Abstract
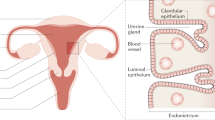
Uterine fibroids (also known as leiomyomas or myomas) are common clonal neoplasms of the uterus. Fibroids have both smooth muscle and fibroblast components, in addition to a substantial amount of fibrous extracellular matrix, which all contribute to the pathogenetic process. Fibroids are extremely heterogeneous in their pathophysiology, size, location and clinical symptomatology. They are also a part of a range of disease in which some variants have facets of malignant behaviour but overall are benign. Risk for fibroids is associated with race; black women have a higher risk of developing fibroids earlier in life than their white counterparts and also develop more-severe forms of the disease. Clinically, fibroids account for one-third to half of all hysterectomies and are associated with substantial morbidity and health care costs for women of reproductive age. Indeed, current treatments are primarily surgical and interventional; approximately three-quarters of all fibroid treatments are hysterectomies. However, clinical innovations are emerging in the use of progesterone receptor modulators as a medical therapy. New information is rapidly accumulating about the genetic subgroups that lead to fibroid formation, which might aid further understanding of the clinical heterogeneity of this disease and lead to individualized treatments. This information is a crucial development given the current lack of high-quality evidence on which to base therapeutic decisions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Stewart, E. A. Uterine Fibroids: the Complete Guide (Johns Hopkins Univ. Press, 2007).
Merrill, R. M. Hysterectomy surveillance in the United States, 1997 through 2005. Med. Sci. Monit. 14, CR24–CR31 (2008).
Borah, B. J., Laughlin-Tommaso, S. K., Myers, E. R., Yao, X. & Stewart, E. A. Association between patient characteristics and treatment procedure among patients with uterine leiomyomas. Obstet. Gynecol. 127, 67–77 (2016).
Cardozo, E. R. et al. The estimated annual cost of uterine leiomyomata in the United States. Am. J. Obstet. Gynecol. 206, 211.e1–211.e9 (2012).
Peddada, S. D. et al. Growth of uterine leiomyomata among premenopausal black and white women. Proc. Natl Acad. Sci. USA 105, 19887–19892 (2008). This paper reports fibroid growth rates using MRI for a racially diverse cohort and examines key differences in growth parameters.
Stewart, E. A. & Nowak, R. A. Leiomyoma-related bleeding: a classic hypothesis updated for the molecular era. Hum. Reprod. Update 2, 295–306 (1996).
Cramer, S. F. & Patel, A. The frequency of uterine leiomyomas. Am. J. Clin. Pathol. 94, 435–438 (1990).
Baird, D. D., Dunson, D. B., Hill, M. C., Cousins, D. & Schectman, J. M. High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am. J. Obstet. Gynecol. 188, 100–107 (2003). This study documents the difference in ultrasonography-diagnosed fibroid disease for black women and the differences between self-report and imaging diagnosis.
Marshall, L. M. et al. Variation in the incidence of uterine leiomyoma among premenopausal women by age and race. Obstet. Gynecol. 90, 967–973 (1997).
Downes, E. et al. The burden of uterine fibroids in five European countries. Eur. J. Obstet. Gynecol. Reprod. Biol. 152, 96–102 (2010).
Borah, B. J., Nicholson, W. K., Bradley, L. & Stewart, E. A. The impact of uterine leiomyomas: a national survey of affected women. Am. J. Obstet. Gynecol. 209, 319.e1–319.e20 (2013).
Laughlin, S. K., Baird, D. D., Savitz, D. A., Herring, A. H. & Hartmann, K. E. Prevalence of uterine leiomyomas in the first trimester of pregnancy: an ultrasound-screening study. Obstet. Gynecol. 113, 630–635 (2009).
Marsh, E. E. et al. Racial differences in fibroid prevalence and ultrasound findings in asymptomatic young women (18–30 years old): a pilot study. Fertil. Steril. 99, 1951–1957 (2013).
Selo-Ojeme, D. et al. The incidence of uterine leiomyoma and other pelvic ultrasonographic findings in 2,034 consecutive women in a north London hospital. J. Obstet. Gynaecol. 28, 421–423 (2008).
Myers, S. L. et al. Self-report versus ultrasound measurement of uterine fibroid status. J. Womens Health (Larchmt) 21, 285–293 (2012).
Wechter, M. E., Stewart, E. A., Myers, E. R., Kho, R. M. & Wu, J. M. Leiomyoma-related hospitalization and surgery: prevalence and predicted growth based on population trends. Am. J. Obstet. Gynecol. 205, 492.e1–492.e5 (2011).
Butt, J., Jeffery, S. T. & Van der Spuy, Z. M. An audit of indications and complications associated with elective hysterectomy at a public service hospital in South Africa. Int. J. Gynaecol. Obstet. 116, 112–116 (2012).
Qi, L. et al. Relationship between hysterectomy and admixture in African American women. Am. J. Obstet. Gynecol. 208, 279.e1–279.e7 (2013).
Wise, L. et al. African ancestry and genetic risk for uterine leiomyomata. Am. J. Epidemiol. 176, 1159–1168 (2012).
Laughlin, S. K. et al. Pregnancy-related fibroid reduction. Fertil. Steril. 94, 2421–2423 (2010).
Huyck, K. L. et al. The impact of race as a risk factor for symptom severity and age at diagnosis of uterine leiomyomata among affected sisters. Am. J. Obstet. Gynecol. 198, 168.e1–168.e9 (2008).
Baird, D. D. & Newbold, R. Prenatal diethylstilbestrol (DES) exposure is associated with uterine leiomyoma development. Reprod. Toxicol. 20, 81–84 (2005).
