Abstract
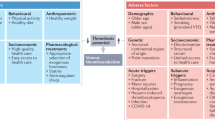
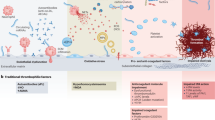
Venous thromboembolism (VTE) encompasses deep-vein thrombosis (DVT) and pulmonary embolism. VTE is the leading cause of lost disability-adjusted life years and the third leading cause of cardiovascular death in the world. DVT leads to post-thrombotic syndrome, whereas pulmonary embolism can cause chronic pulmonary hypertension, both of which reduce quality of life. Genetic and acquired risk factors for thrombosis include non-O blood groups, factor V Leiden mutation, oral contraceptive use, hormone replacement therapy, advanced age, surgery, hospitalization and long-haul travel. A combination of blood stasis, plasma hypercoagulability and endothelial dysfunction is thought to trigger thrombosis, which starts most often in the valve pockets of large veins. Animal studies have revealed pathogenic roles for leukocytes, platelets, tissue factor-positive microvesicles, neutrophil extracellular traps and factors XI and XII. Diagnosis of VTE requires testing and exclusion of other pathologies, and typically involves laboratory measures (such as D-dimer) and diagnostic imaging. VTE is treated with anticoagulants and occasionally with thrombolytics to prevent thrombus extension and to reduce thrombus size. Anticoagulants are also used to reduce recurrence. New therapies with improved safety profiles are needed to prevent and treat venous thrombosis. For an illustrated summary of this Primer, visit: http://go.nature.com/8ZyCuY
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Grant, J. D. et al. Diagnosis and management of upper extremity deep-vein thrombosis in adults. Thromb. Haemost. 108, 1097–1108 (2012).
Virchow, R. Gesammalte Abhandlungen zur Wissenschaftlichen Medizin. 219–732 (in German) (Medinger Sohn & Co., Frankfurt, 1856).
Bagot, C. N. & Arya, R. Virchow and his triad: a question of attribution. Br. J. Haematol. 143, 180–190 (2008).
International Society on Thrombosis and Haemostasis. About World Thrombosis Day [online], (2014).
Naess, I. A. et al. Incidence and mortality of venous thrombosis: a population-based study. J. Thromb. Haemost. 5, 692–699 (2007).
Flinterman, L. E., van Hylckama Vlieg, A., Cannegieter, S. C. & Rosendaal, F. R. Long-term survival in a large cohort of patients with venous thrombosis: incidence and predictors. PLoS Med. 9, e1001155 (2012). This study describes the long-term mortality in a large cohort of Dutch patients with DVT.
Kahn, S. R. et al. The postthrombotic syndrome: evidence-based prevention, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation 130, 1636–1661 (2014).
Cohen, A. T. et al. Venous thromboembolism (VTE) in Europe. The number of VTE events and associated morbidity and mortality. Thromb. Haemost. 98, 756–764 (2007).
ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to the global disease burden. J. Thromb. Haemost. 12, 1580–1590 (2014). This systematic review of the literature shows that VTE is a major burden of disease across low-income, middle-income and high-income countries.
Mackman, N. New insights into the mechanisms of venous thrombosis. J. Clin. Invest. 122, 2331–2336 (2012).
Silverstein, M. D. et al. Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch. Intern. Med. 158, 585–593 (1998).
Reitsma, P. H., Versteeg, H. H. & Middeldorp, S. Mechanistic view of risk factors for venous thromboembolism. Arterioscler. Thromb. Vasc. Biol. 32, 563–568 (2012).
Zakai, N. A. & McClure, L. A. Racial differences in venous thromboembolism. J. Thromb. Haemost. 9, 1877–1882 (2011).
Roach, R. E. et al. The risk of venous thrombosis in individuals with a history of superficial vein thrombosis and acquired venous thrombotic risk factors. Blood 122, 4264–4269 (2013).
Cannegieter, S. C. et al. Risk of venous and arterial thrombotic events in patients diagnosed with superficial vein thrombosis: a nationwide cohort study. Blood 125, 229–235 (2015).
Rosendaal, F. R. & Reitsma, P. H. Genetics of venous thrombosis. J. Thromb. Haemost. 7 (Suppl. 1), 301–304 (2009).
Bezemer, I. D. et al. F9 Malmo, factor IX and deep vein thrombosis. Haematologica 94, 693–699 (2009).
Bezemer, I. D. et al. Gene variants associated with deep vein thrombosis. JAMA 299, 1306–1314 (2008).
Li, Y. et al. Genetic variants associated with deep vein thrombosis: the F11 locus. J. Thromb. Haemost. 7, 1802–1808 (2009).
Austin, H. et al. New gene variants associated with venous thrombosis: a replication study in White and Black Americans. J. Thromb. Haemost. 9, 489–495 (2011).
Andresen, M. S. et al. Mortality and recurrence after treatment of VTE: long term follow-up of patients with good life-expectancy. Thromb. Res. 127, 540–546 (2011).
Wolberg, A. S., Aleman, M. M., Leiderman, K. & Machlus, K. R. Procoagulant activity in hemostasis and thrombosis: Virchow's Triad revisited. Anesth. Analg. 114, 275–285 (2012).
