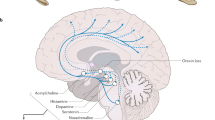
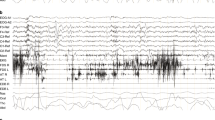
Abstract
Narcolepsy is a chronic sleep disorder that has a typical onset in adolescence and is characterized by excessive daytime sleepiness, which can have severe consequences for the patient. Problems faced by patients with narcolepsy include social stigma associated with this disease, difficulties in obtaining an education and keeping a job, a reduced quality of life and socioeconomic consequences. Two subtypes of narcolepsy have been described (narcolepsy type 1 and narcolepsy type 2), both of which have similar clinical profiles, except for the presence of cataplexy, which occurs only in patients with narcolepsy type 1. The pathogenesis of narcolepsy type 1 is hypothesized to be the autoimmune destruction of the hypocretin-producing neurons in the hypothalamus; this hypothesis is supported by immune-related genetic and environmental factors associated with the disease. However, direct evidence in support of the autoimmune hypothesis is currently unavailable. Diagnosis of narcolepsy encompasses clinical, electrophysiological and biological evaluations, but simpler and faster procedures are needed. Several medications are available for the symptomatic treatment of narcolepsy, all of which have quite good efficacy and safety profiles. However, to date, no treatment hinders or slows disease development. Improved diagnostic tools and increased understanding of the pathogenesis of narcolepsy type 1 are needed and might lead to therapeutic or even preventative interventions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
American Academy of Sleep Medicine. International Classification of Sleep Disorders 3rd edn (American Academy of Sleep Medicine, 2014). Includes current classification of NT1 and NT2 and detailed descriptions of the two narcolepsy subtypes.
Yoss, R. E. & Daly, D. D. Narcolepsy. Med. Clin. North Am. 44, 953–968 (1960).
Lecendreux, M. et al. Attention-deficit/hyperactivity disorder (ADHD) symptoms in pediatric narcolepsy: a cross-sectional study. Sleep 38, 1285–1295 (2015).
Rocca, F. L. et al. Psychosocial profile and quality of life in children with type 1 narcolepsy: a case–control study. Sleep 39, 1389–1398 (2016).
Plazzi, G. et al. Complex movement disorders at disease onset in childhood narcolepsy with cataplexy. Brain 134, 3477–3489 (2011).
Poli, F. et al. High prevalence of precocious puberty and obesity in childhood narcolepsy with cataplexy. Sleep 36, 175–181 (2013).
Andlauer, O. et al. Predictors of hypocretin (orexin) deficiency in narcolepsy without cataplexy. Sleep 35, 1247–1255 (2012).
Knudsen, S., Jennum, P., Alving, J., Sheikh, S. P. & Gammeltoft, S. Validation of the ICSD-2 criteria for CSF hypocretin-1 measurements in the diagnosis of narcolepsy in the Danish population. Sleep 33, 169–176 (2010).
Lopez, R. et al. Temporal changes in the cerebrospinal fluid level of hypocretin-1 and histamine in narcolepsy. Sleep 9 Sept 2016 [epub ahead of print].
Pizza, F. et al. Primary progressive narcolepsy type 1: the other side of the coin. Neurology 83, 2189–2190 (2014).
Peyron, C. et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat. Med. 6, 991–997 (2000). First description of hypocretin deficiency in patients with narcolepsy.
Thannickal, T. C. et al. Reduced number of hypocretin neurons in human narcolepsy. Neuron 27, 469–474 (2000).
Thannickal, T. C., Nienhuis, R. & Siegel, J. M. Localized loss of hypocretin (orexin) cells in narcolepsy without cataplexy. Sleep 32, 993–998 (2009).
Gerashchenko, D. et al. Relationship between CSF hypocretin levels and hypocretin neuronal loss. Exp. Neurol. 184, 1010–1016 (2003).
Nishino, S. et al. CSF hypocretin levels in Guillain–Barré syndrome and other inflammatory neuropathies. Neurology 61, 823–825 (2003).
Plazzi, G. et al. Narcolepsy with cataplexy associated with holoprosencephaly misdiagnosed as epileptic drop attacks. Mov. Disord. 25, 788–790 (2010).
Moghadam, K. K. et al. Narcolepsy is a common phenotype in HSAN IE and ADCA-DN. Brain 137, 1643–1655 (2014).
Silber, M. H., Krahn, L. E., Olson, E. J. & Pankratz, V. S. The epidemiology of narcolepsy in Olmsted County, Minnesota: a population-based study. Sleep 25, 197–202 (2002). One of the largest population-based studies of the prevalence of narcolepsy.
Heier, M. S. et al. Prevalence of narcolepsy with cataplexy in Norway. Acta Neurol. Scand. 120, 276–280 (2009).
Hublin, C., Partinen, M., Kaprio, J., Koskenvuo, M. & Guilleminault, C. Epidemiology of narcolepsy. Sleep 17, S7–S12 (1994).
Hublin, C. et al. The prevalence of narcolepsy: an epidemiological study of the Finnish Twin Cohort. Ann. Neurol. 35, 709–716 (1994).
Ohayon, M. M., Priest, R. G., Zulley, J., Smirne, S. & Paiva, T. Prevalence of narcolepsy symptomatology and diagnosis in the European general population. Neurology 58, 1826–1833 (2002).
Thorpy, M. J. & Krieger, A. C. Delayed diagnosis of narcolepsy: characterization and impact. Sleep Med. 15, 502–507 (2014).
Luca, G. et al. Clinical, polysomnographic and genome-wide association analyses of narcolepsy with cataplexy: a European Narcolepsy Network study. J. Sleep Res. 22, 482–495 (2013). Description of the large European Narcolepsy Network Database.