Lumbiganon, P. et al. Protective effect of depot-medroxyprogesterone acetate on surgically treated uterine leiomyomas: a multicentre case–control study. Br. J. Obstet. Gynaecol. 103, 909–914 (1996).
Wise, L. A. et al. Reproductive factors, hormonal contraception, and risk of uterine leiomyomata in African-American women: a prospective study. Am. J. Epidemiol. 159, 113–123 (2004).
Marshall, L. M. et al. A prospective study of reproductive factors and oral contraceptive use in relation to the risk of uterine leiomyomata. Fertil. Steril. 70, 432–439 (1998).
Wise, L. A. et al. Intake of fruit, vegetables, and carotenoids in relation to risk of uterine leiomyomata. Am. J. Clin. Nutr. 94, 1620–1631 (2011).
Wise, L. A., Radin, R. G., Palmer, J. R., Kumanyika, S. K. & Rosenberg, L. A prospective study of dairy intake and risk of uterine leiomyomata. Am. J. Epidemiol. 171, 221–232 (2010).
Nesby-O'Dell, S. et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: Third National Health and Nutrition Examination Survey: 1988–1994. Am. J. Clin. Nutr. 76, 187–192 (2002).
Eggemoen, A. R. et al. Vitamin D deficiency and supplementation in pregnancy in a multiethnic population-based cohort. BMC Pregnancy Childbirth 16, 7 (2016).
Baird, D. D., Hill, M. C., Schectman, J. M. & Hollis, B. W. Vitamin D and the risk of uterine fibroids. Epidemiology 24, 447–453 (2013). This study examines the link between vitamin D deficiency and fibroids, a key variable that might explain the racial disparities of this disease.
Laughlin, S. K., Schroeder, J. C. & Baird, D. D. New directions in the epidemiology of uterine fibroids. Semin. Reprod. Med. 28, 204–217 (2010).
Faerstein, E., Szklo, M. & Rosenshein, N. Risk factors for uterine leiomyoma: a practice-based case–control study. I. African-American heritage, reproductive history, body size, and smoking. Am. J. Epidemiol. 153, 1–10 (2001).
Moore, K. R., Smith, J. S., Laughlin-Tommaso, S. K. & Baird, D. D. Cervical neoplasia-related factors and decreased prevalence of uterine fibroids among a cohort of African American women. Fertil. Steril. 101, 208–214 (2014).
Bulun, S. E. Uterine fibroids. N. Engl. J. Med. 369, 1344–1355 (2013). This paper provides an updated review of the pathogenesis of fibroids.
Ono, M. et al. Paracrine activation of WNT/β-catenin pathway in uterine leiomyoma stem cells promotes tumor growth. Proc. Natl Acad. Sci. USA 110, 17053–17058 (2013).
Zhou, S. et al. Proteomics identification of annexin A2 as a key mediator in the metastasis and proangiogenesis of endometrial cells in human adenomyosis. Mol. Cell. Proteomics 11, M112.017988 (2012).
Holdsworth-Carson, S. J., Zaitseva, M., Vollenhoven, B. J. & Rogers, P. A. Clonality of smooth muscle and fibroblast cell populations isolated from human fibroid and myometrial tissues. Mol. Hum. Reprod. 20, 250–259 (2014).
Holdsworth-Carson, S. J., Zaitseva, M., Girling, J. E., Vollenhoven, B. J. & Rogers, P. A. Common fibroid-associated genes are differentially expressed in phenotypically dissimilar cell populations isolated from within human fibroids and myometrium. Reproduction 147, 683–692 (2014). This is a key paper delineating the cell types that compose fibroids.
Leppert, P. C. et al. Comparative ultrastructure of collagen fibrils in uterine leiomyomas and normal myometrium. Fertil. Steril. 82 (Suppl. 3), 1182–1187 (2004).
Rogers, R. et al. Mechanical homeostasis is altered in uterine leiomyoma. Am. J. Obstet. Gynecol. 198, 474.e1–474.e11 (2008). This is a key paper linking the ECM of fibroids to its functional significance in this disease.
Ono, M. et al. Role of stem cells in human uterine leiomyoma growth. PLoS ONE 7, e36935 (2012).
Koumas, L., King, A. E., Critchley, H. O., Kelly, R. W. & Phipps, R. P. Fibroblasts heterogeneity: existence of functionally distinct Thy I+ and Thy I, human female reproductive tract fibroblasts. Am. J. Pathol. 159, 925–935 (2001).
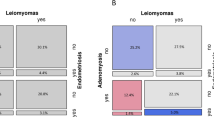
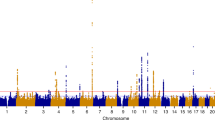
Mehine, M. et al. Characterization of uterine leiomyomas by whole-genome sequencing. N. Engl. J. Med. 369, 43–53 (2013). This paper proposes the key genetic subgroups for fibroids.
Mehine, M., Makinen, N., Heinonen, H. R., Aaltonen, L. A. & Vahteristo, P. Genomics of uterine leiomyomas: insights from high-throughput sequencing. Fertil. Steril. 102, 621–629 (2014).
Makinen, N. et al. MED12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 334, 252–255 (2011).
Halder, S. K. et al. Novel MED12 gene somatic mutations in women from the Southern United States with symptomatic uterine fibroids. Mol. Genet. Genomics 290, 505–511 (2015).
Borggrefe, T. & Yue, X. Interactions between subunits of the Mediator complex with gene-specific transcription factors. Semin. Cell Dev. Biol. 22, 759–768 (2011).
Turunen, M. et al. Uterine leiomyoma-linked MED12 mutations disrupt mediator-associated CDK activity. Cell Rep. 7, 654–660 (2014).