Meier, T. R. et al. Prophylactic P-selectin inhibition with PSI-421 promotes resolution of venous thrombosis without anticoagulation. Thromb. Haemost. 99, 343–351 (2008).
Myers, D. D. Jr. Nonhuman primate models of thrombosis. Thromb. Res. 129 (Suppl. 2), S65–S69 (2012).
Brill, A. et al. von Willebrand factor-mediated platelet adhesion is critical for deep vein thrombosis in mouse models. Blood 117, 1400–1407 (2011).
von Bruhl, M. L. et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 209, 819–835 (2012). This study demonstrates the roles of leukocyte tissue factor, factor XIIa, neutrophils and platelets in a mouse model of thrombosis.
Geddings, J. et al. Strengths and weaknesses of a new mouse model of thrombosis induced by inferior vena cava stenosis: communication from the SSC of the ISTH. J. Thromb. Haemost. 12, 571–573 (2014).
Myers, D. Jr. et al. Selectins influence thrombosis in a mouse model of experimental deep venous thrombosis. J. Surg. Res. 108, 212–221 (2002).
Diaz, J. A. et al. Thrombogenesis with continuous blood flow in the inferior vena cava. A novel mouse model. Thromb. Haemost. 104, 366–375 (2010).
Cooley, B. C., Szema, L., Chen, C. Y., Schwab, J. P. & Schmeling, G. A murine model of deep vein thrombosis: characterization and validation in transgenic mice. Thromb. Haemost. 94, 498–503 (2005).
Wang, X., Smith, P. L., Hsu, M. Y., Ogletree, M. L. & Schumacher, W. A. Murine model of ferric chloride-induced vena cava thrombosis: evidence for effect of potato carboxypeptidase inhibitor. J. Thromb. Haemost. 4, 403–410 (2006).
Aghourian, M. N., Lemarie, C. A. & Blostein, M. D. In vivo monitoring of venous thrombosis in mice. J. Thromb. Haemost. 10, 447–452 (2012).
Cardenas, J. C. et al. Overexpression of the cell cycle inhibitor p16INK4a promotes a prothrombotic phenotype following vascular injury in mice. Arterioscler. Thromb. Vasc. Biol. 31, 827–833 (2011).
Machlus, K. R., Cardenas, J. C., Church, F. C. & Wolberg, A. S. Causal relationship between hyperfibrinogenemia, thrombosis, and resistance to thrombolysis in mice. Blood 117, 4953–4963 (2011).
Machlus, K. R., Lin, F.-C. & Wolberg, A. S. Procoagulant activity induced by vascular injury determines contribution of elevated factor VIII to thrombosis and thrombus stability in mice. Blood 118, 390–398 (2011).
Warlow, C., Ogston, D. & Douglas, A. S. Deep venous thrombosis of the legs after strokes. Part I — incidence and predisposing factors. BMJ 1, 1178–1181 (1976).
Sevitt, S. The structure and growth of valve-pocket thrombi in femoral veins. J. Clin. Pathol. 27, 517–528 (1974).
Hamer, J. D., Malone, P. C. & Silver, I. A. The PO2 in venous valve pockets: its possible bearing on thrombogenesis. Br. J. Surg. 68, 166–170 (1981).
Lin, Z. et al. Krüppel-like factor 2 (KLF2) regulates endothelial thrombotic function. Circ. Res. 96, e48–e57 (2005).
Lin, Z., Hamik, A., Jain, R., Kumar, A. & Jain, M. K. Krüppel-like factor 2 inhibits protease activated receptor-1 expression and thrombin-mediated endothelial activation. Arterioscler. Thromb. Vasc. Biol. 26, 1185–1189 (2006).
Atkins, G. B. & Jain, M. K. Role of Krüppel-like transcription factors in endothelial biology. Circ. Res. 100, 1686–1695 (2007).
Brooks, E. G. et al. Valves of the deep venous system: an overlooked risk factor. Blood 114, 1276–1279 (2009).
Zhou, J., May, L., Liao, P., Gross, P. L. & Weitz, J. I. Inferior vena cava ligation rapidly induces tissue factor expression and venous thrombosis in rats. Arterioscler. Thromb. Vasc. Biol. 29, 863–869 (2009).
Wakefield, T. W. et al. P-selectin and TNF inhibition reduce venous thrombosis inflammation. J. Surg. Res. 64, 26–31 (1996).
Furie, B. & Furie, B. C. Role of platelet P-selectin and microparticle PSGL-1 in thrombus formation. Trends Mol. Med. 10, 171–178 (2004).
Myers, D. D. Jr. et al. P-selectin antagonism causes dose-dependent venous thrombosis inhibition. Thromb. Haemost. 85, 423–429 (2001).
Day, S. M. et al. Macrovascular thrombosis is driven by tissue factor derived primarily from the blood vessel wall. Blood 105, 192–198 (2005).
Mackman, N., Tilley, R. E. & Key, N. S. Role of the extrinsic pathway of blood coagulation in hemostasis and thrombosis. Arterioscler. Thromb. Vasc. Biol. 27, 1687–1693 (2007).
