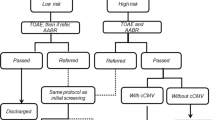
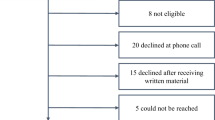
Abstract
Congenital hearing loss (hearing loss that is present at birth) is one of the most prevalent chronic conditions in children. In the majority of developed countries, neonatal hearing screening programmes enable early detection; early intervention will prevent delays in speech and language development and has long-lasting beneficial effects on social and emotional development and quality of life. A diagnosis of hearing loss is usually followed by a search for an underlying aetiology. Congenital hearing loss might be attributed to environmental and prenatal factors, which prevail in low-income settings; congenital infections, particularly cytomegalovirus infection, are also a common risk factor for hearing loss. Genetic causes probably account for the majority of cases in developed countries; mutations can affect any component of the hearing pathway, in particular, inner ear homeostasis (endolymph production and maintenance) and mechano-electrical transduction (the conversion of a mechanical stimulus into electrochemical activity). Once the underlying cause of hearing loss is established, it might direct therapeutic decision making and guide prevention and (genetic) counselling. Management options include specific antimicrobial therapies, surgical treatment of craniofacial abnormalities and implantable or non-implantable hearing devices. An improved understanding of the pathophysiology and molecular mechanisms that underlie hearing loss and increased awareness of recent advances in genetic testing will promote the development of new treatment and screening strategies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Boudewyns, A. et al. Otitis media with effusion: an underestimated cause of hearing loss in infants. Otol. Neurotol. 32, 799–804 (2011).
Rapin, I. & Gravel, J. S. Auditory neuropathy: a biologically inappropriate label unless acoustic nerve involvement is documented. J. Am. Acad. Audiol. 17, 147–150 (2006).
Cone-Wesson, B. & Rance, G. Auditory neuropathy: a brief review. Curr. Opin. Otolaryngol. Head Neck Surg. 8, 421–425 (2000).
Starr, A., Picton, T. W., Sininger, Y., Hood, L. J. & Berlin, C. I. Auditory neuropathy. Brain 119, 741–753 (1996).
Yoshinaga-Itano, C., Sedey, A. L., Coulter, D. K. & Mehl, A. L. Language of early- and later-identified children with hearing loss. Pediatrics 102, 1161–1171 (1998). This paper shows the effect of delayed detection and treatment for congenital hearing loss on speech and language development.
Fortnum, H. & Davis, A. Epidemiology of permanent childhood hearing impairment in Trent Region, 1985–1993. Br. J. Audiol. 31, 409–446 (1997).
American Academy of Pediatrics, Joint Committee on Infant Hearing. Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics 120, 898–921 (2007). This paper presents the guidelines of the American Academy of Pediatrics on the importance of universal neonatal hearing screening, risk factors for congenital hearing loss and management strategies for those who fail the screening test.
Norton, S. J. et al. Identification of neonatal hearing impairment: evaluation of transient evoked otoacoustic emission, distortion product otoacoustic emission, and auditory brain stem response test performance. Ear Hear. 21, 508–528 (2000).
Morton, C. C. & Nance, W. E. Newborn hearing screening — a silent revolution. N. Engl. J. Med. 354, 2151–2164 (2006).
Fortnum, H. M., Summerfield, A. Q., Marshall, D. H., Davis, A. C. & Bamford, J. M. Prevalence of permanent childhood hearing impairment in the United Kingdom and implications for universal neonatal hearing screening: questionnaire based ascertainment study. BMJ 323, 536–540 (2001).
Watkin, P. & Baldwin, M. The longitudinal follow up of a universal neonatal hearing screen: the implications for confirming deafness in childhood. Int. J. Audiol. 51, 519–528 (2012).
Nikolopoulos, T. P. Auditory dyssynchrony or auditory neuropathy: understanding the pathophysiology and exploring methods of treatment. Int. J. Pediatr. Otorhinolaryngol. 78, 171–173 (2014).
Mathers, C., Smith, A. & Concha, M. Global burden of hearing loss in the year 2000. WHOhttp://www.who.int/healthinfo/statistics/bod_hearingloss.pdf (2000).
Driscoll, C., Beswick, R., Doherty, E., D'silva, R. & Cross, A. The validity of family history as a risk factor in pediatric hearing loss. Int. J. Pediatr. Otorhinolaryngol. 79, 654–659 (2015).
van Dommelen, P. & Verkerk, P. H. & van Straaten, H. L. Hearing loss by week of gestation and birth weight in very preterm neonates. J. Pediatr. 166, 840–843.e1 (2015).
van Dommelen, P., Mohangoo, A. D., Verkerk, P. H., van der Ploeg, C. P. & van Straaten, H. L. Risk indicators for hearing loss in infants treated in different neonatal intensive care units. Acta Paediatr. 99, 344–349 (2010).