Markowski, D. N. et al. MED12 mutations in uterine fibroids-their relationship to cytogenetic subgroups. Int. J. Cancer 131, 1528–1536 (2012).
Perot, G. et al. MED12 alterations in both human benign and malignant uterine soft tissue tumors. PLoS ONE 7, e40015 (2012).
Makinen, N. et al. MED12 exon 2 mutations in histopathological uterine leiomyoma variants. Eur. J. Hum. Genet. 21, 1300–1303 (2013).
Gattas, G. J., Quade, B. J., Nowak, R. A. & Morton, C. C. HMGIC expression in human adult and fetal tissues and in uterine leiomyomata. Genes Chromosomes Cancer 25, 316–322 (1999).
Hodge, J. C. et al. Uterine leiomyomata and decreased height: a common HMGA2 predisposition allele. Hum. Genet. 125, 257–263 (2009).
Markowski, D. N. et al. HMGA2 and p14Arf: major roles in cellular senescence of fibroids and therapeutic implications. Anticancer Res. 31, 753–761 (2011).
Peng, Y. et al. Antiproliferative effects by Let-7 repression of high-mobility group A2 in uterine leiomyoma. Mol. Cancer Res. 6, 663–673 (2008).
Moravek, M. B. & Bulun, S. E. Endocrinology of uterine fibroids: steroid hormones, stem cells, and genetic contribution. Curr. Opin. Obstetr. Gynecol. 27, 276–283 (2015).
Bertsch, E. et al. MED12 and HMGA2 mutations: two independent genetic events in uterine leiomyoma and leiomyosarcoma. Mod. Pathol. 27, 1144–1153 (2014).
Zaitseva, M., Vollenhoven, B. J. & Rogers, P. A. Retinoids regulate genes involved in retinoic acid synthesis and transport in human myometrial and fibroid smooth muscle cells. Hum. Reprod. 23, 1076–1086 (2008).
Kurman, R. J. Blaustein's Pathology of the Female Genital Tract (Springer, 2002).
Sanz-Ortega, J., Vocke, C., Stratton, P., Linehan, W. M. & Merino, M. J. Morphologic and molecular characteristics of uterine leiomyomas in hereditary leiomyomatosis and renal cancer (HLRCC) syndrome. Am. J. Surg. Pathol. 37, 74–80 (2013).
Walker, C. L. & Stewart, E. A. Uterine fibroids: the elephant in the room. Science 308, 1589–1592 (2005).
Navarro, A. et al. Genome-wide DNA methylation indicates silencing of tumor suppressor genes in uterine leiomyoma. PLoS ONE 7, e33284 (2012).
Maekawa, R. et al. Genome-wide DNA methylation analysis reveals a potential mechanism for the pathogenesis and development of uterine leiomyomas. PLoS ONE 8, e66632 (2013).
Benassayag, C. et al. Estrogen receptors (ERα/ERβ) in normal and pathological growth of the human myometrium: pregnancy and leiomyoma. Am. J. Physiol. 276, E1112–E1118 (1999).
Hsieh, Y. Y. et al. Estrogen receptor thymine-adenine dinucleotide repeat polymorphism is associated with susceptibility to leiomyoma. Fertil. Steril. 79, 96–99 (2003).
Ishikawa, H. et al. High aromatase expression in uterine leiomyoma tissues of African-American women. J. Clin. Endocrinol. Metab. 94, 1752–1756 (2009).
Donnez, J. et al. Ulipristal acetate versus leuprolide acetate for uterine fibroids. N. Engl. J. Med. 366, 421–432 (2012).
Donnez, J. et al. Long-term treatment of uterine fibroids with ulipristal acetate. Fertil. Steril. 101, 1565–1573.e18 (2014).
Ishikawa, H. et al. Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology 151, 2433–2442 (2010).
Yin, P. et al. Genome-wide progesterone receptor binding: cell type-specific and shared mechanisms in T47D breast cancer cells and primary leiomyoma cells. PLoS ONE 7, e29021 (2012).
Yin, P. et al. Transcription factor KLF11 integrates progesterone receptor signaling and proliferation in uterine leiomyoma cells. Cancer Res. 70, 1722–1730 (2010).
Hoekstra, A. V. et al. Progestins activate the AKT pathway in leiomyoma cells and promote survival. J. Clin. Endocrinol. Metab. 94, 1768–1774 (2009).
Brakta, S., Diamond, J. S., Al-Hendy, A., Diamond, M. P. & Halder, S. K. Role of vitamin D in uterine fibroid biology. Fertil. Steril. 104, 698–706 (2015).
Halder, S. K., Goodwin, J. S. & Al-Hendy, A. 1,25-Dihydroxyvitamin D3 reduces TGF-β3-induced fibrosis-related gene expression in human uterine leiomyoma cells. J. Clin. Endocrinol. Metab. 96, E754–E762 (2011).
Zaitseva, M., Vollenhoven, B. J. & Rogers, P. A. Retinoic acid pathway genes show significantly altered expression in uterine fibroids when compared with normal myometrium. Mol. Hum. Reprod. 13, 577–585 (2007).
Catherino, W. H. & Malik, M. Uterine leiomyomas express a molecular pattern that lowers retinoic acid exposure. Fertil. Steril. 87, 1388–1398 (2007).
Wei, J. et al. Ethnic differences in expression of the dysregulated proteins in uterine leiomyomata. Hum. Reprod. 21, 57–67 (2006).
Lan, M. et al. In vivo evidence of the androgen receptor in association with myometrial cell proliferation and apoptosis. Reprod. Sci. 23, 264–271 (2016).
