoming by saw-scaled or carpet vipers (Echis ocellatus) in northern Nigeria. Toxicon 55, 719–723 (2010).
Abubakar, I. S. et al. Randomised controlled double-blind non-inferiority trial of two antivenoms for saw-scaled or carpet viper (Echis ocellatus) envenoming in Nigeria. PLoS Negl. Trop. Dis. 4, e767 (2010). An example of a randomized controlled, double-blind clinical trial for the evaluation of efficacy and safety of antivenoms in the treatment of snakebite envenomings.
de Silva, H. A. et al. Low-dose adrenaline, promethazine, and hydrocortisone in the prevention of acute adverse reactions to antivenom following snakebite: a randomised, double-blind, placebo-controlled trial. PLoS Med. 8, e1000435 (2011).
Watt, G. et al. Positive response to edrophonium in patients with neurotoxic envenoming by cobras (Naja naja philippinensis). N. Engl. J. Med. 315, 1444–1448 (1986).
Golnik, K. C., Pena, R., Lee, A. G. & Eggenberger, E. R. An ice test for the diagnosis of myasthenia gravis. Ophthalmology 106, 1282–1286 (1999).
Jorge, M. T. et al. Failure of chloramphenicol prophylaxis to reduce the frequency of abscess formation as a complication of envenoming by Bothrops snakes in Brazil: a double-blind randomized controlled trial. Trans. R. Soc. Trop. Med. Hyg. 98, 529–534 (2004).
Darracq, M. A., Cantrell, F. L., Klauk, B. & Thornton, S. L. A chance to cut is not always a chance to cure — fasciotomy in the treatment of rattlesnake envenomation: a retrospective poison center study. Toxicon 101, 23–26 (2015).
Jayawardana, S., Gnanathasan, A., Arambepola, C. & Chang, T. Chronic musculoskeletal disabilities following snake envenoming in Sri Lanka: a population-based study. PLoS Negl. Trop. Dis. 10, e0005103 (2016).
Hasan, S. M. K., Basher, A., Molla, A. A., Sultana, N. K. & Faiz, M. A. The impact of snake bite on household economy in Bangladesh. Trop. Doct. 42, 41–43 (2012).
Vaiyapuri, S. et al. Snakebite and its socio-economic impact on the rural population of Tamil Nadu, India. PLoS ONE 8, e80090 (2013). This paper describes the socioeconomic consequences of snakebites in an impoverished rural setting.
Aye, K.-P. et al. Clinical and laboratory parameters associated with acute kidney injury in patients with snakebite envenomation: a prospective observational study from Myanmar. BMC Nephrol. 18, 92 (2017).
Krishnamurthy, S., Gunasekaran, K., Mahadevan, S., Bobby, Z. & Kumar, A. P. Russell's viper envenomation-associated acute kidney injury in children in southern India. Indian Pediatr. 52, 583–586 (2015).
Muhammed, A. et al. Predictors of depression among patients receiving treatment for snakebite in General Hospital, Kaltungo, Gombe State, Nigeria: August 2015. Int. J. Ment. Health Syst. 11, 26 (2017).
Pawade, B. S. et al. Rapid and selective detection of experimental snake envenomation — use of gold nanoparticle based lateral flow assay. Toxicon 119, 299–306 (2016).
Maduwage, K., O’Leary, M. A. & Isbister, G. K. Diagnosis of snake envenomation using a simple phospholipase A2 assay. Sci. Rep. 4, 4827 (2014).
Harrison, R. A. et al. Research strategies to improve snakebite treatment: challenges and progress. J. Proteomics 74, 1768–1780 (2011). This review discusses some of the main research areas that need to be developed to generate novel diagnostic and therapeutic tools to confront snakebite envenoming.
Harrison, R. & Gutiérrez, J. Priority actions and progress to substantially and sustainably reduce the mortality, morbidity and socioeconomic burden of tropical snakebite. Toxins (Basel) 8, 351 (2016).
Wagstaff, S. C., Laing, G. D., Theakston, R. D. G., Papaspyridis, C. & Harrison, R. A. Bioinformatics and multiepitope DNA immunization to design rational snake antivenom. PLoS Med. 3, e184 (2006).
Casewell, N. R., Wüster, W., Vonk, F. J., Harrison, R. A. & Fry, B. G. Complex cocktails: the evolutionary novelty of venoms. Trends Ecol. Evol. 28, 219–229 (2013).
Ramos, H. R. et al. A heterologous multiepitope DNA prime/recombinant protein boost immunisation strategy for the development of an antiserum against Micrurus corallinus (coral snake) venom. PLoS Negl. Trop. Dis. 10, e0004484 (2016).
Laustsen, A. et al. From fangs to pharmacology: the future of snakebite envenoming therapy. Curr. Pharm. Des. 22, 5270–5293 (2016). A summary of novel therapeutic alternatives to approach snakebite envenomings, including recombinant antibodies and natural and synthetic venom inhibitors.
Engmark, M. et al. High-throughput immuno-profiling of mamba (Dendroaspis) venom toxin epitopes using high-density peptide microarrays. Sci. Rep. 6, 36629 (2016).