Koenighofer, M., Parzefall, T., Ramsebner, R., Lucas, T. & Frei, K. Delayed auditory pathway maturation and prematurity. Wien. Klin. Wochenschr. 127, 440–444 (2015).
Marazita, M. L. et al. Genetic epidemiological studies of early-onset deafness in the U. S. school-age population. Am. J. Med. Genet. 46, 486–491 (1993).
Smith, R. J., Bale, J. F. Jr & White, K. R. Sensorineural hearing loss in children. Lancet 365, 879–890 (2005).
Snoeckx, R. L. et al. GJB2 mutations and degree of hearing loss: a multicenter study. Am. J. Hum. Genet. 77, 945–957 (2005). This paper reports a large multicentre study that is unique in establishing a genotype–phenotype correlation for GJB2 based on a very large set of patients, providing detailed information for many common genotypes.
Cone-Wesson, B. et al. Identification of neonatal hearing impairment: infants with hearing loss. Ear Hear. 21, 488–507 (2000).
Kochhar, A., Hildebrand, M. S. & Smith, R. J. H. Clinical aspects of hereditary hearing loss. Genet. Med. 9, 393–408 (2007).
Grosse, S. D., Ross, D. S. & Dollard, S. C. Congenital cytomegalovirus (CMV) infection as a cause of permanent bilateral hearing loss: a quantitative assessment. J. Clin. Virol. 41, 57–62 (2008). This is an outstanding overview of the overall economic impact of CMV-associated hearing loss.
Goderis, J. et al. Hearing loss and congenital CMV infection: a systematic review. Pediatrics 134, 972–982 (2014).
Banatvala, J. E. & Brown, D. W. Rubella. Lancet 363, 1127–1137 (2004).
Korver, A. M. et al. Causes of permanent childhood hearing impairment. Laryngoscope 121, 409–416 (2011).
Lammens, F., Verhaert, N., Devriendt, K., Debruyne, F. & Desloovere, C. Aetiology of congenital hearing loss: a cohort review of 569 subjects. Int. J. Pediatr. Otorhinolaryngol. 77, 1385–1391 (2013).
Declau, F., Boudewyns, A., Van den Ende, J., Peeters, A. & van den Heyning, P. Etiologic and audiologic evaluations after universal neonatal hearing screening: analysis of 170 referred neonates. Pediatrics 121, 1119–1126 (2008).
Zelante, L. et al. Connexin26 mutations associated with the most common form of non-syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum. Mol. Genet. 6, 1605–1609 (1997).
Riazuddin, S. et al. Tricellulin is a tight-junction protein necessary for hearing. Am. J. Hum. Genet. 79, 1040–1051 (2006).
Wilcox, E. R. et al. Mutations in the gene encoding tight junction claudin-14 cause autosomal recessive deafness DFNB29. Cell 104, 165–172 (2001).
Grimmer, J. F. & Hedlund, G. Vestibular symptoms in children with enlarged vestibular aqueduct anomaly. Int. J. Pediatr. Otorhinolaryngol. 71, 275–282 (2007).
Kim, B. G. et al. Early deterioration of residual hearing in patients with SLC26A4 mutations. Laryngoscope 126, E286–E291 (2016).
Naz, S. et al. Mutations of ESPN cause autosomal recessive deafness and vestibular dysfunction. J. Med. Genet. 41, 591–595 (2004).
Donaudy, F. et al. Espin gene (ESPN) mutations associated with autosomal dominant hearing loss cause defects in microvillar elongation or organisation. J. Med. Genet. 43, 157–161 (2006).
Kitajiri, S. et al. Actin-bundling protein TRIOBP forms resilient rootlets of hair cell stereocilia essential for hearing. Cell 141, 786–798 (2010).
Fettiplace, R. & Kim, K. X. The physiology of mechanoelectrical transduction channels in hearing. Physiol. Rev. 94, 951–986 (2014).
Roux, I. et al. Otoferlin, defective in a human deafness form, is essential for exocytosis at the auditory ribbon synapse. Cell 127, 277–289 (2006). This paper demonstrates that otoferlin interacts with SNARE (soluble NSF (N-ethylmaleimide-sensitive factor) molecules at the afferent ribbon synapses in inner hair cells of the cochlea to trigger exocytosis of the neurotransmitter glutamate.