Wong, J., Gold, E., Johnson, W. & Lee, J. Circulating sex hormones and risk of uterine fibroids: Study of Women's Health Across the Nation (SWAN). J. Clin. Endocrinol. Metab. 101, 123–130 (2016).
Yin, H. et al. Expression profiling of nuclear receptors identifies key roles of NR4A subfamily in uterine fibroids. Mol. Endocrinol. 27, 726–740 (2013).
Zaitseva, M. et al. Aberrant expression and regulation of NR2F2 and CTNNb1 in uterine fibroids. Reproduction 146, 91–102 (2013).
Weston, G. et al. Fibroids display an anti-angiogenic gene expression profile when compared with adjacent myometrium. Mol. Hum. Reprod. 9, 541–549 (2003).
Hodge, J. C. et al. Expression profiling of uterine leiomyomata cytogenetic subgroups reveals distinct signatures in matched myometrium: transcriptional profilingof the t(12;14) and evidence in support of predisposing genetic heterogeneity. Hum. Mol. Genet. 21, 2312–2329 (2012).
Fleischer, R., Weston, G. C., Vollenhoven, B. J. & Rogers, P. A. Pathophysiology of fibroid disease: angiogenesis and regulation of smooth muscle proliferation. Best Pract. Res. Clin. Obstetr. Gynaecol. 22, 603–614 (2008).
Lee, B. S. & Nowak, R. A. Human leiomyoma smooth muscle cells show increased expression of transforming growth factor-β3 (TGFβ3) and altered responses to the antiproliferative effects of TGFβ. J. Clin. Endocrinol. Metab. 86, 913–920 (2001).
Sinclair, D. C., Mastroyannis, A. & Taylor, H. S. Leiomyoma simultaneously impair endometrial BMP-2-mediated decidualization and anticoagulant expression through secretion of TGF-β3. J. Clin. Endocrinol. Metab. 96, 412–421 (2011).
Vollenhoven, B. J., Herington, A. C. & Healy, D. L. Messenger ribonucleic acid expression of the insulin-like growth factors and their binding proteins in uterine fibroids and myometrium. J. Clin. Endocrinol. Metab. 76, 1106–1110 (1993).
Varghese, B. V. et al. Loss of the repressor REST in uterine fibroids promotes aberrant G protein-coupled receptor 10 expression and activates mammalian target of rapamycin pathway. Proc. Natl Acad. Sci. USA 110, 2187–2192 (2013).
Nowak, R. A., Mora, S., Diehl, T., Rhoades, A. R. & Stewart, E. A. Prolactin is an autocrine or paracrine growth factor for human myometrial and leiomyoma cells. Gynecol. Obstet. Invest. 48, 127–132 (1999).
Everitt, J. I., Wolf, D. C., Howe, S. R., Goldsworthy, T. L. & Walker, C. Rodent model of reproductive tract leiomyomata. Clinical and pathological features. Am. J. Pathol. 146, 1556–1567 (1995).
Howe, S. R. et al. Rodent model of reproductive tract leiomyomata. Establishment and characterization of tumor-derived cell lines. Am. J. Pathol. 146, 1568–1579 (1995).
Hassan, M. H. et al. Memy I: a novel murine model for uterine leiomyoma using adenovirus-enhanced human fibroid explants in severe combined immune deficiency mice. Am. J. Obstet. Gynecol. 199, 156.e1–156.e8 (2008).
Wang, G. et al. Nonobese diabetic/severe combined immunodeficient murine xenograft model for human uterine leiomyoma. Fertil. Steril. 101, 1485–1492 (2014).
Machado, S. A. et al. Validation of the aging hen (Gallus gallus domesticus) as an animal model for uterine leiomyomas. Biol. Reprod. 87, 86 (2012).
Munro, M. G., Critchley, H. O., Broder, M. S. & Fraser, I. S. FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age. Int. J. Gynaecol. Obstetr. 113, 3–13 (2011).
Cantuaria, G. H., Angioli, R., Frost, L., Duncan, R. & Penalver, M. A. Comparison of bimanual examination with ultrasound examination before hysterectomy for uterine leiomyoma. Obstet. Gynecol. 92, 109–112 (1998).
Kramer, M. et al. Incidence, risk factors, and temporal trends in severe postpartum hemorrhage. Am J. Obstet. Gynecol. 209, 449.e1–449.e7 (2013).
Stout, M. J. et al. Leiomyomas at routine second-trimester ultrasound examination and adverse obstetric outcomes. Obstet. Gynecol. 116, 1056–1063 (2010).
Ciavattini, A. et al. Number and size of uterine fibroids and obstetrics outcomes. J. Matern. Fetal Neonatal Med. 28, 484–488 (2014).
Ferreira, J. et al. The evolution of fetal presentation during pregnancy: a retrospective, descriptive cross sectional study. Acta Obstet. Gynecol. Scand. 94, 660–663 (2015).
Puri, K., Famuyide, A. O., Erwin, P. J., Stewart, E. A. & Laughlin-Tommaso, S. K. Submucosal fibroids and the relation to heavy menstrual bleeding and anemia. Am J. Obstet. Gynecol. 210, 38.e1–38.e7 (2014).
Myers, E. et al. Management of Uterine Fibroids (Agency for Healthcare Research and Quality, 2001).
Fonseca-Moutinho, J. A., Barbosa, L. S., Torres, D. G. & Nunes, S. M. Abnormal uterine bleeding as a presenting symptoms is related to multiple uterine leiomyoma: an ultrasound-based study. Int. J. Womens Health 18, 689–694 (2013).
Cicinelli, E. et al. Transabdominal sonohysterography, transvaginal sonography, and hysteroscopy in the evaluation of submucous myomas. Obstet. Gynecol. 85, 42–47 (1995).
