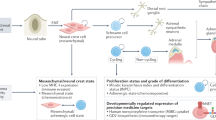
Abstract
Neuroblastoma is the most common extracranial solid tumour occurring in childhood and has a diverse clinical presentation and course depending on the tumour biology. Unique features of these neuroendocrine tumours are the early age of onset, the high frequency of metastatic disease at diagnosis and the tendency for spontaneous regression of tumours in infancy. The most malignant tumours have amplification of the MYCN oncogene (encoding a transcription factor), which is usually associated with poor survival, even in localized disease. Although transgenic mouse models have shown that MYCN overexpression can be a tumour-initiating factor, many other cooperating genes and tumour suppressor genes are still under investigation and might also have a role in tumour development. Segmental chromosome alterations are frequent in neuroblastoma and are associated with worse outcome. The rare familial neuroblastomas are usually associated with germline mutations in ALK, which is mutated in 10–15% of primary tumours, and provides a potential therapeutic target. Risk-stratified therapy has facilitated the reduction of therapy for children with low-risk and intermediate-risk disease. Advances in therapy for patients with high-risk disease include intensive induction chemotherapy and myeloablative chemotherapy, followed by the treatment of minimal residual disease using differentiation therapy and immunotherapy; these have improved 5-year overall survival to 50%. Currently, new approaches targeting the noradrenaline transporter, genetic pathways and the tumour microenvironment hold promise for further improvements in survival and long-term quality of life.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Stiller, C. A. & Parkin, D. M. International variations in the incidence of neuroblastoma. J. Int. Cancer 52, 538–543 (1992).
London, W. B. et al. Evidence for an age cutoff greater than 365 days for neuroblastoma risk group stratification in the Children's Oncology Group. J. Clin. Oncol. 23, 6459–6465 (2005).
Thompson, D. et al. Identification of patient subgroups with markedly disparate rates of MYCN amplification in neuroblastoma: a report from the International Neuroblastoma Risk Group project. Cancer 122, 935–945 (2016).
Schleiermacher, G. et al. Accumulation of segmental alterations determines progression in neuroblastoma. J. Clin. Oncol. 28, 3122–3130 (2010). Comparison at diagnosis and relapse of DNA ploidy and array comparative genomic hybridization for segmental chromosomal aberrations in 394 patients with neuroblastoma showed that further changes evolve at recurrence, even in patients treated with surgery alone, suggesting that patients should be re-evaluated at relapse.
Pugh, T. J. et al. The genetic landscape of high-risk neuroblastoma. Nat. Genet. 45, 279–284 (2013). This comprehensive study using exome, genome and transcriptome analysis identified some important germline and somatic mutations in 240 neuroblastomas, as well as the relatively low frequency of somatic mutations at diagnosis.
George, R. E. et al. Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature 455, 975–978 (2008).
Mosse, Y. P. et al. Identification of ALK as a major familial neuroblastoma predisposition gene. Nature 455, 930–935 (2008). Germline mutations in ALK account for the majority of familial neuroblastoma and somatic mutations are also found in 12% of sporadic neuroblastoma tumours, suggesting ALK as a new therapeutic target.
Kratz, C. P., Rapisuwon, S., Reed, H., Hasle, H. & Rosenberg, P. S. Cancer in Noonan, Costello, cardiofaciocutaneous and LEOPARD syndromes. Am. J. Med. Genet. C Semin. Med. Genet. 157C, 83–89 (2011).
Park, J. R. et al. Children's Oncology Group's 2013 blueprint for research: neuroblastoma. Pediatr. Blood Cancer 60, 985–993 (2013).
Cooper, R. et al. Opsoclonus-myoclonus-ataxia syndrome in neuroblastoma: histopathologic features — a report from the Children's Cancer Group. Med. Pediatr. Oncol. 36, 623–629 (2001).
Rudnick, E. et al. Opsoclonus-myoclonus-ataxia syndrome in neuroblastoma: clinical outcome and antineuronal antibodies-a report from the Children's Cancer Group Study. Med. Pediatr. Oncol. 36, 612–622 (2001).
Cohn, S. L. et al. The International Neuroblastoma Risk Group (INRG) classification system: an INRG Task Force report. J. Clin. Oncol. 27, 289–297 (2009). This is a comprehensive regression tree analysis of 13 potential prognostic factors on an international cohort of 8,800 patients with neuroblastoma, establishing the current basis of international risk stratification.
Matthay, K. K. et al. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis- retinoic acid. N. Engl. J. Med. 341, 1165–1173 (1999). This seminal report of a multicentre randomized controlled study showed the significant improvement in EFS for children treated with myeloablative chemoradiotherapy and purged AHSCT compared with standard-dose chemotherapy, and for subsequent treatment with isotretinoin maintenance compared with no further therapy, with current incorporation into standard care for high-risk neuroblastoma.
Baker, D. L. et al. Outcome after reduced chemotherapy for intermediate-risk neuroblastoma. N. Engl. J. Med. 363, 1313–1323 (2010).
Strother, D. R. et al. Outcome after surgery alone or with restricted use of chemotherapy for patients with low-risk neuroblastoma: results of Children's Oncology Group study P9641. J. Clin. Oncol. 30, 1842–1848 (2012).
Mosse, Y. P. et al. Neuroblastoma in older children, adolescents and young adults: a report from the International Neuroblastoma Risk Group project. Pediatr. Blood Cancer 61, 627–635 (2013).
Maris, J. M. Recent advances in neuroblastoma. N. Engl. J. Med. 362, 2202–2211 (2010).
Schilling, F. H. et al. Neuroblastoma screening at one year of age. N. Engl. J. Med. 346, 1047–1053 (2002).
Woods, W. G. et al. A population-based study of the usefulness of screening for neuroblastoma. Lancet 348, 1682–1687 (1996).