Serra, L., Montagna, P., Mignot, E., Lugaresi, E. & Plazzi, G. Cataplexy features in childhood narcolepsy. Mov. Disord. 23, 858–865 (2008).
Pizza, F. et al. Clinical and polysomnographic course of childhood narcolepsy with cataplexy. Brain 136, 3787–3795 (2013).
Dauvilliers, Y. et al. Age at onset of narcolepsy in two large populations of patients in France and Quebec. Neurology 57, 2029–2033 (2001).
Wing, Y. K. et al. The prevalence of narcolepsy among Chinese in Hong Kong. Ann. Neurol. 51, 578–584 (2002).
Shin, Y. K. et al. Prevalence of narcolepsy-cataplexy in Korean adolescents. Acta Neurol. Scand. 117, 273–278 (2008).
Juji, T., Matsuki, K., Tokunaga, K., Naohara, T. & Honda, Y. Narcolepsy and HLA in the Japanese. Ann. NY Acad. Sci. 540, 106–114 (1988).
Wilner, A. et al. Narcolepsy-cataplexy in Israeli Jews is associated exclusively with the HLA DR2 haplotype. A study at the serological and genomic level. Hum. Immunol. 21, 15–22 (1988).
al Rajeh, S. et al. A community survey of neurological disorders in Saudi Arabia: the Thugbah study. Neuroepidemiology 12, 164–178 (1993).
Longstreth, W. T. Jr et al. Prevalence of narcolepsy in King County, Washington, USA. Sleep Med. 10, 422–426 (2009).
Miller, E. et al. Risk of narcolepsy in children and young people receiving AS03 adjuvanted pandemic A/H1N1 2009 influenza vaccine: retrospective analysis. BMJ 346, f794 (2013).
Liu, R. S. et al. Childhood infections, socioeconomic status, and adult cardiometabolic risk. Pediatrics 137, e20160236 (2016).
Montplaisir, J. & Poirier, G. Narcolepsy in monozygotic twins. Neurology 37, 1089 (1987).
Chabas, D., Taheri, S., Renier, C. & Mignot, E. The genetics of narcolepsy. Annu. Rev. Genomics Hum. Genet. 4, 459–483 (2003).
Ohayon, M. M., Ferini-Strambi, L., Plazzi, G., Smirne, S. & Castronovo, V. Frequency of narcolepsy symptoms and other sleep disorders in narcoleptic patients and their first-degree relatives. J. Sleep Res. 14, 437–445 (2005).
Mignot, E. et al. Complex HLA-DR and -DQ interactions confer risk of narcolepsy-cataplexy in three ethnic groups. Am. J. Hum. Genet. 68, 686–699 (2001). Describes the strong association in NT1 with HLA-DQB1*06:02.
Tafti, M. et al. DQB1 locus alone explains most of the risk and protection in narcolepsy with cataplexy in Europe. Sleep 37, 19–25 (2014).
Ollila, H. M. et al. HLA-DPB1 and HLA class I confer risk of and protection from narcolepsy. Am. J. Hum. Genet. 96, 136–146 (2015). By carefully adjusting for HLA class II effects, these authors discovered HLA class I associations in NT1.
Hor, H. et al. Genome-wide association study identifies new HLA class II haplotypes strongly protective against narcolepsy. Nat. Genet. 42, 786–789 (2010).
Molari, A. & Carcassi, C. HLA and narcolepsy-cataplexy in the Sardinian population. J. Sleep Res. 19, 624–625 (2010).
Han, F. et al. HLA DQB1*06:02 negative narcolepsy with hypocretin/orexin deficiency. Sleep 37, 1601–1608 (2014).
Han, F. et al. Genome wide analysis of narcolepsy in China implicates novel immune loci and reveals changes in association prior to versus after the 2009 H1N1 influenza pandemic. PLoS Genet. 9, e1003880 (2013).
Tafti, M. et al. Narcolepsy-associated HLA class I alleles implicate cell-mediated cytotoxicity. Sleep 39, 581–587 (2016).
Hallmayer, J. et al. Narcolepsy is strongly associated with the T-cell receptor alpha locus. Nat. Genet. 41, 708–711 (2009). First genome-wide association study of NT1 and the discovery of an association with a T cell receptor gene.
Faraco, J. et al. ImmunoChip study implicates antigen presentation to T cells in narcolepsy. PLoS Genet. 9, e1003270 (2013).
Kornum, B. R. et al. Common variants in P2RY11 are associated with narcolepsy. Nat. Genet. 43, 66–71 (2011).
Toyoda, H. et al. A polymorphism in CCR1/CCR3 is associated with narcolepsy. Brain Behav. Immun. 49, 148–155 (2015).
Hor, H. et al. A missense mutation in myelin oligodendrocyte glycoprotein as a cause of familial narcolepsy with cataplexy. Am. J. Hum. Genet. 89, 474–479 (2011).
Nohynek, H. et al. AS03 adjuvanted AH1N1 vaccine associated with an abrupt increase in the incidence of childhood narcolepsy in Finland. PLoS ONE 7, e33536 (2012). One of the first papers to describe the increased risk of narcolepsy following vaccination with Pandemrix.
Bardage, C. et al. Neurological and autoimmune disorders after vaccination against pandemic influenza A (H1N1) with a monovalent adjuvanted vaccine: population based cohort study in Stockholm, Sweden. BMJ 343, d5956 (2011).
Heier, M. S. et al. Incidence of narcolepsy in Norwegian children and adolescents after vaccination against H1N1 influenza A. Sleep Med. 14, 867–871 (2013).