Butenas, S., van't Veer, C., Cawthern, K., Brummel, K. E. & Mann, K. G. Models of blood coagulation. Blood Coag. Fibrinol. 11 (Suppl. 1), S9–S13 (2000).
Allen, G. A. et al. Impact of procoagulant concentration on rate, peak and total thrombin generation in a model system. J. Thromb. Haemost. 2, 402–413 (2004).
Machlus, K. R. et al. Effects of tissue factor, thrombomodulin, and elevated clotting factor levels on thrombin generation in the calibrated automated thrombogram. Thromb. Haemost. 102, 936–944 (2009).
Wichers, I. M. et al. Assessment of coagulation and fibrinolysis in families with unexplained thrombophilia. Thromb. Haemost. 101, 465–470 (2009).
Brandts, A., van Hylckama Vlieg, A., Rosing, J., Baglin, T. P. & Rosendaal, F. R. The risk of venous thrombosis associated with a high endogenous thrombin potential in the absence and presence of activated protein C. J. Thromb. Haemost. 5, 416–418 (2007).
Dargaud, Y., Trzeciak, M. C., Bordet, J. C., Ninet, J. & Negrier, C. Use of calibrated automated thrombinography +/− thrombomodulin to recognise the prothrombotic phenotype. Thromb. Haemost. 96, 562–567 (2006).
van Hylckama Vlieg, A. et al. Elevated endogenous thrombin potential is associated with an increased risk of a first deep venous thrombosis but not with the risk of recurrence. Br. J. Haematol. 138, 769–774 (2007).
Tripodi, A. et al. The endogenous thrombin potential and the risk of venous thromboembolism. Thromb. Res. 121, 353–359 (2007).
ten Cate-Hoek, A. J. et al. Thrombin generation in patients after acute deep-vein thrombosis. Thromb. Haemost. 100, 240–245 (2008).
Hron, G., Kollars, M., Binder, B. R., Eichinger, S. & Kyrle, P. A. Identification of patients at low risk for recurrent venous thromboembolism by measuring thrombin generation. JAMA 296, 397–402 (2006).
Eichinger, S., Hron, G., Kollars, M. & Kyrle, P. A. Prediction of recurrent venous thromboembolism by endogenous thrombin potential and D-dimer. Clin. Chem. 54, 2042–2048 (2008).
Besser, M., Baglin, C., Luddington, R., van Hylckama Vlieg, A. & Baglin, T. High rate of unprovoked recurrent venous thrombosis is associated with high thrombin-generating potential in a prospective cohort study. J. Thromb. Haemost. 6, 1720–1725 (2008).
Tripodi, A. et al. High thrombin generation measured in the presence of thrombomodulin is associated with an increased risk of recurrent venous thromboembolism. J. Thromb. Haemost. 6, 1327–1333 (2008).
Aleman, M. M. et al. Elevated prothrombin promotes venous, but not arterial, thrombosis in mice. Arterioscler. Thromb. Vasc. Biol. 33, 1829–1836 (2013).
Meltzer, M. E., Lisman, T., Doggen, C. J., de Groot, P. G. & Rosendaal, F. R. Synergistic effects of hypofibrinolysis and genetic and acquired risk factors on the risk of a first venous thrombosis. PLoS Med. 5, e97 (2008).
Meltzer, M. E. et al. Venous thrombosis risk associated with plasma hypofibrinolysis is explained by elevated plasma levels of TAFI and PAI-1. Blood 116, 113–121 (2010).
Fay, W. P., Parker, A. C., Condrey, L. R. & Shapiro, A. D. Human plasminogen activator inhibitor-1 (PAI-1) deficiency: characterization of a large kindred with a null mutation in the PAI-1 gene. Blood 90, 204–208 (1997).
Engelmann, B. & Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 13, 34–45 (2013).
Connolly, G. C., Phipps, R. P. & Francis, C. W. Platelets and cancer-associated thrombosis. Seminars Oncol. 41, 302–310 (2014).
Jensvoll, H., Blix, K., Braekkan, S. K. & Hansen, J. B. Platelet count measured prior to cancer development is a risk factor for future symptomatic venous thromboembolism: the Tromso Study. PLoS ONE 9, e92011 (2014).
Zakai, N. A., Wright, J. & Cushman, M. Risk factors for venous thrombosis in medical inpatients: validation of a thrombosis risk score. J. Thromb. Haemost. 2, 2156–2161 (2004).
Khorana, A. A., Francis, C. W., Culakova, E. & Lyman, G. H. Risk factors for chemotherapy-associated venous thromboembolism in a prospective observational study. Cancer 104, 2822–2829 (2005).
Simanek, R. et al. High platelet count associated with venous thromboembolism in cancer patients: results from the Vienna Cancer and Thrombosis Study (CATS). J. Thromb. Haemost. 8, 114–120 (2010).
Castellucci, L. A. et al. Efficacy and safety outcomes of oral anticoagulants and antiplatelet drugs in the secondary prevention of venous thromboembolism: systematic review and network meta-analysis. BMJ 347, f5133 (2013).