Leal, M. C. et al. Hearing loss in infants with microcephaly and evidence of congenital Zika virus infection — Brazil, November 2015–May 2016. MMWR Morb. Mortal. Wkly Rep. 65, 917–919 (2016). Zika virus infection is the latest of the ‘TORCH’ infection complex to be demonstrated as a cause of sensorineural hearing loss. Zika virus infection should be added to the list of infectious diseases that are known to induce hearing loss in infants.
Cannon, M. J. et al. Repeated measures study of weekly and daily cytomegalovirus shedding patterns in saliva and urine of healthy cytomegalovirus-seropositive children. BMC Infect. Dis. 14, 569 (2014).
Kenneson, A. & Cannon, M. J. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev. Med. Virol. 17, 253–276 (2007). This is a detailed analysis of the epidemiology of congenital CMV infection using available literature and previously published work, and is an outstanding review of the range of disabilities and clinical manifestations of congenital infection.
Cohen, B. E., Durstenfeld, A. & Roehm, P. C. Viral causes of hearing loss: a review for hearing health professionals. Trends Hear. 18, 2331216514541361 (2014).
Schleiss, M. R. & Choo, D. I. Mechanisms of congenital cytomegalovirus-induced deafness. Drug Discov. Today Dis. Mech. 2, 105–113 (2006).
Schachtele, S. J., Mutnal, M. B., Schleiss, M. R. & Lokensgard, J. R. Cytomegalovirus-induced sensorineural hearing loss with persistent cochlear inflammation in neonatal mice. J. Neurovirol. 17, 201–211 (2011).
Bradford, R. D. et al. Murine CMV-induced hearing loss is associated with inner ear inflammation and loss of spiral ganglia neurons. PLoS Pathog. 11, e1004774 (2015).
Schraff, S. A. et al. Macrophage inflammatory proteins in cytomegalovirus-related inner ear injury. Otolaryngol. Head Neck Surg. 137, 612–618 (2007).
Schleiss, M. R. Cytomegalovirus in the neonate: immune correlates of infection and protection. Clin. Dev. Immunol. 2013, 501801 (2013).
Enders, G., Daiminger, A., Bader, U., Exler, S. & Enders, M. Intrauterine transmission and clinical outcome of 248 pregnancies with primary cytomegalovirus infection in relation to gestational age. J. Clin. Virol. 52, 244–246 (2011).
Miller, E., Cradock-Watson, J. E. & Pollock, T. M. Consequences of confirmed maternal rubella at successive stages of pregnancy. Lancet 2, 781–784 (1982).
Bouthry, E. et al. Rubella and pregnancy: diagnosis, management and outcomes. Prenat. Diagn. 34, 1246–1253 (2014). This is an outstanding overview of the presentation, epidemiology and management of maternal and fetal rubella virus infection, with an excellent perspective and overview of diagnostic studies.
Sever, J. L., South, M. A. & Shaver, K. A. Delayed manifestations of congenital rubella. Rev. Infect. Dis. 7, S164–S169 (1985).
Lee, J. Y. & Bowden, D. S. Rubella virus replication and links to teratogenicity. Clin. Microbiol. Rev. 13, 571–587 (2000).
Webster, W. S. Teratogen update: congenital rubella. Teratology 58, 13–23 (1998).
Lin, H. C., Shu, M. T., Lee, K. S., Lin, H. Y. & Lin, G. Reducing false positives in newborn hearing screening program: how and why. Otol. Neurotol. 28, 788–792 (2007).
Stephens, D. in Definitions, Protocols and Guidelines in Genetic Hearing Impairment (eds Martini, A., Mazzoli, M., Read, A. & Stephens, D. ) 9–14 (Whurr Publishers, 2001).
Kemp, D. T. Otoacoustic emissions, their origin in cochlear function, and use. Br. Med. Bull. 63, 223–241 (2002).
Rance, G. & Briggs, R. J. Assessment of hearing in infants with moderate to profound impairment: the Melbourne experience with auditory steady-state evoked potential testing. Ann. Otol. Rhinol. Laryngol. Suppl. 189, 22–28 (2002).
Swanepoel, D. & Ebrahim, S. Auditory steady-state response and auditory brainstem response thresholds in children. Eur. Arch. Otorhinolaryngol. 266, 213–219 (2009).
Harlor, A. D. Jr et al. Hearing assessment in infants and children: recommendations beyond neonatal screening. Pediatrics 124, 1252–1263 (2009).
De Leenheer, E. M. et al. Etiological diagnosis in the hearing impaired newborn: proposal of a flow chart. Int. J. Pediatr. Otorhinolaryngol. 75, 27–32 (2011).
Alford, R. L. et al. American College of Medical Genetics and Genomics guideline for the clinical evaluation and etiologic diagnosis of hearing loss. Genet. Med. 16, 347–355 (2014). This paper summarizes protocols for aetiological work-up of congenital hearing loss and advocates the use of comprehensive genetic testing.