Gamazon, E. R. et al. Trans-population analysis of genetic mechanisms of ethnic disparities in neuroblastoma survival. J. Natl Cancer Inst. 105, 302–309 (2013).
Ward, E., DeSantis, C., Robbins, A., Kohler, B. & Jemal, A. Childhood and adolescent cancer statistics, 2014. CA Cancer J. Clin. 64, 83–103 (2014).
Bluhm, E. C., Daniels, J., Pollock, B. H., Olshan, A. F. & Children's Oncology Group. Maternal use of recreational drugs and neuroblastoma in offspring: a report from the Children's Oncology Group (United States). Cancer Causes Control 17, 663–669 (2006).
McCall, E. E., Olshan, A. F. & Daniels, J. L. Maternal hair dye use and risk of neuroblastoma in offspring. Cancer Causes Control 16, 743–748 (2005).
Knudson, A. G. J. & Strong, L. C. Mutation and cancer: neuroblastoma and pheochromocytoma. Am. J. Hum. Genet. 24, 514–522 (1972).
Janoueix-Lerosey, I. et al. Somatic and germline activating mutations of the ALK kinase receptor in neuroblastoma. Nature 455, 967–970 (2008).
Trochet, D. et al. Germline mutations of the paired-like homeobox 2B (PHOX2B) gene in neuroblastoma. Am. J. Hum. Genet. 74, 761–764 (2004).
Amiel, J. et al. Polyalanine expansion and frameshift mutations of the paired-like homeobox gene PHOX2B in congenital central hypoventilation syndrome. Nat. Genet. 33, 459–461 (2003).
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Bosse, K. R. & Maris, J. M. Advances in the translational genomics of neuroblastoma: from improving risk stratification and revealing novel biology to identifying actionable genomic alterations. Cancer 122, 20–33 (2016).
Oldridge, D. A. et al. Genetic predisposition to neuroblastoma mediated by a LMO1 super-enhancer polymorphism. Nature 528, 418–421 (2015).
L'Abbate, A. et al. Genomic organization and evolution of double minutes/homogeneously staining regions with MYC amplification in human cancer. Nucleic Acids Res. 42, 9131–9145 (2014).
Garner, E. F. & Beierle, E. A. Cancer stem cells and their interaction with the tumor microenvironment in neuroblastoma. Cancers (Basel) 8 5 (2015).
Huang, M. & Weiss, W. A. Neuroblastoma and MYCN. Cold Spring Harb. Perspect. Med. 3, a014415 (2013).
Moreau, L. A. et al. Does MYCN amplification manifested as homogeneously staining regions at diagnosis predict a worse outcome in children with neuroblastoma? A Children's Oncology Group study. Clin. Cancer Res. 12, 5693–5697 (2006).
Cox, D., Yuncken, C. & Spriggs, A. I. Minute chromatin bodies in malignant tumours of childhood. Lancet 1, 55–58 (1965).
Kohl, N. E. et al. Transposition and amplification of oncogene-related sequences in human neuroblastomas. Cell 35, 359–367 (1983).
Schwab, M., Varmus, H. E. & Bishop, J. M. Human N-myc gene contributes to neoplastic transformation of mammalian cells in culture. Nature 316, 160–162 (1985). Human MYCN, recently identified as amplified in neuroblastoma cell lines and shown to be amplified in human tumours, was shown to transform normal rat embryo cells into tumours, suggesting the importance of this gene in the pathogenesis of neuroblastoma.
Brodeur, G. M., Seeger, R. C., Schwab, M., Varmus, H. E. & Bishop, J. M. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 1121–1124 (1984).
Seeger, R. C. et al. Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N. Engl. J. Med. 313, 1111–1116 (1985).
Kastan, M. B. & Bartek, J. Cell-cycle checkpoints and cancer. Nature 432, 316–323 (2004).
Weiss, W. A., Aldape, K., Mohapatra, G., Feuerstein, B. G. & Bishop, J. M. Targeted expression of MYCN causes neuroblastoma in transgenic mice. EMBO J. 16, 2985–2995 (1997). The transgenic overexpressing MYCN mouse model for neuroblastoma was shown to have most of the typical features of human neuroblastoma histologically and in molecular characterization, and remains the most widely used spontaneous animal model for investigating the biology and therapy of neuroblastoma.
Armstrong, B. C. & Krystal, G. W. Isolation and characterization of complementary DNA for N-cym, a gene encoded by the DNA strand opposite to N-myc. Cell Growth Differ. 3, 385–390 (1992).
Suenaga, Y. et al. NCYM, a cis-antisense gene of MYCN, encodes a de novo evolved protein that inhibits GSK3β resulting in the stabilization of MYCN in human neuroblastomas. PLoS Genet. 10, e1003996 (2014).
Bresler, S. C. et al. ALK mutations confer differential oncogenic activation and sensitivity to ALK inhibition therapy in neuroblastoma. Cancer Cell 26, 682–694 (2014).
Guan, J. et al. FAM150A and FAM150B are activating ligands for anaplastic lymphoma kinase. eLife 4, e09811 (2015).
Reshetnyak, A. V. et al. Augmentor α and β (FAM150) are ligands of the receptor tyrosine kinases ALK and LTK: hierarchy and specificity of ligand–receptor interactions. Proc. Natl Acad. Sci. USA 112, 15862–15867 (2015).
Berry, T. et al. The ALKF1174L mutation potentiates the oncogenic activity of MYCN in neuroblastoma. Cancer Cell 22, 117–130 (2012).
Zhu, S. et al. Activated ALK collaborates with MYCN in neuroblastoma pathogenesis. Cancer Cell 21, 362–373 (2012).
Mosse, Y. P. et al. Safety and activity of crizotinib for paediatric patients with refractory solid tumours or anaplastic large-cell lymphoma: a Children's Oncology Group phase 1 consortium study. Lancet Oncol. 14, 472–480 (2013).