Becker, E. Jr et al. The added value of transvaginal sonogysterography over transvaginal sonography alone in women with known or suspected leiomyoma. J. Ultrasound Med. 21, 237–247 (2002).
Dueholm, M., Lundorf, E., Hansen, E., Ledertoug, S. & Olesen, F. Evaluaiton of the uterine cavity with magnetic resonance imaging, transvaginal sonography, hysterosonographic examination, and diagnostic hysterocopy. Fertil. Steril. 76, 350–357 (2001).
Dudiak, C. M. et al. Uterine leiomyomas in the infertile patient: preoperative localization with MR imaging versus US and hysterosalpingography. Radiology 167, 627–630 (1988).
Nanda, S., Chadha, N., Sen, J. & Sangwan, K. Transvaginal sonography and saline infusion sonohysterograhy in the evaluation of abnormal uterine bleeding. Aust. N. Z. J. Obstet. Gynaecol. 42, 530–534 (2002).
Spielmann, A. L., Keogh, C., Forster, B. B., Martin, M. L. & Machan, L. S. Comparison of MRI and sonography in the preliminary evaluation for fibroid embolization. Am. J. Roentgenol 187, 1499–1504 (2006).
Toledo, G. & Oliva, E. Smooth muscle tumors of the uterus: a practical approach. Arch. Pathol. Lab. Med. 132, 595–605 (2008).
Clement, P. B. & Young, R. H. Atlas of Gynecologic Surgical Pathology 3rd edn (Elsevier, 2014).
Levy, G., Hill, M. J., Plowden, T. C., Catherino, W. & Armstrong, A. Biomarkers in uterine leiomyoma. Fertil. Steril. 99, 1146–1152 (2013).
Taran, F. A., Weaver, A. L., Coddington, C. C. & Stewart, E. A. Understanding adenomyosis: a case control study. Fertil. Steril. 94, 1223–1228 (2010).
Brosens, J. J., de Souza, N. M. & Barker, F. G. Uterine junctional zone: function and disease. Lancet 346, 558–560 (1995).
Dueholm, M. et al. Reproducibility of evaluation of the uterus by transvaginal sonography, hysterosonographic examination, hysteroscopy and magnetic resonance imaging. Hum. Reprod. 17, 195–200 (2002).
Wright, J. D. et al. Uterine pathology in women undergoing minimally invasive hysterectomy using morcellation. JAMA 312, 1253–1255 (2014).
Matsuda, M. et al. Preoperative diagnosis of usual leiomyoma, atypical leiomyoma, and leiomyosarcoma. Sarcoma 2014, 498682 (2014).
Santos, P. & Cunha, T. Uterine sarcomas: clinical presentation and MRI features. Radiology 21, 4–9 (2015).
US FDA. Updated laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA safety communication. FDAhttp://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm424443.htm (2014).
Stewart, E. A. Clinical practice. Uterine fibroids. N. Engl. J. Med. 372, 1646–1655 (2015). This is an updated review of the clinical diagnosis and treatment of fibroids.
Malik, M., Webb, J. & Catherino, W. H. Retinoic acid treatment of human leiomyoma cells transformed the cell phenotype to one strongly resembling myometrial cells. Clin. Endocrinol. (Oxf.) 69, 462–470 (2008).
Halder, S. K., Sharan, C. & Al-Hendy, A. 1,25-Dihydroxyvitamin D3 treatment shrinks uterine leiomyoma tumors in the Eker rat model. Biol. Reprod. 86, 116 (2012).
Malik, M., Mendoza, M., Payson, M. & Catherino, W. Curcumin, a nutritional supplement with antineoplastic activity, enhances leiomyoma cell apoptosis and decreases fibronectin expression. Fertil. Steril. 91, 2177–2184 (2009).
Lee, T. et al. Inhibitory effects of Scutellaria barbatta D. Don on human unterine leiomyomal smooth muscle cell proliferation through cell cycle analysis. Int. Immunopharmacol. 4, 447–454 (2004).
K.Im, D. et al. Induction of growth inhibition and apoptosis in human leiomyoma cells by isoliquiritigenin. Reprod. Sci. 15, 552–558 (2008).
Roshdy, E. et al. Treatment of symptomatic uterine fibroids with green tea extract: a pilot randomized controlled clinical study. Int. J. Womens Health 5, 477–486 (2013).
Kim, C. et al. Euonymus alatus (Thunb.) Sieb induces apoptosis via mitochondrial pathway as prooxidant in human uterine leiomyomal smooth muscle cells. Int. J. Gynecol. Cancer 16, 843–848 (2006).
Gliklich, R. E. et al. Identification of Future Needs in the Comparative Management of Uterine Fibroid Disease. A Report on the Priority-Setting Process, Preliminary Data Analysis, and Research Plan (Agency for Healthcare Research and Quality, 2011). This is a comprehensive assessment of the evidence, or lack thereof, for treatment of uterine fibroids.
American College of Obstreticians and Gynecologists. ACOG practice bulletin. Alternatives to hysterectomy in the management of leiomyomas. Obstet. Gynecol. 112, 387–400 (2008).
Marret, H. et al. Therapeutic management of uterine fibroid tumors: updated French guidelines. Eur. J. Obstetr. Gynecol. Reprod. Biol. 165, 156–164 (2012).
Perez-Lopez, F. R. et al. EMAS position statement: management of uterine fibroids. Maturitas 79, 106–116 (2014). This is a comprehensive, updated and annotated review of guidelines for fibroid treatment.
Laughlin, S. K., Hartmann, K. E. & Baird, D. D. Postpartum factors and natural fibroid regression. Am. J. Obstet. Gynecol. 204, 496.e1–496.e6 (2011).