Bamiou, D. E., MacArdle, B., Bitner-Glindzicz, M. & Sirimanna, T. Aetiological investigations of hearing loss in childhood: a review. Clin. Otolaryngol. Allied Sci. 25, 98–106 (2000).
Hart, C. K. & Choo, D. I. What is the optimal workup for a child with bilateral sensorineural hearing loss? Laryngoscope 123, 809–810 (2013).
Sloan-Heggen, C. M. et al. Comprehensive genetic testing in the clinical evaluation of 1119 patients with hearing loss. Hum. Genet. 135, 441–450 (2016). This paper demonstrates that comprehensive genetic testing is a foundational diagnostic test, which allows health care providers to make evidence-based decisions in the evaluation of hearing loss, thereby providing better and more cost-effective patient care.
Zong, L. et al. Mutations in apoptosis-inducing factor cause X-linked recessive auditory neuropathy spectrum disorder. J. Med. Genet. 52, 523–531 (2015).
Park, A. H. et al. A diagnostic paradigm including cytomegalovirus testing for idiopathic pediatric sensorineural hearing loss. Laryngoscope 124, 2624–2629 (2014).
Boppana, S. B., Ross, S. A. & Fowler, K. B. Congenital cytomegalovirus infection: clinical outcome. Clin. Infect. Dis. 57, S178–S181 (2013).
Ville, Y. & Leruez-Ville, M. Managing infections in pregnancy. Curr. Opin. Infect. Dis. 27, 251–257 (2014).
Swanson, E. C. & Schleiss, M. R. Congenital cytomegalovirus infection: new prospects for prevention and therapy. Pediatr. Clin. North Am. 60, 335–349 (2013).
Freij, B. J., South, M. A. & Sever, J. L. Maternal rubella and the congenital rubella syndrome. Clin. Perinatol. 15, 247–257 (1988).
Nagasawa, K. et al. Congenital rubella syndrome: a case report on changes in viral load and rubella antibody titers. Pediatrics 137, e20153333 (2016).
Alvarado, J. C. et al. Synergistic effects of free radical scavengers and cochlear vasodilators: a new otoprotective strategy for age-related hearing loss. Front. Aging Neurosci. 7, 86 (2015).
Mukherjea, D. et al. Early investigational drugs for hearing loss. Expert Opin. Investig. Drugs 24, 201–217 (2015).
Chen, G., Zhang, X., Yang, F. & Mu, L. Disposition of nanoparticle-based delivery system via inner ear administration. Curr. Drug Metab. 11, 886–897 (2010).
Fitzpatrick, E. M. et al. Sign language and spoken language for children with hearing loss: a systematic review. Pediatrics 137, e20151974 (2016).
Nittrouer, S. Beyond early intervention: supporting children with CIs through elementary school. Otol. Neurotol. 37, e43–e49 (2016).
Francois, M., Boukhris, M. & Noel-Petroff, N. Schooling of hearing-impaired children and benefit of early diagnosis. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 132, 251–255 (2015).
Kimberlin, D. W. et al. Effect of ganciclovir therapy on hearing in symptomatic congenital cytomegalovirus disease involving the central nervous system: a randomized, controlled trial. J. Pediatr. 143, 16–25 (2003).
Kimberlin, D. W. et al. Valganciclovir for symptomatic congenital cytomegalovirus disease. N. Engl. J. Med. 372, 933–943 (2015). This landmark paper reports the results of a randomized controlled trial, which demonstrates that therapy with oral valganciclovir improves both hearing and neurodevelopmental outcomes in symptomatic congenital CMV infection.
Schleiss, M. R. Developing a vaccine against congenital cytomegalovirus (CMV) infection: what have we learned from animal models? Where should we go next? Future Virol. 8, 1161–1182 (2013).
Royackers, L., Christian, D., Frans, D. & Ermelinde, R. Hearing status in children with congenital cytomegalovirus: up-to-6-years audiological follow-up. Int. J. Pediatr. Otorhinolaryngol. 75, 376–382 (2011).
Oz, H. S. Maternal and congenital toxoplasmosis, currently available and novel therapies in horizon. Front. Microbiol. 5, 385 (2014).
Wright, R. et al. Congenital lymphocytic choriomeningitis virus syndrome: a disease that mimics congenital toxoplasmosis or cytomegalovirus infection. Pediatrics 100, E9 (1997).
Russell, K. et al. Update: interim guidance for the evaluation and management of infants with possible congenital Zika virus infection — United States, August 2016. MMWR Morb. Mortal. Wkly Rep. 65, 870–878 (2016).