Simes, J. et al. Aspirin for the prevention of recurrent venous thromboembolism: the INSPIRE Collaboration. Circulation 130, 1062–1071 (2014).
Riedl, J., Pabinger, I. & Ay, C. Platelets in cancer and thrombosis. Hamostaseologie 34, 54–62 (2014).
McGuinness, C. L. et al. Recruitment of labelled monocytes by experimental venous thrombi. Thromb. Haemost. 85, 1018–1024 (2001).
Blix, K., Jensvoll, H., Braekkan, S. K. & Hansen, J. B. White blood cell count measured prior to cancer development is associated with future risk of venous thromboembolism — the Tromso study. PLoS ONE 8, e73447 (2013).
Connolly, G. C. et al. Leukocytosis, thrombosis and early mortality in cancer patients initiating chemotherapy. Thromb. Res. 126, 113–118 (2010).
Fuchs, T. A. et al. Extracellular DNA traps promote thrombosis. Proc. Natl Acad. Sci. USA 107, 15880–15885 (2010). This study shows that NETs capture platelets and red blood cells, and enhance fibrin deposition.
Savchenko, A. S. et al. Neutrophil extracellular traps form predominantly during the organizing stage of human venous thromboembolism development. J. Thromb. Haemost. 12, 860–870 (2014).
Martinod, K. & Wagner, D. D. Thrombosis: tangled up in NETs. Blood 123, 2768–2776 (2014).
Kannemeier, C. et al. Extracellular RNA constitutes a natural procoagulant cofactor in blood coagulation. Proc. Natl Acad. Sci. USA 104, 6388–6393 (2007).
Martinod, K. et al. Neutrophil histone modification by peptidylarginine deiminase 4 is critical for deep vein thrombosis in mice. Proc. Natl Acad. Sci. USA 110, 8674–8679 (2013).
Massberg, S. et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 16, 887–896 (2010).
Petersen, L. C., Bjorn, S. E. & Nordfang, O. Effect of leukocyte proteinases on tissue factor pathway inhibitor. Thromb. Haemost. 67, 537–541 (1992).
Steppich, B. A., Seitz, I., Busch, G., Stein, A. & Ott, I. Modulation of tissue factor and tissue factor pathway inhibitor-1 by neutrophil proteases. Thromb. Haemost. 100, 1068–1075 (2008).
Patterson, K. A. et al. Rosuvastatin reduced deep vein thrombosis in ApoE gene deleted mice with hyperlipidemia through non-lipid lowering effects. Thromb. Res. 131, 268–276 (2013).
Owens, A. P. 3rd et al. Monocyte tissue factor-dependent activation of coagulation in hypercholesterolemic mice and monkeys is inhibited by simvastatin. J. Clin. Invest. 122, 558–568 (2012).
Glynn, R. J. et al. A randomized trial of rosuvastatin in the prevention of venous thromboembolism. N. Engl. J. Med. 360, 1851–1861 (2009).
Rahimi, K. et al. Effect of statins on venous thromboembolic events: a meta-analysis of published and unpublished evidence from randomised controlled trials. PLoS Med. 9, e1001310 (2012).
Owens, A. P. 3rd & Mackman, N. Microparticles in hemostasis and thrombosis. Circ. Res. 108, 1284–1297 (2011).
Ramacciotti, E. et al. Leukocyte- and platelet-derived microparticles correlate with thrombus weight and tissue factor activity in an experimental mouse model of venous thrombosis. Thromb. Haemost. 101, 748–754 (2009).
Owens, A. P., 3rd & Mackman, N. MP's and VTE's: fact or fiction. Thromb. Res. 128, 505–506 (2011).
Khorana, A. A. et al. Plasma tissue factor may be predictive of venous thromboembolism in pancreatic cancer. J. Thromb. Haemost. 6, 1983–1985 (2008).
Geddings, J. E. & Mackman, N. Tumor-derived tissue factor-positive microparticles and venous thrombosis in cancer patients. Blood 122, 1873–1880 (2013).
Thomas, G. M. et al. Cancer cell-derived microparticles bearing P-selectin glycoprotein ligand 1 accelerate thrombus formation in vivo. J. Exp. Med. 206, 1913–1927 (2009).
Wang, J. G. et al. Tumor-derived tissue factor activates coagulation and enhances thrombosis in a mouse xenograft model of human pancreatic cancer. Blood 119, 5543–5552 (2012).
Ho, C. H. The hemostatic effect of packed red cell transfusion in patients with anemia. Transfusion 38, 1011–1014 (1998).
Khorana, A. A. et al. Blood transfusions, thrombosis, and mortality in hospitalized patients with cancer. Arch. Intern. Med. 168, 2377–2381 (2008).
Gangireddy, C. et al. Risk factors and clinical impact of postoperative symptomatic venous thromboembolism. J. Vasc. Surg. 45, 335–341; discussion 341–332 (2007).
Kumar, M. A. et al. Red blood cell transfusion increases the risk of thrombotic events in patients with subarachnoid hemorrhage. Neurocrit. Care 20, 84–90 (2014).