Flake, G. P. et al. The natural history of uterine leiomyomas: morphometric concordance with concepts of interstitial ischemia and inanosis. Obstet. Gynecol. Int. 2013, 285103 (2013).
Wamsteker, K., Emanuel, M. H. & de Kruif, J. H. Transcervical hysteroscopic resection of submucous fibroids for abnormal uterine bleeding: results regarding the degree of intramural extension. Obstet. Gynecol. 82, 736–740 (1993).
Lethaby, A., Duckitt, K. & Farquhar, C. Non-steroidal anti-inflammatory drugs for heavy menstrual bleeding. Cochrane Database Syst. Rev. 1, CD000400 (2013).
Lukes, A. S. et al. Tranexamic acid treatment for heavy menstrual bleeding: a randomized controlled trial. Obstet. Gynecol. 116, 865–875 (2010).
Eder, S., Baker, J., Gersten, J., Mabey, R. G. & Adomako, T. L. Efficacy and safety of oral tranexamic acid in women with heavy menstrual bleeding and fibroids. Womens Health (Lond. Engl.) 9, 397–403 (2013).
Sangkomkamhang, U. S., Lumbiganon, P., Laopaiboon, M. & Mol, B. W. Progestogens or progestogen-releasing intrauterine systems for uterine fibroids. Cochrane Database Syst. Rev. 2, CD008994 (2013).
Lethaby, A. E., Cooke, I. & Rees, M. Progesterone or progestogen-releasing intrauterine systems for heavy menstrual bleeding. Cochrane Database Syst. Rev. 4, CD002126 (2005).
Kriplani, A., Awasthi, D., Kulshrestha, V. & Agarwal, N. Efficacy of the levonorgestrel-releasing intrauterine system in uterine leiomyoma. Int. J. Gynaecol. Obstet. 116, 35–38 (2012).
Socolov, D. et al. Levonorgestrel releasing-intrauterine system for the treatment of menorrhagia and/or frequent irregular uterine bleeding associated with uterine leiomyoma. Eur. J. Contracept. Reprod. Health Care 16, 480–487 (2011).
Youm, J., Lee, H. J., Kim, S. K., Kim, H. & Jee, B. C. Factors affecting the spontaneous expulsion of the levonorgestrel-releasing intrauterine system. Int. J. Gynaecol. Obstetr. 126, 165–169 (2014).
Mercorio, F. et al. The effect of a levonorgestrel-releasing intrauterine device in the treatment of myoma-related menorrhagia. Contraception 67, 277–280 (2003).
Perez-Lopez, F. R. Long-term consequences of LNG-IUS versus hysterectomy for menorrhagia. Climacteric 17, 308–309 (2014).
Qin, J., Yang, T., Kong, F. & Zhou, Q. Oral contraceptive use and uterine leiomyoma risk: a meta-analysis based on cohort and case–control studies. Arch. Gynecol. Obstet. 288, 139–148 (2013).
Donnez, J. et al. Ulipristal acetate versus placebo for fibroid treatment before surgery. N. Engl. J. Med. 366, 409–420 (2012).
Mutter, G. L. et al. The spectrum of endometrial pathology induced by progesterone receptor modulators. Mod. Pathol. 21, 591–598 (2008).
Dinh, A., Sriprasert, I., Williams, A. R. & Archer, D. F. A review of the endometrial histologic effects of progestins and progesterone receptor modulators in reproductive age women. Contraception 91, 360–367 (2015).
Luyckx, M. et al. First series of 18 pregnancies after ulipristal acetate treatment for uterine fibroids. Fertil. Steril. 102, 1404–1409 (2014).
Steinauer, J., Pritts, E. A., Jackson, R. & Jacoby, A. F. Systematic review of mifepristone for the treatment of uterine leiomyomata. Obstet. Gynecol. 103, 1331–1336 (2004).
Engman, M. et al. GSTM1 gene expression correlates to leiomyoma volume regression in response to mifepristone treatment. PLoS ONE 8, e80114 (2013).
Carr, B. R. et al. An evaluation of the effect of gonadotropin-releasing hormone analogs and medroxyprogesterone acetate on uterine leiomyomata volume by magnetic resonance imaging: a prospective, randomized, double blind, placebo-controlled, crossover trial. J. Clin. Endocrinol. Metab. 76, 1217–1223 (1993).
Perez-Lopez, F. R. Ulipristal acetate in the management of symptomatic uterine fibroids: facts and pending issues. Climacteric 18, 177–181 (2015).
Lethaby, A. E. & Vollenhoven, B. J. An evidence-based approach to hormonal therapies for premenopausal women with fibroids. Best Pract. Res. Clin. Obstetr. Gynaecol. 22, 307–331 (2008).
Muzii, L. et al. GnRH analogue treatment before hysteroscopic resection of submucous myomas: a prospective, randomized, multicenter study. Fertil. Steril. 94, 1496–1499 (2010).
Kamath, M. S., Kalampokas, E. E. & Kalampokas, T. E. Use of GnRH analogues pre-operatively for hysteroscopic resection of submucous fibroids: a systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 177, 11–18 (2014).
Flierman, P. A., Oberye, J. J., van der Hulst, V. P. & de Blok, S. Rapid reduction of leiomyoma volume during treatment with the GnRH antagonist ganirelix. BJOG 112, 638–642 (2005).
Palomba, S. et al. Effectiveness of combined GnRH analogue plus raloxifene administration in the treatment of uterine leiomyomas: a prospective, randomized, single-blind, placebo-controlled clinical trial. Hum. Reprod. 17, 3213–3219 (2002).