McCreery, R. W., Venediktov, R. A., Coleman, J. J. & Leech, H. M. An evidence-based systematic review of amplitude compression in hearing aids for school-age children with hearing loss. Am. J. Audiol. 21, 269–294 (2012).
Xu, J. & Han, W. Improvement of adult BTE hearing aid wearers' front/back localization performance using digital pinna-cue preserving technologies: an evidence-based review. Korean J. Audiol. 18, 97–104 (2014).
Benson, K. B. Audio Engineering Handbook (McGraw-Hill, 1988).
Boothroyd, A., Springer, N., Smith, L. & Schulman, J. Amplitude compression and profound hearing loss. J. Speech Hear. Res. 31, 362–376 (1988).
Moore, B. C. Characterization and simulation of impaired hearing: implications for hearing aid design. Ear Hear. 12, 154S–161S (1991).
National Institute on Deafness and Other Communication Disorders. Charts and tables for hearing statistics. Use of hearing aids by adults with hearing loss. NIDCDhttps://www.nidcd.nih.gov/health/statistics/use-hearing-aids-adults-hearing-loss (updated 30 Sept 2014).
McCormack, A. & Fortnum, H. Why do people fitted with hearing aids not wear them? Int. J. Audiol. 52, 360–368 (2013).
Spivak, L., Sokol, H., Auerbach, C. & Gershkovich, S. Newborn hearing screening follow-up: factors affecting hearing aid fitting by 6 months of age. Am. J. Audiol. 18, 24–33 (2009).
Kasic, J. F. & Fredrickson, J. M. The otologics MET ossicular stimulator. Otolaryngol. Clin. North Am. 34, 501–513 (2001).
Luers, J., Huttenbrink, K., Zahnert, T., Bornitz, M. & Beutner, D. Vibroplasty for mixed and conductive hearing. Otol. Neurotol. 34, 1005–1012 (2013).
Mueller, H. & Hall, J. Audiologists Desk Reference (Singular Publishing Group Inc., 1998).
Gaylor, J. M. et al. Cochlear implantation in adults: a systematic review and meta-analysis. JAMA Otolaryngol. Head Neck Surg. 139, 265–272 (2013).
Roland, J. T. Jr, Gantz, B. J., Waltzman, S. B., Parkinson, A. J. & Multicenter Clinical Trial Group. United States multicenter clinical trial of the cochlear nucleus hybrid implant system. Laryngoscope 126, 175–181 (2016).
Golub, J. S., Won, J. H., Drennan, W. R., Worman, T. D. & Rubinstein, J. T. Spectral and temporal measures in hybrid cochlear implant users: on the mechanism of electroacoustic hearing benefits. Otol. Neurotol. 33, 147–153 (2012).
Fitzpatrick, E. M., Ham, J. & Whittingham, J. Pediatric cochlear implantation: why do children receive implants late? Ear Hear. 36, 688–694 (2015).
Buchman, C. A. et al. Cochlear implantation in children with congenital inner ear malformations. Laryngoscope 114, 309–316 (2004).
Birman, C. S., Elliott, E. J. & Gibson, W. P. Pediatric cochlear implants: additional disabilities prevalence, risk factors, and effect on language outcomes. Otol. Neurotol. 33, 1347–1352 (2012).
Eppsteiner, R. W. et al. Prediction of cochlear implant performance by genetic mutation: the spiral ganglion hypothesis. Hear. Res. 292, 51–58 (2012).
Wu, C. C., Liu, T. C., Wang, S. H., Hsu, C. J. & Wu, C. M. Genetic characteristics in children with cochlear implants and the corresponding auditory performance. Laryngoscope 121, 1287–1293 (2011).
Usami, S. et al. Patients with CDH23 mutations and the 1555A>G mitochondrial mutation are good candidates for electric acoustic stimulation (EAS). Acta Otolaryngol. 132, 377–384 (2012).
Miyagawa, M., Nishio, S. Y. & Usami, S. A. Comprehensive study on the etiology of patients receiving cochlear implantation with special emphasis on genetic epidemiology. Otol. Neurotol. 37, e126–e134 (2016).
Cremers, C. W., Teunissen, E. & Marres, E. H. Classification of congenital aural atresia and results of reconstructive surgery. Adv. Otorhinolaryngol. 40, 9–14 (1988).
Doshi, J. & McDermott, A. L. Bone anchored hearing aids in children. Expert Rev. Med. Devices 12, 73–82 (2015).
Tjellstrom, A., Hakansson, B. & Granstrom, G. Bone-anchored hearing aids: current status in adults and children. Otolaryngol. Clin. North Am. 34, 337–364 (2001).