O'Flanagan, D. et al. Investigation of an association between onset of narcolepsy and vaccination with pandemic influenza vaccine, Ireland April 2009–December 2010. Euro Surveill. 19, 15–25 (2014).
Dauvilliers, Y. et al. Increased risk of narcolepsy in children and adults after pandemic H1N1 vaccination in France. Brain 136, 2486–2496 (2013).
Montplaisir, J. et al. Risk of narcolepsy associated with inactivated adjuvanted (AS03) A/H1N1 (2009) pandemic influenza vaccine in Quebec. PLoS ONE 9, e108489 (2014).
Vaarala, O. et al. Antigenic differences between AS03 adjuvanted influenza A (H1N1) pandemic vaccines: implications for Pandemrix-associated narcolepsy risk. PLoS ONE 9, e114361 (2014).
Picchioni, D., Hope, C. R. & Harsh, J. R. A case–control study of the environmental risk factors for narcolepsy. Neuroepidemiology 29, 185–192 (2007).
Han, F. et al. Narcolepsy onset is seasonal and increased following the 2009 H1N1 pandemic in China. Ann. Neurol. 70, 410–417 (2011).
Han, F., Lin, L., Li, J., Dong, X. S. & Mignot, E. Decreased incidence of childhood narcolepsy 2 years after the 2009 H1N1 winter flu pandemic. Ann. Neurol. 73, 560 (2013).
Melen, K. et al. No serological evidence of influenza A H1N1pdm09 virus infection as a contributing factor in childhood narcolepsy after Pandemrix vaccination campaign in Finland. PLoS ONE 8, e68402 (2013).
Aran, A. et al. Elevated anti-streptococcal antibodies in patients with recent narcolepsy onset. Sleep 32, 979–983 (2009).
Koepsell, T. D., Longstreth, W. T. & Ton, T. G. Medical exposures in youth and the frequency of narcolepsy with cataplexy: a population-based case–control study in genetically predisposed people. J. Sleep Res. 19, 80–86 (2010).
Ambati, A. et al. Increased beta-haemolytic group A streptococcal M6 serotype and streptodornase B-specific cellular immune responses in Swedish narcolepsy cases. J. Intern. Med. 278, 264–276 (2015).
Chang, C. Neonatal autoimmune diseases: a critical review. J. Autoimmun. 38, J223–J238 (2012).
Lin, L. et al. The sleep disorder canine narcolepsy is caused by a mutation in the hypocretin (orexin) receptor 2 gene. Cell 98, 365–376 (1999).
Chemelli, R. M. et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 98, 437–451 (1999).
Nishino, S., Ripley, B., Overeem, S., Lammers, G. J. & Mignot, E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 355, 39–40 (2000).
Mahler, S. V., Moorman, D. E., Smith, R. J., James, M. H. & Aston-Jones, G. Motivational activation: a unifying hypothesis of orexin/hypocretin function. Nat. Neurosci. 17, 1298–1303 (2014). Comprehensive review of the function of the hypocretin system.
Li, J., Hu, Z. & de Lecea, L. The hypocretins/orexins: integrators of multiple physiological functions. Br. J. Pharmacol. 171, 332–350 (2014).
Blouin, A. M. et al. Narp immunostaining of human hypocretin (orexin) neurons: loss in narcolepsy. Neurology 65, 1189–1192 (2005).
Crocker, A. et al. Concomitant loss of dynorphin, NARP, and orexin in narcolepsy. Neurology 65, 1184–1188 (2005).
Savvidou, A. et al. Hypocretin deficiency develops during onset of human narcolepsy with cataplexy. Sleep 36, 147–148 (2013).
Martinez-Orozco, F. J., Vicario, J. L., De Andres, C., Fernandez-Arquero, M. & Peraita-Adrados, R. Comorbidity of narcolepsy type 1 with autoimmune diseases and other immunopathological disorders: a case–control study. J. Clin. Med. Res. 8, 495–505 (2016).
Barateau, L. et al. Comorbidity between central disorders of hypersomnolence and immune-based disorders. Neurology 88, 93–100 (2016).
Plazzi, G. et al. Intravenous high-dose immunoglobulin treatment in recent onset childhood narcolepsy with cataplexy. J. Neurol. 255, 1549–1554 (2008).
Valko, P. O., Khatami, R., Baumann, C. R. & Bassetti, C. L. No persistent effect of intravenous immunoglobulins in patients with narcolepsy with cataplexy. J. Neurol. 255, 1900–1903 (2008).
Fronczek, R., Verschuuren, J. & Lammers, G. J. Response to intravenous immunoglobulins and placebo in a patient with narcolepsy with cataplexy. J. Neurol. 254, 1607–1608 (2007).
Dauvilliers, Y., Abril, B., Mas, E., Michel, F. & Tafti, M. Normalization of hypocretin-1 in narcolepsy after intravenous immunoglobulin treatment. Neurology 73, 1333–1334 (2009).
Bergman, P. et al. Narcolepsy patients have antibodies that stain distinct cell populations in rat brain and influence sleep patterns. Proc. Natl Acad. Sci. USA 111, E3735–E3744 (2014).
Black, J. L. III, Krahn, L. E., Pankratz, V. S. & Silber, M. Search for neuron-specific and nonneuron-specific antibodies in narcoleptic patients with and without HLA DQB1*0602. Sleep 25, 719–723 (2002).
Black, J. L. III et al. Analysis of hypocretin (orexin) antibodies in patients with narcolepsy. Sleep 28, 427–431 (2005).
Deloumeau, A. et al. Increased immune complexes of hypocretin autoantibodies in narcolepsy. PLoS ONE 5, e13320 (2010).