Palomba, S. et al. Raloxifene administration in premenopausal women with uterine leiomyomas: a pilot study. J. Clin. Endocrinol. Metab. 87, 3603–3608 (2002).
Hilario, S. G., Bozzini, N., Borsari, R. & Baracat, E. C. Action of aromatase inhibitor for treatment of uterine leiomyoma in perimenopausal patients. Fertil. Steril. 91, 240–243 (2009).
Varelas, F. K., Papanicolaou, A. N., Vavatsi-Christaki, N., Makedos, G. A. & Vlassis, G. D. The effect of anastrazole on symptomatic uterine leiomyomata. Obstet. Gynecol. 110, 643–649 (2007).
Coutinho, E. M. Gestrinone in the treatment of myomas. Acta Obstet. Gynecol. Scand. Suppl. 150, 39–46 (1989).
Brucker, S. Y. et al. Laparoscopic radiofrequency volumetric thermal ablation of fibroids versus laparoscopic myomectomy. Int. J. Gynaecol. Obstetr. 125, 261–265 (2014).
Bergamini, V. et al. Laparoscopic radiofrequency thermal ablation: a new approach to symptomatic uterine myomas. Am. J. Obstet. Gynecol. 192, 768–773 (2005).
Gupta, J. K., Sinha, A., Lumsden, M. A. & Hickey, M. Uterine artery embolization for symptomatic uterine fibroids. Cochrane Database Syst. Rev. 5, CD005073 (2012).
van der Kooij, S. M., Bipat, S., Hehenkamp, W. J., Ankum, W. M. & Reekers, J. A. Uterine artery embolization versus surgery in the treatment of symptomatic fibroids: a systematic review and metaanalysis. Am J. Obstet. Gynecol. 205, 317.e1–317.e8 (2011).
Mara, M. et al. Midterm clinical and first reproductive results of a randomized controlled trial comparing uterine fibroid embolization and myomectomy. Cardiovasc. Intervent. Radiol. 31, 73–85 (2008).
Pisco, J. M., Duarte, M., Bilhim, T., Cirurgiao, F. & Oliveira, A. G. Pregnancy after uterine fibroid embolization. Fertil. Steril. 95, 1121.e5–1121.e8 (2011).
Hehenkamp, W. J. et al. Loss of ovarian reserve after uterine artery embolization: a randomized comparison with hysterectomy. Hum. Reprod. 22, 1996–2005 (2007).
Rabinovici, J. et al. Pregnancy outcome after magnetic resonance-guided focused ultrasound surgery (MRgFUS) for conservative treatment of uterine fibroids. Fertil. Steril. 93, 199–209 (2010).
Quinn, S. D., Vedelago, J., Gedroyc, W. & Regan, L. Safety and five-year re-intervention following magnetic resonance-guided focused ultrasound (MRgFUS) for uterine fibroids. Eur. J. Obstetr. Gynecol. Reproduct. Biol. 182, 247–251 (2014).
Gorny, K. R. et al. Incidence of additional treatments in women treated with MR-guided focused US for symptomatic uterine fibroids: review of 138 patients with an average follow-up of 2.8 years. J. Vasc. Interv. Radiol. 25, 1506–1512 (2014).
Rocca, W. A. & Ulrich, L. G. Oophorectomy for whom and at what age? Primum non nocere. Maturitas 71, 1–2 (2012).
Rocca, W. A., Grossardt, B. R., Shuster, L. T. & Stewart, E. A. Hysterectomy, oophorectomy, estrogen, and the risk of dementia. Neurodegenerat. Dis. 10, 175–178 (2012).
Ghant, M. S. et al. Beyond the physical: a qualitative assessment of the burden of symptomatic uterine fibroids on women's emotional and psychosocial health. J. Psychosom. Res. 78, 499–503 (2015).
Marsh, E. E., Brocks, M. E., Ghant, M. S., Recht, H. S. & Simon, M. Prevalence and knowledge of heavy menstrual bleeding among African American women. Int. J. Gynaecol. Obstetr. 125, 56–59 (2014).
Stewart, E. A., Nicholson, W. K., Bradley, L. & Borah, B. J. The burden of uterine fibroids for African-American women: results of a national survey. J. Womens Health (Larchmt) 22, 807–816 (2013). This paper articulates the differential clinical burden of fibroids for women of African descent compared with other women.
Spies, J. B. et al. The UFS-QOL, a new disease-specific symptom and health-related quality of life questionnaire for leiomyomata. Obstet. Gynecol. 99, 290–300 (2002).
Harding, G., Coyne, K. S., Thompson, C. L. & Spies, J. B. The responsiveness of the uterine fibroid symptom and health-related quality of life questionnaire (UFS-QOL). Health Qual. Life Outcomes 6, 99 (2008).
Laughlin-Tommaso, S. K., Borah, B. J. & Stewart, E. A. Effect of menses on standardized assessment of sexual dysfunction among women with uterine fibroids: a cohort study. Fertil. Steril. 104, 435–439 (2015).
Coronado, G. D., Marshall, L. M. & Schwartz, S. M. Complications in pregnancy, labor, and delivery with uterine leiomyomas: a population-based study. Obstet. Gynecol. 95, 764–769 (2000).
Klatsky, P. C., Tran, N. D., Caughey, A. B. & Fujimoto, V. Y. Fibroids and reproductive outcomes: a systematic literature review from conception to delivery. Am. J. Obstet. Gynecol. 198, 357–366 (2008).
Johnson, G., MacLehose, R. F., Baird, D. D., Laughlin-Tommaso, S. K. & Hartmann, K. E. Uterine leiomyomata and fecundability in the Right from the Start study. Hum. Reprod. 27, 2991–2997 (2012).