Kiringoda, R. & Lustig, L. A meta-analysis of the complications associated with osseointegrated hearing aids. Otol. Neurotol. 34, 790–794 (2013).
Snik, A. F. et al. Consensus statements on the BAHA system: where do we stand at present? Ann. Otol. Rhinol. Laryngol. Suppl. 195, 2–12 (2005).
Niparko, J. K., Cox, K. M. & Lustig, L. R. Comparison of the bone anchored hearing aid implantable hearing device with contralateral routing of offside signal amplification in the rehabilitation of unilateral deafness. Otol. Neurotol. 24, 73–78 (2003).
Saroul, N., Akkari, M., Pavier, Y., Gilain, L. & Mom, T. Long-term benefit and sound localization in patients with single-sided deafness rehabilitated with an osseointegrated bone-conduction device. Otol. Neurotol. 34, 111–114 (2013).
Schwager, K. Reconstruction of middle ear malformations. GMS Curr. Top. Otorhinolaryngol. Head Neck Surg. 6, Doc01 (2007).
Holden-Pitt, L. & Albertorio, J. Thirty years of the Annual Survey of Deaf and Hard-of-Hearing Children and Youth: a glance over the decades. Am. Ann. Deaf 143, 72–76 (1998).
Lustig, L. R., Leake, P. A., Snyder, R. L. & Rebscher, S. J. Changes in the cat cochlear nucleus following neonatal deafening and chronic intracochlear electrical stimulation. Hear. Res. 74, 29–37 (1994).
Pimperton, H. et al. The impact of universal newborn hearing screening on long-term literacy outcomes: a prospective cohort study. Arch. Dis. Child. 101, 9–15 (2016).
Korver, A. M. et al. Newborn hearing screening versus later hearing screening and developmental outcomes in children with permanent childhood hearing impairment. JAMA 304, 1701–1708 (2010).
Varni, J. W., Seid, M. & Rode, C. A. The PedsQL: measurement model for the pediatric quality of life inventory. Med. Care 37, 126–139 (1999).
Stern-Ginossar, N. et al. Decoding human cytomegalovirus. Science 338, 1088–1093 (2012).
Soderberg-Naucler, C. Treatment of cytomegalovirus infections beyond acute disease to improve human health. Expert Rev. Anti Infect. Ther. 12, 211–222 (2014).
Cheeran, M. C., Lokensgard, J. R. & Schleiss, M. R. Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin. Microbiol. Rev. 22, 99–126 (2009).
Boppana, S. B., Fowler, K. B., Britt, W. J., Stagno, S. & Pass, R. F. Symptomatic congenital cytomegalovirus infection in infants born to mothers with preexisting immunity to cytomegalovirus. Pediatrics 104, 55–60 (1999).
Ross, S. A. et al. Cytomegalovirus reinfections in healthy seroimmune women. J. Infect. Dis. 201, 386–389 (2010).
Boppana, S. B., Rivera, L. B., Fowler, K. B., Mach, M. & Britt, W. J. Intrauterine transmission of cytomegalovirus to infants of women with preconceptional immunity. N. Engl. J. Med. 344, 1366–1371 (2001).
Shearer, A. E. et al. Copy number variants are a common cause of non-syndromic hearing loss. Genome Med. 6, 37 (2014). This paper shows that copy number variants are a common cause of genetic hearing loss. Their involvement in roughly one in five genetic diagnoses mandates their identification in any clinical genetic diagnostic test for hearing loss.
Taylor, K. R. et al. AudioGene: predicting hearing loss genotypes from phenotypes to guide genetic screening. Hum. Mutat. 34, 539–545 (2013).
Nakagawa, T. Strategies for developing novel therapeutics for sensorineural hearing loss. Front. Pharmacol. 5, 206 (2014).
Shibata, S. B. & Raphael, Y. Future approaches for inner ear protection and repair. J. Commun. Disord. 43, 295–310 (2010).
Werner, M., Van De Water, T. R., Hammarsten, P., Arnoldsson, G. & Berggren, D. Morphological and morphometric characterization of direct transdifferentiation of support cells into hair cells in ototoxin-exposed neonatal utricular explants. Hear. Res. 321, 1–11 (2015).
Wan, G., Corfas, G. & Stone, J. S. Inner ear supporting cells: rethinking the silent majority. Semin. Cell Dev. Biol. 24, 448–459 (2013).
Esterberg, R. et al. Fish in a dish: drug discovery for hearing habilitation. Drug Discov. Today Dis. Models 10, e23–e29 (2013).
McCall, A. A. et al. Drug delivery for treatment of inner ear disease: current state of knowledge. EarHear. 31, 156–165 (2010).