Knudsen, S., Mikkelsen, J. D. & Jennum, P. Antibodies in narcolepsy-cataplexy patient serum bind to rat hypocretin neurons. Neuroreport 18, 77–79 (2007).
Overeem, S. et al. Immunohistochemical screening for autoantibodies against lateral hypothalamic neurons in human narcolepsy. J. Neuroimmunol. 174, 187–191 (2006).
Tanaka, S., Honda, Y., Inoue, Y. & Honda, M. Detection of autoantibodies against hypocretin, hcrtrl, and hcrtr2 in narcolepsy: anti-Hcrt system antibody in narcolepsy. Sleep 29, 633–638 (2006).
van der Heide, A., Hegeman-Kleinn, I. M., Peeters, E., Lammers, G. J. & Fronczek, R. Immunohistochemical screening for antibodies in recent onset type 1 narcolepsy and after H1N1 vaccination. J. Neuroimmunol. 283, 58–62 (2015).
Cvetkovic-Lopes, V. et al. Elevated tribbles homolog 2-specific antibody levels in narcolepsy patients. J. Clin. Invest. 120, 713–719 (2010). Describes the presence of autoantibodies that target hypocretin neurons in blood samples from patients with NT1.
Kawashima, M. et al. Anti-tribbles homolog 2 (TRIB2) autoantibodies in narcolepsy are associated with recent onset of cataplexy. Sleep 33, 869–874 (2010).
Toyoda, H. et al. Anti-tribbles homolog 2 autoantibodies in Japanese patients with narcolepsy. Sleep 33, 875–878 (2010).
Lind, A. et al. A/H1N1 antibodies and TRIB2 autoantibodies in narcolepsy patients diagnosed in conjunction with the Pandemrix vaccination campaign in Sweden 2009–2010. J. Autoimmun. 50, 99–106 (2014).
Jego, S. et al. Optogenetic identification of a rapid eye movement sleep modulatory circuit in the hypothalamus. Nat. Neurosci. 16, 1637–1643 (2013).
Ahmed, S. S. et al. Antibodies to influenza nucleoprotein cross-react with human hypocretin receptor 2. Sci. Transl Med. 7, 294ra105 (2015).
Katzav, A. et al. Passive transfer of narcolepsy: anti-TRIB2 autoantibody positive patient IgG causes hypothalamic orexin neuron loss and sleep attacks in mice. J. Autoimmun. 45, 24–30 (2013).
Dauvilliers, Y. et al. Hypothalamic immunopathology in anti-Ma-associated diencephalitis with narcolepsy-cataplexy. JAMA Neurol. 70, 1305–1310 (2013).
Overeem, S. et al. Hypocretin-1 CSF levels in anti-Ma2 associated encephalitis. Neurology 62, 138–140 (2004).
Kallweit, U., Bassetti, C. & Sallusto, F. Hypocretin-reactive CD4+ T cells in narcoleptic patients. Presented at the23rd Congress of the European Sleep Research Society abstr. 202 (2016).
Dauvilliers, Y. et al. Cerebrospinal fluid and serum cytokine profiles in narcolepsy with cataplexy: a case–control study. Brain Behav. Immun. 37, 260–266 (2014).
Lecendreux, M. et al. Impact of cytokine in type 1 narcolepsy: role of pandemic H1N1 vaccination? J. Autoimmun. 60, 20–31 (2015).
Kornum, B. R. et al. Cerebrospinal fluid cytokine levels in type 1 narcolepsy patients very close to onset. Brain Behav. Immun. 49, 54–58 (2015). Describes how it is not possible to detect markers of neuroinflammation in the CSF of patients with NT1 even within 1 month of disease onset.
John, J. et al. Greatly increased numbers of histamine cells in human narcolepsy with cataplexy. Ann. Neurol. 74, 786–793 (2013).
Valko, P. O. et al. Increase of histaminergic tuberomammillary neurons in narcolepsy. Ann. Neurol. 74, 794–804 (2013). References 102 and 103 simultaneously describe that patients with NT1 have increased numbers of histaminergic neurons in the brain.
Shan, L., Dauvilliers, Y. & Siegel, J. M. Interactions of the histamine and hypocretin systems in CNS disorders. Nat. Rev. Neurol. 11, 401–413 (2015).
Dauvilliers, Y. et al. Normal cerebrospinal fluid histamine and tele-methylhistamine levels in hypersomnia conditions. Sleep 35, 1359–1366 (2012).
Sakurai, T. et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92, 573–585 (1998).
Adamantidis, A. R., Zhang, F., Aravanis, A. M., Deisseroth, K. & de Lecea, L. Neural substrates of awakening probed with optogenetic control of hypocretin neurons. Nature 450, 420–424 (2007). Provides optogenetic proof that activation of the hypocretin neurons induces wakefulness.
Tsunematsu, T. et al. Acute optogenetic silencing of orexin/hypocretin neurons induces slow-wave sleep in mice. J. Neurosci. 31, 10529–10539 (2011).
Broughton, R. et al. Excessive daytime sleepiness and the pathophysiology of narcolepsy-cataplexy: a laboratory perspective. Sleep 9, 205–215 (1986).
Saper, C. B., Chou, T. C. & Scammell, T. E. The sleep switch: hypothalamic control of sleep and wakefulness. Trends Neurosci. 24, 726–731 (2001).
Sorensen, G. L., Knudsen, S. & Jennum, P. Sleep transitions in hypocretin-deficient narcolepsy. Sleep 36, 1173–1177 (2013).
Pizza, F. et al. Nocturnal sleep dynamics identify narcolepsy type 1. Sleep 38, 1277–1284 (2015).