Mosse, Y. P. Anaplastic lymphoma kinase as a cancer target in pediatric malignancies. Clin. Cancer Res. 22, 546–552 (2016).
Eleveld, T. F. et al. Relapsed neuroblastomas show frequent RAS–MAPK pathway mutations. Nat. Genet. 47, 864–871 (2015).
Lambertz, I. et al. Upregulation of MAPK negative feedback regulators and RET in mutant ALK neuroblastoma: implications for targeted treatment. Clin. Cancer Res. 21, 3327–3339 (2015).
Cazes, A. et al. Activated Alk triggers prolonged neurogenesis and Ret upregulation providing a therapeutic target in ALK-mutated neuroblastoma. Oncotarget 5, 2688–2702 (2014).
Diskin, S. J. et al. Common variation at 6q16 within HACE1 and LIN28B influences susceptibility to neuroblastoma. Nat. Genet. 44, 1126–1130 (2012).
Molenaar, J. J. et al. LIN28B induces neuroblastoma and enhances MYCN levels via let-7 suppression. Nat. Genet. 44, 1199–1206 (2012).
Schnepp, R. W. et al. A LIN28B–RAN–AURKA signaling network promotes neuroblastoma tumorigenesis. Cancer Cell 28, 599–609 (2015).
Powers, J. T. et al. Multiple mechanisms disrupt the let-7 microRNA family in neuroblastoma. Nature 535, 246–251 (2016).
Valentijn, L. J. et al. TERT rearrangements are frequent in neuroblastoma and identify aggressive tumors. Nat. Genet. 47, 1411–1414 (2015).
Peifer, M. et al. Telomerase activation by genomic rearrangements in high-risk neuroblastoma. Nature 526, 700–704 (2015).
Cheung, N. K. et al. Association of age at diagnosis and genetic mutations in patients with neuroblastoma. JAMA 307, 1062–1071 (2012).
Sausen, M. et al. Integrated genomic analyses identify ARID1A and ARID1B alterations in the childhood cancer neuroblastoma. Nat. Genet. 45, 12–17 (2013).
Kumps, C. et al. Focal DNA copy number changes in neuroblastoma target MYCN regulated genes. PLoS ONE 8, e52321 (2013).
Bown, N. et al. Gain of chromosome arm 17q and adverse outcome in patients with neuroblastoma. N. Engl. J. Med. 340, 1954–1961 (1999).
Attiyeh, E. F. et al. Chromosome 1p and 11q deletions and outcome in neuroblastoma. N. Engl. J. Med. 353, 2243–2253 (2005).
Henrich, K. O., Schwab, M. & Westermann, F. 1p36 tumor suppression — a matter of dosage? Cancer Res. 72, 6079–6088 (2012).
DeNardo, D. G. et al. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov. 1, 54–67 (2011).
Asgharzadeh, S. et al. Clinical significance of tumor-associated inflammatory cells in metastatic neuroblastoma. J. Clin. Oncol. 30, 3525–3532 (2012).
Larsson, K. et al. COX/mPGES-1/PGE2 pathway depicts an inflammatory-dependent high-risk neuroblastoma subset. Proc. Natl Acad. Sci. USA 112, 8070–8075 (2015).
Song, L. et al. Vα24-invariant NKT cells mediate antitumor activity via killing of tumor-associated macrophages. J. Clin. Invest. 119, 1524–1536 (2009).
Egler, R. A., Burlingame, S. M., Nuchtern, J. G. & Russell, H. V. Interleukin-6 and soluble interleukin-6 receptor levels as markers of disease extent and prognosis in neuroblastoma. Clin. Cancer Res. 14, 7028–7034 (2008).
Ara, T. et al. Critical role of STAT3 in IL-6-mediated drug resistance in human neuroblastoma. Cancer Res. 73, 3852–3864 (2013).
Ara, T. et al. Interleukin-6 in the bone marrow microenvironment promotes the growth and survival of neuroblastoma cells. Cancer Res. 69, 329–337 (2009).
Liu, Y. et al. Growth and activation of natural killer cells ex vivo from children with neuroblastoma for adoptive cell therapy. Clin. Cancer Res. 19, 2132–2143 (2013).
Delgado, D. C. et al. Genotypes of NK cell KIR receptors, their ligands, and Fcγ receptors in the response of neuroblastoma patients to Hu14.18-IL2 immunotherapy. Cancer Res. 70, 9554–9561 (2010).
De Bernardi, B. et al. Excellent outcome with reduced treatment for infants with disseminated neuroblastoma without MYCN gene amplification. J. Clin. Oncol. 27, 1034–1040 (2009).
Hiyama, E. et al. Correlating telomerase activity levels with human neuroblastoma outcomes. Nat. Med. 1, 249–255 (1995).
Brodeur, G. M. & Bagatell, R. Mechanisms of neuroblastoma regression. Nat. Rev. Clin. Oncol. 11, 704–713 (2014).
Nakagawara, A. et al. Association between high levels of expression of the TRK gene and favorable outcome in human neuroblastoma. N. Engl. J. Med. 328, 847–854 (1993).
Zhu, Y. et al. Dependence receptor UNC5D mediates nerve growth factor depletion-induced neuroblastoma regression. J. Clin. Invest. 123, 2935–2947 (2013).
Vo, K. T. et al. Clinical, biologic, and prognostic differences on the basis of primary tumor site in neuroblastoma: a report from the international neuroblastoma risk group project. J. Clin. Oncol. 32, 3169–3176 (2014).
DuBois, S. G. et al. Metastatic sites in stage IV and IVS neuroblastoma correlate with age, tumor biology, and survival [see comments]. J. Pediatr. Hematol. Oncol. 21, 181–189 (1999).