Andrews, D. A. & Low, P. S. Role of red blood cells in thrombosis. Curr. Opin. Hematol. 6, 76–82 (1999).
Baskurt, O. K. & Meiselman, H. J. Blood rheology and hemodynamics. Semin. Thromb. Hemost. 29, 435–450 (2003).
Peyrou, V. et al. Contribution of erythrocytes to thrombin generation in whole blood. Thromb. Haemost. 81, 400–406 (1999).
Horne, M. K., 3rd, Cullinane, A. M., Merryman, P. K. & Hoddeson, E. K. The effect of red blood cells on thrombin generation. Br. J. Haematol. 133, 403–408 (2006).
Whelihan, M. F., Zachary, V., Orfeo, T. & Mann, K. G. Prothrombin activation in blood coagulation: the erythrocyte contribution to thrombin generation. Blood 120, 3837–3845 (2012).
Rubin, O. et al. Red blood cell-derived microparticles isolated from blood units initiate and propagate thrombin generation. Transfusion 53, 1744–1754 (2013).
Van Der Meijden, P. E. et al. Platelet- and erythrocyte-derived microparticles trigger thrombin generation via factor XIIa. J. Thromb. Haemost. 10, 1355–1362 (2012).
van Gelder, J. M., Nair, C. H. & Dhall, D. P. The significance of red cell surface area to the permeability of fibrin network. Biorheology 31, 259–275 (1994).
Cines, D. B. et al. Clot contraction: compression of erythrocytes into tightly packed polyhedra and redistribution of platelets and fibrin. Blood 123, 1596–1603 (2014).
Wohner, N. et al. Lytic resistance of fibrin containing red blood cells. Arterioscler. Thromb. Vasc. Biol. 31, 2306–2313 (2011).
Aleman, M. M. et al. Factor XIII activity mediates red blood cell retention in venous thrombi. J. Clin. Invest. 124, 3590–3600 (2014). This study shows that factor XIIIa activity is required to retain red blood cells in venous clots and that inhibition of factor XIIIa reduces clot size.
Varma, M. R. et al. Neutropenia impairs venous thrombosis resolution in the rat. J. Vasc. Surg. 38, 1090–1098 (2003).
Sood, V. et al. Urokinase plasminogen activator independent early experimental thrombus resolution: MMP2 as an alternative mechanism. Thromb. Haemost. 104, 1174–1183 (2010).
Singh, I. et al. Failure of thrombus to resolve in urokinase-type plasminogen activator gene-knockout mice: rescue by normal bone marrow-derived cells. Circulation 107, 869–875 (2003).
Deatrick, K. B. et al. The effect of matrix metalloproteinase 2 and matrix metalloproteinase 2/9 deletion in experimental post-thrombotic vein wall remodeling. J. Vasc. Surg. 58, 1375–1384.e2 (2013).
Wakefield, T. W. et al. Venous thrombosis-associated inflammation and attenuation with neutralizing antibodies to cytokines and adhesion molecules. Arterioscler. Thromb. Vasc. Biol. 15, 258–268 (1995).
Henke, P. K. et al. Targeted deletion of CCR2 impairs deep vein thombosis resolution in a mouse model. J. Immunol. 177, 3388–3397 (2006).
Ali, T. et al. Monocyte recruitment in venous thrombus resolution. J. Vasc. Surg. 43, 601–608 (2006).
Obi, A. T. et al. Plasminogen activator-1 overexpression decreases experimental postthrombotic vein wall fibrosis by a non-vitronectin-dependent mechanism. J. Thromb. Haemost. 12, 1353–1363 (2014).
Baldwin, J. F. et al. The role of urokinase plasminogen activator and plasmin activator inhibitor-1 on vein wall remodeling in experimental deep vein thrombosis. J. Vasc. Surg. 56, 1089–1097 (2012).
Office of the Surgeon General (US); National Heart, Lung, and Blood Institute (US). The surgeon general's call to action to prevent deep vein thrombosis and pulmonary embolism. Rockville (MD): Office of the Surgeon General (US)[online], (2008).
Kahn, S. R. et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 141 (Suppl. 2), e195S–e226S (2012). This paper provides evidence-based guidelines for VTE prevention in non-surgical patients. These guidelines are important because pulmonary embolism is the principal cause of preventable death in hospitalized patients.
Falck-Ytter, Y. et al. Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 141 (Suppl. 2), e278S–e325S (2012).
Wu, O. et al. Screening for thrombophilia in high-risk situations: systematic review and cost-effectiveness analysis. The Thrombosis: Risk and Economic Assessment of Thrombophilia Screening (TREATS) study. Health Technol. Assess. 10, 1–110 (2006).
Kearon, C. Natural history of venous thromboembolism. Circulation 107, I22–I30 (2003).
Levy, M. M., Albuquerque, F. & Pfeifer, J. D. Low incidence of pulmonary embolism associated with upper-extremity deep venous thrombosis. Ann. Vasc. Surg. 26, 964–972 (2012).
Meignan, M. et al. Systematic lung scans reveal a high frequency of silent pulmonary embolism in patients with proximal deep venous thrombosis. Arch. Intern. Med. 160, 159–164 (2000). By performing systematic lung scans in patients presenting with proximal DVT, the authors show that at least 40% of such patients have asymptomatic emboli.