Knudsen, S., Gammeltoft, S. & Jennum, P. J. Rapid eye movement sleep behaviour disorder in patients with narcolepsy is associated with hypocretin-1 deficiency. Brain 133, 568–579 (2010).
Saper, C. B., Fuller, P. M., Pedersen, N. P., Lu, J. & Scammell, T. E. Sleep state switching. Neuron 68, 1023–1042 (2010). Describes in detail how sleep-state switching is regulated and includes both wake-to-sleep transitions and NREM–REM-stage switches.
Overeem, S., Lammers, G. J. & van Dijk, J. G. Weak with laughter. Lancet 354, 838 (1999).
Overeem, S., Lammers, G. J. & van Dijk, J. G. Cataplexy: ‘tonic immobility’ rather than ‘REM-sleep atonia’? Sleep Med. 2, 471–477 (2002).
Schwartz, S. et al. Abnormal activity in hypothalamus and amygdala during humour processing in human narcolepsy with cataplexy. Brain 131, 514–522 (2008).
Meletti, S. et al. The brain correlates of laugh and cataplexy in childhood narcolepsy. J. Neurosci. 35, 11583–11594 (2015).
Kok, S. W. et al. Hypocretin deficiency in narcoleptic humans is associated with abdominal obesity. Obes. Res. 11, 1147–1154 (2003).
Hara, J., Yanagisawa, M. & Sakurai, T. Difference in obesity phenotype between orexin-knockout mice and orexin neuron-deficient mice with same genetic background and environmental conditions. Neurosci. Lett. 380, 239–242 (2005).
van Holst, R. J. et al. Aberrant food choices after satiation in human orexin-deficient narcolepsy type 1. Sleep 39, 1951–1959 (2016).
Fortuyn, H. A. et al. High prevalence of eating disorders in narcolepsy with cataplexy: a case–control study. Sleep 31, 335–341 (2008).
Baimel, C. et al. Orexin/hypocretin role in reward: implications for opioid and other addictions. Br. J. Pharmacol. 172, 334–348 (2015).
Johns, M. W. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep 14, 540–545 (1991).
Nishiyama, T. et al. Criterion validity of the Pittsburgh Sleep Quality Index and Epworth Sleepiness Scale for the diagnosis of sleep disorders. Sleep Med. 15, 422–429 (2014).
Kushida, C. A. et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep 28, 499–521 (2005).
Morgenthaler, T. et al. Practice parameters for the use of actigraphy in the assessment of sleep and sleep disorders: an update for 2007. Sleep 30, 519–529 (2007).
Jennum, P., Ibsen, R., Knudsen, S. & Kjellberg, J. Comorbidity and mortality of narcolepsy: a controlled retro- and prospective national study. Sleep 36, 835–840 (2013).
Plazzi, G., Khatami, R., Serra, L., Pizza, F. & Bassetti, C. L. Pseudocataplexy in narcolepsy with cataplexy. Sleep Med. 11, 591–594 (2010).
Vassalli, A. et al. Electroencephalogram paroxysmal theta characterizes cataplexy in mice and children. Brain 136, 1592–1608 (2013).
Littner, M. R. et al. Practice parameters for clinical use of the multiple sleep latency test and the maintenance of wakefulness test. Sleep 28, 113–121 (2005).
Vogel, G. Studies in psychophysiology of dreams. Arch. Gen. Psychiatry 2, 421–428 (1960).
Dement, W., Rechtschaffen, A. & Gulevich, G. The nature of the narcoleptic sleep attack. Neurology 16, 18–18 (1966).
Trotti, L. M., Staab, B. A. & Rye, D. B. Test–retest reliability of the multiple sleep latency test in narcolepsy without cataplexy and idiopathic hypersomnia. J. Clin. Sleep Med. 9, 789–795 (2013).
Andlauer, O. et al. Nocturnal rapid eye movement sleep latency for identifying patients with narcolepsy/hypocretin deficiency. JAMA Neurol. 70, 891–902 (2013).
Mignot, E. Correlates of sleep-onset REM periods during the multiple sleep latency test in community adults. Brain 129, 1609–1623 (2006).
Marti, I., Valko, P. O., Khatami, R., Bassetti, C. L. & Baumann, C. R. Multiple sleep latency measures in narcolepsy and behaviourally induced insufficient sleep syndrome. Sleep Med. 10, 1146–1150 (2009).
Drakatos, P. et al. Sleep stage sequence analysis of sleep onset REM periods in the hypersomnias. J. Neurol. Neurosurg. Psychiatry 84, 223–227 (2013).
Bourgin, P., Zeitzer, J. M. & Mignot, E. CSF hypocretin-1 assessment in sleep and neurological disorders. Lancet Neurol. 7, 649–662 (2008). Reports on the levels of hypocretin 1 in the CSF in a large range of neurological disorders.
Hirtz, C. et al. From radioimmunoassay to mass spectrometry: a new method to quantify orexin-A (hypocretin-1) in cerebrospinal fluid. Sci. Rep. 6, 25162 (2016).
Billiard, M. et al. Modafinil: a double-blind multicentric study. Sleep 17, S107–S112 (1994).
Broughton, R. J. et al. Randomized, double-blind, placebo-controlled crossover trial of modafinil in the treatment of excessive daytime sleepiness in narcolepsy. Neurology 49, 444–451 (1997). References 141 and 142 describe the efficacy of modafinil for treating excessive daytime sleepiness in narcolepsy.
Volkow, N. D. et al. Effects of modafinil on dopamine and dopamine transporters in the male human brain: clinical implications. JAMA 301, 1148–1154 (2009).
Mitler, M. M., Harsh, J., Hirshkowitz, M. & Guilleminault, C. Long-term efficacy and safety of modafinil (PROVIGIL®) for the treatment of excessive daytime sleepiness associated with narcolepsy. Sleep Med. 1, 231–243 (2000).