Taggart, D. R. et al. Prognostic value of the stage 4S metastatic pattern and tumor biology in patients with metastatic neuroblastoma diagnosed between birth and 18 months of age. J. Clin. Oncol. 29, 4358–4364 (2011).
Hero, B. & Schleiermacher, G. Update on pediatric opsoclonus myoclonus syndrome. Neuropediatrics 44, 324–329 (2013).
Strenger, V. et al. Diagnostic and prognostic impact of urinary catecholamines in neuroblastoma patients. Pediatr. Blood Cancer 48, 504–509 (2007).
Franscini, L. C. et al. Pediatric reference intervals for plasma free and total metanephrines established with a parametric approach: relevance to the diagnosis of neuroblastoma. Pediatr. Blood Cancer 62, 587–593 (2015).
Beiske, K. et al. Consensus criteria for sensitive detection of minimal neuroblastoma cells in bone marrow, blood and stem cell preparations by immunocytology and QRT-PCR: recommendations by the International Neuroblastoma Risk Group Task Force. Br. J. Cancer 100, 1627–1637 (2009).
Viprey, V. F. et al. Neuroblastoma mRNAs predict outcome in children with stage 4 neuroblastoma: a European HR-NBL1/SIOPEN study. J. Clin. Oncol. 32, 1074–1083 (2014).
Monclair, T. et al. The International Neuroblastoma Risk Group (INRG) staging system: an INRG Task Force report. J. Clin. Oncol. 27, 298–303 (2009).
Matthay, K. K. et al. Criteria for evaluation of disease extent by 123I-metaiodobenzylguanidine scans in neuroblastoma: a report for the International Neuroblastoma Risk Group (INRG) Task Force. Br. J. Cancer 102, 1319–1326 (2010).
Dubois, S. G. et al. Evaluation of norepinephrine transporter expression and metaiodobenzylguanidine avidity in neuroblastoma: a report from the Children's Oncology Group. Int. J. Mol. Imaging 2012, 250834 (2012).
Biermann, M. et al. Is there a role for PET-CT and SPECT-CT in pediatric oncology? Acta Radiol. 54, 1037–1045 (2013).
Yanik, G. A. et al. Semiquantitative mIBG scoring as a prognostic indicator in patients with stage 4 neuroblastoma: a report from the Children's Oncology Group. J. Nucl. Med. 54, 541–548 (2013).
Sharp, S. E., Shulkin, B. L., Gelfand, M. J., Salisbury, S. & Furman, W. L. 123I-MIBG scintigraphy and 18F-FDG PET in neuroblastoma. J. Nucl. Med. 50, 1237–1243 (2009).
Liu, Y. L. et al. Diagnostic FDG and FDOPA positron emission tomography scans distinguish the genomic type and treatment outcome of neuroblastoma. Oncotarget 7, 18774–18786 (2016).
Gains, J. E. et al. 177Lu-DOTATATE molecular radiotherapy for childhood neuroblastoma. J. Nucl. Med. 52, 1041–1047 (2011).
Shimada, H. et al. Histopathologic prognostic factors in neuroblastic tumors: definition of subtypes of ganglioneuroblastoma and an age-linked classification of neuroblastomas. J. Natl Cancer Inst. 73, 405–416 (1984). This age-linked histological classification of neuroblastoma using histological type, stroma characteristics and nuclear pathology (mitosis-karyorrhexis index) formed the basis for the current international risk stratification and has remained highly prognostic.
Shimada, H. et al. The International Neuroblastoma Pathology Classification (the Shimada system). Cancer 86, 364–372 (1999).
Shimada, H. et al. International neuroblastoma pathology classification for prognostic evaluation of patients with peripheral neuroblastic tumors: a report from the Children's Cancer Group. Cancer 92, 2451–2461 (2001).
Peuchmaur, M. et al. Revision of the International Neuroblastoma Pathology Classification: confirmation of favorable and unfavorable prognostic subsets in ganglioneuroblastoma, nodular. Cancer 98, 2274–2281 (2003).
Ambros, P. F. et al. International consensus for neuroblastoma molecular diagnostics: report from the International Neuroblastoma Risk Group (INRG) Biology Committee. Br. J. Cancer 100, 1471–1482 (2009).
Bagatell, R. et al. Significance of MYCN amplification in international neuroblastoma staging system stage 1 and 2 neuroblastoma: a report from the International Neuroblastoma Risk Group database. J. Clin. Oncol. 27, 365–370 (2009).
Wang, L. L. et al. Augmented expression of MYC and/or MYCN protein defines highly aggressive MYC-driven neuroblastoma: a Children's Oncology Group study. Br. J. Cancer 113, 57–63 (2015).
Valentijn, L. J. et al. Functional MYCN signature predicts outcome of neuroblastoma irrespective of MYCN amplification. Proc. Natl Acad. Sci. USA 109, 19190–19195 (2012).
Ambros, I. M., Brunner, C., Abbasi, R., Frech, C. & Ambros, P. F. Ultra-high density SNParray in neuroblastoma molecular diagnostics. Front. Oncol. 4, 202 (2014).
Kryh, H. et al. Comprehensive SNP array study of frequently used neuroblastoma cell lines; copy neutral loss of heterozygosity is common in the cell lines but uncommon in primary tumors. BMC Genomics 12, 443 (2011).
Janoueix-Lerosey, I. et al. Overall genomic pattern is a predictor of outcome in neuroblastoma. J. Clin. Oncol. 27, 1026–1033 (2009).
Brodeur, G. M. et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment [see comments]. J. Clin. Oncol. 11, 1466–1477 (1993).
Sawada, T. et al. Mass screening of neuroblastoma in infancy. Am. J. Dis. Child. 136, 710–712 (1982).