Bates, S. M. et al. Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 141 (Suppl. 2), e351S–e418S (2012). This paper provides an evidence-based approach to the diagnosis of DVT.
Huisman, M. V. & Klok, F. A. Diagnostic management of acute deep vein thrombosis and pulmonary embolism. J. Thromb. Haemost. 11, 412–422 (2013).
Linkins, L. A. et al. Selective D-dimer testing for diagnosis of a first suspected episode of deep venous thrombosis: a randomized trial. Ann. Intern. Med. 158, 93–100 (2013).
Righini, M. et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA 311, 1117–1124 (2014).
Authors/Task Force Members et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism: the task force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC) endorsed by the European Respiratory Society (ERS). Eur. Heart J. 35, 3033–3073 (2014).
Kearon, C., Ginsberg, J. S. & Hirsh, J. The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism. Ann. Intern. Med. 129, 1044–1049 (1998).
Klok, F. A., Mos, I. C., Kroft, L. J., de Roos, A. & Huisman, M. V. Computed tomography pulmonary angiography as a single imaging test to rule out pulmonary embolism. Curr. Opin. Pulmonary Med. 17, 380–386 (2011).
Pasha, S. M. et al. NT-pro-BNP levels in patients with acute pulmonary embolism are correlated to right but not left ventricular volume and function. Thromb. Haemost. 108, 367–372 (2012).
Kearon, C. et al. Antithrombotic therapy for VTE disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 141 (Suppl. 2), e419S–e494S (2012). This paper provides evidence-based guidelines for the treatment of VTE.
Schulman, S. et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N. Engl. J. Med. 361, 2342–2352 (2009).
Schulman, S. et al. Treatment of acute venous thromboembolism with dabigatran or warfarin and pooled analysis. Circulation 129, 764–772 (2014).
Hokusai, V. T. E. I. et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N. Engl. J. Med. 369, 1406–1415 (2013).
EINSTEIN Investigators et al. Oral rivaroxaban for symptomatic venous thromboembolism. N. Engl. J. Med. 363, 2499–2510 (2010).
The EINSTEIN–PE Investigators. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N. Engl. J. Med. 366, 1287–1297 (2012).
Agnelli, G. et al. Oral apixaban for the treatment of acute venous thromboembolism. N. Engl. J. Med. 369, 799–808 (2013).
Spyropoulos, A. C. & Lin, J. Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations. J. Manag. Care Pharm. 13, 475–486 (2007).
Ruppert, A., Steinle, T. & Lees, M. Economic burden of venous thromboembolism: a systematic review. J. Med. Econom. 14, 65–74 (2011).
Lee, A. Y. et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N. Engl. J. Med. 349, 146–153 (2003).
Jonas, D. E. & McLeod, H. L. Genetic and clinical factors relating to warfarin dosing. Trends Pharmacol. Sci. 30, 375–386 (2009).
Pirmohamed, M. et al. A randomized trial of genotype-guided dosing of warfarin. N. Engl. J. Med. 369, 2294–2303 (2013).
Kimmel, S. E. et al. A pharmacogenetic versus a clinical algorithm for warfarin dosing. N. Engl. J. Med. 369, 2283–2293 (2013).
Mearns, E. S. et al. Quality of vitamin K antagonist control and outcomes in atrial fibrillation patients: a meta-analysis and meta-regression. Thromb. J. 12, 14 (2014).
van Es, N., Coppens, M., Schulman, S., Middeldorp, S. & Buller, H. R. Direct oral anticoagulants compared with vitamin K antagonists for acute venous thromboembolism: evidence from Phase 3 trials. Blood 124, 1968–1975 (2014).
Meyer, G. et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N. Engl. J. Med. 370, 1402–1411 (2014).
Popuri, R. K. & Vedantham, S. The role of thrombolysis in the clinical management of DVT. Arterioscler. Thromb. Vasc. Biol. 31, 479–484 (2011).
Schulman, S. et al. Extended use of dabigatran, warfarin, or placebo in venous thromboembolism. N. Engl. J. Med. 368, 709–718 (2013).
Noble, S., Lewis, R., Whithers, J., Lewis, S. & Bennett, P. Long-term psychological consequences of symptomatic pulmonary embolism: a qualitative study. BMJ Open 4, e004561 (2014).
Kahn, S. R. et al. Determinants of health-related quality of life during the 2 years following deep vein thrombosis. J. Thromb. Haemost. 6, 1105–1112 (2008).
Ashrani, A. A. et al. Impact of venous thromboembolism, venous stasis syndrome, venous outflow obstruction and venous valvular incompetence on quality of life and activities of daily living: a nested case–control study. Vasc. Med. 15, 387–397 (2010).
van Korlaar, I. et al. Quality of life in venous disease. Thromb. Haemost. 90, 27–35 (2003).
Kahn, S. R. et al. Prospective evaluation of health-related quality of life in patients with deep venous thrombosis. Arch. Intern. Med. 165, 1173–1178 (2005). This study confirms the causal association between PTS and reduced quality of life.