Laustsen, A. H., Johansen, K. H., Engmark, M. & Andersen, M. R. Recombinant snakebite antivenoms: a cost-competitive solution to a neglected tropical disease? PLoS Negl. Trop. Dis. 11, e0005361 (2017).
Lewin, M., Samuel, S., Merkel, J. & Bickler, P. Varespladib (LY315920) appears to be a potent, broad-spectrum, inhibitor of snake venom phospholipase A2 and a possible pre-referral treatment for envenomation. Toxins (Basel) 8, 248 (2016).
O’Brien, J., Lee, S.-H., Onogi, S. & Shea, K. J. Engineering the protein corona of a synthetic polymer nanoparticle for broad-spectrum sequestration and neutralization of venomous biomacromolecules. J. Am. Chem. Soc. 138, 16604–16607 (2016).
Saul, M. E. et al. A pharmacological approach to first aid treatment for snakebite. Nat. Med. 17, 809–811 (2011).
Rucavado, A. et al. Inhibition of local hemorrhage and dermonecrosis induced by Bothrops asper snake venom: effectiveness of early in situ administration of the peptidomimetic metalloproteinase inhibitor batimastat and the chelating agent CaNa2EDTA. Am. J. Trop. Med. Hyg. 63, 313–319 (2000).
Azofeifa, K., Angulo, Y. & Lomonte, B. Ability of fucoidan to prevent muscle necrosis induced by snake venom myotoxins: comparison of high- and low-molecular weight fractions. Toxicon 51, 373–380 (2008).
Prado, N. D. R. et al. Inhibition of the myotoxicity induced by Bothrops jararacussu venom and isolated phospholipases A2 by specific camelid single-domain antibody fragments. PLoS ONE 11, e0151363 (2016).
Richard, G. et al. In vivo neutralization of α-cobratoxin with high-affinity Llama single-domain antibodies (VHHs) and a VHH-Fc antibody. PLoS ONE 8, e69495 (2013).
Gutiérrez, J. M. Improving antivenom availability and accessibility: science, technology, and beyond. Toxicon 60, 676–687 (2012).
Lancet, T. Snake bite — the neglected tropical disease. Lancet 386, 1110 (2015).
King, G. F. (ed.) Venoms to Drugs: Venom as a Source for the Development of Human Therapeutics (Royal Society of Chemistry Publishing, 2015).
Eichberg, S., Sanz, L., Calvete, J. J. & Pla, D. Constructing comprehensive venom proteome reference maps for integrative venomics. Expert Rev. Proteomics 12, 557–573 (2015).
Petras, D., Heiss, P., Süssmuth, R. D. & Calvete, J. J. Venom proteomics of Indonesian king cobra, Ophiophagus hannah: integrating top-down and bottom-up approaches. J. Proteome Res. 14, 2539–2556 (2015).
Petras, D., Heiss, P., Harrison, R. A., Süssmuth, R. D. & Calvete, J. J. Top-down venomics of the East African green mamba, Dendroaspis angusticeps, and the black mamba, Dendroaspis polylepis, highlight the complexity of their toxin arsenals. J. Proteomics 146, 148–164 (2016).
Catherman, A. D., Skinner, O. S. & Kelleher, N. L. Top down proteomics: facts and perspectives. Biochem. Biophys. Res. Commun. 445, 683–693 (2014).
Fornelli, L. et al. Advancing top-down analysis of the human proteome using a benchtop quadrupole-orbitrap mass spectrometer. J. Proteome Res. 16, 609–618 (2016).
Calderón-Celis, F. et al. Elemental mass spectrometry for absolute intact protein quantification without protein-specific standards: application to snake venomics. Anal. Chem. 88, 9699–9706 (2016).
Acknowledgements
The authors thank J. Bravo (Instituto de Biomedicina de Valencia, Consejo Superior de Investigaciones Científicas (CSIC)) for assisting in the elaboration of the structural models shown in Figure 3.
Author information
Authors and Affiliations
Contributions
Introduction (J.M.G.); Epidemiology (A.G.H.); Mechanisms/pathophysiology (J.J.C. and J.M.G.); Diagnosis, screening and prevention (D.A.W.); Management (D.A.W.); Quality of life (D.J.W.); Outlook (J.M.G., R.A.H. and D.J.W.); Overview of the Primer (J.M.G.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Supplementary information S2 (box)
Supplementary file 2 (PDF 132 kb)
Rights and permissions
About this article
Cite this article
Gutiérrez, J., Calvete, J., Habib, A. et al. Snakebite envenoming. Nat Rev Dis Primers 3, 17063 (2017). https://doi.org/10.1038/nrdp.2017.63
Published:
DOI: https://doi.org/10.1038/nrdp.2017.63
This article is cited by
-
Distinct regulatory networks control toxin gene expression in elapid and viperid snakes
BMC Genomics (2024)
-
Tissue damaging toxins in snake venoms: mechanisms of action, pathophysiology and treatment strategies
Communications Biology (2024)
-
Antibody-dependent enhancement of toxicity of myotoxin II from Bothrops asper
Nature Communications (2024)
-
Systematic review and meta-analysis on the efficacy of Indian polyvalent antivenom against the Indian snakes of clinical significance
Archives of Toxicology (2024)
-
Mathematical analysis for the dynamics of snakebite envenoming
Afrika Matematika (2024)