Sawada, T., Nishi, M., Takeda, T. & Iehara, T. Mass screening for neuroblastoma in Japan. Med. Pediatr. Oncol. 31, 429–434 (1998).
Yamamoto, K. et al. Mass screening and age-specific incidence of neuroblastoma in Saitama Prefecture, Japan. J. Clin. Oncol. 13, 2033–2038 (1995).
Hisashige, A. & NBS Evaluation Group. Effectiveness of nationwide screening program for neuroblastoma in Japan. Glob. J. Health Sci. 6, 94–106 (2014).
Hiyama, E. et al. Effectiveness of screening for neuroblastoma at 6 months of age: a retrospective population-based cohort study. Lancet 371, 1173–1180 (2008).
Kerbl, R. et al. Neuroblastoma mass screening in late infancy: insights into the biology of neuroblastic tumors. J. Clin. Oncol. 21, 4228–4234 (2003).
Nakagawara, A. et al. Different genomic and metabolic patterns between mass screening-positive and mass screening-negative later-presenting neuroblastomas. Cancer 68, 2037–2044 (1991).
Schilling, F. H. et al. Children may not benefit from neuroblastoma screening at 1 year of age. Updated results of the population based controlled trial in Germany. Cancer Lett. 197, 19–28 (2003).
Pinto, N. R. et al. Advances in risk classification and treatment strategies for neuroblastoma. J. Clin. Oncol. 33, 3008–3017 (2015).
Nuchtern, J. G. et al. A prospective study of expectant observation as primary therapy for neuroblastoma in young infants: a Children's Oncology Group study. Ann. Surg. 256, 573–580 (2012).
Rubie, H. et al. Excellent outcome with reduced treatment in infants with nonmetastatic and unresectable neuroblastoma without MYCN amplification: results of the prospective INES 99.1. J. Clin. Oncol. 29, 449–455 (2011).
Hero, B. et al. Localized infant neuroblastomas often show spontaneous regression: results of the prospective trials NB95-S and NB97. J. Clin. Oncol. 26, 1504–1510 (2008).
Iehara, T. et al. Successful treatment of infants with localized neuroblastoma based on their MYCN status. Int. J. Clin. Oncol. 18, 389–395 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02176967 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01728155 (2015).
Schmidt, M. L. et al. Favorable prognosis for patients 12 to 18 months of age with stage 4 nonamplified MYCN neuroblastoma: a Children's Cancer Group Study. J. Clin. Oncol. 23, 6474–6480 (2005).
Defferrari, R. et al. Influence of segmental chromosome abnormalities on survival in children over the age of 12 months with unresectable localised peripheral neuroblastic tumours without MYCN amplification. Br. J. Cancer 112, 290–295 (2015).
Kohler, J. A. et al. Treatment of children over the age of one year with unresectable localised neuroblastoma without MYCN amplification: results of the SIOPEN study. Eur. J. Cancer 49, 3671–3679 (2013).
Marachelian, A. et al. The significance of serial histopathology in a residual mass for outcome of intermediate risk stage 3 neuroblastoma. Pediatr. Blood Cancer 58, 675–681 (2012).
Matthay, K. K. et al. Successful treatment of stage III neuroblastoma based on prospective biologic staging: a Children's Cancer Group study. J. Clin. Oncol. 16, 1256–1264 (1998).
Ladenstein, R. et al. Multivariate analysis of risk factors in stage 4 neuroblastoma patients over the age of one year treated with megatherapy and stem-cell transplantation: a report from the European Bone Marrow Transplantation Solid Tumor Registry. J. Clin. Oncol. 16, 953–965 (1998).
Ladenstein, R. et al. Randomized trial of prophylactic granulocyte colony-stimulating factor during rapid COJEC induction in pediatric patients with high-risk neuroblastoma: the European HR-NBL1/SIOPEN study. J. Clin. Oncol. 28, 3516–3524 (2010).
Park, J. R. et al. A phase 3 randomized clinical trial (RCT) of tandem myeloablative autologous stem cell transplant (ASCT) using peripheral blood stem cell (PBSC) as consolidation therapy for high-risk neuroblastoma (HR-NB): a Children's Oncology Group (COG) study. J. Clin. Oncol. Abstr. 34, LBA3 (2016).
Berthold, F. et al. Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lancet Oncol. 6, 649–658 (2005).
Pearson, A. D. et al. High-dose rapid and standard induction chemotherapy for patients aged over 1 year with stage 4 neuroblastoma: a randomised trial. Lancet Oncol. 9, 247–256 (2008).
Zage, P. E. et al. Outcomes of the POG 9340/9341/9342 trials for children with high-risk neuroblastoma: a report from the Children's Oncology Group. Pediatr. Blood Cancer 51, 747–753 (2008).
Kreissman, S. G. et al. Purged versus non-purged peripheral blood stem-cell transplantation for high-risk neuroblastoma (COG A3973): a randomised phase 3 trial. Lancet Oncol. 14, 999–1008 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01704716 (2016).
Garaventa, A. et al. A phase II study of topotecan with vincristine and doxorubicin in children with recurrent/refractory neuroblastoma. Cancer 98, 2488–2494 (2003).
Bagatell, R. et al. Phase II study of irinotecan and temozolomide in children with relapsed or refractory neuroblastoma: a Children's Oncology Group study. J. Clin. Oncol. 29, 208–213 (2011).
Matthay, K. K. et al. Phase II study on the effect of disease sites, age, and prior therapy on response to iodine-131-metaiodobenzylguanidine therapy in refractory neuroblastoma. J. Clin. Oncol. 25, 1054–1060 (2007).
Zhou, M. J. et al. Different outcomes for relapsed versus refractory neuroblastoma after therapy with 131I-metaiodobenzylguanidine (131I-mIBG). Eur. J. Cancer 51, 2465–2472 (2015).