Sidell, D. et al. Combination therapies using an intratympanic polymer gel delivery system in the guinea pig animal model: a safety study. Int. J. Pediatr. Otorhinolaryngol. 84, 132–136 (2016).
Van Kerschaver, E., Boudewyns, A. N., Declau, F., Van de Heyning, P. H. & Wuyts, F. L. Socio-demographic determinants of hearing impairment studied in 103,835 term babies. Eur. J. Public Health 23, 55–60 (2013).
Rohlfs, A. K. et al. Interdisciplinary approach to design, performance, and quality management in a multicenter newborn hearing screening project: introduction, methods, and results of the newborn hearing screening in Hamburg (part I). Eur. J. Pediatr. 169, 1353–1360 (2010).
Colgan, S. et al. The cost-effectiveness of universal newborn screening for bilateral permanent congenital hearing impairment: systematic review. Acad. Pediatr. 12, 171–180 (2012).
Gantt, S. et al. Cost-effectiveness of universal and targeted newborn screening for congenital cytomegalovirus infection. JAMA Pediatr. 170, 1173–1180 (2016). This paper demonstrates the cost-effectiveness of screening for congenital CMV infection, both in the context of universal screening programmes and in a ‘targeted’ screening approach in which infants who fail their neonatal hearing screening are specifically tested for CMV infection. This paper should help to drive the implementation of newborn screening for congenital CMV infection.
Zdebik, A. A., Wangemann, P. & Jentsch, T. J. Potassium ion movement in the inner ear: insights from genetic disease and mouse models. Physiology (Bethesda) 24, 307–316 (2009). This is an excellent overview of inner ear structure and function, particularly with respect to the role of molecular components of the stria vascularis in endolymph production and the recycling of K+ ions.
Jagger, D. J. & Forge, A. Connexins and gap junctions in the inner ear — it's not just about K+ recycling. Cell Tissue Res. 360, 633–644 (2015).
Verhoeven, K. et al. Mutations in the human alpha-tectorin gene cause autosomal dominant non-syndromic hearing impairment. Nat. Genet. 19, 60–62 (1998).
Schraders, M. et al. Mutations of the gene encoding otogelin are a cause of autosomal-recessive nonsyndromic moderate hearing impairment. Am. J. Hum. Genet. 91, 883–889 (2012).
Zheng, J. et al. Carcinoembryonic antigen-related cell adhesion molecule 16 interacts with alpha-tectorin and is mutated in autosomal dominant hearing loss (DFNA4). Proc. Natl Acad. Sci. USA 108, 4218–4223 (2011).
Yariz, K. O. et al. Mutations in OTOGL, encoding the inner ear protein otogelin-like, cause moderate sensorineural hearing loss. Am. J. Hum. Genet. 91, 872–882 (2012).
Grati, M. & Kachar, B. Myosin VIIa and sans localization at stereocilia upper tip-link density implicates these Usher syndrome proteins in mechanotransduction. Proc. Natl Acad. Sci. USA 108, 11476–11481 (2011).
Kazmierczak, P. et al. Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature 449, 87–91 (2007).
Manor, U. et al. Regulation of stereocilia length by myosin XVa and whirlin depends on the actin-regulatory protein Eps8. Curr. Biol. 21, 167–172 (2011).
Delprat, B. et al. Myosin XVa and whirlin, two deafness gene products required for hair bundle growth, are located at the stereocilia tips and interact directly. Hum. Mol. Genet. 14, 401–410 (2005).
Belyantseva, I. A. et al. Myosin-XVa is required for tip localization of whirlin and differential elongation of hair-cell stereocilia. Nat. Cell Biol. 7, 148–156 (2005).
Riazuddin, S. et al. Alterations of the CIB2 calcium- and integrin-binding protein cause Usher syndrome type 1J and nonsyndromic deafness DFNB48. Nat. Genet. 44, 1265–1271 (2012).
Jentsch, T. J. Neuronal KCNQ potassium channels: physiology and role in disease. Nat. Rev. Neurosci. 1, 21–30 (2000).
Nikolopoulos, T. P., Lioumi, D., Stamataki, S. & O'Donoghue, G. M. Evidence-based overview of ophthalmic disorders in deaf children: a literature update. Otol. Neurotol. 27, S1–S24; discussion S20 (2006).
Thiebaut, R., Leproust, S., Chene, G. & Gilbert, R. Effectiveness of prenatal treatment for congenital toxoplasmosis: a meta-analysis of individual patients' data. Lancet 369, 115–122 (2007).
Neu, N., Duchon, J. & Zachariah, P. TORCH infections. Clin. Perinatol. 42, 77–103 (2015).