Klok, F. A. et al. The post-PE syndrome: a new concept for chronic complications of pulmonary embolism. Blood Rev. 28, 221–226 (2014). This comprehensive review introduces the concept of post-pulmonary embolism syndrome as a common outcome, analogous to PTS after DVT.
Kahn, S. R. et al. Compression stockings to prevent post-thrombotic syndrome: a randomised placebo-controlled trial. Lancet 383, 880–888 (2014). This placebo-controlled trial shows that early application of compression stockings after acute DVT fails to prevent PTS.
Kahn, S. R. et al. Graduated compression stockings to treat acute leg pain associated with proximal DVT. A randomised controlled trial. Thromb. Haemost. 112, 1137–1141 (2014).
Brandjes, D. P. et al. Randomised trial of effect of compression stockings in patients with symptomatic proximal-vein thrombosis. Lancet 349, 759–762 (1997).
Prandoni, P. et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann. Intern. Med. 141, 249–256 (2004).
Sorensen, H. T., Mellemkjaer, L., Steffensen, F. H., Olsen, J. H. & Nielsen, G. L. The risk of a diagnosis of cancer after primary deep venous thrombosis or pulmonary embolism. N. Engl. J. Med. 338, 1169–1173 (1998).
Franchini, M. & Mannucci, P. M. Association between venous and arterial thrombosis: clinical implications. Eur. J. Internal Med. 23, 333–337 (2012).
Becattini, C., Vedovati, M. C., Ageno, W., Dentali, F. & Agnelli, G. Incidence of arterial cardiovascular events after venous thromboembolism: a systematic review and a meta-analysis. J. Thromb. Haemost. 8, 891–897 (2010).
Spencer, F. A., Ginsberg, J. S., Chong, A. & Alter, D. A. The relationship between unprovoked venous thromboembolism, age, and acute myocardial infarction. J. Thromb. Haemost. 6, 1507–1513 (2008).
Rectenwald, J. E. et al. Experimental pulmonary embolism: effects of the thrombus and attenuation of pulmonary artery injury by low-molecular-weight heparin. J. Vasc. Surg. 43, 800–808 (2006).
Diaz, J. A. et al. Plasma DNA is elevated in patients with deep vein thrombosis. J. Vasc. Surg. Venous Lymphat. Disord. 1, 341–348.e1 (2013).
Jimenez-Alcazar, M. et al. Impaired DNase1-mediated degradation of neutrophil extracellular traps is associated with acute thrombotic microangiopathies. J. Thromb. Haemost. http://dx.doi.org/10.1111/jth.12796 (2014).
Pabinger, I. & Posch, F. Flamethrowers: blood cells and cancer thrombosis risk. Hematology Am. Soc. Hematol. Educ. Program 410–417 (2014).
Lu, G. et al. A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat. Med. 19, 446–451 (2013).
Honickel, M. et al. Reversal of dabigatran anticoagulation ex vivo: porcine study comparing prothrombin complex concentrates and idarucizumab. Thromb. Haemost. 113, 728–740 (2015).
Mackman, N. Triggers, targets and treatments for thrombosis. Nature 451, 914–918 (2008).
Renne, T. et al. Defective thrombus formation in mice lacking coagulation factor XII. J. Exp. Med. 202, 271–281 (2005).
Kleinschnitz, C. et al. Targeting coagulation factor XII provides protection from pathological thrombosis in cerebral ischemia without interfering with hemostasis. J. Exp. Med. 203, 513–518 (2006).
Howard, E. L., Becker, K. C., Rusconi, C. P. & Becker, R. C. Factor IXa inhibitors as novel anticoagulants. Arterioscler. Thromb. Vasc. Biol. 27, 722–727 (2007).
Schumacher, W. A., Luettgen, J. M., Quan, M. L. & Seiffert, D. A. Inhibition of factor XIa as a new approach to anticoagulation. Arterioscler. Thromb. Vasc. Biol. 30, 388–392 (2010).
Zhang, H. et al. Inhibition of the intrinsic coagulation pathway factor XI by antisense oligonucleotides: a novel antithrombotic strategy with lowered bleeding risk. Blood 116, 4684–4692 (2010).
Muller, F., Gailani, D. & Renne, T. Factor XI and XII as antithrombotic targets. Curr. Opin. Hematol. 18, 349–355 (2011).
Povsic, T. J. et al. Use of the REG1 anticoagulation system in patients with acute coronary syndromes undergoing percutaneous coronary intervention: results from the phase II RADAR-PCI study. EuroIntervention. 10, 431–438 (2014).
Vavalle, J. P. et al. The effect of the REG2 anticoagulation system on thrombin generation kinetics: a pharmacodynamic and pharmacokinetic first-in-human study. J. Thromb. Thrombolysis 38, 275–284 (2014).
Hagedorn, I. et al. Factor XIIa inhibitor recombinant human albumin infestin-4 abolishes occlusive arterial thrombus formation without affecting bleeding. Circulation 121, 1510–1517 (2010).
Decrem, Y. et al. Ir-CPI, a coagulation contact phase inhibitor from the tick Ixodes ricinus, inhibits thrombus formation without impairing hemostasis. J. Exp. Med. 206, 2381–2395 (2009).