Mullassery, D., Farrelly, P. & Losty, P. D. Does aggressive surgical resection improve survival in advanced stage 3 and 4 neuroblastoma? A systematic review and meta-analysis. Pediatr. Hematol. Oncol. 31, 703–716 (2014).
Haas-Kogan, D. A. et al. Impact of radiotherapy for high-risk neuroblastoma: a Children's Cancer Group study. Int. J. Radiat. Oncol. Biol. Phys. 56, 28–39 (2003).
Adkins, E. S. et al. Efficacy of complete resection for high-risk neuroblastoma: a Children's Cancer Group study. J. Pediatr. Surg. 39, 931–936 (2004).
Park, J. R. et al. Outcome of high-risk stage 3 neuroblastoma with myeloablative therapy and 13-cis-retinoic acid: a report from the Children's Oncology Group. Pediatr. Blood Cancer 52, 44–50 (2009).
Simon, T., Haberle, B., Hero, B., von Schweinitz, D. & Berthold, F. Role of surgery in the treatment of patients with stage 4 neuroblastoma age 18 months or older at diagnosis. J. Clin. Oncol. 31, 752–758 (2013).
Wolden, S. L. et al. Local control with multimodality therapy for stage 4 neuroblastoma. Int. J. Radiat. Oncol. Biol. Phys. 46, 969–974 (2000).
Pritchard, J. et al. High dose melphalan in the treatment of advanced neuroblastoma: results of a randomised trial (ENSG-1) by the European Neuroblastoma Study Group. Pediatr. Blood Cancer 44, 348–357 (2005).
Ladenstein, R. L. et al. Busulphan-melphalan as a myeloablative therapy (MAT) for high-risk neuroblastoma: results from the HR-NBL1/SIOPEN trial. J. Clin Oncol. Abstr. 29 (Suppl.), 2 (2011).
George, R. E. et al. High-risk neuroblastoma treated with tandem autologous peripheral-blood stem cell-supported transplantation: long-term survival update. J. Clin. Oncol. 24, 2891–2896 (2006).
Seif, A. E. et al. A pilot study of tandem high-dose chemotherapy with stem cell rescue as consolidation for high-risk neuroblastoma: Children's Oncology Group study ANBL00P1. Bone Marrow Transplant. 48, 947–952 (2013).
Matthay, K. K. et al. Phase I dose escalation of iodine-131-metaiodobenzylguanidine with myeloablative chemotherapy and autologous stem-cell transplantation in refractory neuroblastoma: a new approaches to neuroblastoma therapy consortium study. J. Clin. Oncol. 24, 500–506 (2006).
Yanik, G. A. et al. 131I-metaiodobenzylguanidine with intensive chemotherapy and autologous stem cell transplantation for high-risk neuroblastoma. A new approaches to neuroblastoma therapy (NANT) phase II study. Biol. Blood Marrow Transplant. 21, 673–681 (2015).
Park, J. R. et al. Pilot induction regimen incorporating pharmacokinetically guided topotecan for treatment of newly diagnosed high-risk neuroblastoma: a Children's Oncology Group study. J. Clin. Oncol. 29, 4351–4357 (2011).
Yu, A. L. et al. Anti-GD2 antibody with GM-CSF, interleukin-2, and isotretinoin for neuroblastoma. N. Engl. J. Med. 363, 1324–1334 (2010). This randomized controlled trial in patients with high-risk neuroblastoma after AHSCT showed that maintenance therapy with isotretinoin with the addition of chimeric anti-GD2 monoclonal antibody, GM-CSF and IL-2 resulted in a significantly higher EFS and overall survival than isotretinoin alone.
Siebert, N. et al. Pharmacokinetics and pharmacodynamics of ch14.18/CHO in relapsed/refractory high-risk neuroblastoma patients treated by long-term infusion in combination with IL-2. MAbs 8, 604–616 (2016).
Navid, F. et al. Phase I trial of a novel anti-GD2 monoclonal antibody, Hu14.18K322A, designed to decrease toxicity in children with refractory or recurrent neuroblastoma. J. Clin. Oncol. 32, 1445–1452 (2014).
Shusterman, S. et al. Antitumor activity of hu14.18-IL2 in patients with relapsed/refractory neuroblastoma: a Children's Oncology Group (COG) phase II study. J. Clin. Oncol. 28, 4969–4975 (2010).
Maurer, B. J. et al. Phase I trial of fenretinide delivered orally in a novel organized lipid complex in patients with relapsed/refractory neuroblastoma: a report from the new approaches to neuroblastoma therapy (NANT) consortium. Pediatr. Blood Cancer 60, 1801–1808 (2013).
Kushner, B. H. et al. Phase I trial of a bivalent gangliosides vaccine in combination with β-glucan for high-risk neuroblastoma in second or later remission. Clin. Cancer Res. 20, 1375–1382 (2014).
Bassiri, H. et al. Translational development of difluoromethylornithine (DFMO) for the treatment of neuroblastoma. Transl Pediatr. 4, 226–238 (2015).
London, W. B. et al. Phase II randomized comparison of topotecan plus cyclophosphamide versus topotecan alone in children with recurrent or refractory neuroblastoma: a Children's Oncology Group study. J. Clin. Oncol. 28, 3808–3815 (2010).
Kushner, B. H., Kramer, K., Modak, S. & Cheung, N. K. Irinotecan plus temozolomide for relapsed or refractory neuroblastoma. J. Clin. Oncol. 24, 5271–5276 (2006).
Di Giannatale, A. et al. Phase II study of temozolomide in combination with topotecan (TOTEM) in relapsed or refractory neuroblastoma: a European Innovative Therapies for Children with Cancer-SIOP-European Neuroblastoma study. Eur. J. Cancer 50, 170–177 (2014).