Cutts, F. T. & Vynnycky, E. Modelling the incidence of congenital rubella syndrome in developing countries. Int. J. Epidemiol. 28, 1176–1184 (1999).
James, S. H. & Kimberlin, D. W. Neonatal herpes simplex virus infection: epidemiology and treatment. Clin. Perinatol. 42, 47–59 (2015).
Chau, J., Atashband, S., Chang, E., Westerberg, B. D. & Kozak, F. K. A systematic review of pediatric sensorineural hearing loss in congenital syphilis. Int. J. Pediatr. Otorhinolaryngol. 73, 787–792 (2009).
Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2013. CDChttps://www.cdc.gov/std/stats13/surv2013-print.pdf (2013).
Pessoa, L. & Galvao, V. Clinical aspects of congenital syphilis with Hutchinson's triad. BMJ Case Rep. 2011, bcr1120115130 (2011).
Salviz, M., Montoya, J. G., Nadol, J. B. & Santos, F. Otopathology in congenital toxoplasmosis. Otol. Neurotol. 34, 1165–1169 (2013).
Austeng, M. E. et al. Maternal infection with Toxoplasma gondii in pregnancy and the risk of hearing loss in the offspring. Int. J. Audiol. 49, 65–68 (2010).
McLeod, R. et al. Outcome of treatment for congenital toxoplasmosis, 1981–2004: the National Collaborative Chicago-Based, Congenital Toxoplasmosis Study. Clin. Infect. Dis. 42, 1383–1394 (2006).
Yamamoto, A. Y. et al. Congenital cytomegalovirus infection as a cause of sensorineural hearing loss in a highly immune population. Pediatr. Infect. Dis. J. 30, 1043–1046 (2011).
Manicklal, S., Emery, V. C., Lazzarotto, T., Boppana, S. B. & Gupta, R. K. The “silent” global burden of congenital cytomegalovirus. Clin. Microbiol. Rev. 26, 86–102 (2013).
Westerberg, B. D., Atashband, S. & Kozak, F. K. A systematic review of the incidence of sensorineural hearing loss in neonates exposed to herpes simplex virus (HSV). Int. J. Pediatr. Otorhinolaryngol. 72, 931–937 (2008).
Phillips, J. S., Gaunt, A. & Phillips, D. R. Otosyphilis: a neglected diagnosis? Otol. Neurotol. 35, 1011–1013 (2014).
Acknowledgements
This work was supported by US National Institute on Deafness and Other Communication Disorders grants RO1s DC003544, DC002842 and DC012049 to R.J.H.S. and US National Institute of Child Health and Human Development grants R01s HD044864 and HD079918 to M.R.S. M.A.K.B.-G. is supported by Great Ormond Street Hospital Children's Charity and the National Institute for Health Research Biomedical Research Centre at Great Ormond Street Hospital for Children NHS Foundation Trust and University College London. All authors thank their colleague A. Snik for his input to earlier versions of this manuscript.
Author information
Authors and Affiliations
Contributions
Introduction (G.V.C. and A.N.B.), Epidemiology (A.M.H.K., M.A.K.B.-G. and M.R.S.); Mechanisms/pathophysiology (A.M.H.K., M.A.K.B.-G. and M.R.S.); Diagnosis, screening and prevention (G.V.C., S.U., A.M.H.K., M.R.S. and A.N.B.); Management (M.R.S. and L.R.L.); Quality of life (A.N.B. and A.M.H.K.); Outlook (R.J.H.S. and S.U.); Overview of the Primer (A.M.H.K. and A.N.B.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Korver, A., Smith, R., Van Camp, G. et al. Congenital hearing loss. Nat Rev Dis Primers 3, 16094 (2017). https://doi.org/10.1038/nrdp.2016.94
Published:
DOI: https://doi.org/10.1038/nrdp.2016.94
This article is cited by
-
MPZL2—a common autosomal recessive deafness gene related to moderate sensorineural hearing loss in the Chinese population
BMC Medical Genomics (2024)
-
Low frequency of SLC26A4 c.919-2A > G variant among patients with nonsyndromic hearing loss in Yunnan of Southwest China
BMC Medical Genomics (2024)
-
The genetic basis and the diagnostic yield of genetic testing related to nonsyndromic hearing loss in Qatar
Scientific Reports (2024)
-
Radiological Parameters Predicting the Round Window Niche Visibility through Facial Recess Approach in Cochlear Implantation
Indian Journal of Otolaryngology and Head & Neck Surgery (2024)
-
Neonatal hearing screening - does failure in TEOAE screening matter when the AABR test is passed?
European Archives of Oto-Rhino-Laryngology (2024)