Larsson, M. et al. A factor XIIa inhibitory antibody provides thromboprotection in extracorporeal circulation without increasing bleeding risk. Sci. Transl Med. 6, 222ra217 (2014). This study shows that a factor XIIa-specific antibody reduces activation of coagulation in an extracorporeal membrane oxygenator, suggesting that targeting factor XIIa might reduce thrombosis.
Büller, H. R. et al. Factor XI antisense oligonucleotide for prevention of venous thrombosis. N. Engl. J. Med. 372, 232–240 (2015). This study shows that antisense therapy to decrease factor XI levels reduces VTE in patients undergoing elective primary total knee arthroplasty, without increasing bleeding.
Ramacciotti, E. et al. P-selectin/ PSGL-1 inhibitors versus enoxaparin in the resolution of venous thrombosis: a meta-analysis. Thromb. Res. 125, e138–e142 (2010).
Washington University School of Medicine. The Attract Study [online], (2009).
Ay, C. et al. High concentrations of soluble P-selectin are associated with risk of venous thromboembolism and the P-selectin Thr715 variant. Clin. Chem. 53, 1235–1243 (2007).
Ay, C. et al. High plasma levels of soluble P-selectin are predictive of venous thromboembolism in cancer patients: results from the Vienna Cancer and Thrombosis Study (CATS). Blood 112, 2703–2708 (2008).
Bucciarelli, P. et al. Circulating microparticles and risk of venous thromboembolism. Thromb. Res. 129, 591–597 (2012).
Ay, C., Freyssinet, J. M., Sailer, T., Vormittag, R. & Pabinger, I. Circulating procoagulant microparticles in patients with venous thromboembolism. Thromb. Res. 123, 724–726 (2009).
Huang, W., Goldberg, R. J., Anderson, F. A., Kiefe, C. I. & Spencer, F. A. Secular trends in occurrence of acute venous thromboembolism: the Worcester VTE study Am. J. Med. 127, 829–839.e5 (2014).
Clot Connect. Venous Thromboembolism Resource. [online], (2010).
Kerbauy, M. N., de Moraes, F. Y., Kerbauy, L. N., Conterno Lde, O. & El-Fakhouri, S. Venous thromboprophylaxis in medical patients: an application review. Rev. Assoc. Med. Bras. 59, 258–264 (in Portuguese) (2013).
Anderson, F. A. Jr. et al. Changing clinical practice. Prospective study of the impact of continuing medical education and quality assurance programs on use of prophylaxis for venous thromboembolism. Arch. Intern. Med. 154, 669–677 (1994).
Acknowledgements
The authors thank T. Wakefield, P. Henke, D. Myers and R. Kasthuri for reading the manuscript, and A. Conrad for assistance with Figure 1.
Author information
Authors and Affiliations
Contributions
Introduction (N.M. and A.S.W); Epidemiology (F.R.R.); Mechanisms/pathophysiology (N.M. and A.S.W.); Diagnosis, screening and prevention (I.H.J. and J.I.W.); Management (G.A.); Quality of life (T.B.); Outlook (N.M. and A.S.W.); and overview of the Primer (N.M.).
Corresponding authors
Ethics declarations
Competing interests
G.A. has received personal fees from Boehringer Ingelheim, Bayer Healthcare, Daiichi Sankyo, Sanofi, Pfizer and Bristol–Myers Squibb; all are outside the scope of the submitted work. T.B. has received honoraria for participating in scientific advisory boards for Boehringer Ingelheim and Daiichi Sankyo, and has received an unconditional travel grant from Boehringer Ingelheim. N.M. served as consultant to Merck and Bayer. F.R.R. is listed on several patents for prothrombotic gene variants. J.I.W. served as a consultant to and received honoraria from Boehringer Ingelheim, Bristol–Myers Squibb, Pfizer, Johnson & Johnson, Daiichi Sankyo, ISIS Pharmaceuticals and Portola. A.S.W. served as a consultant to Merck. I.H.J. has no competing financial interests to report.
Rights and permissions
About this article
Cite this article
Wolberg, A., Rosendaal, F., Weitz, J. et al. Venous thrombosis. Nat Rev Dis Primers 1, 15006 (2015). https://doi.org/10.1038/nrdp.2015.6
Published:
DOI: https://doi.org/10.1038/nrdp.2015.6
This article is cited by
-
From compression to diagnosis: identification of superior vena cava syndrome using point-of-care ultrasound in the emergency department
International Journal of Emergency Medicine (2024)
-
Venous anomalies and thromboembolism
Thrombosis Journal (2023)
-
Dynamic predictions of postoperative complications from explainable, uncertainty-aware, and multi-task deep neural networks
Scientific Reports (2023)
-
Nomogram for predicting postoperative deep vein thrombosis in patients with spinal fractures caused by high-energy injuries
Archives of Orthopaedic and Trauma Surgery (2023)
-
Treatment Progress in Diminished Ovarian Reserve: Western and Chinese Medicine
Chinese Journal of Integrative Medicine (2023)