Wilson, J. S., Gains, J. E., Moroz, V., Wheatley, K. & Gaze, M. N. A systematic review of 131I-meta iodobenzylguanidine molecular radiotherapy for neuroblastoma. Eur. J. Cancer 50, 801–815 (2014).
DuBois, S. G. et al. Phase I study of vincristine, irinotecan, and 131I-metaiodobenzylguanidine for patients with relapsed or refractory neuroblastoma: a new approaches to neuroblastoma therapy trial. Clin. Cancer Res. 18, 2679–2686 (2012).
DuBois, S. G. et al. Phase I study of vorinostat as a radiation sensitizer with 131I-metaiodobenzylguanidine (131I-MIBG) for patients with relapsed or refractory neuroblastoma. Clin. Cancer Res. 21, 2715–2721 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01606878 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01742286 (2016).
Infarinato, N. R. et al. The ALK/ROS1 inhibitor PF-06463922 overcomes primary resistance to crizotinib in ALK-driven neuroblastoma. Cancer Discov. 6, 96–107 (2015).
DuBois, S. G. et al. Phase I study of the Aurora A kinase inhibitor alisertib in combination with irinotecan and temozolomide for patients with relapsed or refractory neuroblastoma: a NANT (new approaches to neuroblastoma therapy) trial. J. Clin. Oncol. 34, 1368–1375 (2016).
Puissant, A. et al. Targeting MYCN in neuroblastoma by BET bromodomain inhibition. Cancer Discov. 3, 308–323 (2013).
Henssen, A. et al. Targeting MYCN-driven transcription by BET-bromodomain inhibition. Clin. Cancer Res. 22, 2470–2481 (2015).
Evageliou, N. F. et al. Polyamine antagonist therapies inhibit neuroblastoma initiation and progression. Clin. Cancer Res. 22, 4391–4401 (2016).
Louis, C. U. et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood 118, 6050–6056 (2011).
Kramer, K., Kushner, B., Heller, G. & Cheung, N. K. Neuroblastoma metastatic to the central nervous system. the Memorial Sloan-Kettering Cancer Center Experience and a literature review. Cancer 91, 1510–1519 (2001).
Matthay, K. K. et al. Central nervous system metastases in neuroblastoma: radiologic, clinical, and biologic features in 23 patients. Cancer 98, 155–165 (2003).
Kramer, K. et al. Compartmental intrathecal radioimmunotherapy: results for treatment for metastatic CNS neuroblastoma. J. Neurooncol. 97, 409–418 (2010).
De Bernardi, B. et al. Neuroblastoma with symptomatic epidural compression in the infant: the AIEOP experience. Pediatr. Blood Cancer 61, 1369–1375 (2014).
Mitchell, W. G. et al. Effect of increased immunosuppression on developmental outcome of opsoclonus myoclonus syndrome (OMS). J. Child Neurol. 30, 976–982 (2015).
Bourdeaut, F. et al. VIP hypersecretion as primary or secondary syndrome in neuroblastoma: a retrospective study by the Societe Francaise des Cancers de l'Enfant (SFCE). Pediatr. Blood Cancer 52, 585–590 (2009).
Katzenstein, H. M., Kent, P. M., London, W. B. & Cohn, S. L. Treatment and outcome of 83 children with intraspinal neuroblastoma: the Pediatric Oncology Group experience. J. Clin. Oncol. 19, 1047–1055 (2001).
Laverdiere, C. et al. Long-term outcomes in survivors of neuroblastoma: a report from the Childhood Cancer Survivor Study. J. Natl Cancer Inst. 101, 1131–1140 (2009). This study reports the health outcomes of 954 5-year neuroblastoma survivors who were diagnosed between 1970 and 1986, showing the increase in late mortality, secondary malignancies and chronic health conditions compared with sibling controls.
Wilson, C. L. et al. Renal carcinoma after childhood cancer: a report from the childhood cancer survivor study. J. Natl Cancer Inst. 105, 504–508 (2013).
Cohen, L. E. et al. Late effects in children treated with intensive multimodal therapy for high-risk neuroblastoma: high incidence of endocrine and growth problems. Bone Marrow Transplant. 49, 502–508 (2014).
Gurney, J. G. et al. Hearing loss, quality of life, and academic problems in long-term neuroblastoma survivors: a report from the Children's Oncology Group. Pediatrics 120, e1229–e1236 (2007).
Applebaum, M. A. et al. Second malignancies in patients with neuroblastoma: the effects of risk-based therapy. Pediatr. Blood Cancer 62, 128–133 (2015).
Willi, S. M. et al. Growth in children after bone marrow transplantation for advanced neuroblastoma compared with growth after transplantation for leukemia or aplastic anemia. J. Pediatr. 120, 726–732 (1992).
Hobbie, W. L., Mostoufi, S. M., Carlson, C. A., Gruccio, D. & Ginsberg, J. P. Prevalence of advanced bone age in a cohort of patients who received cis-retinoic acid for high-risk neuroblastoma. Pediatr. Blood Cancer 56, 474–476 (2011).
van Santen, H. M., de Kraker, J., van Eck, B. L., de Vijlder, J. J. & Vulsma, T. High incidence of thyroid dysfunction despite prophylaxis with potassium iodide during 131I-meta-iodobenzylguanidine treatment in children with neuroblastoma. Cancer 94, 2081–2089 (2002).
van Waas, M. et al. Abdominal radiotherapy: a major determinant of metabolic syndrome in nephroblastoma and neuroblastoma survivors. PLoS ONE 7, e52237 (2012).
Meacham, L. R. et al. Diabetes mellitus in long-term survivors of childhood cancer. Increased risk associated with radiation therapy: a report for the Childhood Cancer Survivor Study. Arch. Intern. Med. 169, 1381–1388 (2009).