ACOG Committee on Practice Bulletins-Gynecology. ACOG practice bulletin. Alternatives to hysterectomy in the management of leiomyomas. Obstetr. Gynecol. 112, 387–400 (2008).
Pritts, E. A., Parker, W. H. & Olive, D. L. Fibroids and infertility: an updated systematic review of the evidence. Fertil. Steril. 91, 1215–1223 (2009).
Metwally, M., Cheong, Y. C. & Horne, A. W. Surgical treatment of fibroids for subfertility. Cochrane Database Syst. Rev. 11, CD003857 (2012).
Bosteels, J. et al. Hysteroscopy for treating subfertility associated with suspected major uterine cavity abnormalities. Cochrane Database Syst. Rev. 1, CD009461 (2013).
Fisher, B. From Halsted to prevention and beyond: advances in the management of breast cancer during the twentieth century. Eur. J. Cancer 35, 1963–1973 (1999).
Hodge, J. C., Pearce, K. E., Clayton, A. C., Taran, F. A. & Stewart, E. A. Uterine cellular leiomyomata with chromosome 1p deletions represent a distinct entity. Am J. Obstet. Gynecol. 210, 572.e1–573.e7 (2014).
Ruta, D. A. et al. Assessment of patients with menorrhagia: how valid is a structured clinical history as a measure of health status? Qual. Life Res. 4, 33–40 (1995).
Zakherah, M. S., Sayed, G. H., El-Nashar, S. & Shaaban, M. Pictorial blood loss assessment chart in the evaluation of heavy menstrual bleeding: diagnotic accuracy compared to alkaline hematin. Gynecol. Obstet. Invest. 71, 281–284 (2011).
Moos, R. H. The development of a menstrual distress questionnaire. Psychosomat. Med. 30, 853–867 (1968).
Rosen, R. et al. The Female Sexual Function Index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J. Sex. Marital Ther. 26, 191–208 (2000).
Lukacz, E. S. et al. The use of Visual Analog Scale in urogynecologic research: a psychometric evaluation. Am. J. Obstet. Gynecol. 191, 165–170 (2004).
Melzack, R. The short-form McGill Pain Questionnaire. Pain 30, 191–197 (1987).
Eskenazi, B. et al. Serum dioxin concentrations and risk of uterine leiomyoma in the Seveso Women's Health Study. Am. J. Epidemiol. 166, 79–87 (2007).
Bower, J. K., Schreiner, P. J., Sternfeld, B. & Lewis, C. E. Black–white differences in hysterectomy prevalence: the CARDIA study. Am. J. Public Health 99, 300–307 (2009).
Borgfeldt, C. & Andolf, E. Transvaginal ultrasonographic findings in the uterus and the endometrium: low prevalence of leiomyoma in a random sample of women age 25–40 years. Acta Obstet. Gynecol. Scand. 79, 202–207 (2000).
Acknowledgements
E.A.S. has received research support from the US NIH (R01HD060503, P50HS023418 and R01HD074711) and from Insightec Inc. The authors acknowledge the technical assistance of D. Littlefield.
Author information
Authors and Affiliations
Contributions
Introduction (E.A.S.); Epidemiology (S.K.L.-T.); Mechanisms/pathophysiology (E.A.S., B.V. and D.V.); Diagnosis, screening and prevention (W.H.C.); Management (E.A.S. and S.L.); Quality of life (E.A.S. and S.L.); Outlook (E.A.S.); Overview of Primer (E.A.S.).
Corresponding author
Ethics declarations
Competing interests
E.A.S. is a consultant for AbbVie, Allergan, Astellas Pharma, Bayer Health Care, Gynesonics and Viteava and has received royalties from UpToDate and the Massachusetts Medical Society. S.K.L.-T. has received research support from InSightec and support for consulting from Truven Health Analytics and for serving on the data monitoring board for ULTRA trail from HALT medical. She is an author for UpToDate. W.H.C. has received research support from Bayer Schering Pharma and Patient-Centered Outcomes Research Institute (PCORI), and is a consultant for AbbVie Pharmaceuticals and Actavis. He is an oral boards examiner for the American Board of Obstetrics and Gynecology and a content Review Committee for the American Society of Reproductive Medicine. S.L., B.V. and D.G. declare no competing interests.
Rights and permissions
About this article
Cite this article
Stewart, E., Laughlin-Tommaso, S., Catherino, W. et al. Uterine fibroids. Nat Rev Dis Primers 2, 16043 (2016). https://doi.org/10.1038/nrdp.2016.43
Published:
DOI: https://doi.org/10.1038/nrdp.2016.43
This article is cited by
-
Application of transabdominal ultrasound- and laparoscopy-guided percutaneous microwave ablation for treating uterine fibroids: 24-month follow-up outcomes
Archives of Gynecology and Obstetrics (2024)
-
The efficacy and safety of Chinese herbal medicine Guizhi Fuling capsule combined with low dose mifepristone in the treatment of uterine fibroids: a systematic review and meta-analysis of 28 randomized controlled trials
BMC Complementary Medicine and Therapies (2023)
-
Global, regional, and national time trends in incidence, prevalence, years lived with disability for uterine fibroids, 1990–2019: an age-period-cohort analysis for the global burden of disease 2019 study
BMC Public Health (2023)
-
H3K4me3 mediates uterine leiomyoma pathogenesis via neuronal processes, synapsis components, proliferation, and Wnt/β-catenin and TGF-β pathways
Reproductive Biology and Endocrinology (2023)
-
Potential mechanism of Taohong Siwu Decoction in uterine fibroid treatment based on integrated strategy of network pharmacology and experimental verification
Chinese Medicine (2023)