Mignot, E. J. A practical guide to the therapy of narcolepsy and hypersomnia syndromes. Neurotherapeutics 9, 739–752 (2012).
Aran, A. et al. Clinical and therapeutic aspects of childhood narcolepsy-cataplexy: a retrospective study of 51 children. Sleep 33, 1457–1464 (2010).
Mitler, M. M., Aldrich, M. S., Koob, G. F. & Zarcone, V. P. Narcolepsy and its treatment with stimulants. ASDA standards of practice. Sleep 17, 352–371 (1994).
Mitler, M. M., Shafor, R., Hajdukovich, R., Timms, R. M. & Browman, C. P. Treatment of narcolepsy: objective studies on methylphenidate, pemoline, and protriptyline. Sleep 9, 260–264 (1986).
Auger, R. R. et al. Risks of high-dose stimulants in the treatment of disorders of excessive somnolence: a case–control study. Sleep 28, 667–672 (2005).
Mantyh, W. G., Auger, R. R., Morgenthaler, T. I., Silber, M. H. & Moore, W. R. Examining the frequency of stimulant misuse among patients with primary disorders of hypersomnolence: a retrospective cohort study. J. Clin. Sleep Med. 12, 659–662 (2016).
Maitre, M. The gamma-hydroxybutyrate signalling system in brain: organization and functional implications. Prog. Neurobiol. 51, 337–361 (1997).
Huang, Y. S. & Guilleminault, C. Narcolepsy: action of two gamma-aminobutyric acid type B agonists, baclofen and sodium oxybate. Pediatr. Neurol. 41, 9–16 (2009).
Boscolo-Berto, R. et al. Narcolepsy and effectiveness of gamma-hydroxybutyrate (GHB): a systematic review and meta-analysis of randomized controlled trials. Sleep Med. Rev. 16, 431–443 (2012). Reviews the available evidence of the efficacy of sodium oxybate as a treatment for narcolepsy.
Black, J., Pardi, D., Hornfeldt, C. S. & Inhaber, N. The nightly administration of sodium oxybate results in significant reduction in the nocturnal sleep disruption of patients with narcolepsy. Sleep Med. 10, 829–835 (2009).
Black, J. & Houghton, W. C. Sodium oxybate improves excessive daytime sleepiness in narcolepsy. Sleep 29, 939–946 (2006).
Lammers, G. J. et al. Gammahydroxybutyrate and narcolepsy: a double-blind placebo-controlled study. Sleep 16, 216–220 (1993).
van Schie, M. K. et al. Improved vigilance after sodium oxybate treatment in narcolepsy: a comparison between in-field and in-laboratory measurements. J. Sleep Res. 25, 486–496 (2016).
Bogan, R. K., Roth, T., Schwartz, J. & Miloslavsky, M. Time to response with sodium oxybate for the treatment of excessive daytime sleepiness and cataplexy in patients with narcolepsy. J. Clin. Sleep Med. 11, 427–432 (2015).
Mamelak, M. et al. A 12-week open-label, multicenter study evaluating the safety and patient-reported efficacy of sodium oxybate in patients with narcolepsy and cataplexy. Sleep Med. 16, 52–58 (2015).
Wang, Y. G., Swick, T. J., Carter, L. P., Thorpy, M. J. & Benowitz, N. L. Safety overview of postmarketing and clinical experience of sodium oxybate (Xyrem): abuse, misuse, dependence, and diversion. J. Clin. Sleep Med. 5, 365–371 (2009).
[No authors listed.] The abrupt cessation of therapeutically administered sodium oxybate (GHB) does not cause withdrawal symptoms. J. Toxicol. Clin. Toxicol. 41, 131–135 (2003).
Moller, L. R. & Ostergaard, J. R. Treatment with venlafaxine in six cases of children with narcolepsy and with cataplexy and hypnagogic hallucinations. J. Child Adolesc. Psychopharmacol. 19, 197–201 (2009).
Broderick, M. & Guilleminault, C. Rebound cataplexy after withdrawal from antidepressants. Sleep Med. 10, 403–404 (2009).
Vignatelli, L., D'Alessandro, R. & Candelise, L. Antidepressant drugs for narcolepsy. Cochrane Database Syst. Rev. 1, CD003724 (2008).
Zhang, S. et al. Clinical effect of atomoxetine hydrochloride in 66 children with narcolepsy. Zhonghua Er Ke Za Zhi 53, 760–764 (in Chinese) (2015).
Izzi, F. et al. Effective treatment of narcolepsy-cataplexy with duloxetine: a report of three cases. Sleep Med. 10, 153–154 (2009).
Mansukhani, M. P. & Kotagal, S. Sodium oxybate in the treatment of childhood narcolepsy-cataplexy: a retrospective study. Sleep Med. 13, 606–610 (2012).
Lecendreux, M. et al. Clinical experience suggests that modafinil is an effective and safe treatment for paediatric narcolepsy. J. Sleep Res. 21, 481–483 (2012).
Thorpy, M., Zhao, C. G. & Dauvilliers, Y. Management of narcolepsy during pregnancy. Sleep Med. 14, 367–376 (2013).
Roth, T. et al. Disrupted nighttime sleep in narcolepsy. J. Clin. Sleep Med. 9, 955–965 (2013).
Naumann, A., Bellebaum, C. & Daum, I. Cognitive deficits in narcolepsy. J. Sleep Res. 15, 329–338 (2006).
Van Schie, M. K. M. et al. Sustained attention to response task (SART) shows impaired vigilance in a spectrum of disorders of excessive daytime sleepiness. J. Sleep Res. 21, 390–395 (2012).