Lord, C. J., Tutt, A. N. & Ashworth, A. Synthetic lethality and cancer therapy: lessons learned from the development of PARP inhibitors. Annu. Rev. Med. 66, 455–470 (2015).
Schramm, A. et al. Mutational dynamics between primary and relapse neuroblastomas. Nat. Genet. 47, 872–877 (2015).
Schleiermacher, G. et al. Emergence of new ALK mutations at relapse of neuroblastoma. J. Clin. Oncol. 32, 2727–2734 (2014).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
Bettegowda, C. et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl Med. 6, 224ra224 (2014).
Huang, S. Y. et al. Patient-specific dosimetry using pretherapy [124I]m-iodobenzylguanidine ([124I]mIBG) dynamic PET/CT imaging before [131I]mIBG targeted radionuclide therapy for neuroblastoma. Mol. Imaging Biol. 17, 284–294 (2015).
Doran, M. G. et al. Applying Zr-transferrin to study the pharmacology of inhibitors to BET bromodomain containing proteins. Mol. Pharm. 13, 683–688 (2016).
Gustafson, W. C. et al. Drugging MYCN through an allosteric transition in Aurora kinase A. Cancer Cell 26, 414–427 (2014).
Ross, C. J. et al. Genetic variants in TPMT and COMT are associated with hearing loss in children receiving cisplatin chemotherapy. Nat. Genet. 41, 1345–1349 (2009).
Roco, A., Cayun, J., Contreras, S., Stojanova, J. & Quinones, L. Can pharmacogenetics explain efficacy and safety of cisplatin pharmacotherapy? Front. Genet. 5, 391 (2014).
Banerjee, S. A., Hoppe, P., Brilliant, M. & Chikaraishi, D. M. 5′ flanking sequences of the rat tyrosine hydroxylase gene target accurate tissue-specific, developmental, and transsynaptic expression in transgenic mice. J. Neurosci. 12, 4460–4467 (1992).
Teitz, T. et al. Th-MYCN mice with caspase-8 deficiency develop advanced neuroblastoma with bone marrow metastasis. Cancer Res. 73, 4086–4097 (2013).
Hackett, C. S. et al. Expression quantitative trait loci and receptor pharmacology implicate Arg1 and the GABA-A receptor as therapeutic targets in neuroblastoma. Cell Rep. 9, 1034–1046 (2014).
Althoff, K. et al. A Cre-conditional MYCN-driven neuroblastoma mouse model as an improved tool for preclinical studies. Oncogene 34, 3357–3368 (2015).
Lo Piccolo, M. S., Cheung, N. K. & Cheung, I. Y. GD2 synthase: a new molecular marker for detecting neuroblastoma. Cancer 92, 924–931 (2001).
Wu, Z. L., Schwartz, E., Seeger, R. & Ladisch, S. Expression of GD2 ganglioside by untreated primary human neuroblastomas. Cancer Res. 46, 440–443 (1986).
Cheung, N. K. et al. Key role for myeloid cells: phase II results of anti-GD2 antibody 3F8 plus granulocyte-macrophage colony-stimulating factor for chemoresistant osteomedullary neuroblastoma. J. Int. Cancer 135, 2199–2205 (2014).
Cheung, N. K. et al. FCGR2A polymorphism is correlated with clinical outcome after immunotherapy of neuroblastoma with anti-GD2 antibody and granulocyte macrophage colony-stimulating factor. J. Clin. Oncol. 24, 2885–2890 (2006).
Tarek, N. et al. Unlicensed NK cells target neuroblastoma following anti-GD2 antibody treatment. J. Clin. Invest. 122, 3260–3270 (2012).
Pule, M. A. et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat. Med. 14, 1264–1270 (2008).
Long, A. H. et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat. Med. 21, 581–590 (2015).
Decarolis, B. et al. Iodine-123 metaiodobenzylguanidine scintigraphy scoring allows prediction of outcome in patients with stage 4 neuroblastoma: results of the Cologne interscore comparison study. J. Clin. Oncol. 31, 944–951 (2013).
Bronner, M. E. & Simoes-Costa, M. The neural crest migrating into the twenty-first century. Curr. Top. Dev. Biol. 116, 115–134 (2016).
Simoes-Costa, M. & Bronner, M. E. Establishing neural crest identity: a gene regulatory recipe. Development 142, 242–257 (2015).
Marshall, G. M. et al. The prenatal origins of cancer. Nat. Rev. Cancer 14, 277–289 (2014).
Acknowledgements
The authors thank S. Cho, University of California, San Francisco, USA, for preparing the photomicrographs used in Figure 6.
Author information
Authors and Affiliations
Contributions
Introduction (K.K.M.); Epidemiology (J.M.M.); Mechanisms/pathophysiology (C.L.M. and W.A.W.); Diagnosis, screening and prevention (K.K.M., G.S. and A.N.); Management (K.K.M. and G.S.); Quality of life (L.D.); Outlook (K.K.M. and J.M.M.); Overview of the Primer (K.K.M.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Matthay, K., Maris, J., Schleiermacher, G. et al. Neuroblastoma. Nat Rev Dis Primers 2, 16078 (2016). https://doi.org/10.1038/nrdp.2016.78
Published:
DOI: https://doi.org/10.1038/nrdp.2016.78
This article is cited by
-
Clinical characteristics and prognoses in pediatric neuroblastoma with bone or liver metastasis: data from the SEER 2010–2019
BMC Pediatrics (2024)
-
Approval of DFMO for high-risk neuroblastoma patients demonstrates a step of success to target MYC pathway
British Journal of Cancer (2024)
-
Microfluidic model of the alternative vasculature in neuroblastoma
In vitro models (2024)
-
MUW researcher of the month
Wiener klinische Wochenschrift (2024)
-
Bilateral adrenal neuroblastoma: peculiar pattern of a rare pediatric presentation
Discover Oncology (2024)