Mullington, J. & Broughton, R. Scheduled naps in the management of daytime sleepiness in narcolepsy-cataplexy. Sleep 16, 444–456 (1993).
Rogers, A. E., Aldrich, M. S. & Lin, X. A comparison of three different sleep schedules for reducing daytime sleepiness in narcolepsy. Sleep 24, 385–391 (2001).
Khatami, R. et al. Challenging sleep homeostasis in narcolepsy-cataplexy: implications for non-REM and REM sleep regulation. Sleep 31, 859–867 (2008).
Kapella, M. C. et al. Health-related stigma as a determinant of functioning in young adults with narcolepsy. PLoS ONE 10, e0122478 (2015).
Ohayon, M. M. Narcolepsy is complicated by high medical and psychiatric comorbidities: a comparison with the general population. Sleep Med. 14, 488–492 (2013).
Jennum, P., Ibsen, R., Petersen, E. R., Knudsen, S. & Kjellberg, J. Health, social, and economic consequences of narcolepsy: a controlled national study evaluating the societal effect on patients and their partners. Sleep Med. 13, 1086–1093 (2012).
Dauvilliers, Y. et al. Psychological health in central hypersomnias: the French Harmony study. J. Neurol. Neurosurg. Psychiatry 80, 636–641 (2009).
Dodel, R. et al. The socioeconomic impact of narcolepsy. Sleep 27, 1123–1128 (2004).
Rieger, M., Mayer, G. & Gauggel, S. Attention deficits in patients with narcolepsy. Sleep 26, 36–43 (2003).
Thomann, J., Baumann, C. R., Landolt, H.-P. & Werth, E. Psychomotor vigilance task demonstrates impaired vigilance in disorders with excessive daytime sleepiness. J. Clin. Sleep Med. 10, 1019–1024 (2014).
Philip, P. et al. Maintenance of Wakefulness Test scores and driving performance in sleep disorder patients and controls. Int. J. Psychophysiol. 89, 195–202 (2013).
Pizza, F. et al. Car crashes and central disorders of hypersomnolence: a French study. PLoS ONE 10, e0129386 (2015).
Hegmann, K. T., Andersson, G. B., Greenberg, M. I., Phillips, B. & Rizzo, M. FMCSA's medical review board: five years of progress in commercial driver medical examinations. J. Occup. Environ. Med. 54, 424–430 (2012).
Philip, P. et al. Modafinil improves real driving performance in patients with hypersomnia: a randomized double-blind placebo-controlled crossover clinical trial. Sleep 37, 483–487 (2014).
Pizza, F., Tartarotti, S., Poryazova, R., Baumann, C. R. & Bassetti, C. L. Sleep-disordered breathing and periodic limb movements in narcolepsy with cataplexy: a systematic analysis of 35 consecutive patients. Eur. Neurol. 70, 22–26 (2013).
Sansa, G., Iranzo, A. & Santamaria, J. Obstructive sleep apnea in narcolepsy. Sleep Med. 11, 93–95 (2010).
Donjacour, C. E. et al. Glucose and fat metabolism in narcolepsy and the effect of sodium oxybate: a hyperinsulinemic-euglycemic clamp study. Sleep 37, 795–801 (2014).
Huang, Y. S., Guilleminault, C., Chen, C. H., Lai, P. C. & Hwang, F. M. Narcolepsy-cataplexy and schizophrenia in adolescents. Sleep Med. 15, 15–22 (2014).
Szakacs, A., Hallbook, T., Tideman, P., Darin, N. & Wentz, E. Psychiatric comorbidity and cognitive profile in children with narcolepsy with or without association to the H1N1 influenza vaccination. Sleep 38, 615–621 (2015). Investigates the psychiatric and cognitive problems in children with NT1.
Antelmi, E., Vandi, S., Pizza, F., Liguori, R. & Plazzi, G. Parkinsonian tremor persisting during cataplexy. Sleep Med. 17, 174–176 (2016).
Economou, N. T., Manconi, M., Ghika, J., Raimondi, M. & Bassetti, C. L. Development of Parkinson and Alzheimer diseases in two cases of narcolepsy-cataplexy. Eur. Neurol. 67, 48–50 (2012).
Sorensen, G. L. et al. Attenuated heart rate response is associated with hypocretin deficiency in patients with narcolepsy. Sleep 36, 91–98 (2013).
van der Meijden, W. P. et al. Time- and state-dependent analysis of autonomic control in narcolepsy: higher heart rate with normal heart rate variability independent of sleep fragmentation. J. Sleep Res. 24, 206–214 (2015).
Ohayon, M. M. et al. Increased mortality in narcolepsy. Sleep 37, 439–444 (2014).
Jennum, P., Mayer, G., Ju, Y. E. & Postuma, R. Morbidities in rapid eye movement sleep behavior disorder. Sleep Med. 14, 782–787 (2013).
Tesoriero, C. et al. H1N1 influenza virus induces narcolepsy-like sleep disruption and targets sleep-wake regulatory neurons in mice. Proc. Natl Acad. Sci. USA 113, E368–E377 (2016).
Nishino, S. & Mignot, E. Article reviewed: plasma orexin-A is lower in patients with narcolepsy. Sleep Med. 2, 377–378 (2002).
Pizza, F. et al. Daytime continuous polysomnography predicts MSLT results in hypersomnias of central origin. J. Sleep Res. 22, 32–40 (2013).
Lin, J. S. et al. An inverse agonist of the histamine H3 receptor improves wakefulness in narcolepsy: studies in orexin−/− mice and patients. Neurobiol. Dis. 30, 74–83 (2008).
Dauvilliers, Y. et al. Pitolisant versus placebo or modafinil in patients with narcolepsy: a double-blind, randomised trial. Lancet Neurol. 12, 1068–1075 (2013).
Deadwyler, S. A., Porrino, L., Siegel, J. M. & Hampson, R. E. Systemic and nasal delivery of orexin-A (hypocretin-1) reduces the effects of sleep deprivation on cognitive performance in nonhuman primates. J. Neurosci. 27, 14239–14247 (2007).
Weinhold, S. L. et al. The effect of intranasal orexin-A (hypocretin-1) on sleep, wakefulness and attention in narcolepsy with cataplexy. Behav. Brain Res. 262, 8–13 (2014).
Nagahara, T. et al. Design and synthesis of non-peptide, selective orexin receptor 2 agonists. J. Med. Chem. 58, 7931–7937 (2015).
Hasan, S. et al. How to keep the brain awake? The complex molecular pharmacogenetics of wake promotion. Neuropsychopharmacology 34, 1625–1640 (2009).
Ruoff, C. et al. Effect of oral JZP-110 (ADX-N05) on wakefulness and sleepiness in adults with narcolepsy: a phase 2b study. Sleep 39, 1379–1387 (2016).
Bogan, R. K. et al. Effect of oral JZP-110 (ADX-N05) treatment on wakefulness and sleepiness in adults with narcolepsy. Sleep Med. 16, 1102–1108 (2015).
Hara, J. et al. Genetic ablation of orexin neurons in mice results in narcolepsy, hypophagia, and obesity. Neuron 30, 345–354 (2001).
Tabuchi, S. et al. Conditional ablation of orexin/hypocretin neurons: a new mouse model for the study of narcolepsy and orexin system function. J. Neurosci. 34, 6495–6509 (2014).
Bernard-Valnet, R. et al. CD8 T cell-mediated killing of orexinergic neurons induces a narcolepsy-like phenotype in mice. Proc. Natl Acad. Sci. USA 113, 10956–10961 (2016).
Rose, N. R. & Bona, C. Defining criteria for autoimmune diseases (Witebsky's postulates revisited). Immunol. Today 14, 426–430 (1993).
Mignot, E. Why we sleep: the temporal organization of recovery. PLoS Biol. 6, e106 (2008).
Barateau, L., Lopez, R. & Dauvilliers, Y. Treatment options for narcolepsy. CNS Drugs 30, 369–379 (2016).
Lavie, P. & Peled, R. Narcolepsy is a rare disease in Israel. Sleep 10, 608–609 (1987).
Darwish, M., Kirby, M., Hellriegel, E. T. & Robertson, P. Jr. Armodafinil and modafinil have substantially different pharmacokinetic profiles despite having the same terminal half-lives: analysis of data from three randomized, single-dose, pharmacokinetic studies. Clin. Drug Investig. 29, 613–623 (2009).
Scharf, M. B., Lai, A. A., Branigan, B., Stover, R. & Berkowitz, D. B. Pharmacokinetics of gammahydroxybutyrate (GHB) in narcoleptic patients. Sleep 21, 507–514 (1998).
Acknowledgements
B.R.K. has received funding from the Lundbeck Foundation. S.K. has received funding from the Norwegian Ministry of Health and Care Services. H.M.O. has received funding from the Finnish Cultural Foundation, Jalmari and Rauha Ahokas Foundation, Sigrid Juselius Foundation, Emil Aaltonen Foundation, Päivikki and Sakari Sohlberg Foundation, Orion Farmos Research Foundation and Instrumentarium Science Foundation.
Author information
Authors and Affiliations
Contributions
Introduction (B.R.K.); Epidemiology (H.M.O.); Mechanisms/pathophysiology (S.O., Y.D. and B.R.K.); Diagnosis, screening and prevention (F.P.); Management (S.K., Y.D. and S.O.); Quality of life (S.K. and P.J.J.); Outlook (B.R.K., F.P. and Y.D.); Overview of Primer (B.R.K.).
Corresponding author
Ethics declarations
Competing interests
B.R.K. has received an unrestricted investigator-driven project grant from UCB Pharma. H.M.O. has acted as a consultant for Jazz Pharmaceuticals. P.J.J. has received funds from UCB Pharma. Y.D. has received funds for seminars, board engagements and travel to conferences by UCB Pharma, Jazz Pharmaceuticals, Theranexus, GlaxoSmithKline, Actelion and BioProject. S.O. has received an unrestricted investigator-driven project grant from UCB Pharma and conference travel support from UCB Pharma, Novartis and Boehringer Ingelheim. S.K. and F.P. declare no competing interests.
Rights and permissions
About this article
Cite this article
Kornum, B., Knudsen, S., Ollila, H. et al. Narcolepsy. Nat Rev Dis Primers 3, 16100 (2017). https://doi.org/10.1038/nrdp.2016.100
Published:
DOI: https://doi.org/10.1038/nrdp.2016.100
This article is cited by
-
Genetics of circadian rhythms and sleep in human health and disease
Nature Reviews Genetics (2023)
-
Alexithymia, impulsiveness, emotion, and eating dyscontrol: similarities and differences between narcolepsy type 1 and type 2
Sleep and Biological Rhythms (2023)
-
Evidence of Accidental Dosing Errors with Immediate-Release Sodium Oxybate: Data from the US Food and Drug Administration Adverse Event Reporting System
Drugs - Real World Outcomes (2023)
-
Harriet Tubman’s Hypersomnia: Insights from Historical and Medical Perspectives
Journal of General Internal Medicine (2023)
-
Serum metabolomics study of narcolepsy type 1 based on ultra-performance liquid chromatography–tandem mass spectrometry
Amino Acids (2023)